Abstract
Background
Carbamazepine is an anticonvulsant drug which is monitored in patients due to toxic side effects. At Manchester University NHS Foundation Trust (MFT), carbamazepine is measured using Roche’s Kinetic Interaction of Microparticles in Solution (KIMS) method on the c 702 platform. The assay has an upper limit of linearity of 20 mg/L. Samples with concentrations above this limit should be identified and manually diluted. However, a poor EQA return from UK NEQAS for Tox and TDM Distribution 456 has highlighted an issue with the Roche KIMS assay. Sample A of the distribution had a carbamazepine concentration of 36 mg/L but was underreported by several Roche users. This indicated that the assay was not consistently identifying high concentration samples which required a dilution.
Method
In this investigation, fresh frozen plasma was spiked with carbamazepine concentrations ranging from 15 to 40 mg/L. The spiked samples and EQA material were analysed at two clinical laboratories using the Roche KIMS assay.
Results
Samples spiked with concentrations 20–30 mg/L were not consistently identified for dilution by the analyser. This was observed at both hospital sites. Spike samples and EQA with concentrations >30 mg/L were correctly identified at both sites.
Conclusion
The manual dilution policy has been changed at MFT, so all samples with a carbamazepine level ≥15 mg/L will be manually diluted. The problem was reported to Roche who are investigating the issue further. We would suggest that other laboratories look at validating their dilution protocols.
Keywords
Introduction
Carbamazepine is a widely used anticonvulsant drug that features on the World Health Organization list of essential medicines. 1 It is indicated for the treatment of bipolar disorder, schizophrenia, and epilepsy. 2 Physiologically, carbamazepine preferentially binds voltage-activated sodium channels and increases concentrations of the inhibitory neurotransmitter gamma amino butyric acid (GABA) while also decreasing the excitatory neurotransmitter glutamate. Collectively, these actions help attenuate the development and progression of seizures.
In the circulation, carbamazepine is 70%–80% protein bound. 2 Carbamazepine is metabolized hepatically by cytochrome P450 oxidation to produce pharmacologically active carbamazepine-10,11-epoxide. This metabolism exhibits high inter-individual variability making it difficult to establish a link between dose and concentration, or between concentration and pharmacological effect. 3
Common side effects of carbamazepine include drowsiness, vertigo, blurred vision and slurred speech. 4 Other effects include nausea, vomiting, serious haematological toxicity and hypersensitivity reactions. Due to metabolic inter-individual variability, toxic side effects and its interactions with other drugs, The Royal College of Pathologists recommends that carbamazepine concentrations should be confirmed 2 to 3 months after commencing therapy. 5 Monitoring levels allows clinicians to titrate dosage and achieve optimal therapeutic effects, while avoiding subtherapeutic and toxic levels.
Carbamazepine can be measured by immunoassay which can be performed on automated clinical chemistry platforms. Manchester University NHS Foundation Trust (MFT) analyzes carbamazepine using the Roche Cobas 702 Kinetic Interaction of Microparticles in Solution (KIMS) assay. KIMS is a competitive immunoassay. An analogue of the drug bound to latex beads competes with free drug in the sample for binding to an antibody. The kinetic interaction between the antibodies and the microparticles conjugated to the drug is measured photometrically. The signal produced is inversely proportional to the concentration of analyte in the sample.
