Abstract
Introduction
Repetitive transcranial magnetic stimulation (rTMS) of the left dorsolateral prefrontal cortex (DLPFC) has been used to investigate mood effects in healthy controls (Pascual-Leone and Catala, 1996), to treat major depressive disorder (Martin et al., 2003), and to explore cognitive effects such as the impact on working memory (Mottaghy, 2006). Accurate localisation of this region is therefore important for demonstrating relationships between the DLPFC and behaviour. Furthermore, accuracy as well as reliability are important for localisation in the treatment of depression (Rusjan et al., 2010), which requires multiple separate sessions of localisation and administration. Varied response rates to rTMS across studies for the treatment of depression (Martin et al., 2003) may be related to inconsistent and variable localisation of the site of stimulation.
The DLPFC may be defined anatomically as Brodmann’s (1909) areas 9 and 46 (Rajkowska and Goldman-Rakic, 1995) or on the middle frontal gyrus. Most researchers have targeted the DLPFC with rTMS by using as a reference point the site of stimulation that evokes the maximal response in a contralateral hand muscle such as the abductor pollicis brevis (APB) or the first dorsal interosseus (FDI). Of the 35 studies that we reviewed, 86% located the DLPFC with reference to the motor hand area, 11% used non-stereotaxic MRI guidance, and only 3% used stereotaxic localisation. Based on the presumed location of the central sulcus from the evoked motor response in the hand, the Talairach and Tournoux (1988) atlas coordinates and the 10-20 electrode placement system (Jasper, 1958), the DLPFC was defined as 5 cm anterior to the site of maximal response of the FDI (Pascual-Leone et al., 1996). This quick and inexpensive method became widely used and accepted, even after it was shown to be unreliable in localising the DLPFC (Herwig et al., 2001). Subsequent to Herwig et al.’s (2001) demonstration that this operational localisation of the DLPFC frequently resulted in stimulation of the more posteriorly located premotor cortex, some authors refined the localisation as 6 cm anterior to the site of the APB/FDI (Michael et al., 2003).
Relatively few studies have used stereotaxic or individualised localisation of the DLPFC (Jorge et al., 2004). Yet precise delivery of TMS to the DLPFC will allow greater refinement of its role in mood, depression and cognition, and will potentially lead to more efficacious treatment of depression (Rusjan et al., 2010).
Another alternative is the 10-20 system (Jasper, 1958), which has the advantage of correcting for head size. This method was again investigated by Herwig et al. (2003), who demonstrated that this method also yielded considerable variation. Furthermore, they acknowledge that F3 did not target the left DLPFC and modifications to the conventional placements would be required, such as measuring from F3 1 cm in an antero-lateral direction, or alternatively to target the midpoint of the triangle between F3, F7 and Fp1. While this method warrants further examination in itself, these complications and the extra time required may be unattractive to many researchers and practitioners using TMS.
A recent study (Rusjan et al., 2010) compared stereotaxic and operational methods of localisation of the DLPFC for rTMS. However, this study had a number of limitations. First, they defined the DLPFC with reference to functional neuroimaging abnormalities in psychiatric illness, thus limiting generalisability to the healthy brain. Second, although the studies they reviewed suggested the DLPFC was located at the juncture of areas 9 and 46, they unempirically selected a voxel coordinate at the junction of these two areas. Finally, they selected the operational method of moving 5 cm anterior to the site of the APB even though this method has been demonstrated to be too posterior (Herwig et al., 2001).
The present study aimed to compare the accuracy of a stereotaxic approach with the commonly used method of moving anteriorly in the parasagittal plane from the point of maximal stimulation of the contralateral APB muscle. Most studies have defined the DLPFC as 5 cm anterior to the motor hand area, but this method is too posterior (Herwig et al., 2001). Thus, we operationally defined the DLPFC as 6 cm anterior to the motor hand area. We then proposed a more accurate method of operationally localising the DLPFC for TMS.
