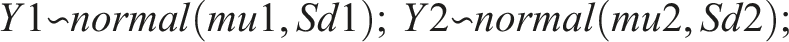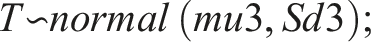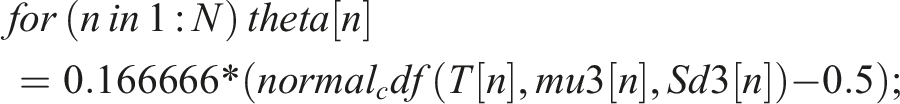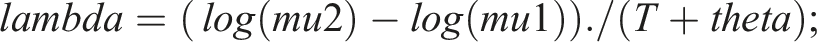Abstract
Background
Phosphatidylethanol (PEth) is formed in erythrocyte membranes after alcohol consumption. When abstaining, the PEth level falls with a rate proportional to its concentration, and a short apparent PEth half-life supports abstinence. We here derive algorithms for calculating unbiased half-lives and confidence intervals (CIs).
Methods
PEth was measured using Acquity UPC2-MS/MS systems in clinical blood samples from out-patients. We identified 6989 individuals having taken two or more PEth samples within 28 days. One measurement pair was randomly selected from everyone. We derived methods for and calculated PEth half-lives and corresponding 95% CIs for exact, rounded, and truncated data, on closed form, Monte Carlo methods and No-U-turn sampling.
Results
The peak of the PEth half-life was at 8.62 days. Peak PEth half-life was 8.72 days for men and 8.47 days for women (P = 0.028) and on age: 8.55 days for age 18–39 years, 8.56 for age 40–59 years and 8.87 for 60+ years (P = 0.026). PEth concentration did not significantly affect half-life. CIs on a closed form performed excellently on exact data, with misclassification of abstinence for 16 out of 6989 observations (0.23%). When rounding or truncating data, misclassification occurred using Monte Carlo methods in 104 (1.5%) and 127 (1.8%) of the observations and using closed form algorithms in 855 (12.2%) and 777 (11.1%).
Conclusion
Unbiased PEth half-lives and CIs can be calculated and put into use in laboratory information systems. Rounding or truncating data used for PEth half-life calculation widened CIs with misinterpretations of alcohol abstinence.
Keywords
Background
Consecutive measurements of phosphatidyl ethanol (PEth) in blood have rapidly become the gold standard for supporting alcohol withdrawal 1 and there is also an association between levels and self-reported intake. 2 PEth forms in erythrocytes in a reaction between ethanol and phosphatidylcholine catalyzed by phospholipase D (PLD),3,4 which is present as two isoenzymes in humans, PLD 1 and 2. These enzymes exhibit variable inter-individual kinetics in catalyzing PEth formation. 5 The individual rate of PEth formation can be assessed by measuring PEth levels in the lysates of freshly isolated erythrocytes incubated ex vivo with standard concentrations of ethanol. 6 As the formation rates differ between individuals, the PEth concentration at steady state will only give a crude estimate of an average alcohol intake. 7 Several major forms of PEth originate from the reaction of phosphatidylcholine with ethanol, 16:0/18:1, 16:0/18:2 or 16:0/20:4, constituting approximately 42%, 26% and 9% of total PEth, respectively. 8 While most of the PEth degrades within a day in most cells and in plasma, human erythrocytes apparently lack the ability to rapidly degrade PEth. 3 Measurements of erythrocyte PEth may thus be used to verify alcohol intake, and to semiquantitatively assess a previous intake. During alcohol abstinence, the apparent elimination PEth half-life in erythrocytes from experimental studies ranged from four to 13 days.9–12 Often, a clinical consultant at the laboratory interprets the results and adds his/hers comments on the report, where the last patient result is compared to the previous one, and where the requester is interested in the likelihood of a new intake since last test However, this interpretation from a pair of measurements is not trivial in a laboratory setting with analytical uncertainty, since uncertainties introduce a bias, and where there are no published methods for obtaining unbiased PEth half-lives and the associated confidence intervals (CIs). We here characterize the uncertainties associated with estimation of PEth half-lives using mathematical models and investigate the impact when the original exact results are rounded or truncated in the laboratory information system. Finally, we investigate the difference between assessing abstinence from PEth half-lives only, or when using the associated CIs.
