Abstract
Keywords
Introduction
Major depressive disorder (MDD) is a leading cause of morbidity affecting 120 million people worldwide (World Health Organisation, 2005). The heritability rate of depression has been estimated as high as 70% (Kendler et al., 1993), with current estimates proposing rates of between 30% and 40% (Sullivan et al., 2000). The overall relative risk for a first-degree relative is two to three (Sullivan et al., 2000), suggesting that genetics may play a significant role in the aetiology of the disorder. The most widely studied gene associated with depression is SLC6A4, which encodes the serotonin transporter (5-HTT) (Levinson, 2009). The S allele leads to lower production of 5-HTT, which decreases the reuptake capacity for serotonin (Lesch et al., 1996). Research on the epidemiology of depression indicates a role for a gene × environment interaction involving the S allele in combination with stressful life events (Caspi et al., 2003; Cervilla et al., 2007; Jacobs et al., 2006; Kendler et al., 2005; Kim et al., 2007; Wilhelm et al., 2006) and the presence of this S allele has also been associated with poor response to anti-depressant medication (see Kemp et al., 2008 for review). Other research implicates a role for the BDNF Val66Met polymorphism in depression (Hwang et al., 2006; Jiang et al. 2005). BDNF, a neuroprotective protein, acts in order to promote neurogenesis: the growth and differentiation of new neurons and synapses (Acheson et al., 1995; Huang and Reichardt, 2001). The BDNF Met allele is associated with inefficient BDNF secretion (Egan et al., 2003) and BDNF is decreased in patients with depression (Castren, 2004, 2005; Chen et al., 2001; Duman and Monteggia, 2006; Karege et al., 2005; Shimizu, 2003). In addition, Met heterozygotes have been reported to be more likely to ruminate, a core feature of depression, than individuals homozygous for the Val allele (Beevers et al., 2009). Researchers have further suggested that alterations in serotonergic activity may reflect underlying plasticity changes subserved by BDNF (Munafo et al., 2005; Urani et al., 2005). Repeated exposure to early life stress (ELS) leads to neuronal loss in regions central for emotion and memory function such as the limbic system, hippocampus and amygdala (Heim and Nemeroff, 1999). These locations are implicated in depression, and BDNF has been shown to directly impact plasticity and growth in these regions (Black, 1999; Conner et al., 1997; Rattiner et al., 2004). However, not all studies have been able to replicate the association between these genetic polymorphisms and depression (Chen et al., 2008; Risch et al., 2009). While it is possible that these contradictory reports relate to underlying genetic heterogeneity, studies remain to be conducted that investigate this possibility. The current study aims to investigate the contribution of 5-HTT and BDNF genetic polymorphisms to subtypes of MDD.
Melancholic depression is a unique subtype of depression characterised by increased hyperarousal and hypercortisolaemia (Gold and Chrousos, 2002) that can lead to neuronal atrophy in the hippocampus (Belmaker and Agam, 2008). This effect can be resolved by antidepressant treatment (Brunoni et al., 2008; Santarelli et al., 2003). Antidepressants that modulate both serotonin and norepinephrine are more efficacious in patients with melancholia (Malhi et al., 2005a), while non-melancholic patients respond well to antidepressants modulating only serotonin (National Institute for Clinical Excellence, 2004). These findings have been reflected in structural and functional models of depression (Malhi et al., 2005b), which support an association between subtypes of depression and neurotransmitter systems. Interestingly, the BDNF Met/Met genotype has been associated with impairments in emotion and cognition similar to those observed in depressed patients (Hasler et al., 2004; Mayberg, 1997) and those with melancholia in particular, including deficits in verbal memory (Dempster et al., 2005; Egan et al., 2003), visual memory (Hariri et al., 2003), working memory and executive functioning (Gatt et al., 2009; Rybakowski et al., 2003). We have previously demonstrated that an interaction between the BDNF Met allele and exposure to ELS predicts neural circuitry and autonomic arousal pathways to depression and anxiety (Gatt et al., 2009), and anxiety comorbidity is a common feature of melancholia (Gold and Chrousos, 2002).
