Abstract
Background
To date, China has no industry standard for reference intervals of paediatric blood biochemical markers. This study aimed to evaluate changes in biochemical markers in the venous blood of healthy children aged 29 days to 12 years, derived from the UniCel DxC 800 system, and establish appropriate reference intervals.
Methods
We analysed venous blood from 1980 healthy children for 20 biochemical markers. Reference intervals were established according to the Clinical and Laboratory Standards Institute C28-A3c guideline and compared with those of adults in China.
Results
All markers except for sodium and chlorine required partitioning by age, but not by sex. The reference intervals of total protein, albumin, globulin, carbon dioxide, urea nitrogen, creatinine and uric acid consistently increased with age in children, but were always lower than those of adults. Children aged 29 days to 12 years had a single combined RI for sodium and chloride, respectively; although the reference intervals in children were similar to those of adults, their upper limits were lower. The reference intervals of direct bilirubin, alanine aminotransferase, aspartate aminotransferase, alkaline phosphatase, potassium, calcium, magnesium, and phosphorus continued to decline with age. The reference intervals of total bilirubin, indirect bilirubin, and gamma-glutamyl transpeptidase initially declined followed by a slight rebound.
Conclusions
While establishing reference intervals, most markers required partitioning by age (aged 29 days to 12 years); the partitioning scheme differed for each marker, and paediatric reference intervals differed from those for adults. It is therefore necessary to establish separate paediatric reference intervals.
Introduction
Blood biochemical markers are widely used in clinical practice to determine whether the body is healthy. They help clinicians to objectively and effectively diagnose, differentially diagnose and monitor diseases. The clinical interpretation of biochemical marker results largely depends on established reference intervals (RIs) derived from clinical studies in healthy populations. To date, many countries and regions have established local RIs for major blood biochemical markers in adults. However, establishing similar RIs for children is difficult.1,2 Researchers in some countries have established RIs for biochemical markers in local children. For instance, Canadian scholars defined the age- and sex-specific reference ranges for biochemical markers in paediatric populations, 3 and German researchers have established age- and sex-specific dynamics in 22 haematological and biochemical analyses from birth to adolescence. 4 It appears that laboratories using the same analysers and reagents can share RIs; however, its applicability needs to be verified for different ethnic groups. 5 There is currently no uniform industry standard for biochemical RIs in Chinese children. In China, clinicians' interpretations of the results of various paediatric biochemical markers mainly refer to the industry standard of ‘reference intervals for common clinical biochemistry tests in adults’ published by the competent health authorities6–8 or foreign literature. However, children are a growing and developing group, and a number of blood biochemical markers change with age; in addition, their RIs differ significantly from those of adults. 9 There are also ethnic differences in serum biomarker concentrations due to the influence of genetic and environmental factors. For instance, the median creatine kinase (CK) activity of healthy Black adult males is approximately double that of Caucasian adult males for genetic factors 10 ; environmental factors largely account for differences in vitamin A concentrations between ethnicities. 11 This may determine possible differences in the RIs of paediatric biochemical markers in different regions. 5 It is therefore necessary to establish local RIs for biochemical markers in children.
In this study, we screened venous blood samples from apparently healthy children (aged 29 days to 12 years) in southwest Fujian, China, for 20 biochemical markers according to the C28-A3c guidelines, 12 and established RIs of blood biochemical markers suitable for local children.
Materials and methods
Ethical approval
The study design was approved by the Institutional Review Board Committee of the hospital; the Institutional Review Board deemed the study exempt from review, and the study adhered to the principles of the Declaration of Helsinki. The need for informed consent was waived by the review board.
Study population
In this study, biochemical marker results of the subjects were based on retrospective data and an analysis of LIS results. Apparently healthy children were selected as study subjects. These children were diagnosed with a finger or toe deformity or underwent preparations for elective surgery for hernia at the Department of Paediatric Surgery of the Women and Children's Hospital, School of Medicine, Xiamen University. All study subjects belonged to the Xiamen, Zhangzhou, Quanzhou, or Longyan areas, and underwent detailed physical examination and disease history consultation during their hospitalization. Patients with a history of heart, liver, kidney, haematological, genetic metabolic, and allergic or autoimmune diseases, acute and/or chronic infections within one month prior to enrolment, intake of any drugs and/or nutrients within the two weeks prior to enrolment, and height and/or weight out of corresponding ranges (mean value plus or minus two standard deviations for reference population of same age and sex) were excluded. 13 A total of 1980 children aged ≥29 days to 12 years were selected between 1 January 2017 and 31 August 2019, including 285 infants (≥29 days–<1 year of age, Ag1), 531 toddlers (≥1–<3 years of age, Ag2), 552 preschool-age children (≥3–<6 years of age, Ag3) and 612 school-age children (≥6–<12 years of age, Ag4).