Methods
Sample preparation
EDTA-anticoagulated whole blood samples were obtained by drawing venous blood into the EDTA-prefilled vacutainer tubes (Beckton Dickinson Norway, Oslo, Norway) and processed as per manufacturer’s instructions. Transportation is usually in the afternoon by car and air to the laboratory. Samples are frozen after arrival at the laboratory and analyzed within 7 days.
PEth measurements
PEth was analyzed as described previously. 13 Briefly, PEth (16:0/18:1) was analyzed in EDTA-anticoagulated whole blood using a Waters Acquity UPC2 ™ Ultra Performance Convergence chromatography system coupled to Waters TQ-S triple quadrupole mass-spectrometer (UPC-MS/MS) (Waters, Milford, MA). The limit of quantification was established at 0.015 µmol/L (10.5 ng/mL) for PEth, as well as PEth values below 0.03 µmol/L were reported as negative. The analytical uncertainty was estimated with a coefficient of variation of 8.7 %, based on internal quality control samples.
Patients
The laboratory does not serve hospitals, but general practice and smaller institutions over all Norway, having most out-reach in Southern and Eastern Norway. The study protocol was approved by the local Regional Ethics Committee (REK sør-øst D, case 25827). The study period was from 2017-11-01 to 2021-01-31. Individuals over 18 years with a pair of PEth measurements within a time frame of 28 days were included in the study. We found 6989 individuals with pairs of PEth measurements meeting criteria for inclusion. Of the 6989 individuals 1367 individuals had rising PEth concentration and classified as a new intake, 1823 individuals had negative (<0.03 µmol/L) PEth tests in both samples and 495 individuals had falling concentrations and a negative second test classified as abstinence. Half-life calculations were performed on the remaining 3304 individuals. Results were entered with four decimal points from the raw data file from the instrument. Results were then either rounded or truncated to two decimal places.
Assessment of alcohol abstinence using PEth half-life estimations
The analytical uncertainty for PEth in our laboratory is roughly proportional to the measurement result and the current coefficient of variation is 8.7%. Before reporting, the result is truncated to two decimal places (in mathematical terms introducing a uniform uncertainty having a min = −0.01 and max = 0). Such truncation is general practice in pharmacology, when the test results used for legal issues are truncated rather than rounded, 14 although this custom differs from the recommended reporting in chemistry. 15 Usually, patients subject to testing use to show up at approximately the same time of the day, so we have assumed that the length of the time interval between sampling can be modeled as days * 24 h/day having a uniform uncertainty with min = −2 h and max = 2 h.
In the present study, we intend to study the effect of rounding and truncating results before calculating half-lives. We further address the differences in assessment whether the assessment is based on the uncertainty of the estimated half-life, in other words, whether the assessment be significantly different if the associated uncertainties are considered. When making an assessment, we follow the recommendations of conformity assessment from The Joint Committee for Guides in Metrology that it is the upper CI (here the 97.5% limit, corresponding to a coverage factor of 1.96). 16 We first perform Monte Carlo simulations 17 to generate estimates with CIs, and we study the effects on truncating or rounding results on the estimated half-life. Monte Carlo simulations, the gold standard, have a draw-back, as simulations are usually not feasible even in a modern-day laboratory information system. We thus seek to derive alternatives to Monte Carlo simulations, where the estimates can be calculated directly without simulations or likelihood optimization in a finite number of standard operations, for example, on a closed-expression form, or in short: closed form. 18 Our derivation of such a closed form expression (i.e., direct calculation of the half-life and its CI from two PEth results and the sampling interval, without simulations) is detailed in the Supplement. What we do, is to apply standard propagation-of-errors to the calculations of half-lives. Such calculations are straightforward for addition or subtraction of numbers, but for multiplication or division as in calculation of half-lives, more complicated, as multiplication or division adds a bias component to the estimate. We also set up a Bayesian model in Rstan,19–21 which may be described as a compiler for a No-U-Turn Sampler (NUTS). Finally, we compare our assessments with assessment based on the calculated half-life alone.