A potential issue with the Roche c702 platform was highlighted following a poor EQA return from UK NEQAS for Tox and TDM Distribution 456 (Figure 1). Sample A of this distribution had a carbamazepine concentration of 36 mg/L. For Roche KIMS users, this would mean a manual 1:1 dilution would have to be performed. However, 15 out of 55 Roche KIMS users reported a result which suggested a dilution had not been performed. Birmingham Quality (UK NEQAS) contacted these 15 participants, 6/15 (40%) had missed the dilution flag and 6/15 (40%) had a value between 19 and 20 mg/L, meaning that the analyser did not identify the sample for dilution. This suggested that a cut-off value of 20 mg/L was too high for the Roche assay and should be lowered. EQA report for UK NEQAS for Tox and TDM Distribution 456, sample A.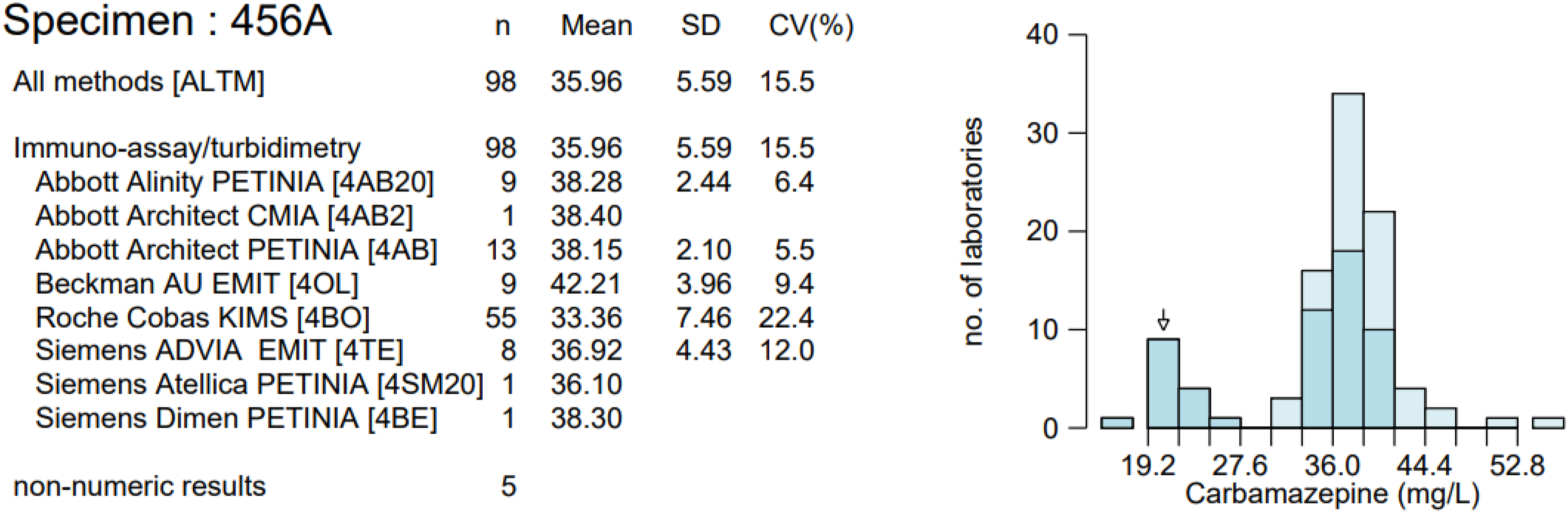
The objective of this study was to show the importance of validating dilution protocols and to determine if high concentration carbamazepine samples were being correctly identified as above the upper limit of linearity by the Roche Cobas c702 platforms.
Method
Fresh frozen plasma was gravimetrically prepared with Cerilliant® certified reference material carbamazepine solution (Sigma Aldrich) to produce samples with concentrations ranging from 10 to 40 mg/L. The samples were analysed by immunoassay using the Roche Cobas c702 module and the Cobas Online TDM Carbamazepine Gen.4 (‘CARB4’) kit). Samples were analysed in replicates of 10 and then diluted 1:1 using Preciset TDM1 Diluent (Roche Diagnostics Limited, Burgess Hill) and re-analysed, in line with the manufacturers recommended procedure. Spiked samples were also analysed at two clinical laboratories using the Roche KIMS assay.
EQA samples from the UK NEQAS for Tox and TDM Distribution 468 were analysed at the same hospital sites and run in replicates of 10. This particular distribution was selected as it had two samples with high carbamazepine concentrations of >30 mg/L.
