Abstract
An elderly patient who presented with 1 week of fever and respiratory symptoms was tested for dengue infection with an Abbott Bioline™ Dengue Duo immunochromatographic assay. Unexpectedly, the control line of the dengue non-structural protein 1 (NS1) component was absent, necessitating result invalidation. It remained absent when the test was repeated on the SD Biosensor Standard™ Q Dengue Duo, but present on the Wells Bio careUSTM Dengue Combo and Asan Easy Test® Dengue DUO assays, suggesting potential interference. Dilution, polyethylene glycol (PEG) precipitation and centrifugation with a 100 kDa filter were performed to reduce/remove the potential interferent. Sera from other patients that showed a control line, and a test line that was either positive or negative for NS1, were used as controls. Upon dilution with negative control serum, a faint control line emerged. PEG precipitation resulted in disappearance of control and test lines in the positive control. Filtration led to emergence of the control line for the patient’s serum but caused the test line for the positive control serum to disappear. Overall, investigations suggested the presence of a high molecular weight (>100 kDa) substance which interferes with chicken IgY-anti-chicken IgY binding at the control line of affected assays. Our results highlight two important points: firstly, some commonly used laboratory procedures (e.g. PEG or filtration) may inadvertently remove the target biomarker (e.g. multimeric NS1) and should be interpreted with appropriate controls. Secondly, alternative kits that use a different antigen-antibody combination for the control line can be considered when similar patients are encountered in future.
Introduction
Dengue fever is the most common arbovirus infection worldwide and contributes to significant global disease burden. 1 Due to ease of use, low cost and short assay time, dengue duo immunochromatographic assays are commonly used to aid presumptive diagnoses of dengue infections. As the name suggests, the assay comprises of two components, designed to detect either IgM or IgG against dengue virus, or the dengue virus non-structural protein 1 (NS1). The combination of the two improves classification of patients into those with primary versus secondary infections, which has implications on clinical course and management. 2
Each component of the dengue duo assay comes with a separate control line and test line. The control line serves as a procedural control, 3 providing a visual signal that results from the binding of labelled antigens that flow with the sample from the conjugate pad to the immobilised antibodies at the control line. According to manufacturers’ instructions for use (IFUs), the test result is invalid if the control line is absent, and an absent control line may be attributable to insufficient sample volume, incorrect procedural techniques or compromised test kits.3–6 It is recommended to repeat the test with a new device should this occur. 3
We recently encountered a patient whose serum unexpectedly failed to produce a control line for the NS1 component of the Abbott Bioline™ Dengue Duo (Dengue NS1 + IgG/IgM) for the first time since its implementation in our laboratory, despite adherence to manufacturer’s instructions, repeated testing using a new device as well as another serum sample from the same patient.
We hypothesised that analytical interference could be the cause of the absent control line, as this is an immunochromatographic assay and several interferences in immunoassays have been identified over the years.7–9 Such interferences can be divided into those that can be detected before analysis (Type 1: represented by lipaemia, haemolysis and icterus), and those that cannot (Type 2: represented by heterophile antibodies, human anti-animal antibodies, macro-complexes and so on). 7
Here, we present the findings from our investigations, including retesting on dengue duo kits from other manufacturers, sample dilution, polyethylene glycol precipitation and filtration centrifugation.
Case report
An elderly patient presented to the National University Hospital in Singapore with fever and a 1-week history of respiratory symptoms. Four days after admission, a dengue duo test was ordered by the clinical team to exclude dengue infection in view of persistent fever whilst on antibiotics as well as thrombocytopaenia, in the backdrop of a rise in dengue infections in the local community at that time. As per manufacturer’s protocol, 3 3 drops of patient serum were dispensed into the specimen well of the NS1 antigen component of the Abbott Bioline™ Dengue Duo (Dengue NS1 Ag + IgG/IgM) kit (Abbott Diagnostics, CA, USA) using the dropper provided. The result was then read after the kit had been left at ambient temperature (20–25°C) for 15 min. Unexpectedly, unlike the control line for the IgM/IgG component, the control line of the NS1 component failed to develop.