The current study examined the contribution of 5-HTT and BDNF genetic polymorphisms to melancholic and non-melancholic depression. On the basis of prior research, we hypothesise that patients with non-melancholia will be characterised by a combination of the 5-HTT S allele and an increased number of ELS events, while melancholia will be characterised by a combination of the 5-HTT S allele, BDNF Met allele and an increased number of ELS events. We further hypothesise that gene by environment interaction effects will predict depression subtype over and above main effects.
Methods
Participants
A total of 128 participants with a primary diagnosis of MDD and 128 healthy control participants were recruited from the community in collaboration with the Brain Resource International Database (Gordon et al., 2005). Exclusion criteria included a history of brain injury (causing loss of consciousness for 10 minutes or more), neurological disorder, psychiatric illness except for depression or a secondary diagnosis of anxiety, current substance dependence disorder or other serious medical condition. Depressed participants with co-morbid anxiety were included to maximise the generalisability of the sample. Secondary diagnoses included generalised anxiety disorder (n = 47), panic disorder (n = 29), and post-traumatic stress disorder (n = 14). All participants were medication free for at least five half-lives. All depressed participants met criteria for MDD as determined by research officers trained and supervised by collaborating psychiatrists using the Mini International Neuropsychological Interview (MINI: Sheehan et al., 1998), a structured psychiatric interview based on the Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition (DSM-IV: American Psychiatric Association, 2000). Severity of depression was measured using the Hamilton Rating Scale for Depression (HRSD: Hamilton, 1960); melancholic features, using CORE Assessment of Psychomotor Change (CORE: Parker et al., 1995; Parker and Hadzi-Pavlovic, 1996); ELS, by the self-report Early Life Stress Questionnaire (ELSQ: Cohen et al., 2006); and self-reports of depression, anxiety and stress, using the Depression, Anxiety and Stress Scales (DASS: Lovibond and Lovibond, 1995). All participants gave written informed consent in accordance with Australian National Health and Medical Research Council (NHMRC) ethical guidelines.
Procedure
After providing consent, trained research officers administered the MINI and HRSD, and completed the CORE. Patients were diagnosed as melancholic (MEL; n = 65) or non-melancholic (NMEL; n = 59) on the basis of the MINI. Participants then completed the DASS and ELSQ at the completion of the clinical interview.
Genotyping
DNA was extracted from cheek swab samples. The 5-HTTPLR genotype was determined by size separation of polymerase chain reaction (PCR) products using a modification of a previously established method (Gatt et al., 2007, 2008; Wilhelm et al., 2006). PCR amplification of participant DNA was undertaken using primers 5’-TGCCGCTCTGAATGCCAGCAC-3’ and 5’-GCGG GATTCTGGTGCCACCTA-3’ in a reaction containing 400 mM betaine, 0.16 mM each of dATP, dCTP and dTTP, and 0.08 mM each of dGTP and 7-deaza-2’deoxyguanosine 5’-triphosphate. PCR amplification conditions were as follows: 94°C for 2 min; 35 cycles of 94°C for 30 s, 65°C for 45 s and 72°C for 90 s; 72°C for 10 min. PCR amplicons were separated by electophoresis on 2% agarose gels. BDNF Val66Met genotype was determined using PCR amplification and restriction digest. PCR amplification of participant DNA was undertaken using primers 5’-TGTATTCCTCCAGCAGAAAGAGAA-3’ and 5’-AA AGAAGCAAACATCCGAGGAC-3’ and standard conditions. Amplified fragments were digested with the restriction enzyme Afl III and digests were separated on 4% agarose gels. Genotypes were scored independently by two researchers.
