Abstract
Proprotein Convertase Subtilisin-Kexin type 9 (PCSK9) is a key regulator of lipid metabolism, binding to the low-density lipoprotein receptor (LDLR) on the cell surface and preventing its recycling, thereby reducing clearance of LDL cholesterol (LDLc) from the circulation. For this reason, it constitutes an alternative therapeutic target for the control of hypercholesterolaemia, with the development of monoclonal antibodies against PCSK9 occurring within 12 years of the protein’s discovery. Recent research has also suggested an inflammatory role played by PCSK9, with elevated plasma levels identified in critical illnesses such as sepsis and Acute Respiratory Distress Syndrome, where PCSK9 is thought to reduce bacterial endotoxin clearance and may exacerbate inflammation. Further work is required in order to clarify the exact role played by PCSK9 in extra-hepatic tissues, and the potential benefits of its pharmacological inhibition.
Introduction
The discovery of the Proprotein Convertase Subtilisin-Kexin type 9 (PCSK9) enzyme in 2003 and the subsequent development of inhibitory drugs have revolutionised the understanding of lipid metabolism and the management of hypercholesterolaemia and cardiovascular risk. 1 Gain of function (GOF) mutations in the PCSK9 gene account for around 1% of cases of the genetic lipid disorder familial hypercholesterolaemia, leading to the demonstration of the enzyme’s role in regulating low-density lipoprotein cholesterol (LDLc). 2 Subsequently, loss of function (LOF) mutations have been shown to be more common than GOF in the healthy population and to have no association with adverse effects3–6 prompting the development of PCSK9 inhibition strategies as a novel opportunity for safe lowering of LDLc. Monoclonal antibodies (mAbs) targeting PCSK9 activity were approved within 12 years of the protein’s discovery. Evidence of LDLc lowering and morbidity/mortality benefits cemented the role of these drugs in the cardiovascular arena.7–10 A small interfering ribonucleic acid called inclisiran is also available to inhibit PCSK9 in lipid management. 11
Highly expressed in the liver, the role of PCSK9 in regulating lipids is both intra- and extracellular; however, the LDLc lowering effect of PCSK9 inhibition is thought to occur primarily through extracellular binding to the low-density lipoprotein receptor (LDLR).
12
The LDLR was demonstrated in 1976 to be the primary means of LDLc removal from circulation.
13
Circulating LDLc binds to the cell surface LDLR before the complex is internalised within coated vesicles and joins the endosomal compartment.4,10,13 On descent towards merging with the lysosome, the lowering pH induces dissociation of the LDLR from its target, allowing it to be recycled back to the cell surface, and LDLc to undergo further processing (Figure 1). In the 20-h lifespan of the LDLR, this recycling process occurs every 10−15 min, allowing multiple rounds of LDLc degradation.
14
In contrast, when PCSK9 binds to the LDLR, its structure changes from a closed to an open conformation, targeting it to the lysosomal compartment for degradation along with LDLc (Figure 1).
15
The affinity of PCSK9 for the LDLR is dependent on calcium concentration and considerably increased at a lower pH, suggesting that PCSK9 chaperone activity impedes the LDLR from dissociating from its target in the endosomal compartment.15–17 The subsequent reduction in LDLR expression on the cell surface leads to an increase in plasma levels of LDLc and, if persistent, to increased risk of cardiovascular events. While primarily described in hepatocytes, PCSK9 has also been shown to degrade LDLR levels in other cell types such as vascular endothelial and smooth muscle cells, pancreatic islet cells, and macrophages.18–21 The role of PCSK9 in lipid metabolism and LDLR degradation. Circulating LDLc binds to the cell surface LDLR in hepatocytes, and the complex is internalised within coated vesicles. On descent towards merging with the lysosome, the lowering pH induces dissociation of the LDLR from its target, allowing it to be recycled back to the cell surface while LDLc is degraded in the lysosome. In contrast, when the CAT domain of PCSK9 binds to the LDLR EGFR-A domain, this dissociation is prevented, targeting the entire complex to the lysosomal compartment for degradation along with LDLc. Intracellularly, nascent PCSK9 can also bind to the LDLR in the trans-Golgi network and target it towards the lysosome for degradation. Pre-pro PCSK9 undergoes autocatalytic cleavage in the endoplasmic reticulum, following which the prodomain noncovalently associates with CAT domain, masking catalytic activity. After maturation in the Golgi apparatus, PCSK9 is secreted into circulation, where it can act to degrade cell surface LDLR expression or undergo a second proteolytic cleavage by Furin. Created in BioRender.com.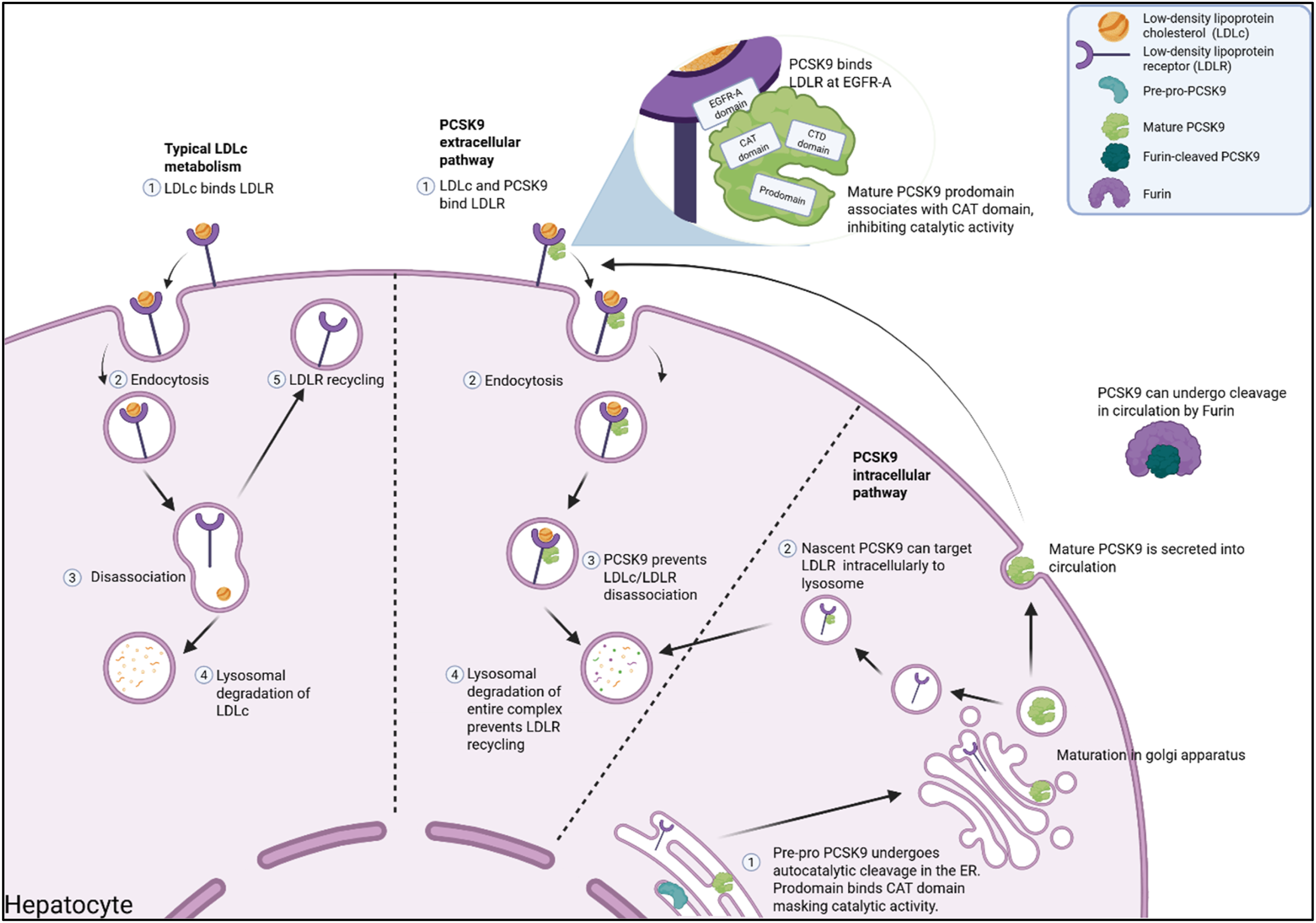