To illustrate the effect of uncertainty on decisions we chose a half-life of 13 days (the longest half-life published in a scientific report) and compared the apparent lower and upper 95% confidence limits to this half-life. When the upper 95% confidence limit of apparent PEth half-life is shorter than 13 days it may be concluded with “abstinence.” When the lower 95% confidence limit of apparent half-life is longer than 13 days, it may be concluded with “new intake of alcohol.” The rest of the samples having confidence intervals overlapping 13 days were considered “inconclusive.”
The derivation of expressions for half-life on closed form has direct, real-life applications, as it may be the base of an automated routine in a laboratory information system for the assessment of alcohol intake based on two measurements. The expressions we derived are not exact but approximations, hence the need to verify those expressions to gold standard Monte Carlo simulations. The findings may be generalized two other laboratory measurements where estimation of half-life is important, that is, the monitoring of cancer by using repeated serum tumor marker measurements.
Bayesian modeling in Rstan
Bayesian modeling is gaining popularity. We set up a Bayesian model in Rstan, which is the R interface to Stan. Stan is an open-source No-U-turn sampler (NUTS), and is increasingly replacing BUGS, Open BUGS and JAGS as the Bayesian sampler of choice. Bayesian modeling is somewhat demanding, as one must decide whether the “half-life” is an inherent characteristic of the individual (and thus the observed results depend on the inherent “half-life”), or if “half-life” simply is a calculation done post sampling. We chose the latter, and the model was:
Finally, half-life was obtained as a posterior estimate:
Results
