Abstract
Introduction
Bipolar disorder (BD) is a lifelong illness characterized by periods of recurrent depression and mania. It presents clinically in a myriad of forms with a symptom profile that can vary considerably from one individual to the next (Alda et al., 2009; Malhi et al., 2009c). However, it is bipolar depression that exerts greater disability and generally requires more sophisticated treatment (Malhi et al., 2003, 2009b, 2009c). More critical, however, is the need to confer mood stability and provide effective prophylaxis (Malhi et al., 2012), and it is in this context that lithium truly distinguishes itself.
Rediscovered 60 years ago by John Cade for the treatment of BD (Cade, 1949), lithium has been tested and trialled repeatedly against placebo and new comparators. The migration of anticonvulsants and then antipsychotics into the realm of lithium (Malhi et al., 2011a) has steadily expanded the pharmacological armamentarium for the treatment of BD, making it significantly more complex (Malhi et al., 2012). An additional level of sophistication has been brought about by the introduction of newly developed psychological therapies, social interventions and educational approaches (Malhi et al., 2009c, 2009e). Remarkably, despite such dramatic changes lithium has managed to retain its salience, as evidenced by its prominent positioning in recent sets of guidelines (Malhi et al., 2009d; NICE, 2006; Yatham et al., 2005). Its use for more than half a century, beginning with the treatment of BD and later in antidepressant augmentation, has generated a wealth of empirical evidence in addition to which it has been found to have a unique therapeutic profile that includes for instance its anti-suicidal and neuroprotective properties (Grunze et al., 2009; Malhi and Gershon, 2009; van Erp et al., 2011). Despite being a veteran amongst putative mood stabilizers its ‘gold standard’ status has been protected by a raft of new data that have been spurred by an increase in the detection and diagnosis of BD.
The reasons for this growth, in particular over the past decade, are the subject of much debate but the emergence of new treatments for BD is undoubtedly either a cause or a consequence (Malhi et al., 2011a). Most of these ‘novel’ medications are in fact ‘old drugs’ with a new indication, and the majority have initially focused on the acute phases of the illness, in particular mania. Of note, thus far none have managed to improve upon lithium, especially in terms of long-term treatment and prophylaxis (Goodwin and Malhi, 2007; Malhi et al., 2011a). By translating the Rosetta Stone of its mechanism of action into an understanding of the core pathophysiology of BD, lithium remains the critical key to unlocking the aetiology of the disorder.
In clinical practice the use of lithium has diminished somewhat in favour of other agents (Malhi, 2010) and therefore doctors, especially those who have trained more recently, need to be reacquainted with its use. Therefore, this review provides a comprehensive and evidence-based practical guide to the administration of lithium therapy, in particular for the treatment of BD.
Methods
A comprehensive search of peer-reviewed publications using MEDLINE, EMBASE and PsychINFO was conducted regarding the use of lithium in the treatment of BD. Keywords used to identify relevant articles and limit our search strategy included: adherence, bipolar depression, bipolar disorder, compliance, dosing schedules, hypomania, indications, lithium, maintenance therapy, mania, pharmacodynamics, pharmacokinetics, prophylaxis, regime, response, safety, side effects, suicide, tolerability and toxicity. In addition, literature known to the authors from books and book chapters was also scrutinized and the findings from the literature were then synthesized.
The paper is structured as follows. Initially, an outline of the science of lithium therapy is provided so as to aid an understanding of how lithium interacts with key systems within the body. The putative mechanisms of action of lithium are also briefly considered with respect to its psychotropic pharmacology. The therapeutic profile of lithium is then carefully reviewed examining both its efficacy, in particular in BD, and its tolerability is discussed next in the context of potential side effects, toxicity and poor compliance. Finally, the use of lithium in clinical practice is outlined with particular emphasis on practical aspects such as oral dosing, plasma monitoring and the tailoring of treatment to individual needs.
The science of lithium therapy
Preparations
Clinically, lithium is administered as a salt in the form of lithium carbonate, lithium citrate, lithium chloride or lithium sulfate. Conjugation of lithium in these various forms alters its pharmacokinetics. In a study that compared the various lithium salts, it was found that the chloride and sulfate preparations reach peak plasma concentrations 1 hour after oral administration as compared to 4 hours for the carbonate preparation (Altamura et al., 1977). This is because lithium carbonate is the least water-soluble of the various salts and as such is absorbed more slowly in the upper gastrointestinal tract than the others (Altamura et al., 1977; Grandjean and Aubry, 2009a). All three salts have similar pharmacokinetic properties with respect to volume of distribution, bioavailability and half-life (Altamura et al., 1977; Widerlov, 1976) and both immediate or slow release forms of lithium carbonate are currently available in Australia.
Slow release (SR) medications produce relatively more stable plasma lithium levels as compared to immediate release (IR) preparations because the latter are generally thought to result in greater peaks and troughs, but the evidence for this is somewhat equivocal. Some researchers have postulated that the peak plasma concentration of a SR lithium formulation is more likely to be delayed rather than lowered (Fyro et al., 1970; Johnson and Hunt, 1984; Perry et al., 1981), but there is also evidence to suggest that peak plasma concentrations of SR lithium preparations are lower than those of IR preparations (Grandjean and Aubry, 2009a; Shelley and Silverstone, 1986), even though elimination rates do not differ significantly (Shelley and Silverstone, 1986). Further, it should be noted that plasma lithium concentrations in patients taking SR lithium are not constant across individuals possibly because of individual differences in gastrointestinal tract functioning (Fyro et al., 1970).
Pharmacokinetics
Lithium is a naturally occurring ion that consequently is water-soluble, does not bind to plasma proteins and is able to cross the blood–brain barrier. It is absorbed predominantly through the stomach (Fyro et al., 1970) without undergoing metabolism but is excreted almost exclusively by the kidney (Gitlin, 1999; Sproule et al., 2000) (Figure 1). Therefore, before commencing lithium therapy it is important to consider renal function and this can be assessed by measuring urea and electrolytes along with determining the creatinine clearance as indicated (see Table 2). The latter is a product of muscle protein metabolism that is released into the blood circulation at a constant rate and distributed in total body water. Creatinine is therefore filtered passively through the glomerulus in proportion to the glomerular filtration rate (Pachoreck, 2001) and if this decreases the excretion of lithium is impaired and its plasma levels increase, resulting in toxicity (see later).
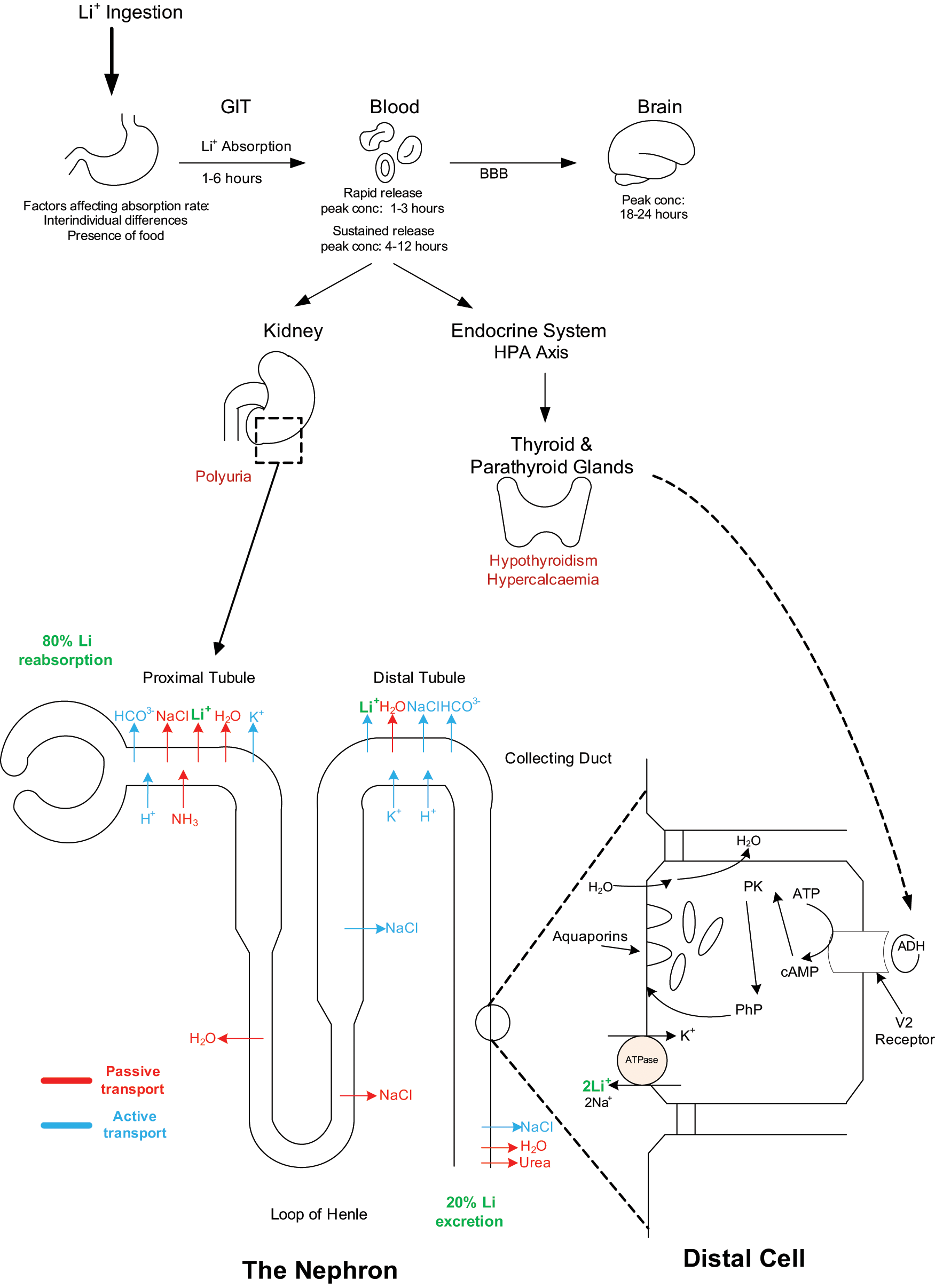
Pharmacokinetics and pharmacodynamics of lithium. This figure shows the effects of lithium on particular organs in the body as well as its movement through the nephron. Pharmacokinetics: Following
Peak lithium plasma levels are reached 1–3 hours after administration of IR preparations (Altamura et al., 1977) and 4–12 hours after SR preparations (Carson and Roberts, 2001; Chen et al., 2004). Lithium in the brain peaks about 24 hours after ingestion, and this is primarily due to the lower permeability of the blood–brain barrier (Chen et al., 2004). After a day or so of lithium administration the mean ratio of its concentration in plasma to cerebrospinal fluid is 3.6:1 (Sproule et al., 2000).