Materials and methods
Participants
Participants were 18 (10 female) healthy people who were recruited through advertisements placed in local university and hospital departments. Their mean (SD) age was 25.89 (6.05) (range = 20–47) years. Seventeen of the participants were right-handed as determined by the Edinburgh Handedness Inventory (Oldfield, 1971). Participants were excluded if they reported any history of neurologic illness. In addition, participants were administered the Structured Clinical Interview for DSM-IV-TR (First et al., 2002) and were excluded if they met criteria for any lifetime psychiatric diagnosis. The study was approved by the Southern Health Research Ethics Committee and Monash University.
Image acquisition
A high-resolution, three-dimensional, T1-weighted image comprising 144 contiguous slices of 1 mm thickness and 1 mm × 1 mm pixel dimensions, with anatomical volume covering the whole brain, was acquired on a 1.5 T Siemens Magnetom. Parameters were repetition time 10.5 milliseconds, echo time 2.2 milliseconds, inversion time 350 milliseconds, flip angle 20 degrees, field of view 25 cm and matrix 256 × 256. Preceding the MRI scan, vitamin E capsules were placed on at least seven points of the scalp based on the standard international 10-20 system (Jasper, 1958) to act as fiducial markers. Specifically, capsules were always placed at Cz, the nasion, the inion, anterior to both tragi, and 50% of the distance between Cz and each tragus.
Transcranial magnetic stimulation
Using a Magstim Transcranial Magnetic Stimulator with a 70 mm figure-of-eight coil, the site of maximal stimulation of the right APB and the resting motor threshold were established using electromyography (Dantec Counterpoint EMG; Dantec, Skovlunde, Denmark) and Laboratory Virtual Instrumentation Engineering Workbench (LabVIEW; http://www.ni.com/labview/). Each participant’s resting motor threshold was defined as the lowest intensity at which an evoked potential of at least 100 µv magnitude was observed in the APB in at least five out of ten consecutive trials. The threshold was found using the method of limits. The point on the scalp where the application of TMS produced a response in the contralateral APB (referred to hereafter as the ‘APB’) was marked and the position was verified with subsequent restimulation. Subsequent to confirmation of the site of the APB, a second mark was placed 6 cm anterior to this location in the parasagittal plane (referred to hereafter as the ‘operational DLPFC’).
Stereotaxic localisation
The left DLPFC was defined stereotaxically by taking separately the arithmetic means of the
The participant’s head was then registered to their native space MRI using the MiniBIRD Model 800 (Ascension Technology Corporation, Burlington, VT, USA), MRIcro (http://www.sph.sc.edu/comd/rorden/mricro.html), and MRIreg (http://www.sph.sc.edu/comd/rorden/mrireg.html). The coordinates of each of the fiducial markers from the vitamin E capsules on the MRI were entered into MRIcro and MRIreg before the participant was restrained in a head rest. The points on the scalp corresponding to the location of the fiducial markers were then coregistered with the MiniBIRD. The MiniBIRD is reported to have a static resolution of 0.5 mm. Once coregistration was complete, the coordinates of the site of the APB and the site 6 cm anterior to this were extracted from MRIcro. The location on the scalp that was closest to the target DLPFC was identified with the MiniBIRD and marked (referred to hereafter as the ‘stereotaxic DLPFC’). Finally, the coordinates of this point were recorded.