Estimates from Monte Carlo sampling and closed form compared to estimates obtained without considering uncertainty
Classification of paired PEth measurements into negative, abstinent, inconclusive, or new intake using Monte Carlo simulations (MC) and closed form (CF) calculations.
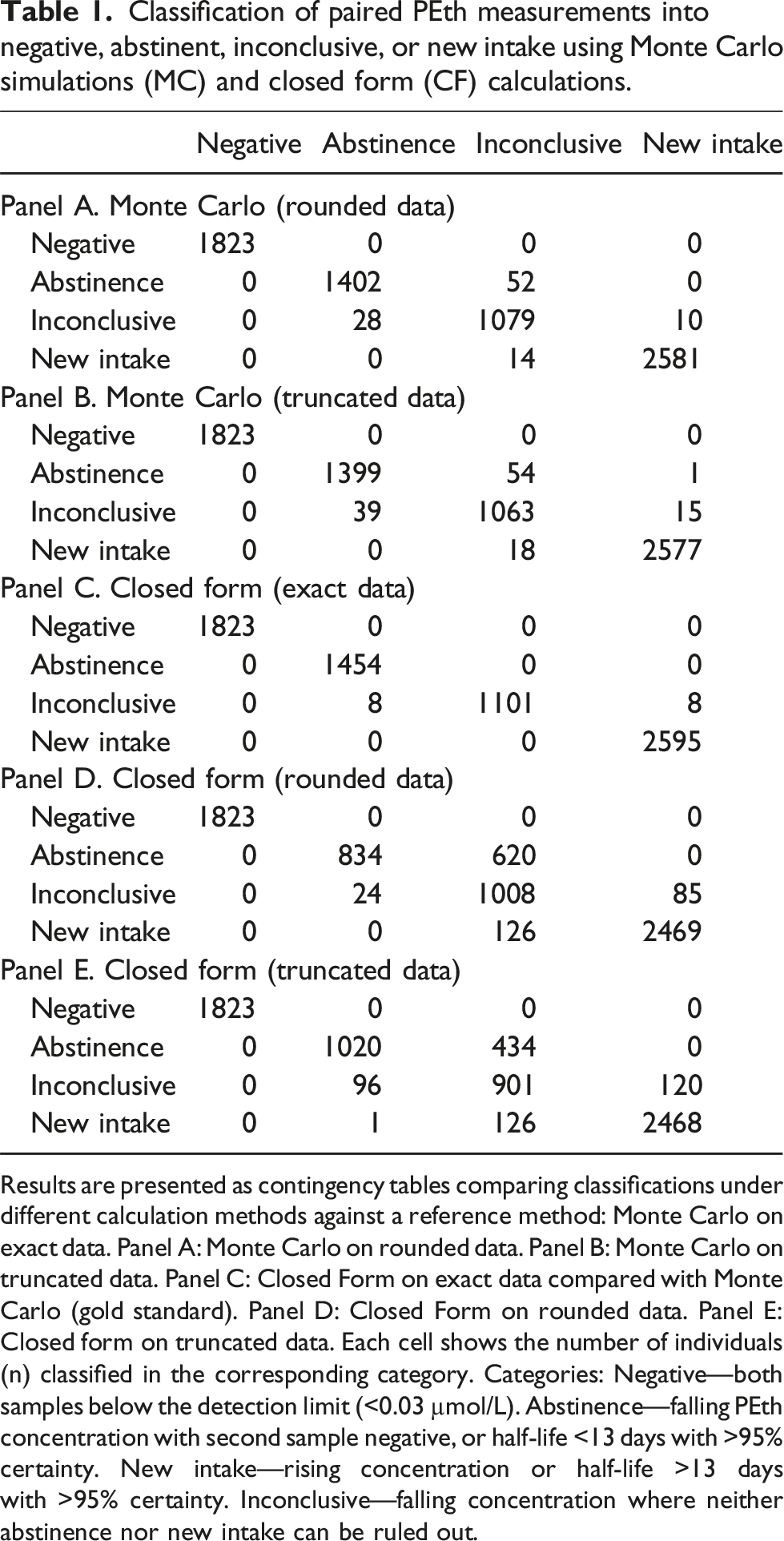
Results are presented as contingency tables comparing classifications under different calculation methods against a reference method: Monte Carlo on exact data. Panel A: Monte Carlo on rounded data. Panel B: Monte Carlo on truncated data. Panel C: Closed Form on exact data compared with Monte Carlo (gold standard). Panel D: Closed Form on rounded data. Panel E: Closed form on truncated data. Each cell shows the number of individuals (n) classified in the corresponding category. Categories: Negative—both samples below the detection limit (<0.03 µmol/L). Abstinence—falling PEth concentration with second sample negative, or half-life <13 days with >95% certainty. New intake—rising concentration or half-life >13 days with >95% certainty. Inconclusive—falling concentration where neither abstinence nor new intake can be ruled out.
Distribution of half-lives in the patient population
One may expect that a large subset of patients subject to testing do abstain from alcohol, with half-lives corresponding to half-lives obtained in an experimental study
9
with a mean half-life of 7.6 days. We plotted a histogram of all half-lives (Figure 1) and calculated the peak. The peak in our study is slightly higher, with 8.62 days (95% CI 8.47–8.79). It has been discussed whether the half-life is longer when the PEth concentration is low. In our study the peak density of the half-life was 8.23 days (7.2–17.4) when PEth was >1.00 (Very high), 8.51 days (8.12–8.94) when 0.30 ≤ PEth<1.00 (High), 8.56 days (8.35–8.78) when 0.15 ≤ PEth <0.30 (Moderately high) and 8.72 days (8.52–8.93) when 0.03 ≤ PEth <0.15 (Within normal). There was a trend of slightly prolongated PEth elimination at lower concentrations of PEth but the differences were not significant. Women had a significantly (P = 0.026) shorter PEth half-life than men: 8.47 days (8.29–8.67) versus 8.73 (8.56–8.92) On increasing age, PEth half-lives increased, from 8.55 days (8.34–8.80) for age 18 to 39, 8.56 days (8.33–8.78) for age 40 to 59, to 8.87 days (8.65–9.10) for 60+ years. Higher PEth half-life for 60+ years was significant (P = 0.026). There was a trend of slightly prolongated PEth elimination at lower concentrations of PEth. A.) The peak density of the half-life was 8.23 days when PEth was >1.00 (very high), 8.51 days when 0.30 ≤ PEth<1.00 (high), 8.56 days when 0.15 ≤ PEth <0.30 (moderately high), and 8.72 days when 0.03 ≤ PEth <0.15 (within normal). B.) Women had shorter PEth half-life than men: 8.47 days versus 8.73 days (P = 0.026). C.) On increasing age, PEth half-lives increased, from 8.55 days for age 18 to 39, 8.56 days for age 40 to 59, to 8.87 days for 60+ years (P = 0.026).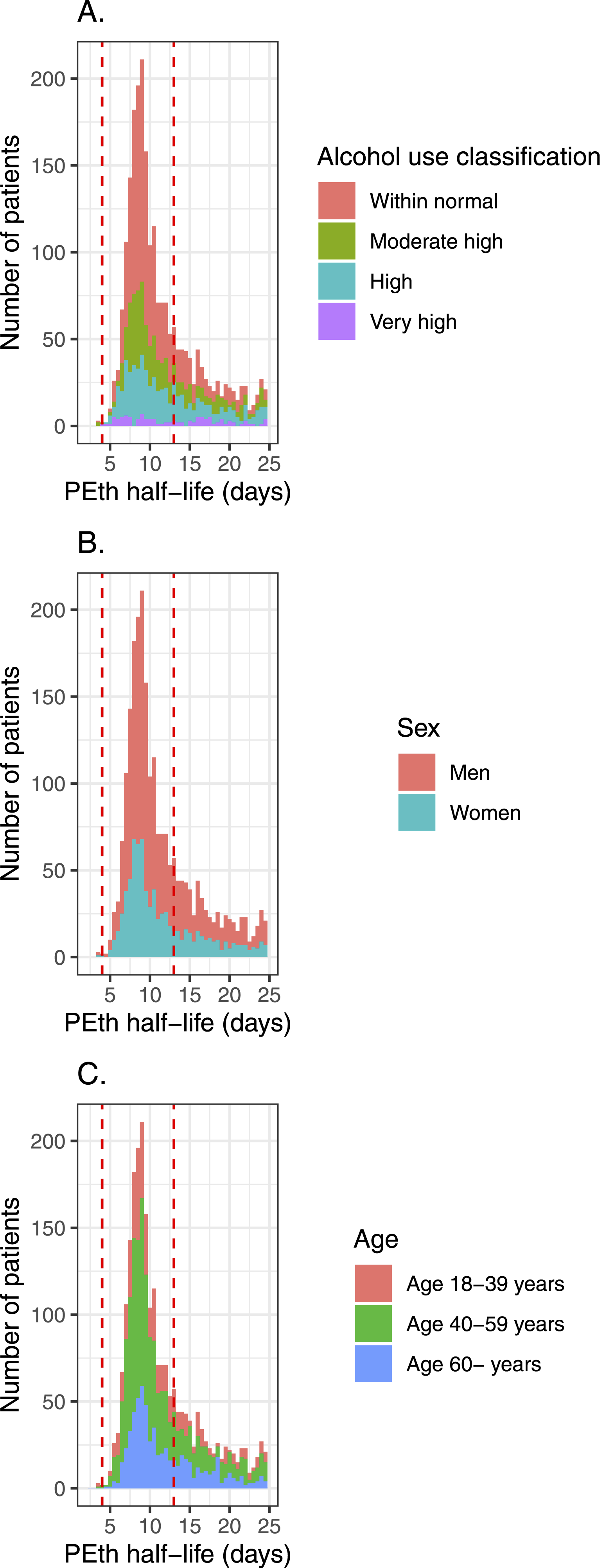
Classification of patients taking measurement uncertainty into account
Using our estimates of measurement uncertainty (using a coverage factor of 1.96), we may classify observations into abstinence (PEth half-life is shorter than 13 days with more than 95% certainty) or new intake (PEth half-life is longer than 13 days with more than 95% certainty). Observations compatible with a half-life both shorter and longer than 13 days are classified as inconclusive. We see that this classification quite differs from a classification based on PEth half-life estimates (Figure 2). In a sensitivity analysis, we tested the effect of shortening the limit for the upper CI of a PEth half-life that is considered a new intake. When the limit is set to 13 days, 