BD has higher rates of primary thyroid abnormalities than the general population (Bauer et al., 2008), but treatment with lithium increases the likelihood of transient subclinical hypothyroidism (T3/T4 are normal but thyroid-stimulating hormone (TSH) is elevated). With longer term use overt hypothyroidism (reduced T3/T4 and elevated TSH) occurs in approximately 10% of patients and this figure is higher still in women (13%), and especially in those who start treatment between the ages of 40 and 60 years (20%) (Johnston and Eagles, 1999; Lazarus, 1998).
With similar properties to sodium and potassium ions, lithium has an entry point into renal cells but transport out of cells is more limited, and gradually this results in intracellular accumulation (Timmer and Sands, 1999). Specifically, lithium is filtered completely in the glomeruli, and approximately 80% is reabsorbed in the proximal tubules (see Figure 1). This obviates the need for further absorption in the loop of Henle and therefore the remainder accumulates in the collecting tubule (Eyer et al., 2006; Gitlin, 1999; Sproule et al., 2000).
Pharmacologically, lithium diminishes the response of cyclic adenosine monophosphate (cAMP) to antidiuretic hormone (ADH), which in turn limits the ability of the kidney to store free water. In conjunction with the accumulation of lithium in the collecting tubules, the effect of lithium on ADH decreases the ability of the kidneys to concentrate urine (Gitlin, 1999). This predisposes individuals to the development of diabetes insipidus that can ultimately affect 10% of lithium-treated patients (Bendz and Aurell, 1999). Though typically regarded a long-term side effect of lithium therapy, longitudinal studies suggest that a detectable impairment of renal concentrating ability may emerge within the first few years of exposure to lithium (Gitlin, 1999).
Pharmacodynamics
The pharmacodynamic properties of lithium are influenced by factors such as age, renal function, pregnancy and lactation as well as concomitant medication. All have an effect on plasma lithium levels. Clinicians therefore need to adjust the oral dose of lithium according to the circumstances of each individual ‘and monitor the plasma levels closely to ensure a therapeutic level is maintained and the risk of toxicity is minimized.
Age
With age, renal function declines (Lindeman et al., 1985), and for those on lithium this translates to diminishing lithium clearance. Medical co-morbidities, such as chronic heart failure and hypertension that compromise renal clearance, are also increasingly more likely (Fliser et al., 1997) and therefore, clinically, lower oral doses are needed to achieve therapeutic plasma levels in the elderly (Eastham et al., 1998).
Further, elderly patients are often on multiple medications that can affect the concentration of lithium. For example, the net loss of sodium with diuretics leads to increased reabsorption in the proximal tubules. Thus, diuretics increase the plasma levels of lithium (Thomsen and Leyssac, 1986) and therefore elderly patients generally require lower oral doses of lithium (about 25–30% lower) to achieve therapeutic plasma concentrations. Of note, levels should be monitored more closely when there are changes to primary or comorbid treatment (Eastham et al., 1998; Wijeratne and Draper, 2011; Wilkinson et al., 2002).
Renal function
The risk of lithium intoxication increases greatly for patients with abnormal renal function. In the case of acute renal dysfunction, the dose of lithium should be decreased and its plasma levels titrated to prevent toxicity. Renal insufficiency and nephritic syndrome are relative contraindications to lithium therapy (Grandjean and Aubry, 2009a; Morriss et al., 2008); however, moderate renal dysfunction can be managed provided lithium levels are closely monitored. This option should be considered in cases where lithium has been shown to be an effective prophylactic treatment (Gash et al., 1995).
Pregnancy and lactation
Bipolar disorder has implications for women as regards their ability to maintain wellness and have a family. Women of childbearing age and pregnant women with BD that have been exposed to lithium in the first trimester of pregnancy possibly incur an increased risk of congenital heart failure (Malhi et al., 2009c). Specifically, the risk of Ebstein’s anomaly is thought to be slightly elevated in the first trimester (4–12%) as compared to the general population (2–4%) (Cohen et al., 1994; Viguera and Cohen, 1998); however, recently this figure has been considered to be an overestimation (McKnight et al., 2012). In practice, the risk–benefit ratio of lithium therapy during the first trimester needs to be considered carefully on a case-by-case basis.
Further, all pregnant women with BD should have their mood monitored closely as they are a population at increased risk of relapse (Malhi et al., 2009c). This is because during pregnancy the glomerular filtration rate increases and this in turn increases lithium clearance by up to 50%. The plasma lithium levels therefore fall significantly and put the mother at potential risk of relapse (Llewellyn et al., 1998). Further, following childbirth, the mother’s blood volume rapidly returns to normal along with the rate of renal clearance of medications. Therefore, pregnant women need to reduce the dose of lithium or temporarily stop treatment 1 or 2 days prior to the expected delivery date (Grandjean and Aubry, 2009a), or have a treatment plan to increase hydration (Llewellyn et al., 1998) in order to prevent lithium intoxication. Post delivery, mothers should reinstitute treatment immediately as the risk of relapse at this time is more than threefold higher (80% risk of relapse as compared to 24% in non-pregnant women) (Viguera and Cohen, 1998).
Like many drugs, lithium is excreted in breast milk and as a consequence plasma levels in infants whose mothers are taking lithium are often high (30–50% that of the mother) (Viguera et al., 2007; Yoshida et al., 1999). Hence, mothers who require lithium therapy may need to reconsider breastfeeding in the context of an individualized risk–benefit assessment (Grandjean and Aubry, 2009a). Should the mother continue to breastfeed, the infant needs to be monitored closely for toxicity as the effects of lithium on infants are not known (Grandjean and Aubry, 2009b; Yoshida et al., 1999).
Co-medication
Co-administered medications can potentially increase lithium retention in the body. For instance, diuretics increase water clearance and this in turn decreases lithium clearance, especially those diuretics that act by depleting sodium (Okusa and Crystal, 1994). Non-steroidal anti-inflammatory drugs (Phelan et al., 2003), angiotensin converting enzyme (ACE) inhibitors (Finley et al., 1996) and withdrawal of caffeine (Mester et al., 1995) have also been shown to increase plasma lithium concentrations, although the mechanism by which this occurs is not yet well known (Grandjean and Aubry, 2009a; Okusa and Crystal, 1994; Phelan et al., 2003). Nevertheless, lithium dosage should be reduced when co-prescribing with these medications (Grandjean and Aubry, 2009b).
Biology and mechanisms of action
Neurotransmitters
The precise mechanisms by which lithium achieves its many therapeutic benefits is not yet fully understood; however, a substantive body of evidence implicates a number of neutrotransmitters.