PCSK9 structure and function
Found on chromosome 1p32.3 and 4C7, the PCSK9 gene is well conserved amongst higher mammals.1,22 The protein is encoded by 15 exons and is processed from its 74 kDa precursor to the mature, 62 kDa form in under two hours.16,23 Its structure consists of a signal peptide and prodomain, a catalytic (CAT) domain well conserved among proprotein convertases, and a C-terminal domain (CTD) that is unique within the family. 16 The maturation and secretion of PCSK9 involves three stages. First, the signal peptide must be cleaved, following which PCSK9 undergoes autocleavage through the CAT domain at the FAQ152/SIPK site in the endoplasmic reticulum.1,22,24 This self-cleavage is thought to also involve the CTD and is essential for PCSK9 maturation and secretion. Following autocleavage, the 14 kDa prodomain noncovalently associates with the CAT domain, masking catalytic activity.22,25 The final step in PCSK9 secretion is its transportation into circulation via the Golgi complex (Figure 1).
Mature, secreted PCSK9 can undergo a second proteolytic cleavage by Furin, resulting in the dissociation of the prodomain and segment 153−218 (Figure 1). 26 This truncated (55 kDa), ‘inactive’ form of PCSK9 can only be formed in the circulation, and recent mass spectrometry analysis has revealed that up to 40% of circulating PCSK9 is Furin-cleaved.26–28 Due to its lack of a prodomain, once internalised, Furin-cleaved PCSK9 cannot be secreted and has a shorter half-life compared to the intact form. 29 The ability of Furin-cleaved PCSK9 to lower cell surface LDLR levels is unclear. Furin-cleaved PCSK9 is reported by some to be less active and have approximately half of the affinity for the LDLR as that of the intact, mature form.26,28,30 Unusually for an enzyme, the catalytic activity of PCSK9 is not required for LDLR degradation, rationalising the existence of GOF variants.19,25
The CAT subunit of PCSK9 can bind the LDLR at its Epidermal Growth Factor Receptor A (EGFR-A) domain, remaining bound in the endosomal compartment and facilitating LDLR degradation.12,22 The positively charged CTD of PCSK9 also participates in this process, interacting with the negatively charged ligand binding repeats of the LDLR intracellularly to traffic the complex to the lysosome. 31 Recent experimental evidence has indicated the possibility that a third partner is required for efficient LDLR degradation, binding the CTD and allowing the complex to be targeted to the late endosome. 19 Not all of the essential partners required for PCSK9-LDLR trafficking have been identified; however, cyclase associated protein 1 (CAP-1) has been implicated as an important positive regulator of PCSK9 function, mediating its endocytosis and LDLR degradation capabilities. 32 PCSK9 can also act to facilitate LDLR expression using the same CAT subunit/EGFR-A domain interaction. Pre-pro-PCSK9 will bind the LDLR precursor in the endoplasmic reticulum, facilitating its transport to the Golgi apparatus for maturation. This interaction is mutually beneficial, allowing pre-pro-PCSK9 to undergo autocleavage for maturation and secretion. The function of this process remains unknown, as PCSK9 chaperone activity is not essential to LDLR maturation and occurs less frequently than PCSK9-mediated degradation. 33 While the extracellular pathway is the primary route of LDLR degradation by PCSK9, this process is also implemented intracellularly.34,35 Mechanistic studies have identified a role played by nascent PCSK9, where binding to the LDLR will target it from the trans-Golgi network towards the lysosome for degradation (Figure 1). 34 Despite being a minor route for LDLR degradation, this intracellular pathway must be considered when it comes to therapeutic inhibition of PCSK9.