The distance between the locations of interest (i.e. the stereotaxic DLPFC, operational DLPFC, and the APB) and the target DLPFC were calculated using the Euclidian distance:
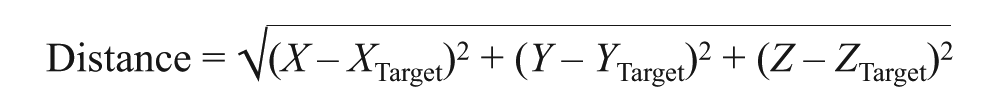
where
Conversion from ‘scalp’ coordinates to ‘brain’ coordinates
Given the coordinates we obtained reflected points on the scalp, we estimated the corresponding area of the brain that would have been stimulated by obtaining the nearest voxel within the brain. This was achieved by employing a binary brain mask supplied in SPM5. The voxel within the mask that was closest to each coordinate was extracted and this was taken to be the voxel that would most likely have been stimulated by the application of rTMS at the corresponding scalp coordinate. When more than one within-brain voxel was equidistant to a given point, the arithmetic mean was taken for the
Development of an empirically based method of operational localisation
To determine whether there could be a more accurate method of operational localisation of the DLPFC, we undertook a number of steps. First, we identified the extra-skull coordinate that was closest to our functional neuroimaging defined target DLPFC. This was achieved by using the Brain Extraction Tool (Smith, 2002) function within FSL (http://www.fmrib.ox.ac.uk/fsl/) (Smith et al., 2004) to create a binary extra-skull mask. Then the extra-skull coordinate within this mask that was closest to the target DLPFC (i.e. −38 30 30) was extracted, analogously to the approach mentioned above that was used to identify the closest within-brain voxel of the scalp coordinates obtained with the MiniBIRD. Second, the translational distances in the
Results
Scalp coordinates
The mean (SD) of the
Mean (standard deviation) of
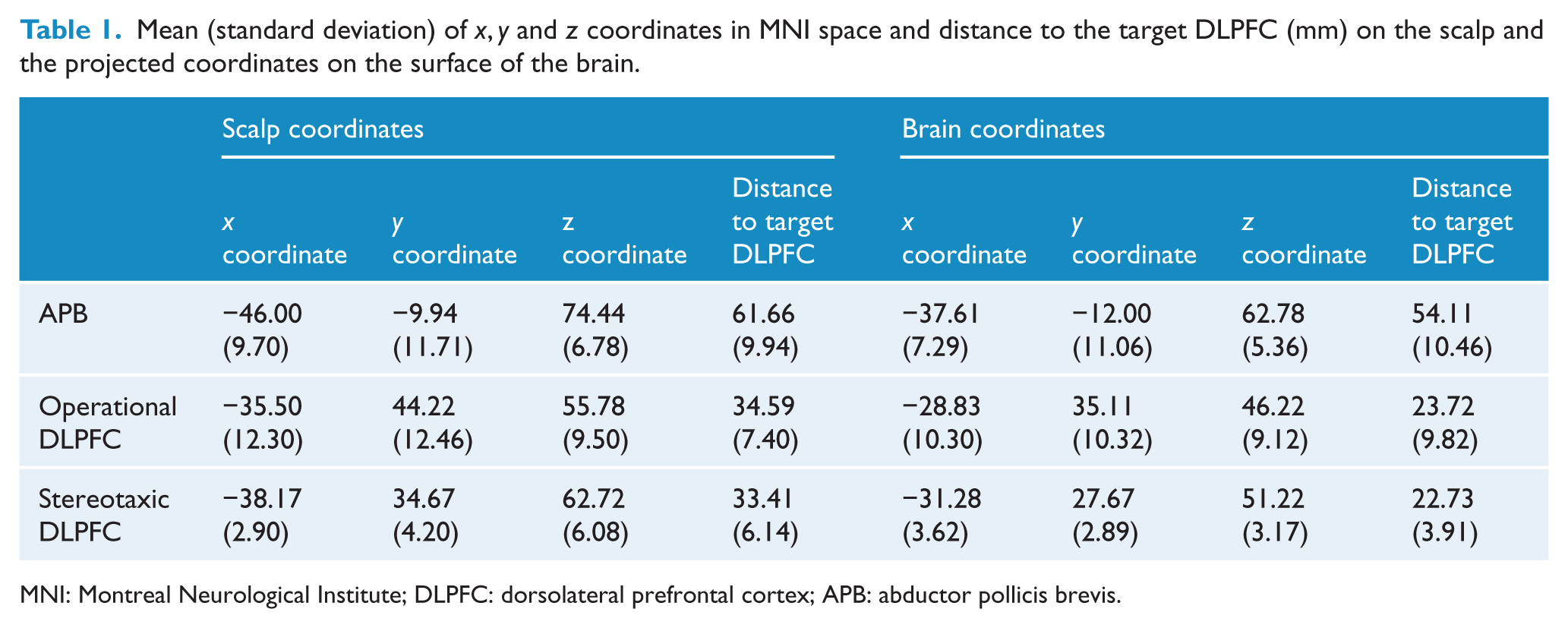
MNI: Montreal Neurological Institute; DLPFC: dorsolateral prefrontal cortex; APB: abductor pollicis brevis.