Statistical analyses
All statistical analyses were performed using SPSS (Version 17; SPSS Inc., Chicago, IL, USA). Initially, group characteristics were examined using one-way analyses of variance (i.e. 3 Group (MEL, NMEL, CTRL) × 2 Genotype (LL, SL or SS; VV, VM or MM) and follow-up post-hoc tests using Tukey’s HSD on total ELS events and chi square analyses on genotype. Next we conducted a mediation analysis ((G × G) × E mediation model) using the Preacher and Hayes method (Preacher and Hayes, 2008) to confirm a mediating role of ELS in the relationship between 5-HTTPLR × BDNF polymorphism combinations and subtype (MEL, NMEL, CTRL), in which age and gender were entered as covariates. The Preacher and Hayes (2008) SPSS Macro was configured for 5000 bootstrap samples and 95% confidence. Finally, we examined whether ELS, genotype and their interaction were able to predict depression subtype using a series of binary logistic regressions using the enter method such that all the variables were entered at the same time. Logistic regression was chosen above multinomial regression as values related to diagnosis are ordered and independent categories. Thus, using a binary model we were able to test each category independently. This method was chosen to test our primary hypothesis: an interaction between the predictors (e.g. y = b0 + b1(A) …)(e.g. for A and B in y = b0 + b1(A) + b2(B) + b3(A*B) + error) over and above the main effects (i.e. A and B). All main and interaction effects were therefore tested in the model. The regressions were completed with the following reference categories for dependent variables: 1. NMEL = 0, MEL = 1; 2. CTRL = 0; MEL = 1; and 3. CTRL = 0; NMEL = 1. Analyses focused on whether interaction effects were significant over and above main effects using the following regression equation: ln(odds (dependent variable)) = b0 + b1(BDNF)1 + b2(5-HTT)2 + b3(ELS)3 + b4(BDNF × ELS)4 + b5(5-HTT × ELS)5 + b6(BDNF × 5-HTT × ELS)6 + b7(age)7 + b8(gender)8. The BDNF and 5-HTT alleles were coded as a binary variable (BDNF Val66Met: V66M:VV versus (VM or MM); 5-HTTPLR:LL versus (SL or SS)) to examine the effects of the BDNF M allele and 5-HTT S allele as vulnerability factors. In order to confirm our hypothesis that variance could not be better explained by a diagnosis of MDD regardless of subtype, regression models were also tested contrasting CTRL and MDD (NMEL + MEL) (reference categories of 0 and 1, respectively). Significant effects were set at p < 0.05, while findings were labelled as trends if p < 0.1 and p > 0.05.
Results
Clinical data
Significant differences were observed between groups (MEL, NMEL, CTRL) on the HRSD (F(2) = 844.671, p < 0.001), DASS-D (F(2) = 4633.332, p < 0.001), DASS-A (F(2) = 103.463, p < 0.001), DASS-S (F(2) = 255.588, p < 0.001) and the CORE total score (F(2) = 77.247, p < 0.001), such that patients with melancholia displayed increased scores on all measures than those with non-melancholia and controls, respectively (see Table 1 for means and standard deviations of clinical data). Increased levels of depression severity, psychomotor disturbance and anxious arousal are consistent with current conceptualisations of melancholia (Gold and Chrousos, 2002; Hickie, 1996). There were no significant differences for diagnostic group (MEL, NMEL, CTRL) on gender (χ2(2) = 1.345, p = 0.510) or age (F(2) = 1.401, p = 0.248) (see Table 1 for age and gender demographics).
Mean and standard deviations (M ± SD) of clinical data by group, including age, gender, and early life stress demographics.