Sample collection and processing
Fasting was not required; venous blood was collected using a needle (model specification: flexible connection 0.7, manufactured by Weihai Weigao Blood Collection Supplies Co. Ltd, China) into PET evacuated tubes (Fuzhou Chang Geng Medical Devices Co. Ltd, China) containing separation gel/accelerator. Each evacuated tube was filled with 3 ml venous blood, gently inverted back and forth four to five times after blood collection, and centrifuged after standing for 20 min. Serum was obtained by centrifugation at 1500
Laboratory assays
Automatic biochemical analysers (UniCel DxC 800 system, Beckman Coulter Company, USA), auxiliary reagents, calibration materials, and quality control materials were used. Biochemical testing was performed by an ISO 15189 accredited laboratory in strict accordance with operating instructions. The following biochemical markers were assessed: total protein (TP), albumin (ALB), globulin (GLB), total bilirubin (TBIL), direct bilirubin (DBIL), indirect bilirubin (IBIL), alanine aminotransferase (ALT), aspartate aminotransferase (AST), gamma-glutamyl transpeptidase (GGT), alkaline phosphatase (ALP), potassium (K), sodium (Na), chloride (Cl), calcium (Ca), carbon dioxide (CO2), magnesium (Mg), phosphorus (PO4), urea nitrogen (BUN), creatinine (CREA) and uric acid (URIC). All biochemical tests underwent automated interference analysis for haemolysis, icterus and lipaemia. When haemolysis reached 1 (+)
14
and icterus reached 2 (+),
14
the specimen was deemed unacceptable for inclusion. When lipaemia reached 2 (+),
14
the specimens were centrifuged again for 10 min at 10,000
Statistical analysis and determination of RIs
All data were analysed according to the Clinical and Laboratory Standards Institute (CLSI) C28-A3c guidelines.
10
Statistical analysis was performed using SPSS 22.0 software (IBM, Armonk, New York, United States). First, we visually inspected data to identify suspected age and sex partitions using scatterplots, and removed outliers using boxplots. Second, we statistically confirmed the age and sex grouping using the Harris and Boyd method
