Abstract
Background
Cystatin C (CysC) is a biomarker used to assess kidney function. Produced by all nucleated cells, it is freely filtered by the glomeruli, and its serum levels increase rapidly following kidney injury. Establishing reference intervals (RIs) is fundamental for the clinical application of CysC, as these parameters vary with geographic region and ethnic origin. This study aimed to determine RIs for CysC in healthy children aged 5–11 years from public schools in Santa Maria, southern Brazil.
Methods
We enrolled 134 healthy children (aged 5–11 years, both sexes). Cystatin C levels were measured via immunoturbidimetry, and RIs were defined by the 2.5th and 97.5th percentiles.
Results
The RI for serum CysC was 0.59 to 1.10 mg/L. Sex-specific analysis revealed CysC RIs of 0.53–1.10 mg/L for females and 0.61–1.10 mg/L for males.
Conclusions
These findings provide valuable insights for paediatric clinical decision-making in a previously unstudied population and underscore the need for further research to validate and refine the clinical applications of CysC in paediatrics.
Introduction
Chronic kidney disease (CKD) is a major global public health concern, affecting approximately 850 million people worldwide. In adults, CKD is often associated with diabetes and obesity, whereas in children, it is more commonly linked to congenital anomalies of the kidney and urinary tract. 1 These conditions are typically asymptomatic during early stages, delaying diagnosis and increasing the risk of severe outcomes such as seizures, growth retardation, and stroke.
Serum creatinine remains the primary renal biomarker due to its widespread availability and cost-effectiveness. However, its concentration is influenced by muscle mass, age, gender, physical activity, and protein intake, making it less reliable in growing children. 2
Given these limitations, interest is growing in alternative markers like cystatin C (CysC), a non-glycosylated, low-molecular-weight protein produced at a constant rate by all nucleated cells. Unlike creatinine, CysC is unaffected by muscle mass and is freely filtered by the glomeruli. Its levels rise rapidly in renal dysfunction, and it may detect glomerular filtration rate changes earlier than creatinine. 2
Establishing reference intervals (RIs) is crucial for clinical application, particularly in paediatrics where physiological variability is high. CysC RIs vary significantly with age, sex, ethnicity, and geographic region, underscoring the need for population-specific data.3,4 While adult reference values are well-established, paediatric RIs remain scarce. This study therefore aimed to establish reference intervals for serum CysC in a cohort of healthy Brazilian children.
Methodology
This cross-sectional study enrolled 134 healthy children aged 5 to 11 years from public schools in southern Brazil. Participants were included after written informed consent from parents/guardians and child assent, excluding those with renal, hepatic, infectious, inflammatory, or malignant diseases. The study was approved by the local Research Ethics Committee (number 29792120.9.0000.5346).
After an 8-h fast, venous blood was collected into anticoagulant-free tubes. Serum was separated by centrifugation (2800 r/min for 15 min), aliquoted, and stored at −80°C until analysis. CysC levels were measured using an immunoturbidimetric commercial assay (Biotécnica, Brazil) traceable to ERM-DA471/IFCC on a BS380 analyzer (Mindray, China). The assay range was 0.20–8.00 mg/L with a limit of detection of 0.15 mg/L. Within- and between-run CVs were 3.5% and 5.6%, respectively. Calibration was performed using a five-point calibrator set traceable to ERM-DA471/IFCC. Serum creatinine was measured using an IDMS-traceable modified Jaffe method on a Dimension XL 200 analyzer (Siemens, Germany). Reference intervals were determined using the 2.5th and 97.5th percentiles, per IFCC recommendations. 5
Statistical analyses used Statistica v.14. The Shapiro–Wilk test assessed normality. As variables were non-parametric, data are presented as medians and interquartile ranges, compared using the Mann–Whitney U test. The outlier detection using Tukey’s method and 90% confidence intervals (Cis) for the percentiles, calculated via bootstrap (1000 replicates). A P-value <.05 was considered significant.
Results
Reference interval results for cystatin c in the overall study population and stratified by sex.