As the test result is invalid if the control line is absent, and as an absent control line may be attributable to insufficient sample volume, incorrect procedural techniques or compromised test kits, we repeated the test with the same sample on a new kit (Figure 1(a)), and subsequently with another serum sample from the same patient collected at a different time (data not shown). In spite of this, the NS1 control line remained absent. Test results obtained on dengue duo kits from 4 different manufacturers. The control line was absent for the NS1 antigen component (highlighted in rectangles) when the Abbott (a) and SD Biosensor (b) dengue duo kits were used, but present when the Wells Bio (c) and Asan (d) kits were used. The antigen-antibody combination used for the NS1 component by each manufacturer, and either presence/absence of the NS1 control line, is indicated below the picture.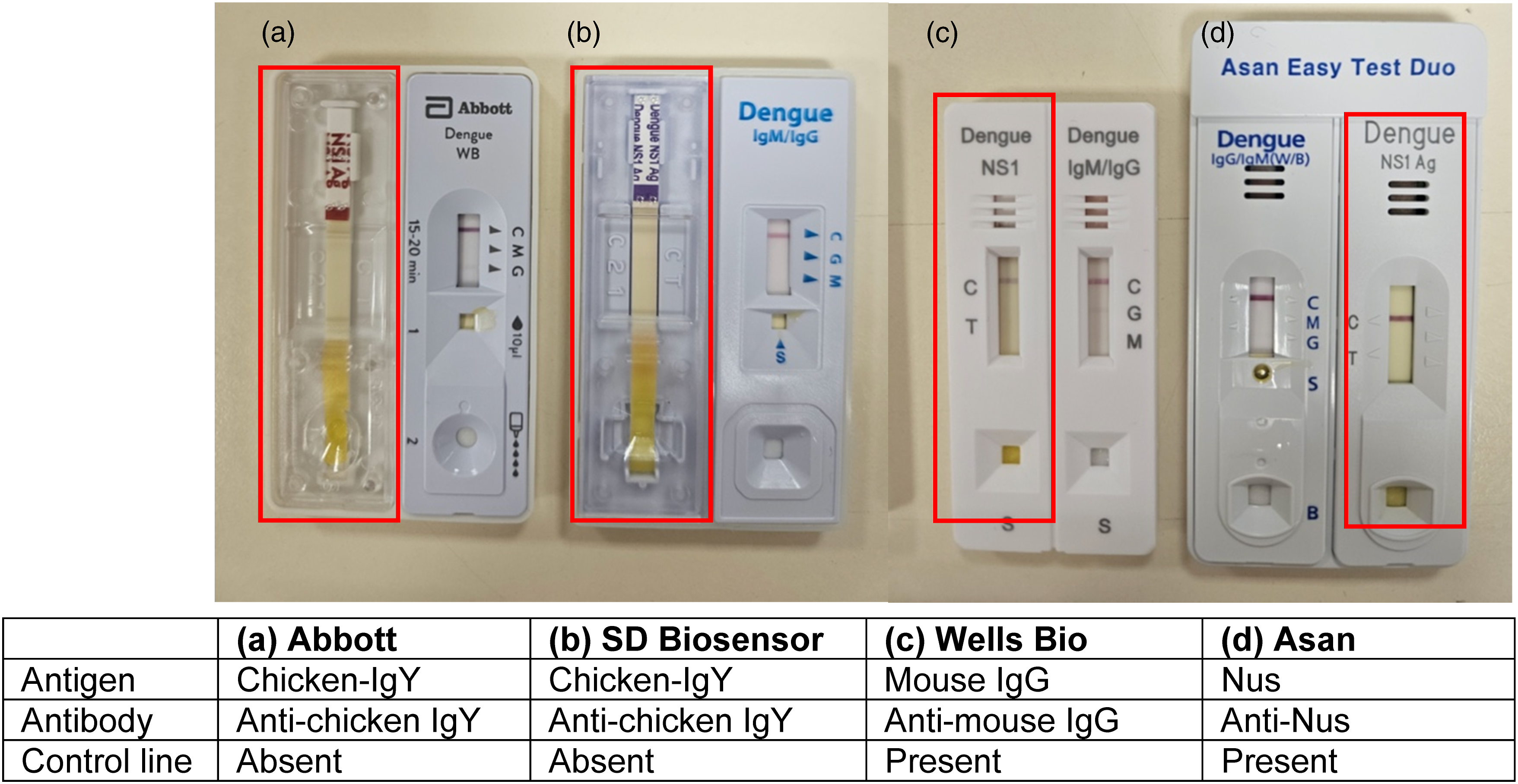
We suspected assay interference which was reproducible on a different sample from the same patient, and on a new kit. First, we retested the patient’s sample using dengue duo assays from other manufacturers that employed either the same or a different combination of antigen and antibody for the NS1 control line. Information on the antigen-antibody combination was either obtained from the IFU or via personal communication with representatives of local distributors of the kits.
Like in the Abbott kit, 3 the NS1 control line in the SD Biosensor Standard™ Q Dengue Duo kit (SD Biosensor, Suwon, Republic of Korea) consists of immobilised anti-chicken IgY antibodies bound to chicken IgY-gold colloid from the conjugate pad (personal communication, SPD Scientific Pte Ltd, 15 March 2024). The NS1 control line was likewise absent when the SD Biosensor kit was used (Figure 1(b)).