Results
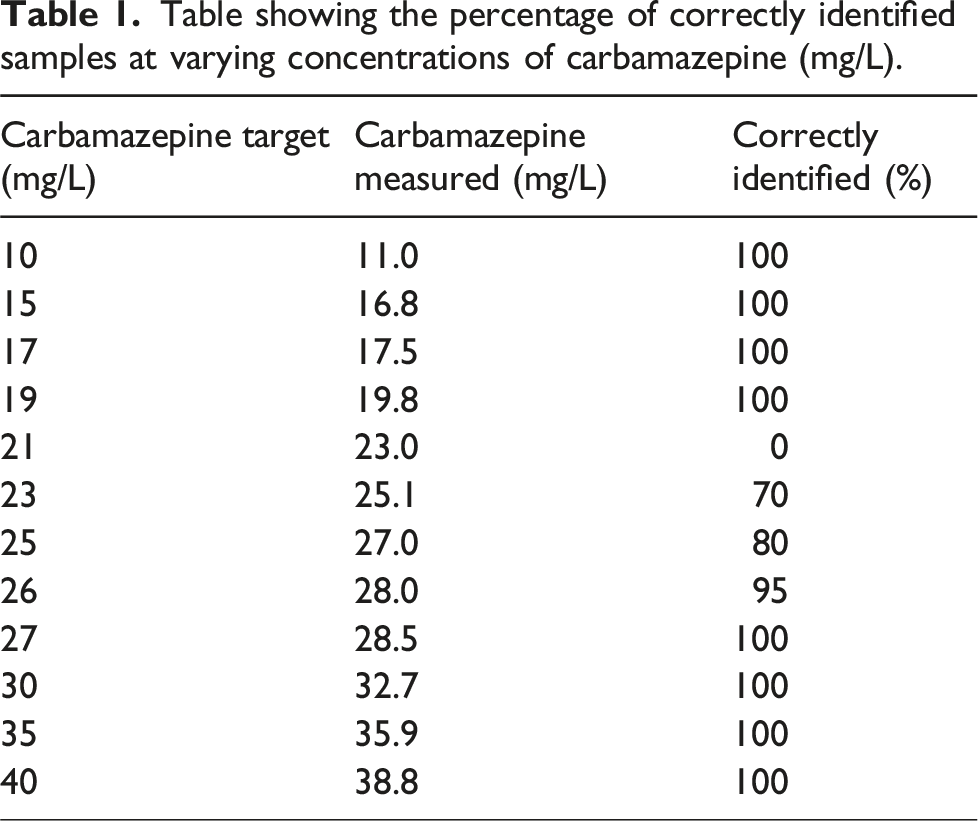
Table showing the percentage of correctly identified samples at varying concentrations of carbamazepine (mg/L).
The carbamazepine assay showed similar performance across two clinical laboratory sites (Figure 2). However, the Roche assay at Site 2 showed poorer performance at a carbamazepine concentration