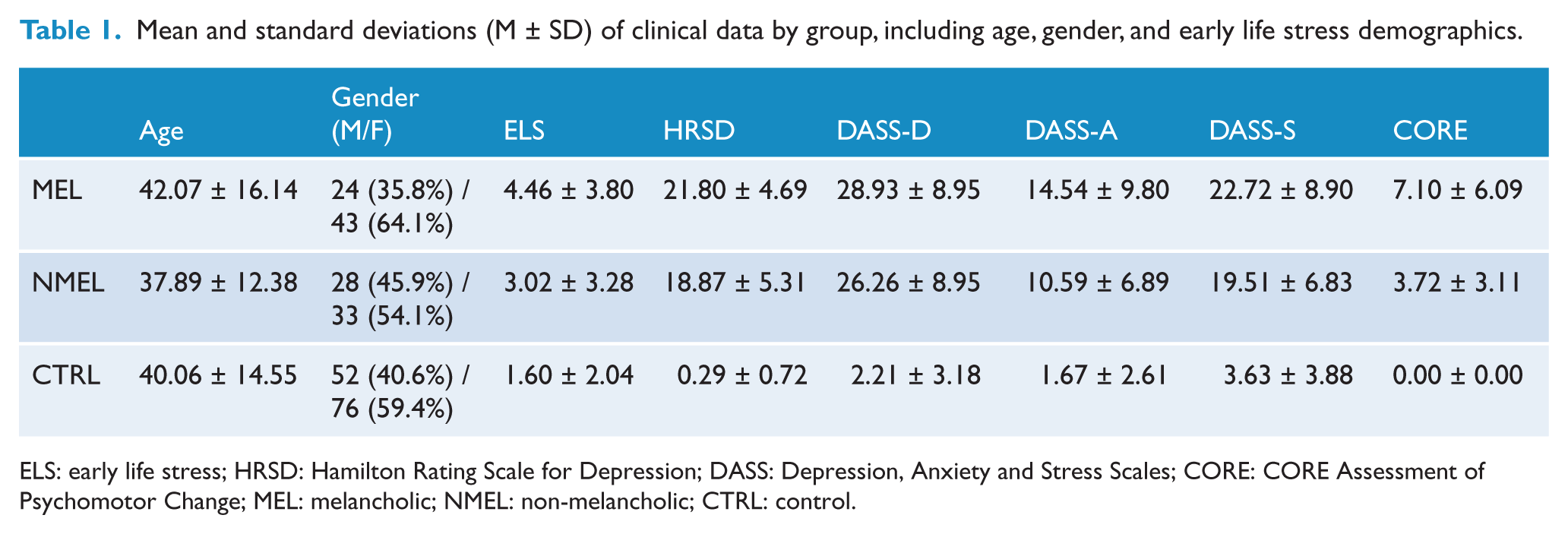
ELS: early life stress; HRSD: Hamilton Rating Scale for Depression; DASS: Depression, Anxiety and Stress Scales; CORE: CORE Assessment of Psychomotor Change; MEL: melancholic; NMEL: non-melancholic; CTRL: control.
Group characteristics: early life stress
There were significant differences across diagnostic groups (MEL, NMEL, CTRL) for the total number of ELS events (F(2) = 22.103, p < 0.001). The melancholic group had a greater number of ELS events (mean = 4.463, SD = 3.795), followed by the non-melancholic (mean = 3.016, SD = 3.279) and control (mean = 1.594, SD = 2.037) groups. The number of ELS events differed significantly between all groups (melancholic versus non-melancholic patients: MD = 1.446, SD = 0.513, Tukey’s HSD, p < 0.05; melancholic patients versus controls: MD = 2.869, SD = 0.437, p < 0.001; and non-melancholic patients versus controls: MD = 1.423, SD = 0.451, p < 0.01). The most common forms of ELS for the melancholic group were family conflict (47.8%), emotional abuse (46.3%), bullying (43.3%) and divorce (37.8%); for the non-melancholic group: domestic violence (32.8%), family conflict (32.8%), divorce (32.8%) and bullying (27.9%); and for controls: family conflict (14.8%), divorce (14.8%), emotional abuse (13.3%) and bullying (12.5%).
Group characteristics: genotype
The genotype frequencies were for BDNF: 64.6% VV (n = 155), 31.3% VM (n = 75) and 4.2% MM (n = 10) and for 5-HTT: 30.8% LL (n = 74), 50.8% SL (n = 122) and 18.3% SS (n = 44). Both were in Hardy-Weinberg equilibrium (BDNF: χ2(1) = 0.06, p = 0.190; 5-HTT: χ2(1) = 0.26, p = 0.390). No significant relationships were revealed between genotype (BDNF: χ2(2) = 3.057, p = 0.217; 5-HTT: χ2(2) = 0.828, p = 0.661) and group (MEL, NMEL, CTRL). Genotype frequencies are listed in Table 2.
Genotype frequencies and number of vulnerability alleles (i.e. BDNF Met allele and/or 5-HTT S allele) by group.

MEL: melancholic; NMEL: non-melancholic; CTRL: control.