Glutamate
For instance, lithium has been shown to have effects on the excitatory neurotransmitter glutamate, the concentrations of which appear to be increased in mania and decreased in depression (Berk and Malhi, 2011). Studies on both monkey and mouse cerebrocortical slices suggest that acute lithium administration increases the availability of glutamate in the post-synaptic neuron via NMDA (N-methyl
Dopamine
Dopamine (DA) is another neurotransmitter that has been found to increase in patients with mania and hence is a potential target for the action of lithium (Post et al., 1980). Indeed, studies in rats have shown that, when treated with a DA re-uptake inhibitor, lithium-treated rats had lower levels of DA compared to those that were lithium naïve (Ferrie et al., 2005, 2006). This suggests that lithium modulates the synthesis and/or release of DA in the presynaptic terminal. In addition, lithium may also exert its effects on post-synaptic DA transmission by attenuating G-protein functioning and inhibiting downstream second messenger systems such as adenlyl cyclase and cyclic adenosine monophosphate pathways (Manji and Lenox, 2000).
GABA
A third neurotransmitter that lithium increases the concentration of within the brain is the inhibitory neurotransmitter γ-aminobutyric acid (GABA), and theoretically this change might decrease neural over-excitation and thereby alleviate mania (Brunello and Tascedda, 2003; Lenox et al., 1998; Post et al., 2003).
Second messenger signaling
GSK-3
Glycogen synthase kinase-3 (GSK-3) is an important downstream target of monoaminergic neurotransmission and has direct involvement in modulating synaptic plasticity, cell structure and resilience, and has been recently identified as a prominent target for lithium’s therapeutic and neuroprotective effects (Berk et al., 2010b; Ikonomov and Manji, 1999; Machado-Vieira et al., 2009b; O’Brien and Klein, 2009). Increased GSK-3 has been shown to be associated with behaviours of mania in mice (Marmol, 2008) and lithium has been shown to directly inhibit GSK-3, contributing therefore to its anti-manic properties (Ikonomov and Manji, 1999; Machado-Vieira et al., 2009b). However, in some individuals BD is increasingly being regarded as a neurodegenerative disease with patients displaying atrophy in multiple brain regions compared to healthy controls (Savitz and Drevets, 2009). In this context the inhibition of GSK-3 by lithium perhaps explains its neuroprotective properties (Tajes et al., 2009), in particular its defence against cell death caused by excessive excitatory neurotransmission such as during a manic episode (Chiu and Chuang, 2010; Post et al., 2003).
Inositol
One of the most widely accepted hypotheses for lithium’s mode of action is the ‘inositol depletion hypothesis’. Briefly, inositol is responsible for maintaining myo-inositol levels (mI), which in turn maintain phospholipid concentrations of cell membranes. Phospholipids are important for the binding of secondary messengers such as diaglycerol and inositol triphosphate (Manji and Lenox, 2000), which are involved in receptor-mediated signal transduction within the central nervous system (CNS) (Machado-Vieira et al., 2009a; Ikonomov and Manji, 1999). Hence, the maintenance of mI levels is important for the efficiency of cellular signaling (Deranieh and Greenberg, 2009).
In both in vivo and in vitro studies, lithium has been shown to interfere with the recycling of inositol, reducing its availability and thus reducing cellular signaling (Manji and Lenox, 2000). It is hypothesized that lithium acts on regions of the brain where inositol is in excess, and indeed preliminary evidence from magnetic resonance imaging studies shows that mI levels are increased in mania and depression but unchanged in euthymic patients (Chen et al., 1999; Ikonomov and Manji, 1999). Furthermore, lithium does not inhibit inositol in euthymic patients, which lends support to the hypothesis that lithium only reduces the levels of inositol when these are in excess (Silverstone et al., 2005).
Clinical profile
Efficacy within BD
Acute mania
The efficacy of lithium as a first-line treatment in acute mania has been demonstrated in clinical trials spanning several decades (Baldessarini and Tondo, 2000). The evidence from robust randomized controlled trials suggests that lithium is efficacious in the treatment of mania when compared to valproate (Bowden et al., 2010; Yildiz et al., 2011), and antipsychotics such as olanzapine and risperidone (Segal et al., 1998; Shafti, 2010; Yildiz et al., 2011) or placebo (Geddes et al., 2004; Gershon et al., 2009; Grunze et al., 2009; Malhi et al., 2009a; Yildiz et al., 2011). Consequently, the majority of clinical practice guidelines recommend lithium monotherapy as a first-line option for the treatment of acute mania (Malhi et al., 2009a, 2009d; NICE, 2006; Yatham et al., 2005, 2006) even though in practice its anti-manic action can take up to 6–10 days to take full effect. This is significantly slower than most atypical antipsychotics and so in reality lithium in combination with an antipsychotic is a better option for the treatment of more severe mania than monotherapy of either agent (Grunze et al., 2009; Malhi et al., 2009a; Tohen et al., 2002).
Acute bipolar depression
The evidence for lithium monotherapy in the treatment of acute depression is not as impressive as for acute mania partly because lithium takes 6–8 weeks to achieve an antidepressant effect, but also because in practice depression often has important psychological and social contributors to its variance, and requires a correspondingly broader therapeutic approach. Studies have shown that lithium is more likely to be effective when used to treat ‘classic’ BD (Gershon et al., 2009) in which there are discrete episodes of recurrent bipolar depression separated by distinct periods of remission. Therefore, the evidence from bipolar depression randomized controlled trials is skewed by high withdrawal rates because of prior exposure that obscures efficacy, and the inclusion of patients without bipolar I (Fountoulakis et al., 2008a). Indeed, recent reviews acknowledge a lack of good quality randomized controlled trials for lithium in bipolar depression, making it difficult to evaluate its efficacy using meta-analyses (Van Lieshout and MacQueen, 2010; Vieta et al., 2010).
Interestingly, more recent clinical trials show that when compared with placebo lithium fails to achieve significant superiority (Bhagwagar and Goodwin, 2002; Fountoulakis, 2010; Van Lieshout and MacQueen, 2010). Nevertheless, lithium is widely regarded as an important agent in the treatment of bipolar depression by current clinical practice guidelines (Grandjean and Aubry, 2009c; Vieta et al., 2010; Yatham et al., 2009).
Maintenance and prophylaxis
Lithium monotherapy has demonstrated superiority in the prophylaxis of mania, depression, and suicidal behaviour (Malhi et al., 2009a; Young and Hammond, 2007) both in earlier studies conducted in the 1970s that specifically appraised lithium and in more recent studies where it has been used as a comparator for newer agents (Coryell, 2009). Starting maintenance early provides the best likelihood of good long-term outcomes (Freeman and Freeman, 2006; Geddes et al., 2004) and the BALANCE study has underscored the real world effectiveness of lithium as a true prophylactic agent (Geddes et al., 2010). A recent meta-analysis of clinical trials showed that lithium therapy reduces the overall risk of relapse by 40–61% and that its effect in preventing a manic relapse is more pronounced than for depression (40% decreased risk compared with 22%) (Geddes et al., 2004). Further, abrupt cessation or reduction of lithium levels during maintenance therapy can cause a recurrence of symptoms and lead to relapse in 60–80% of patients (Cavanagh et al., 2004; Klein et al., 1992; Perlis et al., 2002; Silverstone, 2000; Yatham et al., 2005).
Recent reviews of prospective placebo controlled trials in well-characterized BD populations that achieved plasma lithium concentrations of 0.8–1.2 mmol/L, report effective prophylaxis (Grandjean and Aubry, 2009c), but an optimal effect may be achievable at lower doses (Perlis et al., 2002).
Rapid cycling and mixed states
Rapid cycling, defined by the occurrence of four or more episodes per year, occurs in 13–20% of bipolar patients (Goldberg and Berk, 2010). Conditions such as hypothyroidism, antidepressants and substance abuse possibly contribute to the development of rapid cycling (Coryell, 2005; Yatham et al., 2005), and this is a concern because patients that are rapid cycling are often less responsive and less adherent to treatment than those with conventional patterns of BD. Further, in clinical practice, rapid cycling presentations can be difficult to reliably differentiate from mixed episodes and manifestations of borderline personality (Malhi et al., 2009a). The traditional view is that lithium is less effective in rapid cycling and mixed states, especially in terms of preventing relapse, though there is evidence that it decreases symptom severity and reduces morbidity (Coryell, 2005; Grandjean and Aubry, 2009a; Muzina, 2009; Suppes et al., 2008). Therefore, on balance, current wisdom advocates the use of lithium for rapid cycling in combination with other psychotropic drugs, particularly atypical antipsychotics (Coryell, 2005; Grandjean and Aubry, 2009c; Muzina, 2009).
Positioning in guidelines
The positioning of lithium in the majority of clinical practice guidelines reflects its efficacy in treating the respective phases of BD (Figure 2). By definition, treatment guidelines are designed to inform clinicians of best practice and the evidence for this is usually based on clinical trial data. This has some inherent limitations. First, the ‘types’ of BD studied in clinical trials has undergone significant transformation with a shift towards BD II and bipolar spectrum disorders; second, most clinical trials are of relatively short duration (12–16 weeks) and therefore do not reflect the reality of lifelong treatment; third, there is a paucity of high-quality data from which to extrapolate to real-world management (Nivoli et al., 2011); and lastly, because of service filter effects, most trials in academic tertiary centres in the developed world unwittingly concentrate on the most ill and refractory individuals, skewing results towards the negative. It is because of this that there are many discrepancies between guidelines and therefore in addition to using the evidence it is important to draw on experience to create individualized treatment regimens (Malhi et al., 2009c). In this context, lithium has a substantive empirical evidence base and has therefore managed to retain prominence in most clinical practice guidelines.