15
when Z > Z*, grouping was considered (Note: s1 is the standard deviation of the first group; s2 is the standard deviation of the second group;
Results
A total of 1980 subjects were included in the present study. Among the 20 markers, most were measured directly by the instrument, except for GLB and IBIL, which were obtained from instrument calculation. Scatterplots of the biochemical markers are shown in Figure 1(a) to (c).
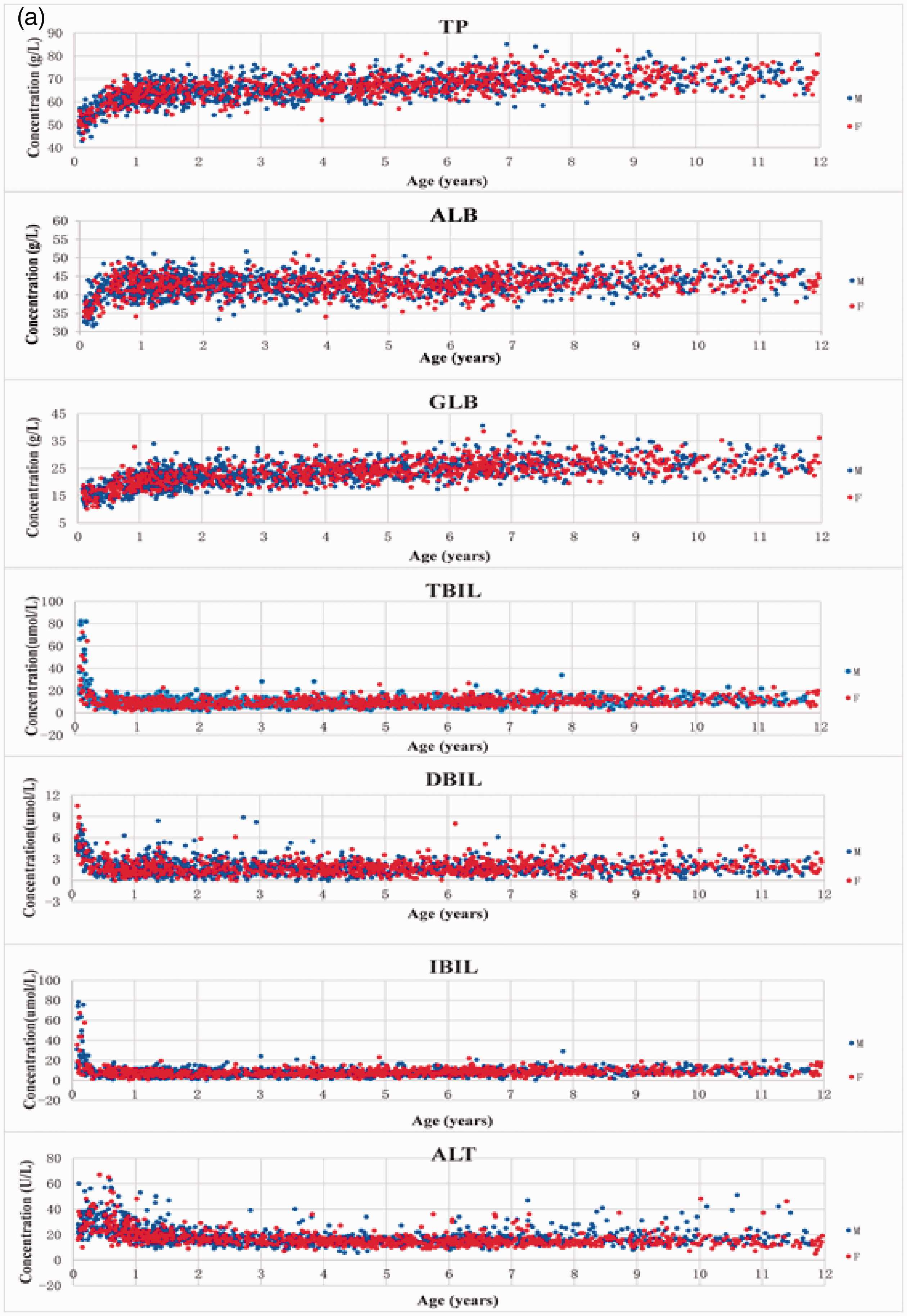
(a) Scatterplots of the biochemical markers (TP, ALB, GLB, TBIL, DBIL, IBIL, ALT). Note: M: male; F: female. (b) Scatterplots of the biochemical markers (AST, GGT, ALP, K, Na, Cl and Ca). Note: M: male; F: female. (c) Scatterplots of the biochemical markers (CO2, Mg, PO4, BUN, CREA and URIC). Note: M: male; F: female.Note. Please refer to the online version of the article to view the figure in colour.
Based on the scatterplots of the biochemical markers, we divided the children into the following groups: infants (≥29 days–< 1 year of age, Ag1 [≥29 days–< 6 months, Ag1a; ≥6 months–< 1 year, Ag1b; ≥29 days–< 3 months, Ag1c; ≥3 months–< 1 year, Ag1d]); toddlers (≥1–<3 years of age, Ag2); preschool-age children (≥3–<6 years of age, Ag3) and school-age children (≥6–<12 years, Ag4). Age and sex partitions of each marker were determined using the Harris and Boyd method. See Supplemental Tables 2 and 3 for details.
As depicted in Supplemental Table 2, all 20 markers did not need to be grouped by sex. For male–female comparisons in each age group, the Z value was less than the Z* value. As presented in Supplemental Table 3, most markers required grouping by age except for Na and Cl, as the markers indicated that the Z value was greater than the Z* value among different age groups.
We calculated the RIs for all partitions and then calculated 90% CIs for the upper and lower limits of each RI with the weighted data. The RIs of the 20 biochemical markers are presented in Table 1.
Age-specific reference intervals for 20 biochemical markers.