In contrast, the Wells Bio careUS™ Dengue Combo NS1 & IgM/IgG kit (Wells Bio, Seoul, Republic of Korea) employs a mouse IgG-anti-mouse IgG combination (personal communication, Unison Collaborative Pte Ltd, 2 April 2024), and the Asan Easy Test® Dengue DUO kit (Asan Pharmaceutical, Seoul, Republic of Korea) uses a Nus-anti-Nus combination (personal communication, Biomedix Pte Ltd, 18 April 2024) for the control line of the NS1 component. For these two kits, the NS1 control line appeared when the index patient’s serum was applied (Figure 1(c) and (d)). Consistent with these findings, the control line of the IgM/IgG component in the Abbott kit, which uses a mouse IgG-anti-mouse IgG combination, 3 is also unaffected by the interferent.
To reduce the concentration of the interferent, we diluted the index patient’s serum with serum from another patient who had tested negative for NS1 antigen but had a control line present on the Abbott kit (‘Negative NS1 control’) in a 1:1 ratio before retesting on the Abbott kit. This time, a faint band emerged at the NS1 control line.
Next, polyethylene glycol (25% w/v PEG 6000, Sigma-Aldrich, MO, USA) precipitation was performed on the index patient’s serum and negative NS1 control serum in order to remove high molecular weight proteins, such as immunoglobulins, which could be the source of interference. However, it was observed that a purple line formed at the distal edge of the conjugate pad within the detection zone of the assay for both samples when the PEG supernatant was applied to a test kit. This is possibly due to increased sample viscosity and precipitation of test components by PEG, leading to retention of the gold colloid signal molecules at the conjugate pad.
As an alternative to PEG precipitation, we centrifuged the index serum and negative NS1 control serum using Amicon® Ultra-15 100 kDa filters (Merck Millipore, MA, USA) prior to applying the filtrate to the test kit. This resulted in emergence of the control line for both samples, suggesting that the interferent is high in molecular weight (>100 kDa) and can be removed by filtration. However, when serum from a patient who was positive for NS1 antigen (‘Positive NS1 control’) was filtered and tested, the control line remained present, but the test line disappeared. This finding was somewhat unexpected as NS1 has a molecular weight ranging from 46 to 55 kDa, depending on glycosylation status. 10 However, recent studies suggest that secreted NS1 antigen can in fact be dimeric, tetrameric or even hexameric. 11 Therefore, we postulate that filtration caused the unintentional removal of NS1 in addition to the interferent, resulting in disappearance of the test line.
Effects of Dilution, PEG Treatment and Filtration of the Index Patient’s Serum on Dengue Duo Test Results.
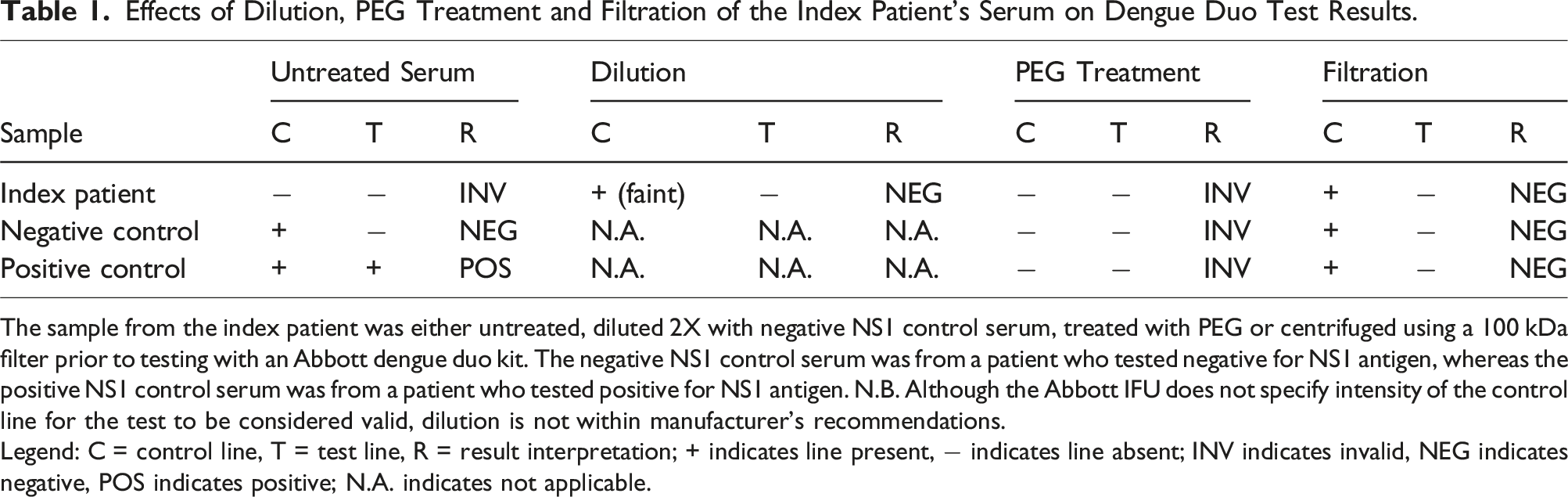
The sample from the index patient was either untreated, diluted 2X with negative NS1 control serum, treated with PEG or centrifuged using a 100 kDa filter prior to testing with an Abbott dengue duo kit. The negative NS1 control serum was from a patient who tested negative for NS1 antigen, whereas the positive NS1 control serum was from a patient who tested positive for NS1 antigen. N.B. Although the Abbott IFU does not specify intensity of the control line for the test to be considered valid, dilution is not within manufacturer’s recommendations.
