Abstract
Background
The Roche Cobas c Tina-quant C-Reactive Protein (CRP) IV method has shown persistent, significant negative bias compared to other methodologies in the UK NEQAS external quality assurance (EQA) scheme. The aim of this study was to compare CRP concentrations determined by this restandardized 4th generation assay with another commercially available method to assess the extent of any bias and impact on clinical cut-offs.
Methods
CRP concentrations in 38 anonymized patient serum samples were determined by a particle-enhanced immunoturbidimetric assay on the Roche Cobas c701 module and a latex assay on the Beckman Coulter AU5800 analyser over a concentration range of 1–308 mg/L.
Results
97.4% samples analysed by the Roche Tina-quant CRP IV method demonstrated a negative bias compared to the Beckman AU latex method. The mean absolute bias was −4.12 mg/L (range: −19.13–10.93 mg/L; 95% CI: −17.25–9.01 mg/L). The mean relative difference was −15.5% (range: -40–4%; 95% CI: −33.03–1.94%). This bias was seen across the range of samples assayed, including at clinically significant cut-offs of 5 mg/L (−24% bias), 20 mg/L (−5%) and 100 mg/L (−13%).
Conclusion
The negative bias of the Roche method demonstrated in the EQA scheme appears to reflect genuine differences in patient results, rather than an EQA matrix effect, despite re-standardization of the CRP assay. This report indicates that clinical cut-offs and reference ranges may be method-dependent, which should be reflected in published guidance and interpretation provided by laboratories.
Introduction
C-Reactive Protein (CRP) is a sensitive acute-phase protein produced in response to inflammatory stimuli, including bacterial infections. CRP concentrations aid clinical decision making; guidance by NICE instructs the use of CRP to determine prescription of antibiotics in patients with respiratory infections. At CRP concentrations <20 mg/L, no treatment is needed; at concentrations 20–100 mg/L, treatment should be considered if symptoms worsen; at concentrations >100 mg/L, antibiotic therapy should be offered. 1 The Gloucestershire Community Acquired Pneumonia Pathway uses the NICE-recommended cut-offs to manage patients in primary care and limit hospital admissions.
The Biochemistry laboratory at Gloucestershire Hospitals NHS Foundation Trust uses a UKAS-accredited Roche Cobas c Tina-quant CRP IV method (CRP Gen IV, Roche Diagnostics GmbH, Mannheim, Germany) to analyse 284,000 samples annually. Historically, the Roche Cobas c method has shown persistent, significant negative bias compared to other methodologies in the UK NEQAS CRP external quality assurance (EQA) scheme. 2 EQA is an essential aspect of laboratory quality management, allowing comparison of analytical performance between different analysers, sites and methodologies.
From 2016 to 2022, UK NEQAS excluded the method from the calculation of the “all laboratory trimmed mean” (ALTM) for the scheme, although the Roche Cobas c Tina-quant CRP IV method was still compared to the ALTM and was the largest user group (290 users). The ALTM was calculated from the remaining nine methodologies also assessed by the scheme (274 users). 2 Although now included in the ALTM, the assay still demonstrates a negative bias and it is not known whether CRP concentrations in patient samples are also subject to the apparent method bias.
The Roche Tina-quant CRP IV assay is standardized to the reference material ERM-DA474/IFCC. Roche have stated that negative bias in EQA schemes is not seen in patient samples in their own studies. The implication is that any bias in the assay performance in EQA schemes is due to the EQA matrix effect.
Since patient results obtained by any method are compared to the same clinical cut-offs, 1 it is essential that bias between methods is known so that service users are aware of the impact on result interpretation. The aim of this study was to compare CRP concentrations in patient samples (to remove any EQA matrix effect) determined by the Roche methodology with a different commercially available method to assess the extent of any bias and thus the impact on clinical cut-offs of different assays.