of 27 mg/L. Graph comparing the percentage of correctly identified samples requiring a dilution on two hospital sites.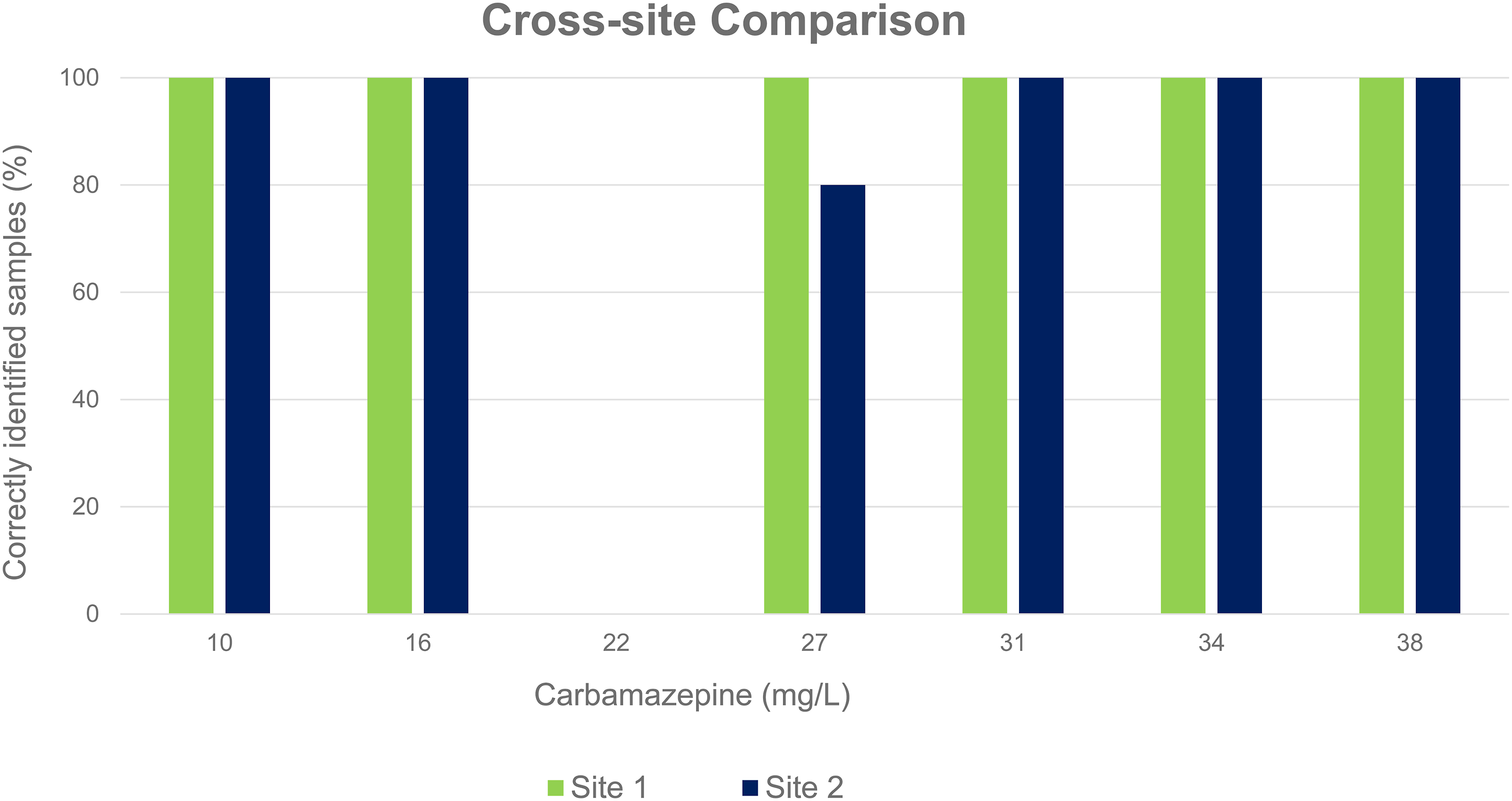
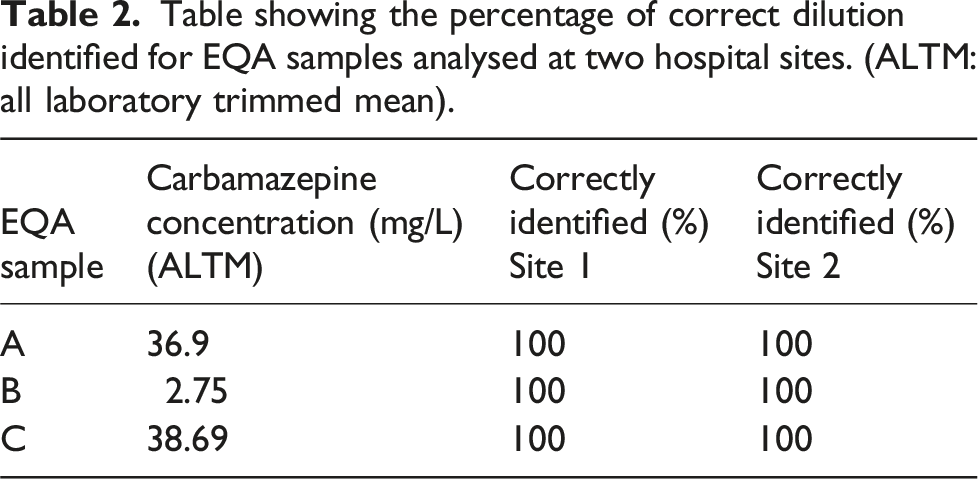
Table showing the percentage of correct dilution identified for EQA samples analysed at two hospital sites. (ALTM: all laboratory trimmed mean).