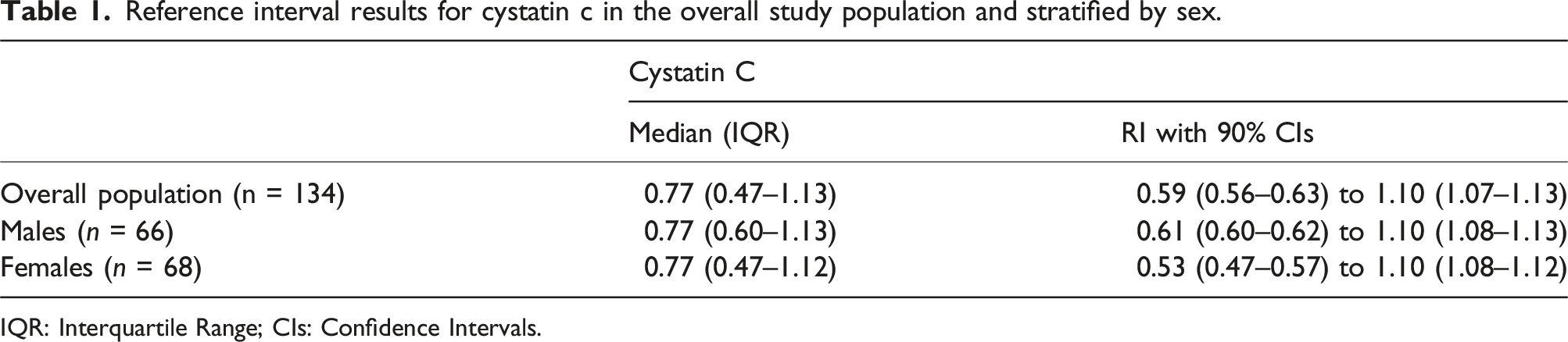
IQR: Interquartile Range; CIs: Confidence Intervals.
Discussion
This study establishes the first reference values for CysC in a Brazilian paediatric population. Our determined RI of 0.59–1.10 mg/L aligns closely with reports from other countries. For example, Kurajica et al. reported an RI of 0.61–1.08 mg/L for Croatian children aged 8–18 years. 2 The Canadian CALIPER study found similar values, with an RI of 0.62–1.11 mg/L6. Ziegelasch et al. evaluated CysC by age and sex, observing lower and upper limits of 0.65–1.08 mg/L for 6-year-old boys and 0.67–1.07 mg/L for girls. Our study found slightly lower values for females, but this may be explained by differences in population characteristics. 3 Cai et al. also documented increasing CysC levels in boys from age six, consistent with our findings that male children showed slightly higher lower limits than females. 4 Although sex-specific reference values were generated, they should be interpreted as descriptive given the limited subgroup size (n < 120).
These comparisons highlight the need for region-specific RIs, as serum biomarker concentrations are influenced by genetic, environmental, nutritional, and methodological factors. 5 International guidelines support CysC as an alternative to creatinine, but its clinical integration requires reliable, population-specific reference data, a critical need in paediatrics, where growth and developmental changes can affect biomarker interpretation. 5 Brazil’s geographic and ethnic diversity reinforces the need for population-specific reference intervals, as biomarker levels vary across distinct demographic groups. The reference ranges presented in this study converge with other available paediatric data such as the CALIPER study. 6 Since CysC is not significantly affected by variables such as muscle mass, the eGFR results obtained from the use of this variable will allow for a more reliable assessment of renal function regardless of differences in the body composition of the individuals evaluated.
We established reference intervals for serum cystatin C in healthy children from southern Brazil. Our findings support CysC as a valuable biomarker for estimating GFR in children, offering advantages over creatinine through reduced susceptibility to non-GFR influences and greater sensitivity to early renal impairment. The determined RI CysC (0.59–1.10 mg/L) aligns with international values, reinforcing its clinical applicability. As paediatric RIs are scarce in South America, our results fill an important gap, contributing to more accurate renal assessment. The use of the modified Jaffe method for creatinine determination constitutes a limitation, since it presents lower analytical specificity compared with enzymatic methods. However, this is still the most widely used method in laboratories worldwide. Further multi-centre studies with larger cohorts are necessary to validate and standardize CysC RIs globally for integration into clinical practice.
Supplemental Material
Supplemental Material - Reference intervals for serum cystatin C in Brazilian children aged 5–11 years: A short report
Supplemental Material for Reference intervals for serum cystatin C in Brazilian children aged 5–11 years: A short report by NF Jacobi, ACM Ciceri, LE de Oliveira, ISO Tioda, M Kaefer, SA de Oliveira, LB Pasqualoto, BG Rossato, RN Moresco, C Paniz, and José Antonio Mainardi de Carvalho in Annals of Clinical Biochemistry.
Footnotes
Acknowledgements
We thank the Municipal Department of Education and the City Hall of Santa Maria for their collaboration in this study. This work was supported by a scholarship from the Coordination for the Improvement of Higher Education Personnel (CAPES).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval was granted by the Research Ethics Committee of the Federal University of Santa Maria (CAEE 29792120.9.0000.5346).
Guarantor
José Antonio Mainardi de Carvalho.
Contributorship
All authors contributed substantially to the work reported. N.F.J., A.C.M.C., and L.E.O. participated in study design, data collection, recruited of patients and laboratory analyses. I.S.O.T., M.K., S.A.O. and L.B.P.,contributed to sample collection, data analysis and interpretation. B.G.R. and R.N.M. assisted in statistical analysis and manuscript preparation. C.P. and J.A.M.C. supervised the study, critically reviewed the manuscript, and approved the final version. All authors reviewed and approved the final manuscript and agree to be accountable for all aspects of the work.
Data Availability Statement
Data will be made available on request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