PCSK9 inhibitors
Given the observation that LOF mutations in PCSK9 can confer benefits such as LDLc reduction and significantly decreased atherosclerosis development, efforts were turned towards the development of agents to therapeutically inhibit PCSK9 function and/or expression. 5 The most widely used drugs in this area are monoclonal antibodies specific for the PCSK9 catalytic subunit. These prevent its interaction with the EGFR-A domain of the LDLR.36,37 Two human mAbs, alirocumab and evolocumab, have demonstrated a consistent and lasting ability to lower plasma LDLc and PCSK9 (by up to 50−70% and 100%, respectively), and are currently licensed for use in the United States and European Union.38–40 Long-term trials (alirocumab: 78 weeks and evolocumab: 52 weeks) have demonstrated that both mAbs are safe and well-tolerated, with minor side effects of oropharyngeal pain, nasopharyngitis, rhinorrhoea, and injection site reactions occurring with a frequency of between 1:10 and 1:100.38,39,41,42 The morbidity and mortality benefits of these mAbs are also widely reported in the literature. Long-term follow up of atherosclerosis patients receiving evolocumab has demonstrated a 15−20% reduction in the likelihood of major cardiovascular events and 23% decreased risk of cardiac death in comparison to those on placebo. 9 Given the life-long nature of these therapeutics, their relatively frequent administration, and high production costs however, funding restrictions have limited their widespread adoption across the United Kingdom, and they remain largely within the realm of specialist lipid and cardiology clinics. 43
Due to the intracellular pathway of PCSK9, approaches to lowering its mRNA expression have also been investigated.9,10 Small interfering RNAs (siRNAs) such as Inclisiran, administered twice a year via lipid nanoparticles, have exhibited success in humans. 44 Inclisiran has been demonstrated to lower circulating PCSK9 by 70% and plasma LDLc levels by 40% after one dose. 45 Direct comparisons of siRNA and mAb therapy for PCSK9 inhibition in humans has not yet been performed; however, around 30% of circulating PCSK9 remains after using the siRNA approach.10,45
The lack of harm observed in individuals with PCSK9 LOF variants has prompted the consideration of more permanent solutions for PCSK9 inhibition.4–6 Peptide and virus-like particle-based vaccines generating PCSK9-specific antibodies functional for up to one year have been demonstrated to lower plasma LDLc by up to 50% and reduce biochemical measures of inflammation in the plasma of atherosclerotic mice. 46 Vaccination strategies such as these, perhaps administered annually, could constitute a more affordable approach than mAbs, with the added potential to prevent hypercholesterolaemia and atheroma development altogether in younger patients. 6 Permanent PCSK9 inhibition has also been investigated using CRISPR (Clustered Regularly Interspaced Short Palindromic Repeats)/Cas9 genome editing, demonstrating efficient plasma cholesterol reduction of 35−40% and no off-target mutagenesis; however, only short-term efficacy and safety have been observed thus far in murine models.47,48 The possibility of so far unidentified long-term adverse consequences to permanent inhibition of PCSK9 must also be considered, however. It has been suggested that a consistent deficiency in PCSK9 could potentially reduce the ability of the liver to regenerate and could enhance viral uptake and severity of infections such as hepatitis C.49–51
Lipids and inflammation
Lipid metabolism and inflammatory pathways are heavily interlinked.52–54 Cholesterol and its lipoprotein carriers have immunomodulatory capabilities, including the binding and neutralisation of endotoxin to prevent activation of toll-like receptors (TLRs). 55 Reduced cholesterol concentration has also been demonstrated to influence natural killer cell function, reducing their cytotoxicity. 56 Key receptors regulating the immune response such as T- and B-cell receptors (T/BCRs) and TLRs are localised within lipid rafts, with higher cholesterol concentration promoting raft TLR4/9 expression and immune synapse formation between TCRs and immune complexes.57,58 High-density lipoprotein (HDL) is capable of reducing vascular pro-inflammatory mechanisms and downregulating vascular and intracellular adhesion molecules such as VCAM-1 and ICAM-1 in animal models, through suppression of NF-KB activity. 59 Transcription factors involved in lipid metabolism can also have an impact on inflammatory gene expression through interaction with transcription factors controlling inflammatory pathways. Peroxisome proliferator-activated receptors, Liver X receptors, and Farnesoid X receptors can all inhibit inflammatory responses and macrophage activation through a combination of physical interaction with transcription factors such as NF-KB or indirect binding of transcriptional corepressors (transrepression).60–62 Increasing interest in recent years has promoted research into the potential of modulating lipid transport in critical illnesses such as sepsis, where reductions in total cholesterol, LDLc and HDL are well recognised, often correlating with mortality risk.63–68 In Acute Respiratory Distress Syndrome (ARDS), disproportionate levels of pro- and anti-inflammatory lipid mediators have been suggested by both pre-clinical and clinical research to contribute to disease severity and mortality, with significantly lower plasma lipid concentrations, particularly polyunsaturated fatty acids, present in a so-called ‘hyperinflammatory phenotype’. 69 The relationship between the plasma lipid profile and inflammation in ARDS is complex, and further work investigating the balance between pro- and anti-inflammatory lipid concentrations in the condition is warranted.69,70