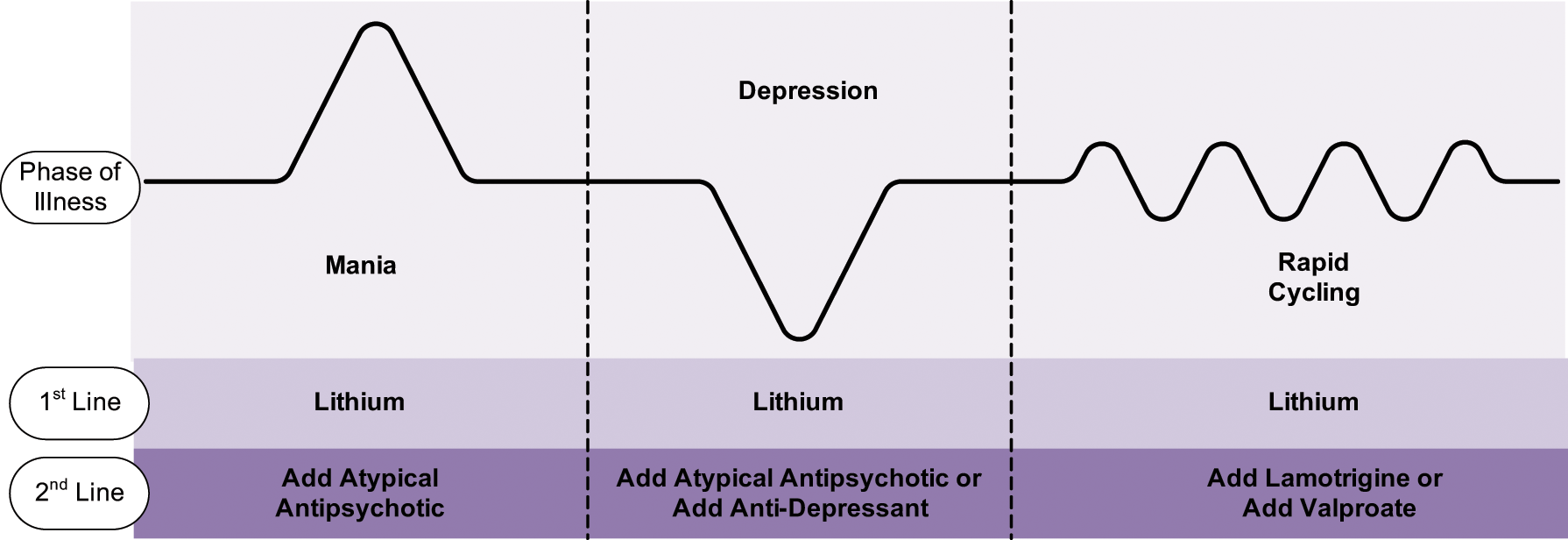
Summary of the current recommendations for lithium use in phases of illness of BD. Lithium is recommended as a first-line treatment by all guidelines across all phases of the illness. Mania: Lithium is recommended in combination with an atypical antipsychotic for severely ill patients (APA, 2002; Malhi et al., 2009d; NICE, 2006; Yatham et al., 2005) or monotherapy for less ill patients (APA, 2002; NICE, 2006). Depression: Lithium monotherapy is recommended first-line, and the addition of an atypical antipsychotic or second mood stabilizer should be considered if the patient is not already on treatment (APA, 2002; Malhi et al., 2009d; NICE, 2006). Rapid cycling: Lithium monotherapy is not as effective in rapid cycling BD but is recommended as a first-line option in combination with other treatments (Yatham et al., 2005). It is also a valid second-line treatment for this phase after atypical antipsychotic medications or valproate have been trialled (APA, 2002; Malhi et al., 2009d; NICE, 2006).
Specificity within BD
Lithium is particularly suited to the treatment of ‘classic’ (manic-depressive) bipolar disorder. Its specificity for presentations characterized by recognizable recurrent episodes of mania or depression with clearly established euthymia in between means that it is less suited to complex manifestations of bipolarity (Gershon et al., 2009; Malhi et al., 2012). A corollary view, conceptualizing lithium response as a quality biomarker of core bipolarity, sheds an awkward light on the broader bipolar spectrum.
Efficacy in other disorders
Major (unipolar) depression
Lithium is superior to placebo in the maintenance phase of unipolar depression (Malhi et al., 2009a) and is effective in preventing relapse across a range of plasma levels (Cipriani et al., 2006; Hopkins and Gelenberg, 2000). Its efficacy has not been compared to other drugs in detail (Cipriani et al., 2006; Gershon et al., 2009) and therefore conventional antidepressants are used first-line for the treatment of major depression with lithium added for augmentation (Malhi et al., 2009a). In a naturalistic study that examined recurrences in patients with unipolar and bipolar depression, 59% of unipolar patients and 51% of bipolar patients had no recurrences in the 5 years after lithium prophylaxis was commenced, and 73.7% of the unipolar patients and 65.9% of bipolar patients reported a good outcome (Lepkifker et al., 2007).
Schizophrenia
The evidence for lithium use in schizophrenia is relatively weak with little impact on the core symptoms of the illness. Its principal therapeutic effect is on mood and it benefits those with affective and mood-related symptoms (Gershon et al., 2009; Malhi et al., 2009a). This again points to its potential role as a biological probe.
Additional properties
Suicide / suicidal behaviour
Suicide is a significant contributor to increased mortality rates in BD (Silverstone, 2000). The risk of suicide or suicide attempts in BD are about 10 times higher than in the general population (Baldessarini et al., 2006) and so of the major benefits of long-term lithium therapy is its demonstrated ability to reduce the risk of suicide/suicidal behaviour (Baldessarini et al., 2006; Bowden, 2000; Gershon et al., 2009; Goodwin et al., 2003; Malhi et al., 2009a, 2009c) provided patients adhere to treatment (Silverstone, 2000). In patients, the risk of suicide is six times greater for those not taking lithium as compared to those who are (Baldessarini et al., 2006) and, therefore, clinically, lithium reduces the risk of death and suicide by 60% and the risk of self-harm by 70% (Cipriani et al., 2005; Goodwin et al., 2003). Further, patients who do not respond well to lithium prophylaxis in terms of relapse are still less likely to attempt suicide (Muller-Oerlinghausen, 2001). This unique ‘anti-suicidal action’ of lithium remains an enigma, though it has been postulated that lithium targets impulsivity and aggression, both of which are thought to play a role in acts of suicide and self-harm (Baldessarini et al., 2006; Gershon et al., 2009; Young and Hammond, 2007).
Neuroprotective effects
As previously discussed, while it is not known exactly how lithium exerts its therapeutic effects, its actions on neurotransmitter release and cellular signalling cascades, promote neuronal viability by protecting them against neurotoxicity and apoptosis (Berk, 2009; Berk et al., 2011b; Post et al., 2003). The presence of increased neural glutamate (as found in mania) activates NMDA receptors and this produces excitatory neurotoxicity. Lithium protects against this process by inactivating these receptors in cultures of rat cerebellar granule cells (Nonaka et al., 1998). Lithium also increases the release of the inhibitory neurotransmitter, GABA, which mediates a reduction of excitatory neurotransmission and potentially prevents neurotoxicity and cell death (Brunello and Tascedda, 2003; Dixon and Hokin, 1998; Lenox et al., 1998; Post et al., 2003). In addition, lithium treatment increases neuroprotective proteins such as BDNF and Bcl-2, and decreases pro-apoptotic enzymes such as GSK-3. Together, these changes to cellular metabolism, neurotransmitters and factors not only prevent cell death, but over the long term produce changes that increase neurotrophy and alter gene transcription (Ikonomov and Manji, 1999; Machado-Vieira et al., 2009b; Marmol, 2008). Interestingly, N-acetyl-aspartate (NAA), a maker of neuronal function and viability, has been found to be increased at therapeutic levels of lithium (Marmol, 2008).
Evidence for neuroprotection also comes from neurostructural findings that show increased grey matter volumes in lithium-treated bipolar patients compared to those not treated (Machado-Vieira et al., 2009b). Of note, BD patients treated with lithium have larger hippocampal and amygdala volumes than healthy controls and those patients not on lithium therapy (Hallahan et al., 2011). Further, one study has shown that the efficacy of prophylactic treatment is related to executive function such that excellent lithium responders have better executive function than poor, or non-lithium responders. In addition, lithium may also reduce the risk of dementia (Kessing et al., 2010). The putative neuroprotective effects of lithium clearly warrant further investigation (Rybakowski et al., 2009).