Discussion
This investigation has shown that there is a lack of consistency in identifying high concentration carbamazepine samples by the Roche carbamazepine KIMS assay between 20 and 30 mg/L. Roche’s KIMS assay has an upper limit of linearity of 20 mg/L; thus, any results above this concentration should be identified, and a manual 1:1 dilution performed. Samples with spike concentrations greater than 30 mg/L were correctly identified by the analyser on each replicate analysed. However, spiked samples with concentrations ranging between 20 and 30 mg/L were not consistently identified. Precision data from January to September 2024 confirms that the assay was performing within the specifications set by the manufacturer, with the coefficient of variation (CV) being <3% across all three levels of internal quality control (IQC) during this time period. This suggests that patient samples with carbamazepine concentrations ranging from 20 to 30 mg/L may have been falsely reported as lower than their true value.
EQA samples which had been analysed at both sites were correctly identified for each replicate analysed, including the two high concentration samples. As these samples had concentrations ≥30 mg/L, this agreed with the pattern observed in the spiked plasma samples. However, when distribution 468 was first analysed at Site 1, the EQA return showed a negative bias of −47.2% for sample A. A similar pattern was observed in a previous distribution, 456, in which sample A had a carbamazepine concentration of 36 mg/L. Site 1’s EQA return showed a large negative bias of −37.2% for sample A. Furthermore, Birmingham Quality (UK NEQAS) has reported that a number of Roche KIMS users had returned similar results. Between-laboratory agreement plots show that Roche KIMS users have greater %CVs at carbamazepine concentrations >30 mg/L than that of other methods (Figure 3). Therefore, while the results of this investigation indicate that the Roche KIMS assay does not consistently identify samples with carbamazepine concentrations ranging from 20 to 30 mg/L, the results of previous EQA returns evidence that the issue is also occurring at higher concentrations. Plot of carbamazepine between-laboratory agreement. ALTM: all laboratory trimmed mean; MLTM: method laboratory trimmed mean. Filled in points = Distribution 456.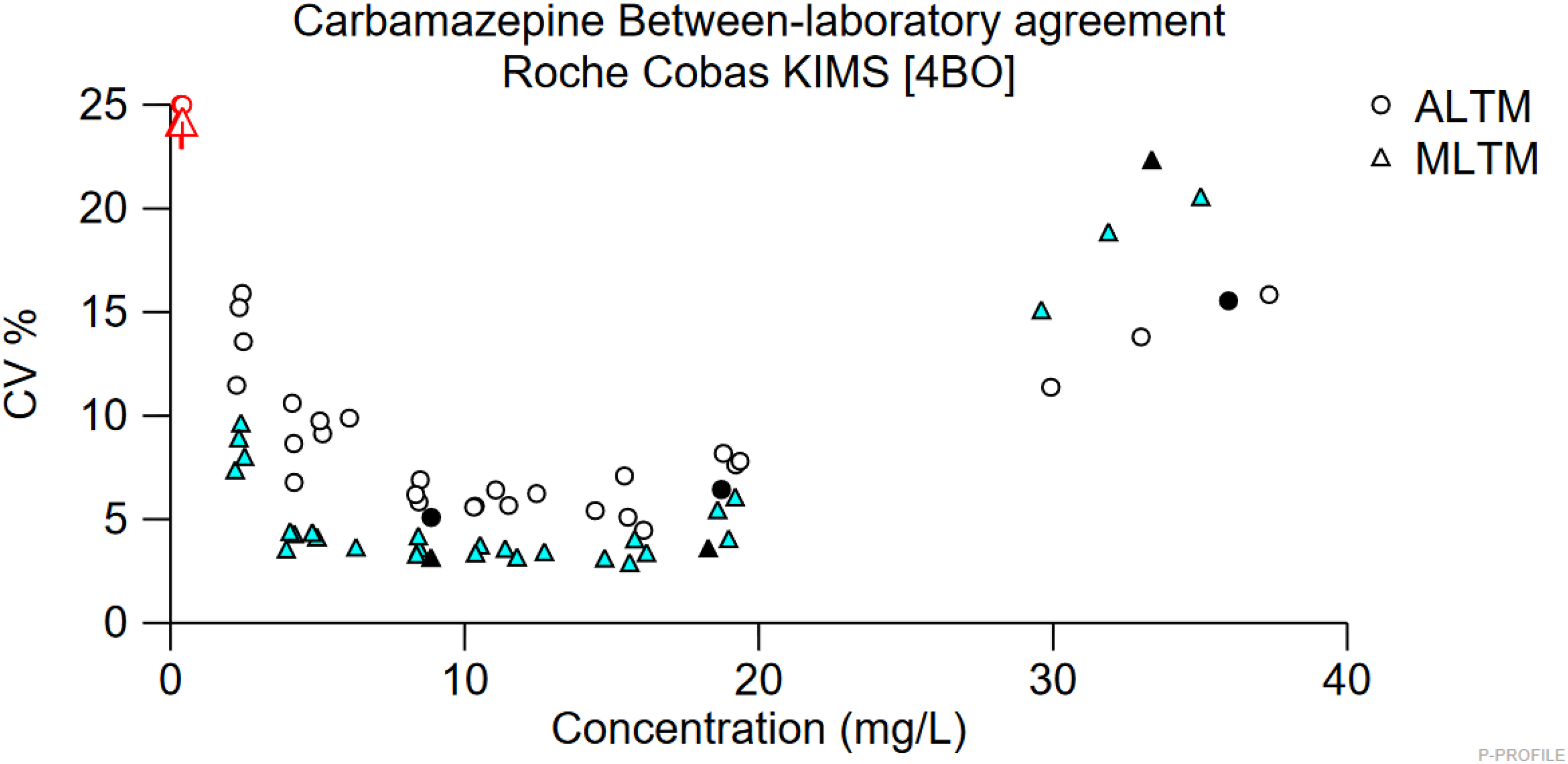