The localisation of each of the APB, the operational DLPFC and the stereotaxic DLPFC for each participant, as well as the target DLPFC, are shown in Figure 1.
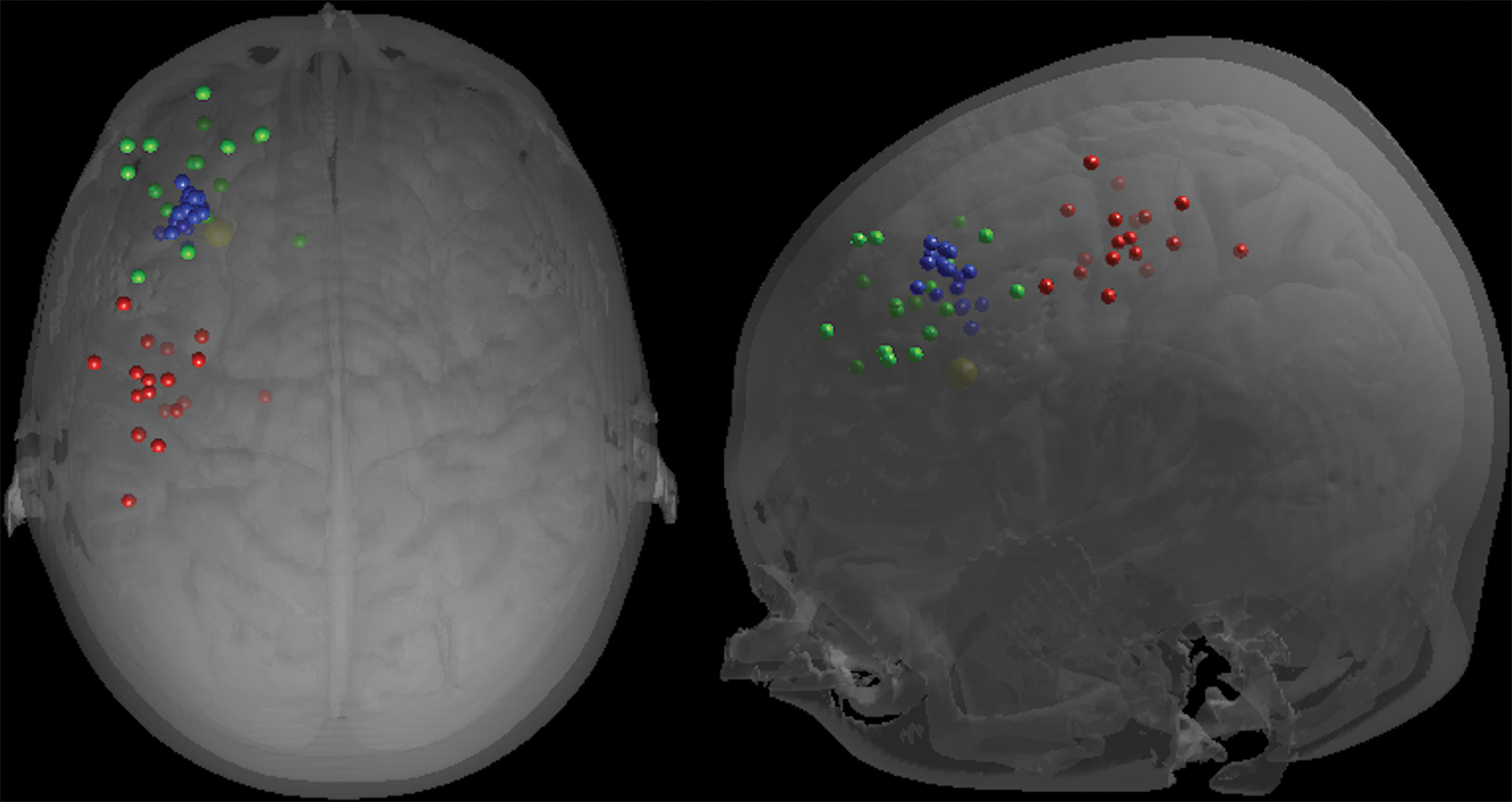
Location of the APB (red), operational DLPFC (green) and stereotaxic DLPFC (blue) on the scalp for each participant, and target DLPFC (yellow) displayed