Method
Surplus serum from 38 samples collected into BD VacutainerTM SSTTM II Advance Tubes (Beckton Dickinson, Plymouth, UK) received in the laboratory over the course of 1 day in May 2021 was anonymized and aliquoted following CRP concentration determination by a particle-enhanced immunoturbidimetric assay 3 on the Roche Cobas c701 module. The concentration range was 1–308 mg/L (assay measuring range: 0.3–700 mg/L), with the majority of samples chosen around the consensus reference interval for adults of 5 mg/L3,4,5 and the clinical decision limits of 20 mg/L and 100 mg/L1. No samples were diluted prior to analysis.
Samples were stored at 4–6°C until transportation to Southwest Pathology Services, Taunton, overnight at room temperature the day after initial analysis. Samples were then analysed using the Beckman AU5800 Latex CRP method (Beckman Coulter, Brea, California, USA). Beckman AU is included in UK NEQAS’s ALTM calculations. 2 Roche state stability of 14 days at room temperature and 21 days at 2–8°C 4 ; Beckman state stability of 11 days at room temperature and 56 days at 2–8°C. 4
Statistical analysis was performed on Microsoft Excel (Microsoft Corporation, WA, USA).
Results
As shown in Figure 1, 97.4% of samples analysed on the Roche Cobas c701 demonstrated a negative bias compared to the Beckman AU5800. The mean absolute bias was −4.12 mg/L (range: −19.13–10.93 mg/L; 95% CI: −17.25–9.01 mg/L). The mean relative difference was −15.5% (range: −40–4%; 95% CI: −33.03–1.94%). Paired t-test analysis demonstrated a significant difference (p = 0.003) between the two data sets.
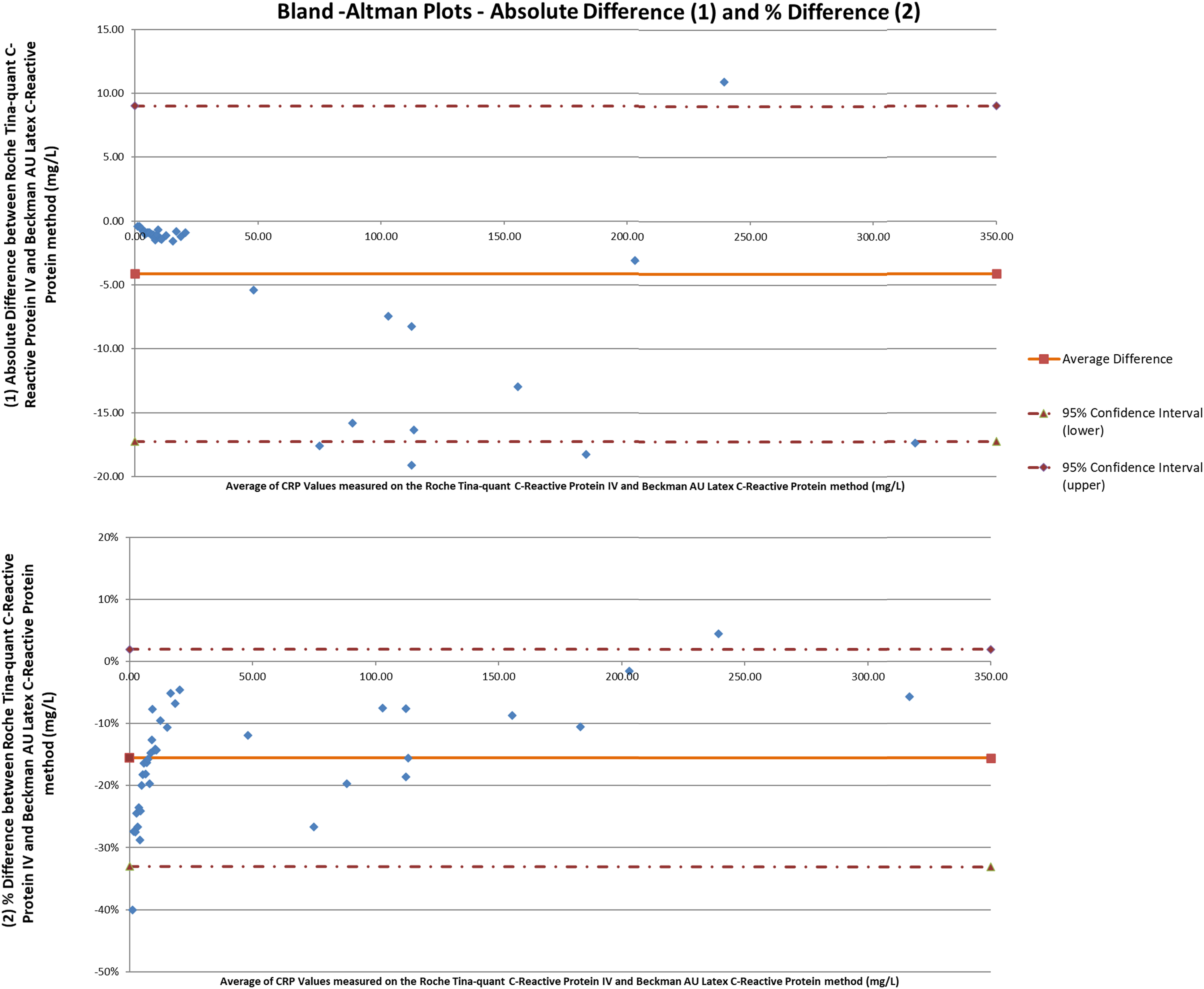
Bland-Altman plots showing the absolute (1) and percentage difference (2) in CRP measurement (mg/L) between Roche Tina-quant CRP IV and Beckman AU5800 Latex CRP methods.
Discussion
This study has demonstrated a clear negative bias on 97.4% of samples assayed by the Roche Tina-quant CRP IV method compared to the Beckman AU5800 Latex method. This bias is seen across the range of samples assayed, including at clinically significant cut-offs of 5 mg/L (bias of −24% demonstrated), 20 mg/L (−5%) and 100 mg/L (−13%). As CRP is reported as integers, these biases may re-categorise patients around clinical cut-offs, influencing the treatment protocol followed.