Tolerability
Side effects
Lithium is associated with both acute and chronic side effects that can limit tolerability but, in practice, for the majority of patients the impact of these is transient and modest (Malhi and Berk, 2012).
Acute
Upon initiating lithium, side effects such as tremor, general fatigue, diarrhoea, thirst, polyuria, nausea, headache and vomiting are common but the effects are usually transient (Table 1) (Fyro et al., 1970; Lydiard and Gelenberg, 1982) and usually last no more than a day or so, but in some patients troublesome side effects can persist for days or weeks (Grandjean and Aubry, 2009b). The majority of these side effects are associated with rapid increases of plasma lithium concentration and therefore can also occur when the dose of lithium is raised (Fyro et al., 1970; Karniol et al., 1978; Yatham et al., 2005).
Transient and acute side effects of lithium therapy.

Chronic
In the management of BD, lithium is usually prescribed long term and therefore chronic side effects may emerge following years and decades of treatment.
Renal
Long-term treatment with lithium can impair the renal concentrating ability and cause polyuria and secondary thirst (Gitlin, 1999; Grandjean and Aubry, 2009b; Schou and Vestergaard, 1988). In most cases this effect is modest and renal function is readily restored upon cessation of lithium but in a minority of patients chronic lithium use induces renal insufficiency. The precise mechanism by which this occurs is not known but it warrants careful monitoring as it can potentially lead to end-stage renal failure (Bendz, 1983; Gitlin, 1999; Markowitz et al., 2000). There is a relatively small risk of renal toxicity with long-term lithium use. Specifically, end-stage renal failure occurs in 0.53% of lithium-treated individuals compared to 0.2% in the general population (Coresh et al., 2003) but it is important to note that in the vast majority of bipolar patients the trajectory of decrease in glomerular function with long-term lithium treatment is similar to that associated with normal ageing (Gitlin, 1999; Silverstone, 2000). Further, the risks for renal disease such as diabetes are generally more common in bipolar cohorts, and changes may not emerge solely because of lithium use. (See Table 2 for renal monitoring recommendations.)
Lithium therapy monitoring recommendations.
Endocrine
Lithium affects both thyroid and parathyroid gland functioning. In the thyroid gland its accumulation alters the stimulation of thyroglobulin and inhibits iodine uptake, iodotyrosine coupling and thyroxine secretion. Reduction of the latter leads to an increase in TSH and the typical clinical picture of hypothyroidism emerges (Lombardi et al., 1993). Clinically significant hypothyroidism is seen in 8–19% of patients on lithium, as compared to 0.5–1% in the general population (Grandjean and Aubry, 2009b; Johnston and Eagles, 1999), especially in women and the elderly and more so within the first 2 years of commencing treatment (Vincent et al., 1993). However, even independent of treatment, thyroid abnormalities are more common in bipolar disorder (Chakrabarti, 2011). The detection and management of hypothyroidism is important because it increases the likelihood of developing depression and rapid cycling (Silverstone, 2000; Vincent et al., 1993; Yatham et al., 2005) and therefore patients with hypothyroidism on lithium therapy should be prescribed thyroxine (Freeman and Freeman, 2006; Yatham et al., 2005).
Patients on lithium are prone to develop hypercalcaemia secondary to elevated parathyroid hormone (PTH) levels. This occurs because lithium decreases PTH calcitonin binding (Kusalic and Engelsmann, 1999) which results in a compensatory decrease in urinary calcium excretion and is associated with a decrease in bone resorption (Grandjean and Aubry, 2009b; Mak et al., 1998).
Both thyroid and parathyroid abnormalities are relatively common with long-term lithium therapy and therefore both thyroid function and blood calcium levels warrant routine assessment (see Table 2).
Obesity
Long-term treatment with lithium usually results in noticeable weight gain. An increase of 1–2 kg over the first 12 weeks of treatment is common and in some individuals this increase may continue. Overall, 5% or more of patients on lithium (Keck et al., 2003; Yatham et al., 2005) report noticeable weight gain but this is modest in comparison to other psychotropic medications (Allison et al., 1999; Keck et al., 2003).
BD predisposes to obesity because of hypersomnia, hyperphagia and decreased physical activity, especially when patients are depressed. Treatment with lithium adds to this by virtue of its direct effects on appetite, thyroid function and cellular glucose uptake, all of which encourage weight gain (Keck et al., 2003). Compounding this further, patients on lithium often experience polydipsia and consume drinks with high sugar and calorie content (Grandjean and Aubry, 2009b; Livingstone and Rampes, 2006). Therefore, when commencing lithium it is important to anticipate weight gain (see Table 2) and to put in place preventative lifestyle changes that include diet and exercise.
Cardiac
Patients on long-term lithium therapy may manifest electrocardiogram (ECG) changes such as QTc interval prolongation, increased PR interval duration, diffuse T-wave inversion and sinus bradycardia (Hsu et al., 2005; Roose et al., 1979; Rosenqvist et al., 1993; Steckler, 1994; Timmer and Sands, 1999). Most of these changes are reversible and disappear when lithium is withdrawn, but occasionally cardiac abnormalities can persist, and rarely may precipitate clinically significant arrhythmias (Mamiya et al., 2005; Rosenqvist et al., 1993).
Neurological
The neurotoxic effects of lithium are related to treatment duration (Oakley et al., 2001). A reduction in peripheral nerve conduction velocities (Chang et al., 1990) results in neurological symptoms and the central effects can be observed on electroencephalography (EEG) (Fountoulakis et al., 2008b). Clinically, the effects of lithium manifest as tremor and changes in cognition (Grandjean and Aubry, 2009b; Kropf and Muller-Oerlinghausen, 1979; Squire et al., 1980; Wingo et al., 2009).
Tremor
Tremor is a relatively common and troubling side effect of lithium therapy that can sometimes persist long term (Lydiard and Gelenberg, 1982). It occurs at therapeutic levels and can therefore be treated by reducing the dose of lithium or administering beta-blockers (Carroll et al., 1987; Kato et al., 1996; Vestergaard et al., 1988).
Cognition
The effects of lithium on cognition are disputed with short-term studies in healthy subjects finding no discernible effects (Karniol et al., 1978; Kropf and Muller-Oerlinghausen, 1979) but long-term exposure seemingly producing changes in learning and memory (Grandjean and Aubry, 2009b; Kropf and Muller-Oerlinghausen, 1979; Squire et al., 1980; Wingo et al., 2009). Others have suggested that cognition is preserved in lithium responders (Rybakowski and Suwalska, 2010) and clearly this warrants further research.
Toxicity
Acute
In acute intoxication the plasma levels of lithium can reach potentially lethal levels (> 2.0 mmol/L), especially once lithium excretion reaches its upper limit, after which it accumulates rapidly and leads to precipitous worsening of symptoms (Grandjean and Aubry, 2009b). However, in some instances, even such high plasma levels may lead to relatively mild symptoms, and individuals recover without permanent neurological damage (Chen et al., 2004). This is because lithium can take up to 24 hours to cross the blood–brain barrier into the brain. In counterpoint, elderly individuals can manifest lithium toxicity at therapeutic levels (Shulman, 2010) and therefore it is essential to monitor plasma lithium levels closely and avoid toxicity but, if it occurs, prompt action may still prevent serious damage.
Chronic
If lithium is found to be effective and the individual is well suited to lithium therapy, it is usually administered for years and sometimes decades to maintain mood stability and prevent the recurrence of symptoms (see previous discussion on ‘Maintenance and prophylaxis’). However, with life-long treatment lithium can gradually accumulate within the brain and lead to chronic toxicity. This occurs despite plasma concentrations remaining within the therapeutic range because lithium has a longer half-life in brain tissue than in plasma (Chen et al., 2004). Toxicity because of chronic lithium use is also subject to other factors including change in lithium dose, individual tolerance and impaired renal function, especially in aging individuals (Chen et al., 2004; Grandjean and Aubry, 2009b; Oakley et al., 2001). Therefore, it is important to routinely consider the possibility of chronic toxicity in patients being prescribed lithium long term.
Treatment
In a recent study (Eyer et al., 2006) that examined patients with chronic and acute on chronic lithium toxicity, haemodialysis was found to be the most effective method for reducing lithium in the blood, whereas diuretics were partially effective at increasing renal clearance. One limitation of this study, however, was that patients were also taking other medications known to alter renal function (Eyer et al., 2006). Nevertheless, haemodialysis can be used to treat lithium toxicity and, in addition, thiazide diuretics, ACE inhibitors or non-steroidal anti-inflammatory medications may confer some prophylactic benefits and perhaps even prevent or limit the risk of renal dysfunction, or episodes of intoxication (Finley et al., 1995; Gitlin, 1999; Malhi and Tanious, 2011). In this regard, potassium (20–50 mmol/day) is also potentially useful in reducing the effects of lithium toxicity on the kidney (Batlle et al., 1985; Gitlin, 1999) and there is preclinical evidence (from animal studies) that N-acetyl cysteine may prevent lithium-induced renal harm (Efrati et al., 2005).