The pro-inflammatory effect of lipids is also well documented, particularly through diet induced ‘metabolic inflammation’ reflected in higher circulating inflammatory markers such as high sensitivity C-reactive protein (hsCRP) in obese individuals and the secretion of pro-inflammatory cytokines by macrophages upon the expansion of adipose tissue.71,72 The uptake of modified lipoproteins by resident liver macrophages (Kupffer cells) can induce hepatic inflammation, whereas similar mechanisms by infiltrating monocytes in vessel walls can amplify signalling pathways such as NF-KB or c-Jun N-terminal kinase, and shift M2 macrophages towards the inflammatory M1 phenotype.73,74 The accumulation of cholesterol crystals in the vessel wall in hypercholesterolaemia and subsequent uptake by macrophages has also been demonstrated to activate the NLRP3 inflammasome, leading to pro-IL-1β cleavage by caspase 1 and subsequent IL-1β release. 75 Hyperlipidaemia is a crucial risk factor for the development of atherosclerosis, which is increasingly being considered an inflammatory disease due to the impact of inflammation on plaque development. Carried by atherogenic lipoproteins, lipids enter the arterial wall and accumulate, while oxidised LDL (oxLDL) activates endothelial cells to produce adhesion molecules that attract inflammatory cells such as neutrophils.76,77 The uptake of oxLDL initiates the conversion of macrophages to foam cells, which locally amplify inflammatory signalling and contribute to ‘foamy plaque’ formation. 78 In viral infection, the LDLR has been proposed as a route of entry for viruses such as Hepatitis C, whereas infection with dengue virus promotes cellular cholesterol synthesis for enhanced viral packaging and suppression of the antiviral type 1 interferon response.50,79
Statins and inflammation
Statins are widely prescribed for the management of hypercholesterolaemia and cardiovascular risk. They act through competitive inhibition of the enzyme hydroxymethylglutaryl-CoA (HMG-CoA) reductase, thus downregulating hepatic cholesterol biosynthesis. Expression of LDLRs on hepatocyte surfaces increases as a result of lowered intracellular cholesterol concentration, promoting the clearance of circulating LDLc and, in time, lowering the risk of subsequent cardiovascular events. 80 Statins also demonstrate effects outside of LDLc reduction, including anti-inflammatory, anti-proliferative, and anti-thrombotic abilities. Statin use illustrates the potential for therapeutic manipulation of lipid metabolism for immunomodulation.81–83
In pulmonary pathologies particularly, statins have exhibited capabilities such as modulation of the endothelium, promotion of endothelial repair following inflammation, and attenuation of vascular leakage.84,85 Several potential mechanisms have been proposed for the anti-inflammatory properties exhibited by statins in respiratory disease, such as reduced inflammatory cytokine release following disturbances in prenylation. The activation of transcription factors NF-KB, AP-1, and HIF-1α are all inhibited by statins, preventing the expression of pro-inflammatory cytokines such as TNF-α, IL-6, and IL-8. 86 In addition, experimental evidence has demonstrated the ability of statins to interfere with leucocyte infiltration to target organs and T-cell antigen presentation, suppressing Th17 cell production of IL-17.87,88 The cholesterol lowering effect of statins can also have an impact on their anti-inflammatory properties due to the resultant instability of lipid rafts, which can act as platforms for the interaction of immune cells by aggregating key receptors upon ligand binding.55–58,89,90
The therapeutic potential demonstrated by statins for the treatment of critical illness has been investigated through clinical studies such as the hydroxymethylglutaryl-CoA reductase inhibition with simvastatin in acute lung injury to reduce pulmonary dysfunction (HARP) trial. 91 Promising initial results prompted larger, multicentre, randomised, placebo-controlled trials (such as HARP-2 and the Stains for Acutely Injured Lungs from Sepsis or SAILS trial) which failed to demonstrate an improvement in clinical outcomes.92,93 Secondary analyses of the HARP-2 clinical trial, however, highlighted improved survival with simvastatin of patients belonging to the hyperinflammatory phenotype of ARDS. 94 Differential clinical outcomes and responses to ARDS treatment strategies such as fluid management have also been observed between ARDS phenotypes, suggesting that inflammatory phenotyping of critically ill patients could identify a patient cohort whose biology more closely reflects pre-clinical study observations.94,95 The precise mechanism of benefit for statins in the treatment of critical illness remains unclear, especially as their reduction in cholesterol biosynthesis promotes the expression of PCSK9 through the sterol regulatory element binding protein (SREBP). 96 A meta-analysis of 15 statin clinical trials has found that statin therapy increased mean plasma PCSK9 levels by 41 ng/mL.96,97 Concerns that PCSK9 activity could cause standard doses of statins to lose effectiveness have also been raised, with hepatocyte cells under hypoxia demonstrating reduced responses to simvastatin and mevastatin when supplemented with PCSK9. 79 Combining statin therapy with PCSK9 inhibitors is commonplace in lipid management but could also have substantial benefits for the treatment of dysregulated inflammation, with PCSK9 knockout mice demonstrating increased sensitivity to statins in experimental work. 98 These findings highlight the complexity of the inflammatory role of PCSK9.