1117 individuals would be considered inconclusive and 1228 having a new intake. Shortening the limit to 11 days reduces the number of individuals considered to be inconclusive to 901 and increases the number of individuals having a new intake to 1444. For 9 days, the numbers are 612 and 1733.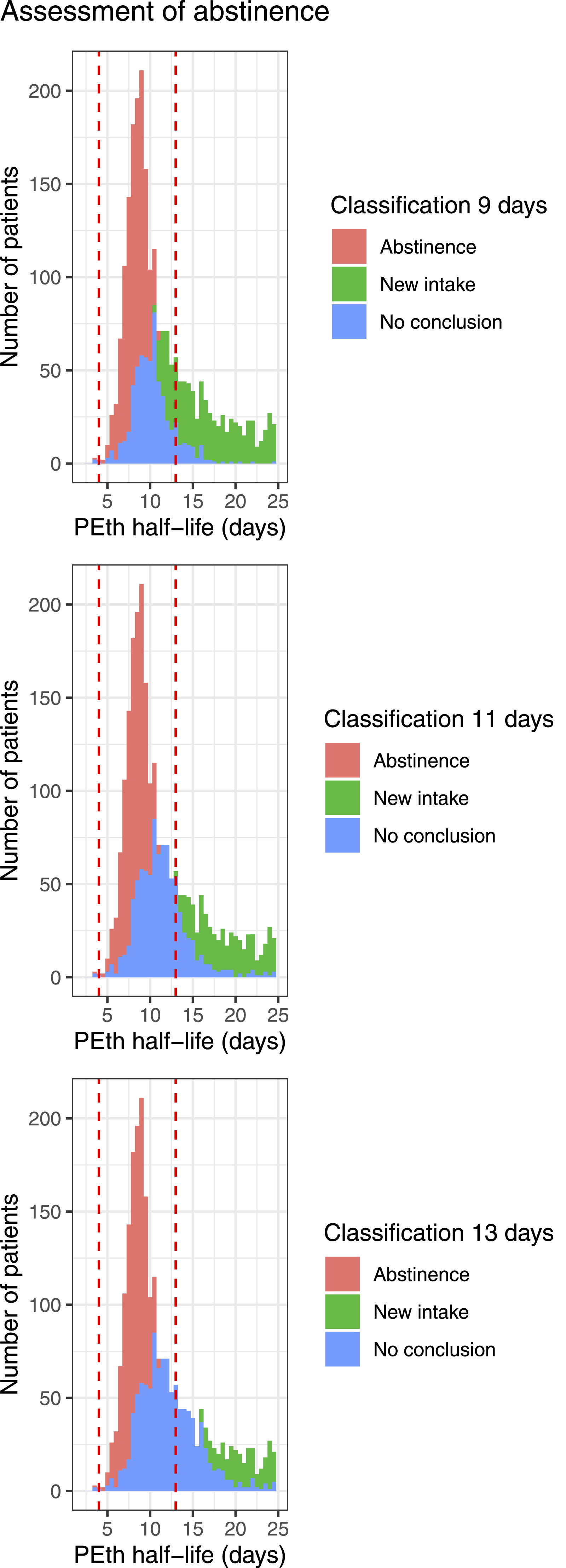
We did a sensitivity analysis to illustrate the effect of shortening the limit for the upper CI of a PEth half-life that is considered a new intake (Figure 2). When the limit is set to 13 days, 1117 individuals would be considered inconclusive and 1228 having a new intake. Shortening the limit to 11 days reduces the number of individuals considered to be inconclusive to 901 and increases the number of individuals having a new intake to 1444. For 9 days the numbers are 612 and 1733. However, if the penultimate sample has a PEth concentration in blood below the cut-off limit for reporting, this will always trigger the outcome “abstinence,” regardless of the estimated half-life.
Bayesian modeling in Rstan
Calculation was slow, and we didn’t see any obvious gains for Bayesian modeling, since none of the parameters had to be estimated simultaneously. The results agreed well with Monte Carlo estimates (Figure 3d Supplement):
Validation of the CIs obtained on closed form
For each pair of PEth measurements the following calculations were performed. We first calculated best possible estimates for half-lives and their associated CIs using the results with four decimal points and Monte Carlo Simulation (N = 10,000)., We then performed Monte Carlo Simulations for results rounded to two decimal points shown in Panel A (Table 1) and truncated to two decimal points (Panel B). Finally, we calculated the approximations on Closed Form and compared them with Monte Carlo results, as presented in Panels C, D and E (Table 1).