on an individual 3D-rendered image viewed superiorly (
When comparing the distance between the operational DLPFC and the target DLPFC and the distance between the stereotaxic DLPFC and the target DLPFC, Levene’s test for equality of variances was not significant,
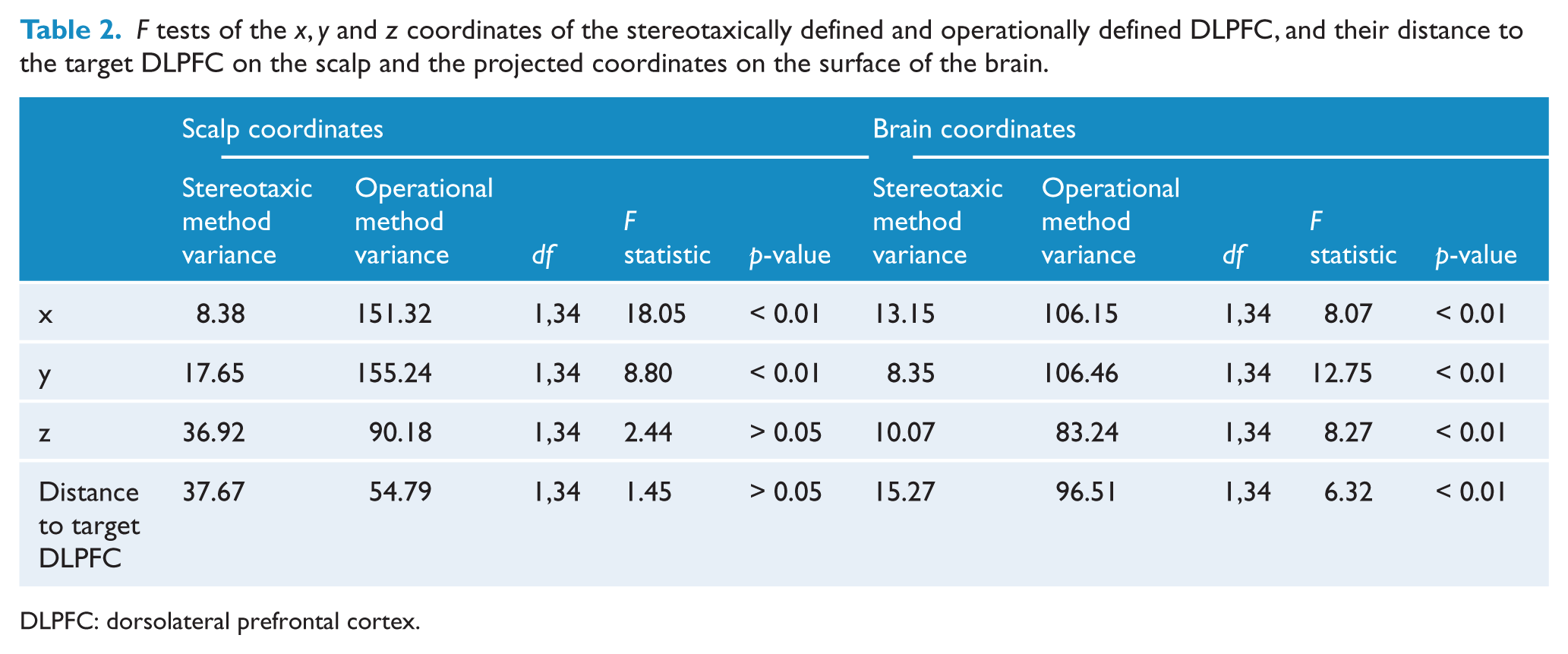
DLPFC: dorsolateral prefrontal cortex.