PCSK9 and inflammation
Outside of the lipid regulatory role of PCSK9, experimental and clinical evidence has recently identified an inflammatory role of the protein. PCSK9 expression has been demonstrated to directly impact pro-inflammatory signalling pathways, elevating the production of pro-inflammatory cytokines.99,100 In addition, levels of the protein in critically ill patient populations have been significantly associated with clinical outcomes such as mortality and ICU-free days.99–102 This identification of PCSK9 as a potential inflammatory mediator has prompted investigation into its role in the pathogenesis of critical illnesses such as sepsis and ARDS, and the potential application of PCSK9 inhibiting therapies within their care.
PCSK9 inhibition has been demonstrated in murine models to promote a broad attenuation of the inflammatory cascade, with its inhibition downregulating mononuclear cell expression of IL-6, TNF-α, and IL-1β mRNA and serum levels of CXCL-1, -10, -13, and complement factor C5a in APOE*3Leiden.CETP mice.103–105 PCSK9 null mice have exhibited a diminished inflammatory response to lipopolysaccharide (LPS) administration, while PCSK9 overexpression in human macrophages decreases concentrations of anti-inflammatory markers such as Arg-1 and IL-10. 106 Incubation of primary human macrophages and macrophage-like cell lines (such as THP-1s) with physiologically relevant increasing concentrations of recombinant PCSK9 (0.25−2.5 µg/ml) has been demonstrated to increase gene expression of the pro-inflammatory cytokines TNF-α and IL-1β in a dose dependent manner.104,106 PCSK9 elevation has also been demonstrated to enhance monocyte chemotaxis and circulating monocyte recruitment from the bone marrow.107,108 Recent work has identified a potential role of the Janus kinase (JAK) pathway in PCSK9-mediated inflammation, with JAK inhibition reducing TNF-α production by THP-1 macrophages incubated with the protein, although the mechanism through which PCSK9 interacts with the JAK/STAT pathway requires further investigation. 106
Research into the mechanisms through which PCSK9 mediates inflammation has demonstrated the involvement of the TLR4/NF-KB signalling pathway (Figure 2).104,106,109–111 The expression of many pro-inflammatory cytokines is under the control of this pathway, with TLR4 challenge by pathogen- and damage-associated molecular patterns inducing downstream signalling to allow nuclear translocation of NF-KB subunits and the upregulation of inflammatory markers such as TNF-α and IL-1β.104,106 In unstimulated cells, NF-KB p50/p65 subunits are bound their inhibitor (inhibitor of KB or IKB) in the cytoplasm. Upon inflammatory stimulation, successive downstream signalling events converge on the activation of the IKB kinase complex (IKK), allowing nuclear localisation of NF-KB subunits.
112
PCSK9 stimulation of primary human macrophages and THP-1 cells results in nuclear translocation of p65 and induction of IL-1β, IL-6, TNF-α, CXCL2, and MCP1 mRNA.
106
PCSK9 binding of TLR4 has been demonstrated experimentally to facilitate NF-KB mediated inflammation; however, the protein has also been shown to activate the NF-KB pathway through other routes. CAP-1, a binding partner of PCSK9, has recently been shown in primary human macrophages to not only mediate LDLR degradation by the protein but also to contribute to NF-KB activation through recruitment and phosphorylation of downstream molecules such as PKCδ and Syk, activation of the PI3/Akt pathway, and IKB degradation (Figure 2).
113
Furthermore, the binding affinity of PCSK9 is stronger for CAP-1 than for TLR4, and recent work has also suggested that PCSK9/TLR4 interaction is dependent on CAP-1 presence. In CAP-1 deficient THP-1 cells, PCSK9/TLR4 interaction and inflammatory cytokine secretion was significantly diminished. Inflammatory cytokine production remained unchanged in TLR4 deficient cells, highlighting the crucial role played by CAP-1 in PCSK9-mediated inflammation.
113
LDLR presence has also been observed to play a role in the pro-inflammatory effect of PCSK9, markedly increasing PCSK9 induced TNF-α expression; however, a significant effect was also demonstrated in macrophages derived from LDLR null mice.
106