Clinical application
Initiating therapy
Given that lithium has a relatively narrow therapeutic index, it is important to maintain lithium levels within a specific range for each individual patient. This requires achieving a balance between effectiveness and adverse effects. Earlier guidelines (Figure 3) suggest that when initiating lithium therapy de novo it should be administered in small divided doses to minimise side effects, and then titrated gradually to achieve plasma levels of 0.5–1.2 mmol/L, while monitoring for tolerability (American Psychiatric Association (APA), 2002). Further, lithium naïve patients should aim to achieve plasma levels of 0.8–1.0 mmol/L, especially for prophylaxis against manic episodes (Hopkins and Gelenberg, 2000). The National Institute for Health and Clinical Excellence (NICE, 2006) proposes a narrower starting dose of 0.6–0.8 mmol/L, and more recent studies indicate that these suggested levels confer reasonable prophylaxis and are associated with an acceptable side effect profile (Severus et al., 2008).
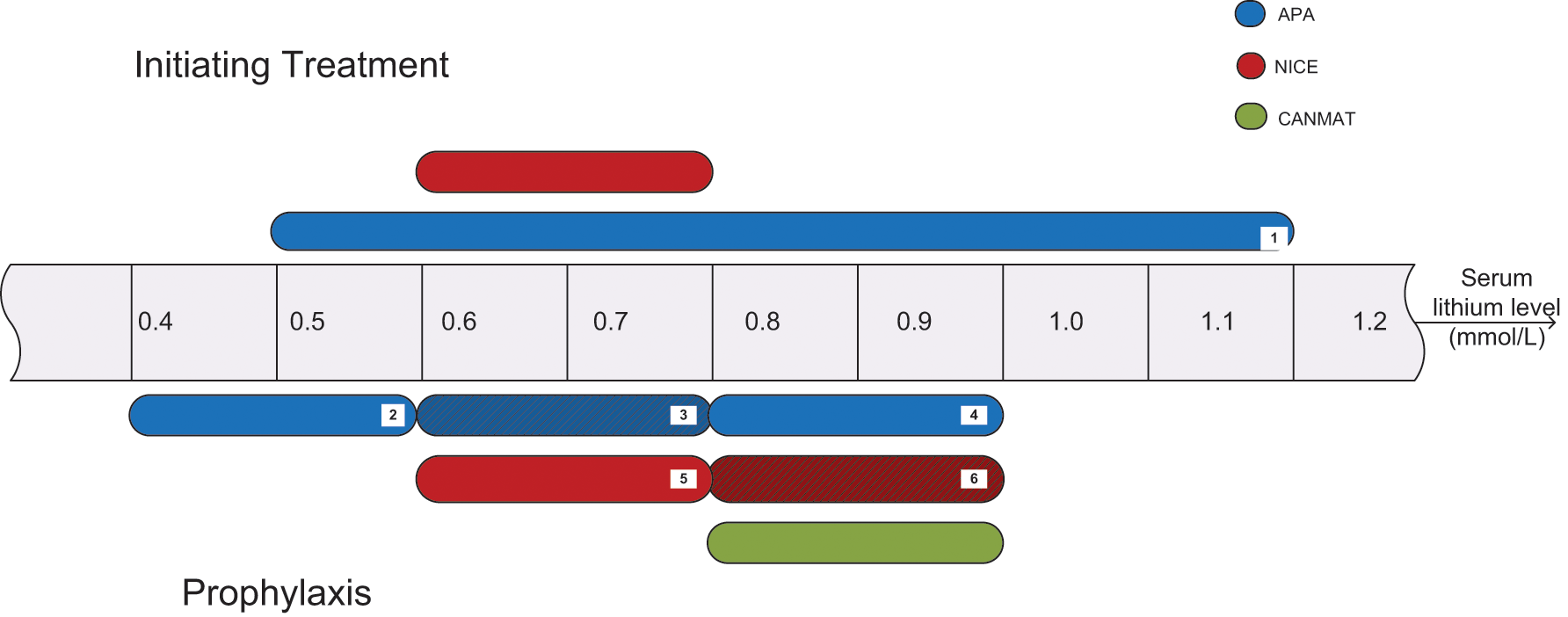
Recommended lithium levels for initiating treatment and prophylaxis (adapted from Malhi and Tanious, 2011). The figure shows the recommended plasma lithium concentrations for initiation of treatment and prophylaxis in patients with BD (APA, 2002; NICE, 2006; Yatham et al., 2005). [1] Recommendation is to start with low divided doses and increase to a plasma lithium concentration of 1.2 mmol/L depending on adverse effects; as concentrations reach this upper therapeutic range, the patient should be monitored and assessed for toxicity; [2] concentration range that may be sufficient for prophylaxis; [3] concentration range commonly chosen by psychiatrists but that has not been formally studied; [4] concentration range that is more efficacious for controlling symptoms, especially mania, although adverse effects are less well tolerated; [5] recommended concentration range for stabilizing patients; [6] recommended concentration range for patients previously taking lithium who have relapsed. (APA: American Psychiatric Association; CANMAT: Canadian Network for Mood and Anxiety Treatments; NICE: National Institute for Health and Clinical Excellence (UK).)
Therefore, clinically, after stabilizing the plasma levels of lithium to 0.6–0.8mmol/L, the oral dose can then be increased to achieve higher plasma levels if breakthrough symptoms occur (NICE, 2006). It is this higher range of lithium plasma levels that the Canadian Network for Mood and Anxiety Treatments (CANMAT) guideline advocates for maintenance of BD with lithium therapy (Yatham et al., 2006). The APA also suggests that higher plasma lithium levels (0.8–1.0 mmol/L) may be used for controlling symptoms but that relatively low levels (0.4–0.6 mmol/L) may provide sufficient prophylaxis long term (APA, 2002; Severus et al., 2008).
Maintenance therapy
Target lithium levels
The primary aim of maintenance therapy is to prevent the recurrence of symptoms whilst minimizing side effects and maintaining compliance. Interestingly, a review of different dosing schedules has demonstrated a dose–response relationship between lithium and the recurrence of symptoms wherein even low levels of lithium may be protective (Hopkins and Gelenberg, 2000). However, the evidence for ‘high’ versus ‘low’ lithium levels in prophylaxis is inconsistent. Predictably, studies report both high rates of relapse with ‘low’ plasma lithium levels (0.4–0.6 mmol/L), presumably because of lack of efficacy, and low rates of relapse with ‘high’ levels (0.8–1.1 mmol/L), but contrary to this some studies have found that high plasma lithium levels result in greater dropouts because of adverse effects (Silverstone, 2000). In either context, in clinical practice it is important to avoid the abrupt cessation of treatment with lithium, as this is likely to precipitate a relapse of the illness (Mander and Loudon, 1988).
In sum, plasma lithium concentrations should be optimised to the individual symptom and tolerability profile of patients. BD patients likely to develop depressive episodes may benefit from prophylactic levels of 0.4–0.8 mmol/L, whereas in those more likely to develop mania, levels of 0.6–1.0 mmol/L may be more effective (Kleindienst et al., 2005, 2007; Severus et al., 2009). Equally, an individual with tolerability issues at a higher level may derive more benefit from low-dose lithium therapy.
Administration
Most of the current recommendations for lithium administration suggest divided daily doses. One of the main arguments against a single-dose (SD) regime is that compared to a multiple-dose (MD) schedule, there are greater peaks in plasma lithium levels, which are not well tolerated because of associated side effects, and deeper troughs that make breakthrough symptoms more likely. However, since pharmacokinetic properties remain unchanged with either regime (Perry et al., 1981), in practice, no clinically significant differences have been found with respect to side-effect profile and recurrence of mania (Ljubicic et al., 2008). Further, plasma lithium levels remain within the therapeutic range with both SD and MD regimes (Gitlin, 1999; Hunter, 1988; Ljubicic et al., 2008), and because lithium exerts its therapeutic effects within the CNS there is another ‘buffer’ between plasma levels and therapeutic effect that needs to be considered when comparing dosing schedules. Specifically, lithium has a longer half-life in neural tissues and, therefore, lithium once-daily, or even every alternate day, may, in some cases, be sufficient to maintain an effective brain lithium concentration over a 48-hour period when used for prophylaxis, especially given the lag between plasma and brain level adjustment (Ljubicic et al., 2008; Plenge et al., 1994) (Figure 4).