There was no significant difference in the
To examine the differences between the APB and the stereotaxic DLPFC, we report the total distance as well as the
Mean (standard deviation) of the total distance and the
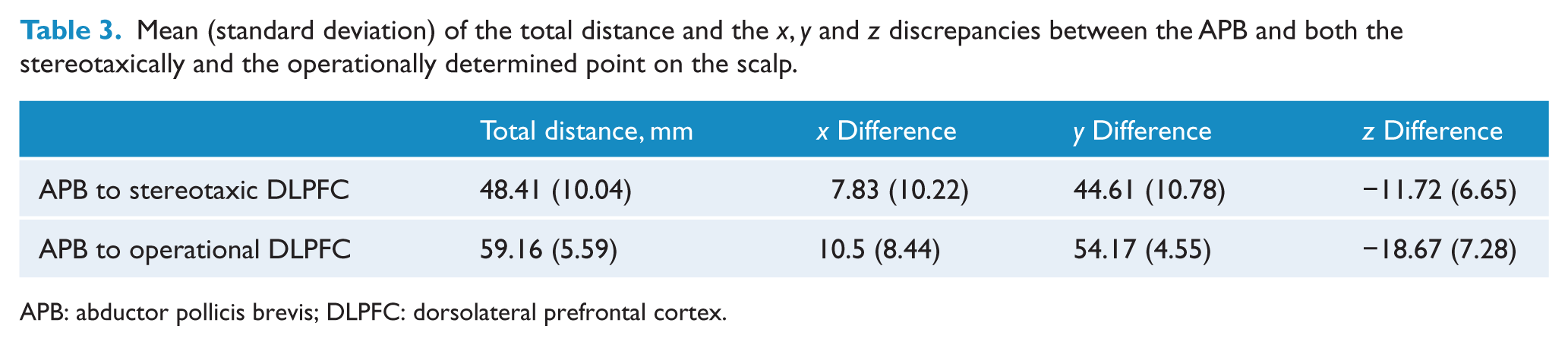
APB: abductor pollicis brevis; DLPFC: dorsolateral prefrontal cortex.
There was a significant difference between the stereotaxic and operational DLPFC in their distance from the APB,
Brain coordinates
The mean (SD) of the
The localisation of the APB, the operational DLPFC and the stereotaxic DLPFC for each participant, as well as the target DLPFC, is shown in Figure 2.
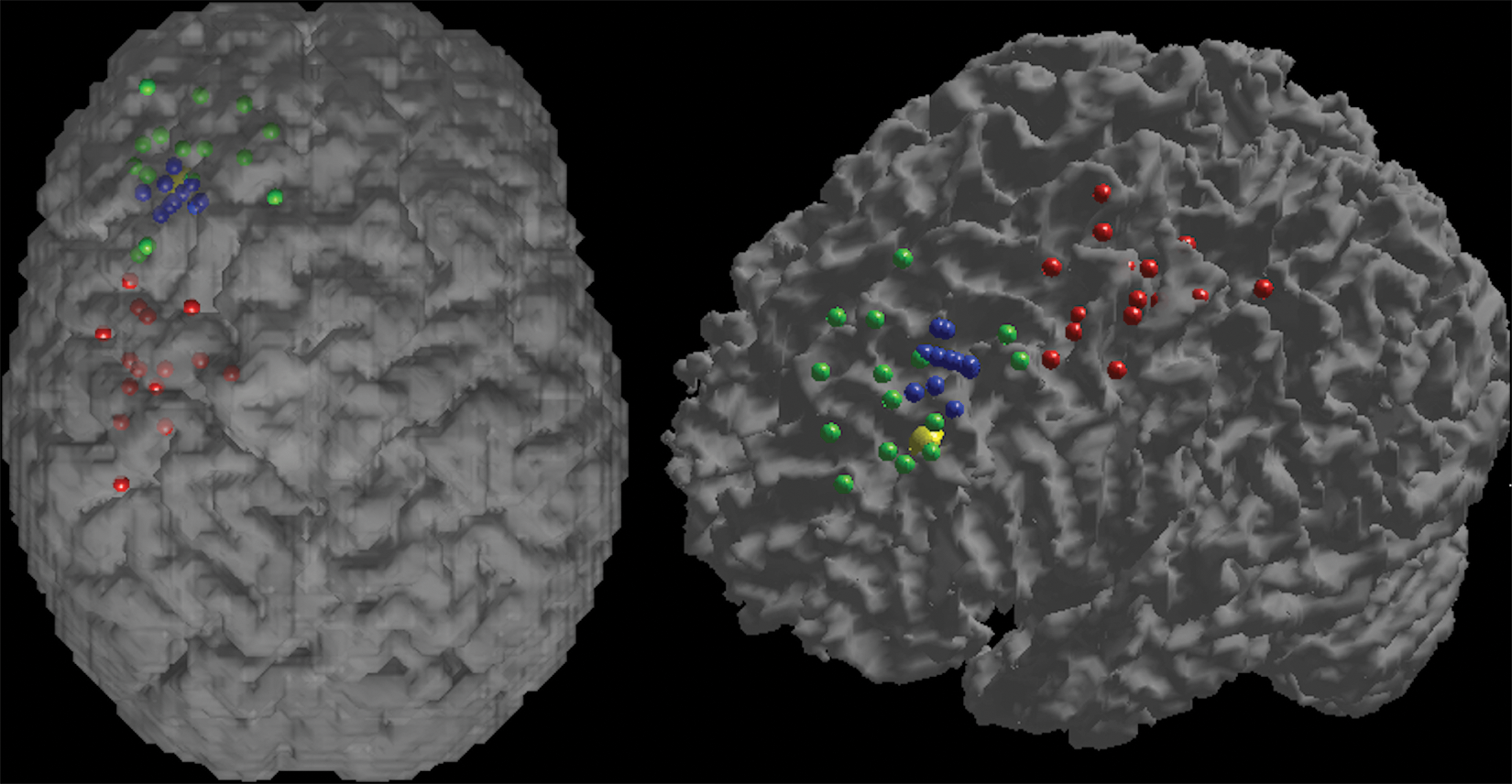
Location of the APB (red), operational DLPFC (green), stereotaxic DLPFC (blue) and target DLPFC (yellow) displayed on an individual 3D-rendered brain viewed superiorly (
When comparing the distance between the operational DLPFC and the target DLPFC with the distance between the stereotaxic DLPFC and the target DLPFC, Levene’s test for equality of variances was significant,
Levene’s test for equality of variances revealed significant differences between the stereotaxic DLPFC and the operational DLPFC for the
There was no significant difference in the
We used the Talairach Daemon (Lancaster et al., 2000) to determine to which gyrus each projected coordinate corresponded. The stereotaxic technique targeted the middle frontal gyrus in 94% (17/18) and the superior frontal gyrus in 6% (1/18) of participants. In contrast, the operational method targeted the superior frontal gyrus in 67% (12/18) and the middle frontal gyrus in 33% (6/18) of participants.
An empirically based method of operational localisation