These data suggest that other transmembrane protein receptors such as the very low-density lipoprotein receptor (VLDLR), Apolipoprotein E receptor 2 (ApoER2), and Leucine Rich Pentatricopeptide Repeat (LRP-130) could participate in the pro-inflammatory response to PCSK9.106,111,114–116 PCSK9-mediated inflammatory signalling in macrophages. Binding of PCSK9 to TLR4 recruits adaptor proteins such as MyD88, initiating phosphorylation and degradation of IKBα. Phosphorylation and nuclear translocation of NF-KB subunits p50/p65 initiates the expression of pro-inflammatory cytokines such as TNF-α, IL-6, and IL-8. CAP-1, a binding partner of PCSK9, recruits and phosphorylates downstream signalling molecules PKCδ and Syk, which activate the PI3/Akt pathway. Akt initiates NF-KB signalling through IKBα phosphorylation, also phosphorylating SREBP. Phosphorylated SREBP can activate the NF-KB pathway through IKBα degradation and induce PCSK9 expression. Increased PCSK9 expression and the resultant degradation of cell surface LDLRs can contribute to reduced clearance of bacterial endotoxins such as LPS, exacerbating inflammation. Created in BioRender.com.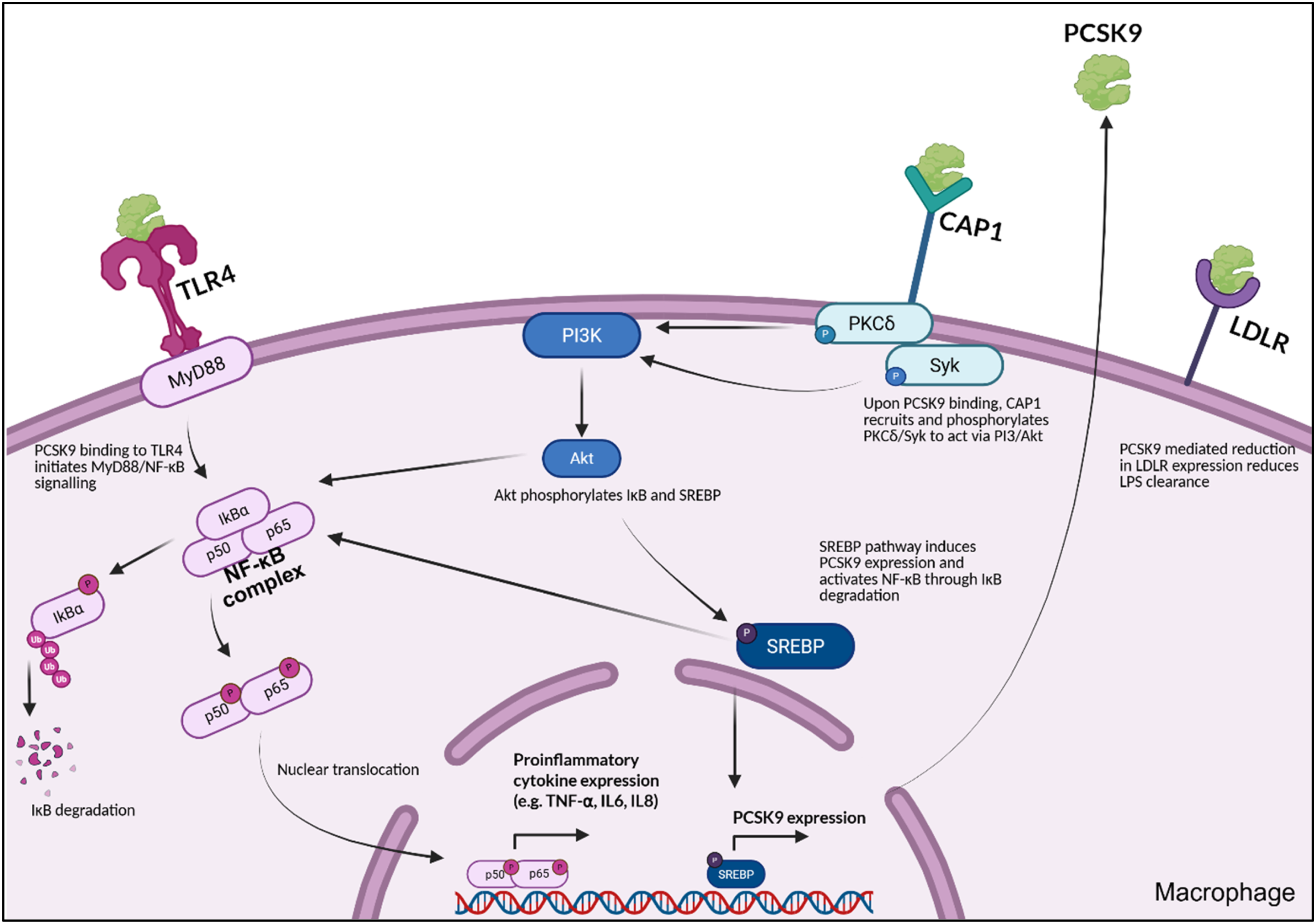
PCSK9 is one of 33 genes regulated by the SREBP family, with lipid loading reducing its expression in inflammatory cells such as macrophages.97,111,117 As cellular cholesterol concentration decreases, SREBP-2 activation results in PCSK9 production and cholesterol biosynthesis, a process reported to be involved in the exocytosis of viruses such as SARS-CoV-2. 111 Interaction between the SREBP-2 and NF- KB pathways (Figure 2) has been proposed to result in inflammation as well as vascular and organ damage, with PCSK9 levels in the peripheral blood mononuclear cells of Coronavirus Disease 2019 (COVID-19) patients correlating with severity of disease.111,118,119 PCSK9 supplementation has also been demonstrated to impact the effectiveness of standard doses of therapies such as statins in hypoxic conditions, reducing the potency of both simvastatin and mevastatin to clear virus in Huh7 cells cultured at 5% O2 compared to normoxia controls. 79 Elevated circulating PCSK9 concentrations have been observed in cohorts of patients with critical illnesses such as sepsis, where it has been hypothesised that a decline in plasma cholesterol during sepsis onset results in a compensatory rise in PCSK9 production. 101 Conversely, alternative reports have suggested that inflammation can directly induce hepatic expression of PCSK9, suggesting the potential of a cyclical relationship between PCSK9 expression and systemic inflammation in critical illness.101,120
PCSK9 and bacterial infection
The similarities between the mechanisms of transport and clearance of pathogenic phospholipids and cholesterol have prompted much investigation into the role of PCSK9 in the inflammatory response to bacterial endotoxins. 100 It has been demonstrated that the ability of PCSK9 to reduce hepatic clearance of bacterial endotoxins such as LPS through LDLR downregulation is a key contributor to its inflammatory role, with increasing PCSK9 levels trapping endotoxin in the circulation and increasing the likelihood of inflammatory organ failure. 101 PCSK9 levels in patients presenting with suspected early sepsis have been observed to be significantly higher at presentation in those who subsequently develop respiratory or cardiovascular failure compared to those with no organ dysfunction, with levels of the protein continuing to increase post admission. 101 Therapies such as statins have, however, been observed to increase circulating PCSK9 levels and LDLR expression with chronic use, highlighting the need to differentiate causality and association in observational studies79,100,101 Attenuated inflammatory responses to sepsis and improved survival have been observed following PCSK9 inhibition in murine models, with PCSK9 knockout mice exhibiting 55% lower plasma endotoxin concentrations 6-h post LPS administration compared to controls. 100 Fluorescently labelled LPS has been utilised to demonstrate the impact that increasing doses of extracellular PCSK9 can have on endotoxin uptake by human liver cells (and therefore clearance from circulation). At extracellular PCSK9 concentrations of 3 µg/mL, LDLR expression and fluorescent LPS uptake within HepG2 cells has been observed to decrease by 65%. In other reports, a linear reduction in endotoxin clearance was observed with rising extracellular PCSK9 levels in the range of 0.25–1 µg/mL.100,101 The necessity of the LDLR for this process has been exhibited through PCSK9 inhibition in LDLR knockout and background control mice, where a lack of LDLR presence was observed to eliminate the anti-inflammatory effects of PCSK9 inhibition on the response to LPS injection. 100 A similar reduced inflammatory response to LPS has been shown in healthy human volunteers with PCSK9 LOF alleles, with significantly decreased IL-6 concentrations observed 2-h post LPS injection in those carrying a LOF PCSK9 allele compared to patients without. 100 No difference in baseline IL-6 concentration was observed prior to inflammatory stimulus in this cohort.