Discussion
PEth has rapidly becoming the marker of choice in assessing alcohol intake and supporting alcohol withdrawal. PEth is a specific biomarker for alcohol intake, is stable on transportation and storage for several days 22 and yields high enough sensitivity to detect a single alcohol intake. 23 However, there are few articles discussing how to use and report PEth measurements in clinical practice. Helander proposes cut-offs from 0.03 to 0.05 µmol/L for reporting and 0.30 µmol/L as an indication of high-risk consumption.24,25 In Norway guidelines have recommended a cut-off of 0.03 µmol/L to indicate that alcohol has been consumed during foregoing days. 26 None of the guidelines contain recommendations on how falling levels should be interpretated, and there is no comment on differences in PEth half-lives between individuals. Van Uytfanghe used self-reports and train a decision tree model for assessing abstinence 27 which led to a harmonization document (2022 Consensus of Basel, Society of PEth Research) for the interpretation of phosphatidylethanol concentrations in the clinical and forensic setting.28,29 The consensus defines the target measurand (PEth 16:0/18:1 in whole blood), cut-off concentrations of 20 ng/mL (0.028 µmol/L) and 200 ng/mL (0.28 µmol/L) for intake and high-risk consumption, as well as minimal requirements for the applied analytical method (i.e., accuracy and precision within 15%). Finally, recent reports point at faster elimination in individuals with a high erythrocyte turnover,30,31 but it remains to see the consequences of those observations for the interpretation of PEth results.
In a study based on laboratory data, we cannot deduce which of the subjects that consumed alcohol between the PEth measurements. However, the apparent half-life corresponding to the peak of the distribution, should be a fairly measurement of the PEth half-life in a person abstaining from alcohol. It is also possible to from those measurements calculate sensitivity and specificity of the testing protocol, that is, what is the minimal sensible time span between two PEth measurements, to able to conclude on abstinence.
In our laboratory we have always truncated results for tests with a possible medico-legal outcome to the desired decimal places, which is also the case in other laboratories (personal communication). The rationale is that by rounding, one half of the cases get a reported result that is higher than the original read-out. But the origins of this tradition are difficult to trace. In older textbooks we find a different approach where subtraction of three analytical standard deviations from test results is suggested. 32 Alternatively, laboratories round the results, but increase the cut-off with between one and three analytical standard deviations.
In this study, we have modeled analytic uncertainty as proportional to the measured concentration. This follows common practice of reporting analytical and biological uncertainty as proportional and estimated at a single level, as in databases of uncertainties for laboratory tests. 33 However, this is a generalization, since it implies that the analytical error approximates 0 when the analyte concentration is small. The subject is discussed by Martin, 15 who proposes a more realistic model of analytical uncertainty with a constant part (dominating at small concentrations of the analyte) and a proportional part (dominating at large concentrations), and finally, Martin notes the possibility of creating a vector of uncertainties, where each individual measurement may have its own uncertainty, modeled or obtained by replicated or taken off set from other studies. We think that the model of Martin is more accurate than assuming just a proportional analytical uncertainty. However, we believe that we are somewhat secured by the cut-off, as low measurements below cut-off are not reported quantitatively but simply as “negative.” If one would extend the measurement and report lower concentrations, it might not be feasible to do without modeling with both a constant and a proportional component of analytical uncertainty.
We first assessed the effect on rounding and truncating results on the calculation of the half-life. The derived expressions on closed form return unbiased estimates of the half-life. However, the resulting uncertainty (e.g., the root-square-mean deviation of the estimate) is larger after rounding or truncation, due to the inevitable addition of the uncertainty of the rounding or truncating procedure. We have then assessed the estimated uncertainties of the calculated half-lives, having found a good agreement between the CIs estimated by Monte Carlo simulations, or obtained by closed form with exact data. On contrary, CIs for the estimated half-lives for rounded or truncated results tend to be larger than by Monte Carlo simulations. One potential explanation may be that the normal approximations of the (uniform) rounding, and truncation procedures are fair when those procedures are relatively small compared to the change in concentrations (having a normal distribution). A longer estimated elimination half-life inevitably means a smaller change in concentrations between the two sample occasions, and accordingly that rounding, and truncation will have a larger impact on the CIs. The same applies to samples with low PEth concentrations, where rounding and truncation have larger effects.