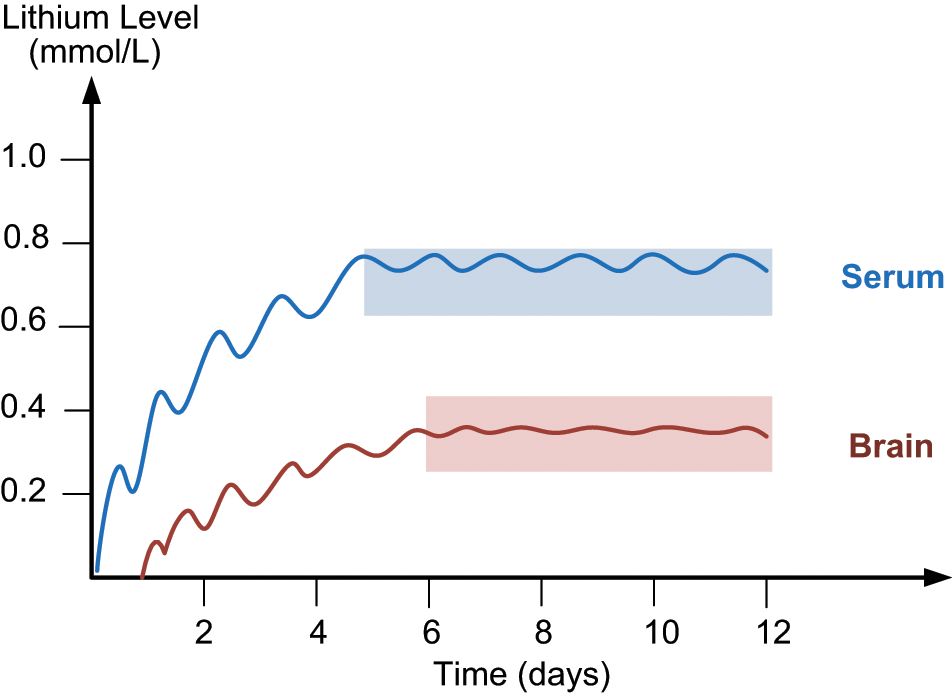
Plasma and brain lithium levels. At therapeutic plasma lithium levels (0.8 mmol/L) the level of lithium in the brain is approximately 0.4 mmol/L (the ratio of brain:plasma lithium levels is approx 0.5:1) (Soares et al., 2000). Of note: lithium levels in the brain peak approximately 24 hours after they peak in plasma (Chen et al., 2004).
An advantage of a daily schedule for lithium compared to a twice-daily schedule is that it causes less polyuria with long-term administration and hence decreases the chance of permanent renal damage (Bendz, 1983; Gitlin, 1999). In addition, since lithium clearance is lower overnight, a single evening dose can be reduced by 25% (Ljubicic et al., 2008). Such a reduction in the oral dose of lithium significantly decreases the likelihood of side effects (Coppen et al., 1983) and therefore, if in practice it is well tolerated, a once-daily trial should be considered (Gitlin, 1999; Ljubicic et al., 2008; Malhi and Tanious, 2011).
Alternate-day dosing may further reduce side effects and the risk of renal damage. Early studies that maintained lithium levels within the therapeutic range showed promise, but recent research (Jensen et al., 1990; Plenge et al., 1999) suggests that alternate-day dosing confers a threefold increase in the risk of relapse as compared to daily dosing, and that plasma levels drop below 0.5 mmol/L. Further, from a practical perspective it is much harder to adhere to irregular medication regimes.
In sum, a once-daily dosing schedule confers several advantages. First, a low, single dose of lithium at night reduces the likelihood of peak side effects, such as fatigue and sedation (Ljubicic et al., 2008). Second, reducing the oral dose diminishes the overall side-effect burden (Bowen et al., 1991) and reduces the likelihood of glomerular impairment long term. Finally, daily dosing is advantageous because it enhances adherence and this is important in achieving long-term efficacy.
Adherence
Long-term maintenance of plasma lithium levels is critical for effective prophylaxis of BD. Continuity of maintenance treatment is particularly important because cessation is a strong predictor of suicide risk (Young and Hammond, 2007) and is the main reason for recurrences
Reasons for poor adherence
The reasons for poor adherence are complex and varied. Side effects are a commonly cited reason that can be quite incapacitating. However, tolerability is not the major driver of adherence; indeed, stigma and a lack of acceptance and understanding play a significant role. Commonly reported side effects of lithium therapy that impair compliance include weight gain, polyuria, tremor and fatigue (Rosa et al., 2007). Weight gain is troublesome, in particular for females (Fakhoury et al., 2001; Kessing et al., 2007; Silverstone, 2000; Yatham et al., 2005) and, not surprisingly, individuals who report multiple side effects are also more likely to be less satisfied with their medication and management overall (Fakhoury et al., 2001). Figure 5 attempts to depict graphically the relationship between treatment adherence, risk of relapse and plasma lithium levels.
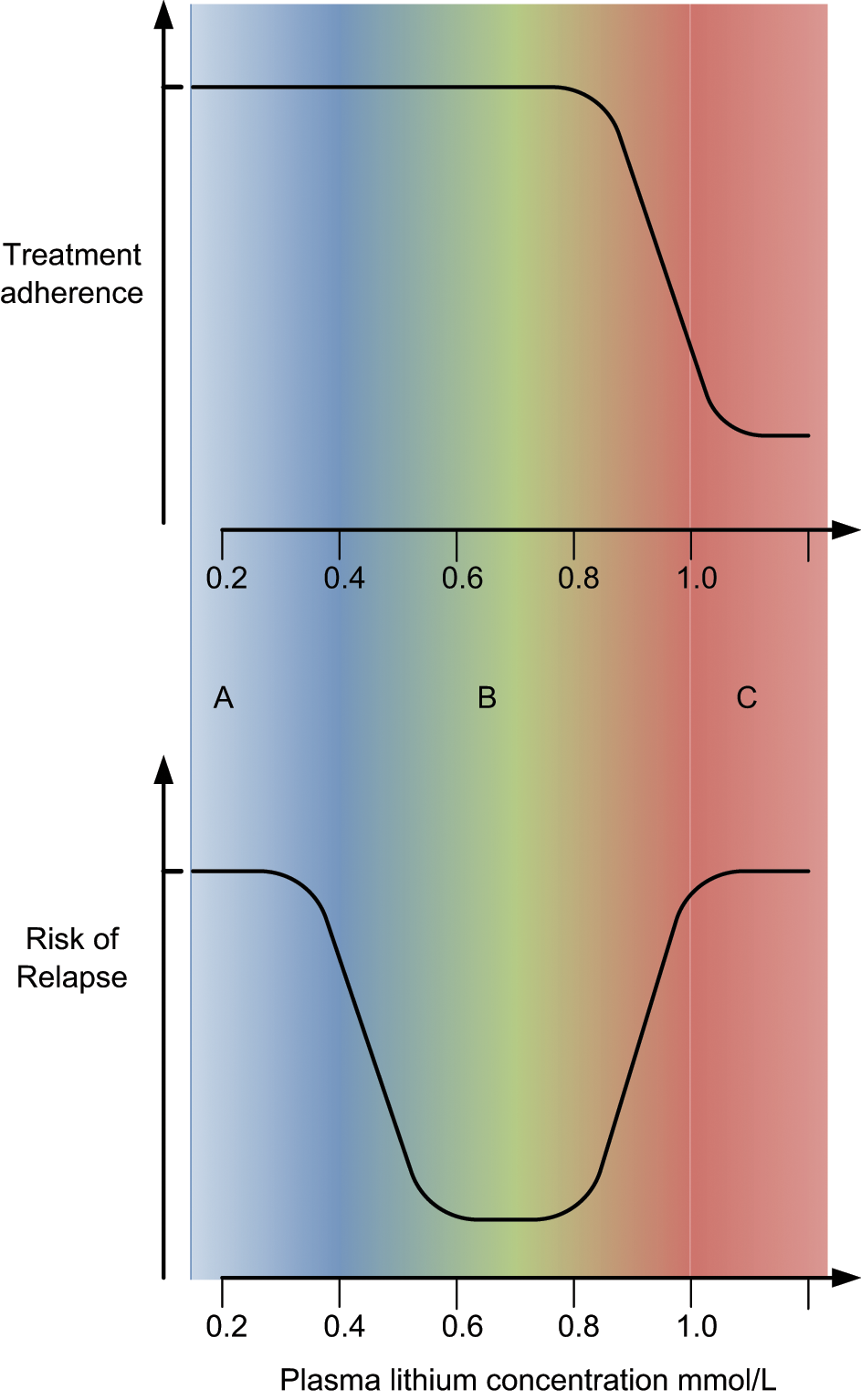
Treatment adherence and risk of relapse. A: (BLUE) Low plasma lithium levels produce better treatment adherence, but the risk of relapse is high because low lithium levels often provide insufficient prophylaxis; B: (GREEN) this range of lithium plasma levels is optimal as it achieves prophylaxis and the risk of relapse is low; C: (RED) high plasma lithium levels result in poor treatment adherence because the likelihood of troublesome side effects is greater. Clinically, intermittent adherence is the most common form of non-adherence and this means that lithium levels are often fluctuating and prophylaxis is suboptimal.