LPS is found on the outer membrane of Gram-negative bacteria such as Escherichia coli, and has three components. 121 The O-specific side chain, comprised of oligosaccharide units, is attached to the exterior of the polysaccharide core, while the inner section of the core contains heptose and 2-keto-3-deoxyoctonic acid.121,122 While the core region remains relatively conserved among bacterial species, the lipid A portion of LPS exhibits substantial structural diversity and is predominantly responsible for the immunogenicity of LPS. Attached to the core by a ketosidic bond, lipid A consists of a phosphorylated glucosamine disaccharide backbone with linked fatty acid chains. 122 LPS elicits a cellular response through the LPS binding protein (LBP), as well as membrane anchored co-receptors MD-2 and CD-14. 123 This facilitates the binding of TLR4 to LPS, activating two signalling cascades. The first pathway recruits intracellular adaptor proteins such as MyD88, activating downstream signalling pathways that eventually allow the nuclear translocation of transcription factors such as NF-KB, CREB, and AP-1, regulating the expression of pro-inflammatory cytokines. The second pathway involves the endosomal encapsulation of TLR4, resulting in NF-KB induced type 1 interferon production.123,124
Pulmonary infection is often cited as a leading cause of ARDS, through lung infection due to Gram-negative organisms, resulting in injury to the alveolar epithelium and increased vascular endothelial permeability. 125 The clearance of LPS from the circulation is accomplished primarily by the liver through the LDLR, while lipoproteins can also act in its neutralisation by binding LPS and allowing its phagocytosis through immune cells such as macrophages.126,127 PCSK9-mediated reduction in LDLR levels on hepatocytes can decrease hepatic uptake of LPS, exacerbating systemic inflammation in critical illnesses and resulting in phagocytic cells becoming the primary route of LDL clearance. 128 The internalisation of LPS-bound LDL can induce macrophage apoptosis or the formation of foam cells, which are heavily involved in chronic inflammation and the pathogenesis of atherosclerosis. 129 In addition, consistent endotoxin clearance through exposure to continuously high levels of LPS can result in the failure of Kupffer cells to recognise and clear other microorganisms in a septic host. 130 The role of PCSK9 in LPS clearance requires fine balance, however, as the action of PCSK9 inhibitors can reduce the neutralisation ability of LDL on LPS, resulting in excessive systemic inflammation.
PCSK9 in Acute Respiratory Distress Syndrome and sepsis
The emerging role of PCSK9 in inflammation and infection has driven interest in PCSK9 as a therapeutic target in ARDS and sepsis, conditions associated with widespread and dysregulated inflammation.100–102,131,132
ARDS accounts for 10% of intensive care unit admissions, with a mortality rate of 30−40%.133,134 Damage to the alveolar epithelial/endothelial barrier results in the build-up of protein rich fluid in the airspaces of the lungs, resulting in hypoxaemia. 135 ARDS typically occurs in response to an inflammatory trigger such as pneumonia, severe trauma, or aspiration of gastric contents. 134 Despite decades of research into the epidemiology and pathophysiology of the condition, the specific mechanisms behind ARDS pathogenesis are poorly understood, and effective pharmacological therapies have yet to be identified.133,134 The heterogeneity of ARDS has been suggested as one contributory factor to the failure of pharmacological therapies to date, leading to the identification of two biological phenotypes. These phenotypes, termed ‘hypo’- and ‘hyper’-inflammatory, have unique inflammatory profiles and clinical characteristics, with differential responses to several therapies including statins. 94
Sepsis is one of the leading causes of mortality worldwide.136,137 Characterised by a dysregulated immune response to infection and life-threatening organ dysfunction, the pathogenesis and manifestations of sepsis are varied, with septic shock being its most severe form. 138 Excessive stimulation of pathogen recognition receptors such as TLRs results in overactivation of multiple downstream signalling pathways, elevating production of pro-inflammatory cytokines and promoting widespread tissue damage and organ dysfunction.139,140 At the cellular and molecular level, imbalances in pathophysiological processes such as coagulation, mitochondrial function, and autophagy can all contribute to changes in organ function characteristic of sepsis.141–143 Overproduction of inflammatory cytokines such as IL-6 can enhance the expression of acute-phase proteins by the liver, which is additionally activated by the translocation of LPS and microorganisms originating from the gut lumen. 144 Despite progress in the understanding, pathophysiology, and immune mechanisms involved in sepsis in recent decades, the condition accounts for up to 20% of deaths worldwide, and specific therapeutic interventions have yet to be developed. 136 Only supportive therapies have demonstrated an impact on mortality, highlighting the need for novel pharmacological therapies for the condition. 145 In sepsis-induced multiple organ dysfunction, the lungs are often the primarily affected organ system, and there is considerable overlap between sepsis and ARDS, with the incidence of ARDS reported to be up to 40% in septic shock.146,147