Relationship between genotype and subtype: mediation by early life stress
With 239 valid cases and age and gender entered as covariates, the Preacher and Hayes (2008) bootstrap results for indirect effects demonstrated a significant ab path (genotype–environment), 95% bias corrected and accelerated confidence interval (0.010, 0.075). The summary for the subtype reference categories model was significant, F(4, 234) = 4.22, p = 0.003. The a path (β = −0.55, t = −2.717, p = 0.007) and b path (β = −0.070, t = −4.000, p < 0.001) were significant. The c path (genotype–phenotype) was not significant (p = 0.667) but, as signified by the bootstrap results, c’ path was significantly less robust than the c path (p = 0.806). There were no significant effects of the covariates age (β = 0.001, t = 0.346, p = 0.730) and gender (β = −0.065, t = −0.605, p = 0.556) on the model. Therefore, the relationship between 5-HTTPLR × BDNF polymorphism combinations and subtype reference categories are mediated by an indirect effect of ELS (see Figure 1).
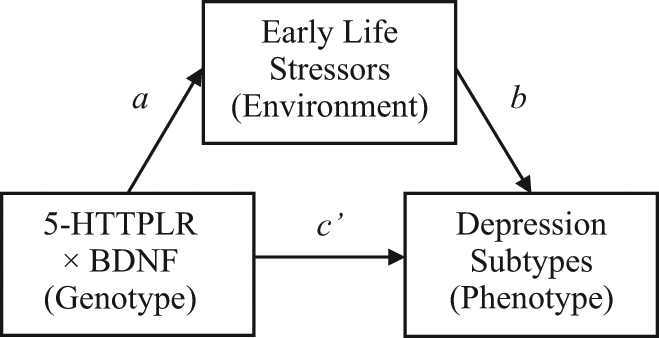
A mediation model of the indirect effect of early life stressors on the relationship between a 5-HTTPLR × BDNF and depression subtypes.
The contribution of genotype and early life stress to depression heterogeneity
For the main and interaction effects, models BDNF × ELS, 5-HTT × ELS and BDNF × 5-HTT × ELS significantly contributed to non-melancholia relative to controls only (see Table 3). Neither main nor interaction effects were found in the combined model for melancholia. The odds ratio revealed that an interaction between BDNF and ELS increased risk of non-melancholia by 3.327, whereas for the interaction between 5-HTT and ELS it was by 2.406. The interaction between BDNF, 5-HTT and ELS, whilst significant, only resulted in an odds ratio of 0.523. No variable was observed to contribute to the model comparing MDD patients (melancholic and non-melancholic depression combined) with controls.
Logistic regression values for non-melancholia versus controls.
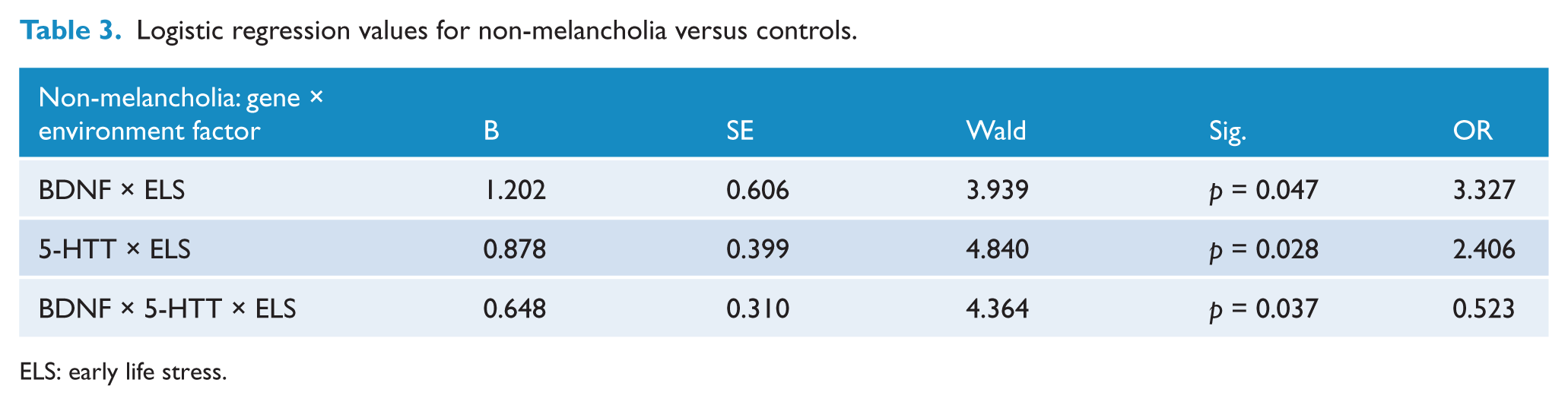
ELS: early life stress.