The extra-brain coordinate closest to the target DLPFC was −51 43 43. The translational distances in the
Discussion
Accurate localisation of the DLPFC is important for investigation of the behavioural effects of rTMS. Brodmann’s area 46 was measured as approximately 21 mm × 22 mm and area 9 as approximately 27 mm × 25 mm in the anterior–posterior and dorsal–ventral axes, respectively (Rajkowska and Goldman-Rakic, 1995); thus, localisation should at least be within this magnitude of resolution. We examined localisation of the DLPFC for rTMS and showed that operational and stereotaxic determination of the site of stimulation yield differential localisation. Specifically, we showed that while the absolute distance to the target DLPFC was not significantly different between the two methods, there was significantly different localisation in the
Our results are consistent with Herwig et al. (2001) in the finding that moving anteriorly in a parasagittal plane from the site of the APB tends to target more dorsally situated Brodmann areas (i.e. 6 and 8). We also showed that the operational definition of 6 cm anterior to the site of the APB was 16 mm more dorsal than the DLPFC, as defined with functional neuroimaging. A recent study (Rusjan, 2010) defined the DLPFC based on the combination of functional neuroimaging abnormalities in depression and the cytoarchitectural atlas of Brodmann’s areas 9 and 46 (Rajkowska and Goldman-Rakic, 1995). The present study has the advantages of objectively defining the DLPFC and is more applicable to studies examining cognitive and mood effects in healthy participants given we defined the DLPFC in this population.