Serious toxicity complications are unusual at concentrations <25 mg/L. 6 Concentrations >40 mg/L are associated with life-threatening toxicity and increasingly severe features including coma, respiratory depression and convulsions. 7 Paediatrics may be at a greater risk of these effects at lower serum concentrations than adults. 8 MFT’s phoning limit for carbamazepine is ≥ 25 mg/L, in line with locally agreed guidance. This investigation has shown that samples with carbamazepine concentrations between 25 and 30 mg/L are not consistently identified by the analyser for dilution. If this were to occur in a patient sample, the result reported would be significantly lower than its true value. This would prevent urgent results from being communicated to clinical staff. This poses a dangerous risk to patients.
Conclusion
This investigation showed that the Roche KIMS carbamazepine assay does not consistently identify samples for dilution between 20 and 30 mg/L. This issue was observed at two different hospital sites. Concentrations ≥30 mg/L were correctly identified on a number of replicates across multiple sites. This varied from what had been seen in past EQA schemes which revealed several Roche users had underreported samples with concentrations greater than 30 mg/L.
Due to this investigation, it was agreed that all MFT samples with a carbamazepine concentration of ≥15 mg/L would be manually diluted, to ensure that high results were not being reported incorrectly at lower concentrations. Lowering the cut-off for dilution ensures that patients results are accurately reported and that patient care can be appropriately managed as a result. The problem was reported to Roche who are investigating the issue further.
This issue appears to only affect the Roche KIMS assay system, but it has also been observed on other KIMS assays, for example, the Roche KIMS Gentamicin assay for EQA specimens with a concentration of >10 mg/L which is the concentration for dilution. We would suggest that you validate your dilution protocols in your laboratory.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
N/A.
Guarantor
James M Hawley.
Contributorship
JMH conceived the study. AAS and CGR undertook the investigations. RM takes responsibility for the EQA data supplied by Birmingham Quality (UK NEQAS). AAS wrote the first draft. All authors reviewed, edited and approved the final version of the manuscript.