We then tested for misclassifications. Misclassifications, for example, when the pair of observations is given the wrong interpretation, may result both from the procedures of rounding or truncation, or from a non-optimal procedure for estimating the CIs. We find that there is a close agreement between the gold standard (i.e., exact data and a Monte Carlo procedure) against the closed form estimate obtained from exact results. Both rounding and truncation result in increasing misclassification, even if a Monte Carlo procedure is used. Using a CI obtained on closed form, will clearly increase misclassifications. As noted previously, misclassifications are skewed, not symmetrical. Noteworthy, most PEth half-life estimations in studies performed on abstaining volunteers were done with no reference to how calculation of PEth half-life was done, for example, if data had been rounded or truncated, and how many digits were used in calculations.
Our results clearly show that in case of PEth, the method used to calculate half-lives (with associated CIs) infer various levels of systematic bias and has significant effects on the calculated half-life estimate. Rounding or truncation further adds a stochastic effect and rounding or truncation thus should be avoided when possible.
Limitations
We have calculated the peaks of the PEth distributions, as there are few real-world studies published. A significant limitation is the lack of other reference tests, such as other markers for alcohol abuse, or patient information on alcohol intake. However, the estimated peaks show quite small differences for elimination regarding PEth concentrations, age and gender. A second limitation is that we cannot assess the variability of PEth elimination, not between patients and nor within patients. Good studies for patient (not research subject) variability using unbiased calculation methods are surely needed.
Conclusion
We have shown that estimates of half-lives and their associated uncertainties may be calculated by Monte Carlo procedures or on a closed form from pairs of exact, rounded, or truncated PEth measurements. Such calculations may be added to a laboratory information system or could be assessed independently on a web site. We show that rounding and truncation result in a slight bias in the calculated half-life, and the expressions derived by us may compensate for that. The estimated half-lives and uncertainties may be used to assess abstinence. However, the calculations of uncertainties are less accurate on rounded or truncated results, due to introduction of a stochastic error. Rounding and truncation thus will always lead to increasing misclassification. Assessment of abstinence should thus be preferably based on exact results, avoiding rounding and truncation. Although the calculation methods seem complex, they can easily be put into clinical use. Further, measuring PEth with short time intervals give high uncertainty in the estimated apparent PEth half-life. Our calculations may provide guidance to a least time frame between samples.
Supplemental Material
Supplemental Material - Estimation of half-life with confidence interval for Phosphatidylethanol (PEth) in blood based on two consecutive measurements
Supplemental Material for Estimation of half-life with confidence interval for Phosphatidylethanol (PEth) in blood based on two consecutive measurements by Johan Bjerner, Michael Sovershaev, Elisabeth Wiik Vigerust, Thor Hilberg, Johannes Østrem Fjøse, and Tom Rune Karlsen in Annals of Clinical Biochemistry.
Footnotes
Acknowledgements
We thank Trude Sandvik and Asgeir Husa for having set up LC-MS/MS methods for PEth and analyzing samples daily. We thank Torleif Trydal and Aleksandar Djordjevic for fruitful discussions, on alcohol misuse, PEth-testing, and measurement uncertainty. Finally, the Swedish Board for Accreditation and Conformity Assessment (SWEDAC) is acknowledged for providing the first author with seminars and working experience in calibration and measurement uncertainty.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
The study protocol was approved by the local Regional Ethics Committee (REK sør-øst D, case 25827).
Contributorship
Johan Bjerner: Study design, statistics, collection of data, calculation scripts, and manuscript preparation. Michael Sovershaev: Study design, application to ethics committee, and manuscript preparation. Elisabeth Wiik Vigerust: Study design, expertise in pharmacology and toxicology, and manuscript preparation. Thor Hilberg: Study design, application to ethics committee, expertise in pharmacology and toxicology, and manuscript preparation. Johannes Østrem Fjøse: Study design, statistics, and manuscript preparation. Tom Rune Karlsen: Study design, statistics, and manuscript preparation.
Guarantor
The corresponding author is acting as guarantor of this study.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.