One unexpected finding was that the stereotaxic method was even more dorsal than the operational method: 21 mm dorsal to the functional neuroimaging-defined DLPFC. One possible reason for this is that there could be more than one point on the scalp that is equidistant from the stereotaxic DLPFC, which was subcortical in the white matter of the middle frontal gyrus. Owing to the morphology of the scalp/skull, the closest point to the DLPFC was located on the dorsal aspect on the scalp rather than a more lateral point, which would have presumably targeted the DLPFC more accurately. Consistent with this idea was our finding that the stereotaxic method was more accurate in the
While stereotaxic methods are more reliable than operational methods of localisation, the availability, the efficiency and the financial appeal of the latter mean that they will likely continue to be used. Considering that, there is a need for a more accurate method of operational localisation. Herwig et al. (2001) showed that moving 5 cm anterior to the site of the APB was dorsal to the DLPFC, and we showed a similar result defining the DLPFC 6 cm anterior to the site of the APB. Both of these results suggest that researchers could more accurately localise the DLPFC as anterior and ventrolateral from the site of the APB. We provide an empirical estimate of a more accurate method of operational localisation. We suggest that the DLPFC should be defined as 5 mm lateral, 53 mm anterior and 31 mm inferior to the site of the APB. However, caution must be used in interpreting this finding. We derived these coordinates in stereotaxic space and it cannot be assured that this would equate to identical areas when measuring these distances on the scalp given the irregularity of the morphology of the skull; measuring 53 mm anterior in a parasagittal plane will also cause displacement in the other axes given the three-dimensional nature of the skull. This issue is inherent to any operational method with reference to the APB. In a practical sense it is particularly difficult to translate in the
A potential source of variance using any operational method is the localisation of the APB. To our knowledge this variance has not been systematically studied previously. Our study showed the standard deviation of the APB in the
Lotze et al. (2003) proposed an alternative method of projecting coordinates from the scalp surface to the brain by fitting an ellipsoid to the head. Our method of extracting the nearest voxel has the advantages of being simpler to calculate and not making assumptions about head shape and direction of the field.
An important issue is whether more accurate localisation of the DLPFC results in more efficacious effects such as antidepressant treatment. This has long been theorised to be true, but limited evidence exists to support the claim (Fitzgerald et al., 2009). Future research will be needed to determine in general whether better localisation of the DLPFC is more efficacious and specifically whether our empirically based method of operational localisation is more effective than the existing 5 cm or 6 cm methods.
In the present study we have focussed the left DLPFC because it has been more extensively used in rTMS studies of depression, cognition and mood effects in healthy controls. Given asymmetries in neuroanatomy, a similar procedure could be repeated for the right hemisphere. A potential limitation of our target site was that we defined this based on functional imaging studies of cognitive paradigms. It is plausible that emotional-based paradigms such as mood induction studies would yield a different subregion of the DLPFC, although this has never been investigated to our knowledge. Future research could determine whether cognitive and emotional paradigms activate different subregions of the DLPFC and whether delivery of TMS to these sites produces different effects on mood and cognition (Herwig et al., 2003).
Large individual differences in anatomy render operational localisation, with reference to the site of the APB, inaccurate and unreliable. The current study represents the first specific attempt to compare a stereotaxic method with an operational method of localisation of the DLPFC for TMS. It has been proposed that more precise coil positioning may lead to a superior antidepressant effect of rTMS (Herwig et al., 2001; Rusjan et al., 2010), and the same principle can be used to more accurately study acute mood effects in healthy controls and cognitive effects such as working memory. We suggest that the DLPFC should be defined as 5 mm lateral, 53 mm anterior and 31 mm inferior to the site of the APB for TMS.
Footnotes
Acknowledgements
The authors would like to thank Dr Vladimir Dubaj for his assistance with the operation of the TMS and the MiniBIRD.
Funding
The study was supported by departmental funds.
Declaration of Interest
The authors report no conflicts of interest. The authors alone are responsible for the content and writing of this paper.