This study contradicts the conclusions of the method comparison of patient samples performed by the company that there is comparability of CRP results between different assays, which did not include the Beckman platform. The negative bias of the Roche method in the EQA scheme appears to reflect genuine differences seen in patient results, rather than a matrix effect limited to EQA specimens. EQA data shows that the Beckman method was negatively biased to the ALTM (but included in it) at the time Roche were excluded due to persistent negative bias.
The clinical significance of this study is that clinical cut-offs may be method-dependent and, if so, this should be reflected in published guidance and interpretation provided by laboratories. Further, since the two methods studied have the same reference range of <5 mg/L3,4 the implication that the results from each assay are interchangeable may not be correct.
This study was limited by the number of samples used and by comparing results to only one analytical platform. Future studies could compare patient results across all commercially available CRP assays.
Conclusion
This study demonstrates a significant negative bias between the Roche Tina-quant CRP IV method and another commercially available method. The difference seen in patient samples is evidence that the deviation in EQA performance is not due to the matrix effect and has not been resolved by the re-standardization of the CRP assay. Based on this data, laboratories using the Roche platform should consider the appropriateness of local and national clinical cut-offs and the CRP reference range. Service users should be advised of the methodology used for CRP analysis so that informed decisions can be made at relevant clinical cut-offs to ensure the best patient care can be provided.
Footnotes
Acknowledgements
We would like to acknowledge Callum Scott and Hannah Bastug for undertaking sample analysis. We would like to acknowledge UK NEQAS for agreeing to the use of their report for this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Guarantor
ST.
Contributorship
ES conceived the idea to publish. ES and CW analysed the data. All authors wrote the first draft. CW prepared the figures. ES wrote the abstract. All authors reviewed and edited the manuscript including revisions and approved the final version of the article.