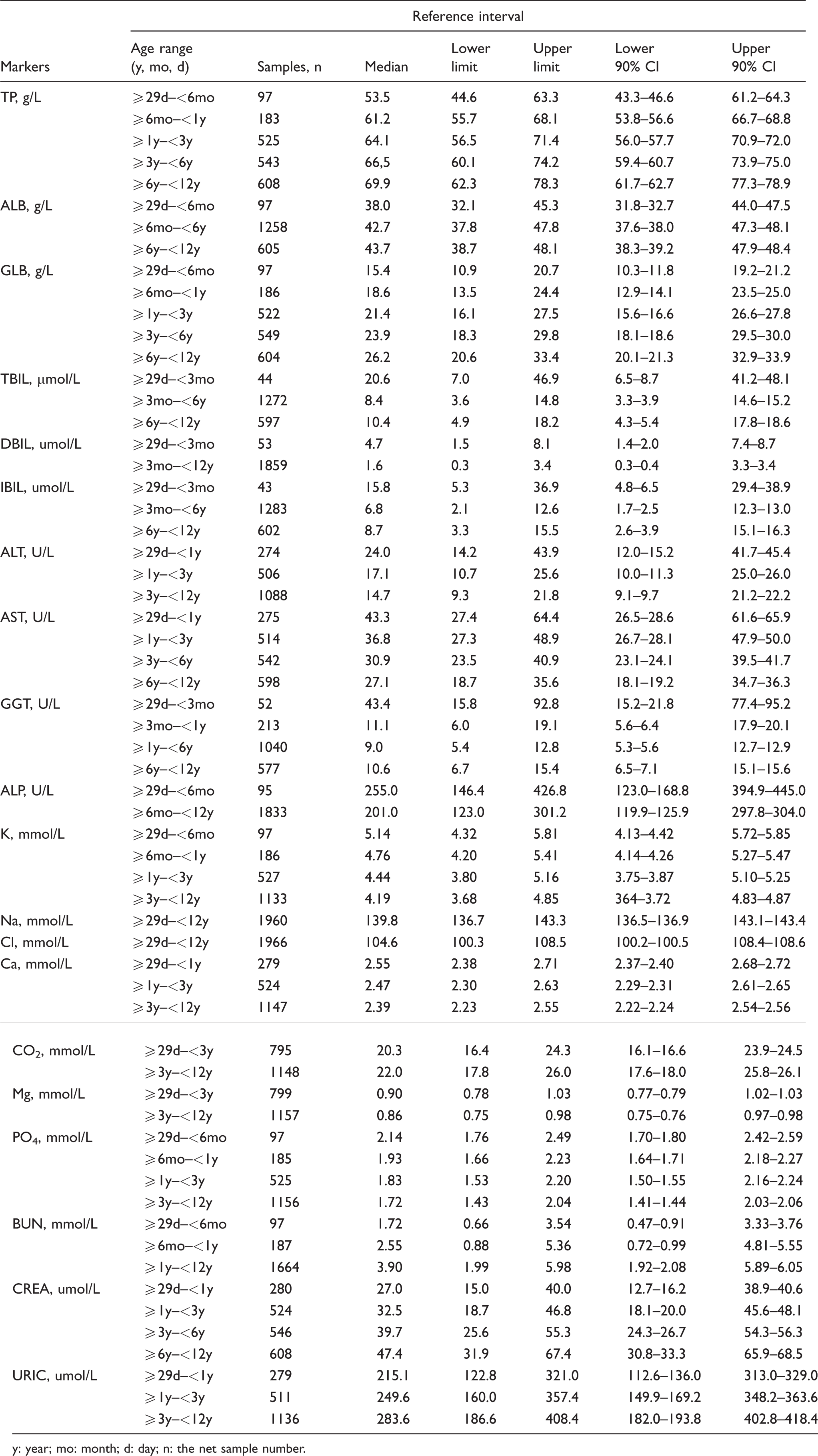
y: year; mo: month; d: day; n: the net sample number.