Discussion
The current study reveals that the BDNF and 5-HTT genotypes contribute to increased risk for non-melancholia, but not melancholia, when an individual is exposed to ELS. Contrary to our expectations, depressed patients with BDNF Met and 5-HTT S alleles are more likely to meet criteria for non-melancholic depression rather than melancholia. These preliminary findings indicate that different combinations of genotype may affect the phenotype of depression. The 5-HTT S allele may contribute to symptoms of depression (i.e. depressed mood, sleep irregularities and appetite alterations) congruent with both subtypes (Malhi et al., 2005b; Nutt, 2008), while the Met allele may contribute to the additional symptoms observed in more severe cases of depression that may require pharmacological treatment (i.e. working memory and executive cognitive dysfunction) (Parker et al., 2001; Stewart et al., 1989) in the context of ELS. Interestingly, prior research has indicated that personality dysfunction may contribute to non-melancholia compared to melancholia (Rubino et al., 2009), and such dysfunction may relate to 5-HTT and BDNF polymorphisms. The 5-HTT S allele may be a genetic substrate for the latent personality trait, neuroticism (or negative affectivity), and psychopathology in individuals with this vulnerability allele may develop under conditions of stress (Caspi et al., 2010). Furthermore, we have previously reported (Gatt et al., 2009) that an interaction between the BDNF Met allele and ELS may lead to higher levels of neuroticism and higher depression through bodily arousal. It is important to note that we are not concluding that there is no genetic basis to the melancholic subtype. Instead, we suggest that melancholia may involve other mechanisms not investigated in this study. Alternatively, it is possible that the lack of findings for patients with melancholia reflects a Type II error due to small sample size. Regardless, an important conclusion that can be drawn from the findings reported here is that non-melancholia may well have an underlying biological basis, consistent with structural and functional models of depression (Malhi et al., 2005b).
While the Met and S alleles provide a vulnerability to the non-melancholic subtype, their link to reduced function only predicts non-melancholia in combination with ELS, consistent with increasing recognition of the interaction between genetic and environmental factors (Kaufman et al., 2006). We observed that while ELS contributes to both melancholic and non-melancholic depression, patients with the melancholic subtype experience a greater number of ELS events (i.e. overall melancholic patients have more ELS). This further suggests that there are mechanisms other than BDNF and 5-HTT vulnerabilities alongside ELS that contribute to melancholic depression. Indeed, antidepressants that modulate both serotonin and norepinephrine are more efficacious in patients with melancholia (Malhi et al., 2005a), while non-melancholic patients respond well to antidepressants modulating only serotonin (National Institute for Clinical Excellence, 2004). Anhedonia symptoms have also been linked to deficits in the dopamine neurotransmitter system, which may be specific to melancholia (Malhi et al., 2005a; Stein, 2008).