In addition to tolerability issues, individuals taking lithium also express frustration about needing to take medications when they are symptom-free. This is associated with denial of their illness and it contributes to a perceived lack of efficacy and loss of energy and productivity. Further, some patients report missing the ‘highs’ of hypomania (Grandjean and Aubry, 2009a; Malhi et al., 2010; Rosa et al., 2007; Scott and Pope, 2002). All of these issues are more evident in those who have been recently diagnosed with BD, younger individuals and those taking lithium long term. Interestingly, individuals who do not have a strong doctor–patient alliance and those who have received less education about the disorder and its treatment are also likely to adhere to medication (Kleindienst and Greil, 2004; Rosa et al., 2007).
Enhancing adherence
Enhancing adherence requires a multifaceted approach that involves education and monitoring in the context of a therapeutic alliance (Berk et al., 2004, 2010a). It has been shown that patients who are informed about their illness and the importance of long-term treatment, have better compliance (Colom et al., 2003; Even et al., 2007). Other strategies that enhance adherence to treatment include educating the family and friends of the patient to recognise the early signs of relapse and institute suitable measures to curtail stressors (Rosa et al., 2007; Berk et al., 2011a). Further, encouraging patients to make a commitment to treatment prior to commencement (Rosa et al., 2007; Silverstone, 2000) and coupling pharmacotherapy with psychotherapy has been shown to greatly improve patient outcomes (Colom et al., 2003; Even et al., 2007; Yatham et al., 2005).
The close monitoring of patients improves adherence in two ways. First, it allows tailoring of the therapeutic dose to suit the individual, so that therapeutic benefit is optimised and the likelihood of side effects is reduced (Silverstone, 2000). Second, regular monitoring increases clinical contact and therefore patients are likely to receive more frequent supervision and better education concerning their illness and its management (Rosa et al., 2007).
Individualising lithium therapy
Lithium is a suitable first-line treatment option in the management of BD that is particularly efficacious when applied selectively (Berk and Malhi, 2011). As mentioned already, those with a classic picture of BD are most likely to be responsive, whereas those with mixed presentations, rapid cycling and less easily defined manifestations are likely to respond poorly (Gershon et al., 2009).
Predictors of a poor response to lithium include comorbid substance misuse and a history of more than three episodes of a predominantly depressive presentation. A positive family history, administration early in the course of the illness and a beneficial prior response are predictors of a good response (Frye et al., 1998). Using such indicators, amongst others, the application of lithium can be individualized. Further, as has been discussed in detail, the plasma levels of lithium can be monitored and adjusted according to mood state and the needs of the individual (Malhi et al., 2011b). To assist with this, the lithiumeter can be used to determine the optimal plasma levels required from initiation through to maintenance, taking into consideration the predominant mood state (Figure 6).
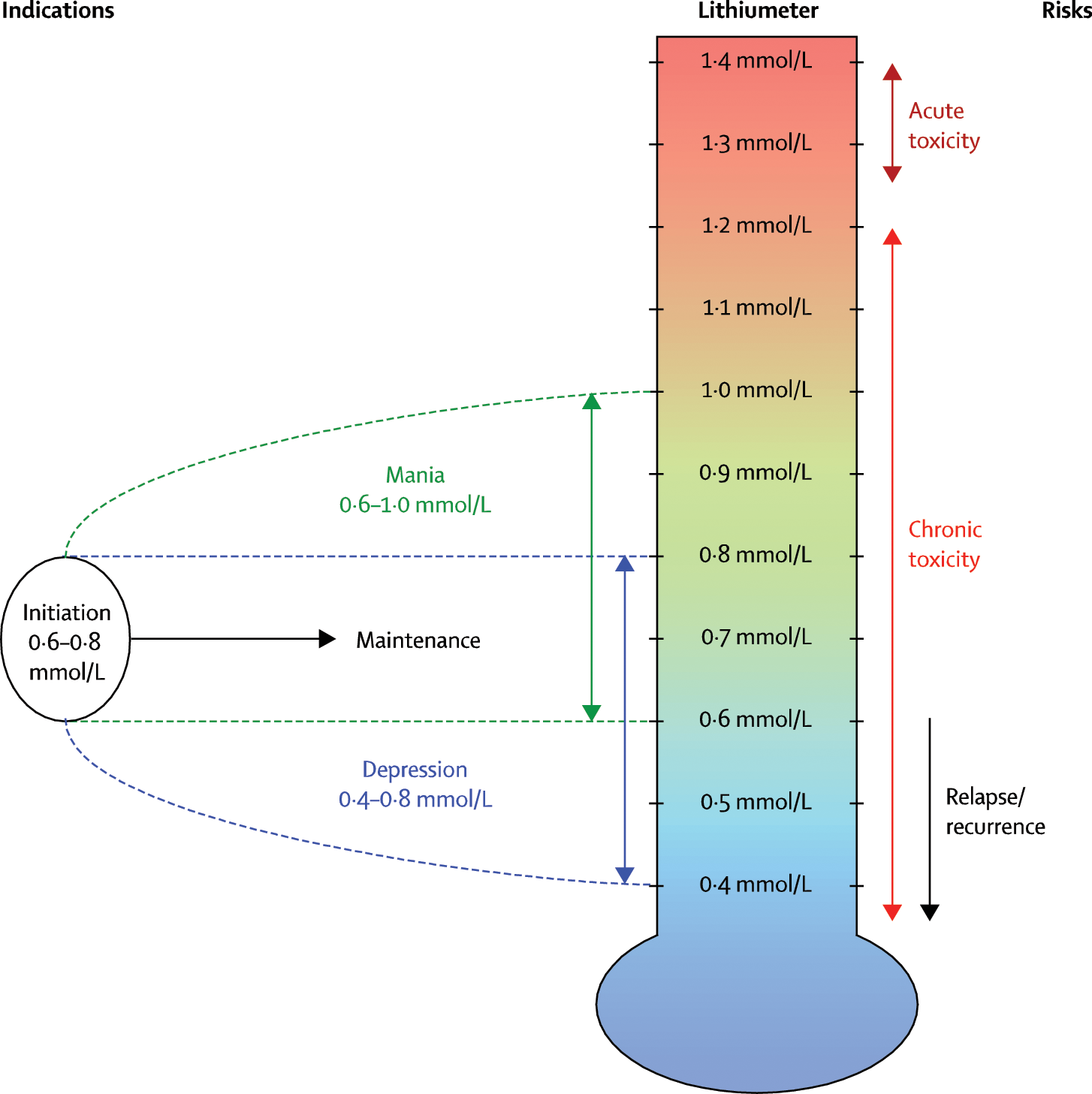
The lithiumeter. The lithiumeter depicts indications and risks associated with lithium according to its blood plasma levels. Optimal levels for treatment initiation is between 0.6 and 0.8 mmol/L, and should be individually tailored on the basis of recurrences and polarity of symptoms. Indications: This depicts suitable plasma levels for initiation and maintenance therapy in relation to the phases of illness, which are shown on the left. Prophylaxis for depression (shown in blue) might be achieved satisfactorily at lower levels than for mania (shown in green). Risks: The risks associated with lithium therapy are illustrated on the right in red. Acute toxicity is likely at levels above 1.2 mmol/L but, over time, even much lower therapeutic levels of lithium can lead to chronic toxicity, especially in elderly individuals. However, relapse/recurrence is more likely at lower plasma lithium levels. Reproduced with permission from Malhi and Berk (2012).
Conclusion
Recent evidence (Geddes et al., 2010; McKnight et al., 2012) has once again positioned lithium centre stage in the management of BD. Used judiciously, the side effects of this magical element can be minimised and significant long-term efficacy can be achieved. Within the growing BD armamentarium, lithium is one of the few agents that not only offers effective management of the illness, but is also potentially anti-suicidal and neuroprotective. To gain full benefit, however, lithium has to be prescribed to specific patients at the correct dosage and in the appropriate manner. Thus, the practice of lithium therapy must be borne out of a sound understanding of its clinical science.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Declaration of interest
GSM has received research support from AstraZeneca, Eli Lilly, Organon, Pfizer, Servier and Wyeth; has been a speaker for AstraZeneca, Eli Lilly, Janssen-Cilag, Lundbeck, Pfizer, Ranbaxy, Servier, and Wyeth; and has been a consultant for AstraZeneca, Eli Lilly, Janssen-Cilag, Lundbeck, and, Servier. MB has received research support from the Medical Benefits Fund of Australia, Bristol-Myers Squibb, Eli Lilly, GlaxoSmithKline, Organon, Novartis, Mayne Pharma, and Servier; has been a speaker for AstraZeneca, Bristol-Myers Squibb, Eli Lilly, GlaxoSmithKline, Janssen-Cilag, Lundbeck, Merck, Pfizer, Sanofi-Synthelabo, Servier, Solvay, and Wyeth; and has served as a consultant to AstraZeneca, Bristol-Myers Squibb, Eli Lilly, GlaxoSmithKline, Janssen-Cilag, Lundbeck, and Servier.