Discussion
The RIs of various biochemical markers for children have been confirmed to be significantly different from those for adults. 9 Establishing RIs suitable for local children is currently an urgent task for medical laboratories in many countries. Obtaining a large sample size of healthy children for blood collection is a considerable challenge for research institutions. Under normal circumstances, fasting venous blood samples are usually obtained while establishing the RIs of blood markers in a specific population. The duration of fasting is not uniform, but is generally advised for at least 8 h. 17 The implementation of fasting for 8 h is not common among children, and in particular, among newborns and infants. Local children are usually examined for the 20 biochemical markers by obtaining blood samples at random. Therefore, this study retrospectively analysed the results of 20 biochemical markers in random venous blood samples in apparently healthy children in southwest Fujian province, China, and established RIs for the markers suitable for local paediatric populations. In the existing literature, large blood sample sizes for establishing RIs are mostly achieved by recruiting healthy volunteers. Since this is very difficult to obtain in paediatric populations, there are few areas that have established independent RIs for children. In this study, blood samples were taken from children undergoing elective surgery, who had to undergo various pre-surgical blood tests. They only suffered from finger or toe deformities, or hernia. Organs related to the biochemical markers developed normally, and they were selected according to the strict exclusion criteria during inclusion in the study. This provides a new concept and choice for selecting research objects to establish RIs.
Among the 20 biochemical markers, TP, ALB, GLB, TBIL, DBIL, IBIL, ALT, AST, GGT and ALP are commonly used clinical markers, that reflect human liver function, and K, Na, Ca, Mg, Cl, CO2, PO4 are electrolytes in body fluids, that maintain the osmotic pressure and normal distribution of body fluids. BUN, CREA and URIC are commonly used clinical indicators for evaluating kidney function. In this study, we observed that each marker measured in the same age range (aged 29 days to 12 years) did not need to be grouped by sex, but all measured markers except for Na and Cl required grouping by age. The RIs of TP, ALB, GLB, CO2, BUN, CREA and URIC increase with age and continue to rise, but are always lower than those of adults. This may be attributed to the fact that albumin is synthesized by the liver, globulin is synthesized by the human mononuclear phagocytic system, 18 CO2 is determined by the respiratory and metabolic factors, URIC is mainly generated by the human liver, bone marrow and muscle, among others, BUN is the product of protein metabolism and CREA is the product of muscle metabolism in the human body. In summary, children are smaller and thus have less muscle mass, and smaller kidneys and livers than adults; therefore, the levels of production of the above markers were lower than those of adults. The variation trends of these markers are consistent with the findings of the Canadian study by Colantonio et al. 2 (in the same age group), but the RIs established did not overlap completely. Children aged 29 days to 12 years had a single combined RI for Na (136.7–143.3 mmol/L) and Cl (100.3–108.5 mmol/L), and the RIs were nearly similar to those of adults (137–147 mmol/L and 99–110 mmol/L, respectively); however, the upper limit of the RIs was lower. The findings of this study were consistent with those found by Adeli et al. 9 in Canada. However, in that study, the RIs for children and adults nearly overlapped (135–143 mmol/L vs. 136–143 mmol/L and 100–107 mmol/L vs. 101–108 mmol/L, respectively), while in this study, their upper limits varied; the reason for the difference needs further investigation. The RIs of DBIL, ALT, AST, ALP, K, Ca, Mg and PO4 continued to decline with age, and the RIs of TBIL, IBIL and GGT initially declined, followed by a slight rebound. These phenomena were related to the increasing ability to metabolize and regulate these markers following the maturity of various organs with increasing age. They were also related to various changes and fluctuations in the growth and development of children. The variation trends of the RIs of AST, Ca, Mg and PO4 were consistent with those described by Zierk et al. 4 from Germany and Adeli et al. 9 from Canada. The variation trends of the RIs of ALT and GGT were different from those observed by Zierk et al. 4 in a German cohort but were consistent with those described by Adeli et al. 9 from Canada. In our study, the reference value of ALT was higher in the age group of 29 days to 1 year, and the RI was wide. In the age group of 1 to 12 years, the reference value decreased with age, and the RI narrowed. In the age group of 29 days to 6 years, the reference value of GGT decreased and the RI narrowed with the increase of age. In the age group of 6 to 12 years, the reference value increased slightly, and the RI slightly widened. In the study by Zierk et al., 4 the reference value of ALT decreased in the age range of 0 to 5 years, and the RI narrowed with increasing age; in the age range of 5 to 12 years, the reference value increased slightly, and the RI widened slightly with increasing age. In the age range of 0 to 12 years, the reference value of GGT slowly increases, and the RI slowly shifts upward with increasing age. The variation trend of the RI of ALP in this study was consistent with those observed by Zierk et al. 4 and Adeli et al. 9 in the age group of less than 10 years; however, the RI was not widened and not slightly higher in those aged 10–12 years. The differences in the starting time nodes of the growth and development stages in children may be related to race, nutritional status and the living environment. The variation trends of the RIs of bilirubin in this study were similar to those observed by Colantonio et al. 2 in Canada; however, the age grouping scheme was obviously different. The key grouping time node in this study was 3 months, while those of the study by Colantonio et al. were 15 days and 1 year. Significant changes and fluctuations in the growth and development of children highlight the importance of determining age-specific paediatric RIs.