The presence of the BDNF Met allele may also contribute to non-melancholia considering its influence on both serotonin and norepinephrine production (Santarelli et al., 2003). The Met allele of the BDNF polymorphism has been linked to lower levels of activity-dependent secretion (Egan et al., 2003) along with deficiencies in neuropsychological performance tasks (Dempster et al., 2005; Egan et al., 2003; Gatt et al., 2007; Hariri et al., 2003; Hasler et al., 2004; Mayberg, 1997; Rybakowski et al., 2003). These functions, such as working memory and executive cognitive functioning contribute to resilience and effective coping mechanisms. Thus, when a Met carrier is exposed to ELS (i.e. environmental factors) they are potentially less able to utilise problem-solving strategies to manage their distress leading to a non-melancholic depression. This is also consistent with findings that repeated exposure to ELS leads to neuronal loss in regions consistently implicated in depression (Heim and Nemeroff, 1999); areas which have been shown to be directly affected by BDNF (Black, 1999; Conner et al., 1997; Rattiner et al., 2004). BDNF is thus required as a protective factor against neuronal atrophy. Hence, a reduction of BDNF in non-melancholic depression may lead to neurocognitive deficits that in turn lead to reduced coping abilities in the context of stress. As the production of serotonin has been linked to changes in BDNF (Munafo et al., 2005; Urani et al., 2005), this relationship may be specific to non-melancholia such that stress precipitates a disruption in neural circuitry in those individuals with 5-HTT and BDNF vulnerability. Indeed, Met alleles and S alleles have previously predicted an increased risk for depression in maltreated children when ELS is considered (Kaufman et al., 2006; see also Harkness and Monroe, 2006). Previous research has also demonstrated that higher levels of stress sensitivity are associated with lower influence of negative life events on future episodes of depression (Wichers et al., 2009). This may be due to sensitisation that leads to more autonomous onsets of depression later in life (i.e. the kindling hypothesis (Kendler et al., 2000; Post, 1992)). The kindling hypothesis proposes that the neurobiology of affective disorders changes as a function of exposure to stress and time (Kendler et al., 2000). Our results support this proposal for non-melancholic depression in particular, a disorder associated with generally less severe depression and younger age of onset; and that this effect may be driven by vulnerability in BDNF and 5-HTT polymorphisms, exacerbated by repeated exposure to ELS.
The key finding from the current study is that BDNF and 5-HTT polymorphisms contribute to the development of non-melancholic depression after exposure to ELS. It is possible that non-melancholic patients have difficulties utilising effective coping mechanisms which may contribute to further reductions in resilience after exposure to ELS. The combination of these genetic risk factors and environmental predisposition toward stress may contribute to depression with non-melancholia but not melancholia. However, the number of ELS events may play a greater role in the development of melancholia than in non-melancholia. We further suggest there may be alternative genetic mechanisms contributing to the development of the melancholic subtype that were not examined in this study. With further research, additional contributing factors such as neuropsychological function and psychophysiological substrates of depression subtypes could be examined.
The current study has a number of limitations. First, we restricted our focus to 5-HTT and BDNF genotypes, and the lack of results for patients with melancholia indicates that additional genetic mechanisms may be involved. Second, the preliminary nature of our findings should be noted and the limitations of a small sample size considered. However, we suggest that a lack of study power as a function of a small sample size is usually associated with false negatives (i.e. a Type II error) rather than false positives (i.e. a Type I error). Our findings therefore support a role for genetic factors in the development of non-melancholia. Future studies should further examine differential gene × environment interactions that may underlie melancholia versus non-melancholia, rather than patients diagnosed with MDD per se.
Footnotes
Acknowledgements
We also acknowledge the data and support provided by BRAINnet; ![]() , under the governance of the BRAINnet Foundation. BRAINnet is the scientific network that coordinates access to the Brain Resource International Database for independent scientific purposes.
, under the governance of the BRAINnet Foundation. BRAINnet is the scientific network that coordinates access to the Brain Resource International Database for independent scientific purposes.
Sponsors played no role in analysis and interpretation of data, writing of the manuscript or in the decision to submit the paper for publication.
The authors CRQ, TO and AHK had full access to all of the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis
Funding
CRQ and TO were supported by an Australian Postgraduate Award, and AHK by a National Health and Medical Research Council Career Development Award Fellowship (571101). This research was supported by a National Health and Medical Research Council (NHMRC) Project Grant (464863) and an Australian Research Council Discovery Grant (DP0987332).
Declaration of interest
AH has been awarded research funding from the NHMRC, Australian Rotary, Perpetual Trustees, Eli Lilly Australia, Janssen-Cilag Australia and the Schizophrenia Fellowship of NSW. AWFH has also received consultancy fees from Organon Australia, Eli Lilly and Lundbeck Australia. AWFH has received payments for educational sessions run for Astra Zeneca, Janssen-Cilag, Eli Lilly and Organon. AWFH has also run educational sessions for a number of medical education companies including Wellmark Australia, Reed Business Information and CME LLC.
Johnson and Johnson Pharmaceutical Research and Development, RED Europe also supported the project from which data in the current study were drawn.