Legend: C = control line, T = test line, R = result interpretation; + indicates line present, − indicates line absent; INV indicates invalid, NEG indicates negative, POS indicates positive; N.A. indicates not applicable.
Discussion
Immunochromatographic assays have become widely used in the healthcare, food and environmental industries, and at home for self-testing in recent years. 12 The validity of the result is contingent on the presence of the control line, which indicates that sample flow has occurred. To our knowledge, this is the first report of an absent control line in an immunochromatographic assay that is not a result of insufficient sample volume, improper test technique or compromised test kits, all of which are reasons suggested by manufacturers. The clinical implication of control line failure is a potentially missed diagnosis of dengue infection, as the result becomes unreportable.
We have systematically performed multiple troubleshooting steps including retesting on dengue duo kits from other manufacturers, sample dilution, PEG precipitation and filtration, as suggested in the algorithm for troubleshooting immunoassay interference outlined by Wauthier and colleagues. 9
Retesting the sample showed that the NS1 control line was affected for only the kits that employed a chicken IgY-anti-chicken IgY combination (Abbott and SD Biosensor), but not those that employed other antigen-antibody combinations at the control line (Wells Bio and Asan). Although dilution resulted in appearance of a faint control line, we note that this result may not be reportable as sample dilution is not within manufacturer’s guidelines. Precipitation caused the retention of the gold colloidal signal at the conjugate pad in both the index patient as well as control samples, leading to an unreadable result. Finally, filtration removed the interferent, but also caused the inadvertent removal of the high molecular weight NS1 multimer, leading to disappearance of the test line in positive NS1 control samples.
Taken together, our results suggest the presence of a high molecular weight substance that interferes with the binding of chicken IgY to anti-chicken IgY, resulting in failure of control line to develop for the NS1 component of affected kits.
Interestingly, this patient was the first patient we encountered who had a negative control line, but proved to not be the last. During the process of manuscript preparation, we encountered another patient whose serum also failed to produce a negative NS1 control line on the Abbott kit. This time, we tested the serum on a Wells Bio kit with the knowledge that it uses a different antibody-antigen combination and the NS1 control line appeared (data not shown).
Our case report highlights that some commonly-performed laboratory procedures to remove interference such as PEG precipitation or filtration may inadvertently remove the target biomarker, and that results should be interpreted with the appropriate negative and positive controls. Further, should similar patients be encountered in future, kits from other manufacturers that employ a different antibody-antigen configuration at the control line can be tested if available.
However, our report has a number of limitations. Firstly, although the dengue duo has been in use for at least a decade at our laboratory, this is the first time we observed a negative control line, and only for 2 patients thus far. This is likely a rare phenomenon, as we have not been able to find reports of similar cases occurring elsewhere in the literature.
Secondly, we have not conclusively identified the exact nature of the interferent. Potential high molecular weight interferents include heterophile antibodies and human anti-animal antibodies. Heterophile antibodies are thought to arise from the natural process of antibody diversification, without a history of immunoglobulins or other well defined immunogens, producing weak, polyreactive antibodies. 13 In contrast, human anti-animal antibodies are usually associated with exposure to animal proteins in a social or therapeutic setting. 14
As the interferent seemed to affect the chicken IgY-anti-chicken IgY combination but not other antigen-antibody combinations, we speculate that human anti-animal antibodies targeting chicken antigens could be the reason. Although both patients had never undergone treatment or imaging with monoclonal antibodies, they had a common history of prior prolonged exposure to chickens: the first patient had been a chicken rice seller, and the second patient had worked on a chicken farm in his youth. Further studies with gel filtration chromatography, protein A/G to target immunoglobulins or heterophile blocking tubes to remove heterophile antibodies may help to elucidate the true nature of this interferent. 9
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The study does not meet the definition of human subjects research and was not reviewed by our hospital’s Institutional Review Board.
Guarantor
LO.
Contributorship
SXS performed the experiments and wrote the first draft of the manuscript. ATC and JSY contributed to the data. LO and TPL conceptualised the study, and together with SKS and SS, helped to revise the manuscript. All authors critically reviewed the manuscript and approved the final version of the manuscript.