In summary, our study established RIs of TP, ALB, GLB, TBIL, DBIL, IBIL, ALT, AST, GGT, ALP, K, Na, Cl, Ca, CO2, Mg, PO4, BUN, CREA and URIC for children in southwest Fujian, China, and is the first to perform this investigation in China. The obtained data confirmed that the RIs of the 20 selected markers for children (aged 29 days to 12 years) deviated from the RIs defined for adults. Even among the children in this study, the RIs of most markers differed across various age groups. Therefore, it is necessary to establish separate RIs for children. There was a limitation in our study, in that the sample sizes of the neonatal and adolescent age groups were relatively small, and thus the corresponding RIs could not be established. Differences caused by ethnicity and differences in methodology, detection systems, and reagents may affect the applicability of selected RIs.3,5,19
Conclusions
While establishing RIs, most markers required partitioning by age (aged 29 days to 12 years); the partitioning scheme differed for each marker, and paediatric RIs differed from those for adults. It is therefore necessary to establish separate paediatric RIs. We recommend that qualified and capable laboratories establish suitable individual RIs. Laboratories that cannot establish their own RIs should select those established by other laboratories and use these after verifying their suitability to their own conditions.
Supplemental Material
sj-pdf-1-acb-10.1177_0004563220965386 - Supplemental material for Establishing reference intervals of 20 biochemical markers for children in Southwestern Fujian, China based on the UniCel DxC 800 system
Supplemental material, sj-pdf-1-acb-10.1177_0004563220965386 for Establishing reference intervals of 20 biochemical markers for children in Southwestern Fujian, China based on the UniCel DxC 800 system by Jiming Li, Huifen Zhang, Haichen Chen, Yuebin Gan, Juan Li and Huibin Huang in Annals of Clinical Biochemistry
Supplemental Material
sj-pdf-2-acb-10.1177_0004563220965386 - Supplemental material for Establishing reference intervals of 20 biochemical markers for children in Southwestern Fujian, China based on the UniCel DxC 800 system
Supplemental material, sj-pdf-2-acb-10.1177_0004563220965386 for Establishing reference intervals of 20 biochemical markers for children in Southwestern Fujian, China based on the UniCel DxC 800 system by Jiming Li, Huifen Zhang, Haichen Chen, Yuebin Gan, Juan Li and Huibin Huang in Annals of Clinical Biochemistry
Supplemental Material
sj-pdf-3-acb-10.1177_0004563220965386 - Supplemental material for Establishing reference intervals of 20 biochemical markers for children in Southwestern Fujian, China based on the UniCel DxC 800 system
Supplemental material, sj-pdf-3-acb-10.1177_0004563220965386 for Establishing reference intervals of 20 biochemical markers for children in Southwestern Fujian, China based on the UniCel DxC 800 system by Jiming Li, Huifen Zhang, Haichen Chen, Yuebin Gan, Juan Li and Huibin Huang in Annals of Clinical Biochemistry
Footnotes
Acknowledgements
We would like to thank the paediatric surgeons who offered their help for screening the study subjects; we would also like to thank all the participants involved in the study and the staff of the Medical Laboratory, Women and Children's Hospital, School of Medicine, Xiamen University.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The study design was approved by the Institutional Review Board Committee of the hospital. (REC number: KY-2020–048). The Institutional Review Board deemed the study exempt from review, and the study adhered to the principles of the Declaration of Helsinki. The need for informed consent was waived by the review board.
Guarantor
JL.
Contributorship
HZ and JL researched the literature and conceived the study. HC and JL were involved in protocol development, obtaining ethical approval, and data analysis. JL wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