Severe COVID-19 can result in ARDS. In a pilot clinical trial, patients hospitalised for COVID-19 with hypoxia (ratio of arterial oxygen partial pressure to fractional inspired oxygen or PaO2/FiO2 ratio <300 mmHg) and typical opacities on chest X-ray were randomised to a single dose of evolocumab or placebo. Those receiving evolocumab (n = 30) showed a trend to lower rate of intubation or mortality at 30 days than those receiving placebo (n = 30), with the benefit appearing higher in those with higher IL-6 at baseline. The study was a feasibility pilot, not powered for outcomes 99 but the data suggest that a clinical trial of PCSK9 inhibitors in critical illness is feasible and that certain more inflamed cohorts may benefit more, in keeping with the concept of personalisation according to inflammatory phenotypes within critical illnesses such as ARDS.94,99 In another cohort of ARDS patients, Metkus et al. demonstrated a significant correlation between elevated levels of PCSK9 and poorer clinical outcomes, such as fewer ICU-free and ventilator-free days. 102 Larger, adequately powered, clinical trials are required to determine the safety and efficacy of PCSK9 inhibition in critically ill patients. The identification of a conceptual model of PCSK9 as an inhibitor of LPS clearance in sepsis has prompted the suggestion that the protein could be involved in endotoxin clearance in the alveolar airspace after lung injury, given its constitutive expression by pulmonary endothelial and type I/II alveolar epithelial cells reported by the Human Protein Atlas.102,148 This suggestion is further substantiated by the recent observation that the Angiotensin Converting Enzyme-2 (ACE2) receptor, highly expressed by alveolar epithelial cells, can be targeted by extracellular PCSK9 for degradation, impeding LDLR downregulation and suggesting a stronger binding affinity of PCSK9 for ACE2. 149 This would suggest that the association between PCSK9 inhibition and reduced inflammation may be partly due to enhanced ACE2 activity and subsequently elevated anti-inflammatory angiotensin levels. The role of ACE2 in the entry of respiratory viruses such as SARS-CoV-2, however, would imply that its PCSK9-mediated degradation would be beneficial in the context of infection, rather than detrimental, highlighting how the impact of PCSK9 in viral infection is still emerging.106,149
PCSK9 has also been demonstrated to reduce surface expression of the epithelial sodium channel (ENaC) in transfected HEK 293T cells. Coimmunoprecipitation studies have suggested that PCSK9 and ENaC interact intracellularly in their immature forms, inhibiting the ENaC exocytosis and enhancing its proteasomal degradation. 150 The catalytic activity of PCSK9 is not required for this reduction in ENaC expression, and given the lack of homologous motifs, it has been suggested that PCSK9 binds ENaC through a different binding mechanism than to the LDLR, possibly facilitated by an adaptor protein. 150 This finding has yet to be explored in type II alveolar epithelial cells; however, given that impaired ENaC dependent fluid clearance is a key feature of ARDS, further work could determine if this mechanism contributes to the emerging deleterious role of PCSK9 in ARDS. 151
In genotyped human septic shock cohorts, PCSK9 LOF alleles have been observed to be significantly associated with improved survival and lower concentrations of pro-inflammatory cytokines, with the GOF genotype exhibiting the opposite effect.100,101 Elevated circulating PCSK9 levels have been demonstrated to significantly increase the risk of 28-day mortality in sepsis, highlighting its potential as a prognostic tool. 152 Significant association of higher plasma PCSK9 concentrations with the development of respiratory and cardiovascular failure has also been observed in sepsis. 101 Randomised studies investigating the potential impact of PCSK9 inhibitor administration on critical care cohorts such as ARDS (non-COVID-19) and sepsis patients have yet to be performed, but the increasing body of evidence that reduced PCSK9 activity could confer protection against inflammation and organ failure in these conditions warrants further study.
Conclusions
Advances in the therapeutic regulation of lipid metabolism triggered by the discovery of PCSK9 have the potential to be replicated in inflammatory diseases such as sepsis and ARDS. There is increasing evidence of a pro-inflammatory role played by PCSK9, including elevated levels in sepsis and ARDS patients, a correlation between concentrations of PCSK9 and pro-inflammatory cytokines, and a survival benefit in critically ill patients associated with loss of PCSK9 function. More research is warranted in order to understand the role of PCSK9 outside of lipid regulation, including patterns of expression and function in extra-hepatic tissues in health and disease states. Given that PCSK9 inhibiting drugs are well-tolerated and because there is a lack of effective targeted treatments for sepsis and ARDS, PCSK9 inhibitors (chiefly mAbs) represent an exciting potential therapy for life-threatening inflammatory conditions such as sepsis and ARDS which merit further study.
Footnotes
Acknowledgements
This article was prepared at the invitation of the Clinical Sciences Reviews Committee of the Association for Clinical Biochemistry and Laboratory Medicine.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: PH has received speaking honoraria from Amgen, Internis Pharmaceuticals Ltd, and Amarin. PH has been paid for advisory board work for Chiesi Ltd and Novartis.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a Department for the Economy Northern Ireland Ph.D. studentship awarded to EC, and an Association of Physicians of GB and Ireland Young Investigator’s award awarded to JS.
Ethical approval
Not required.
Guarantor
PH.
Contributorship
EC wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
