Abstract
Background
With the recent outbreak in West Africa, hospitals worldwide have been developing protocols for suspect of cases of Ebola virus disease. Patients with Ebola virus disease present with a severe gastroenteritis leading to dehydration and electrolyte abnormalities and as such, routine chemistry analysis is essential for patient management. While point-of-care testing can be used with additional precautions for rapid chemistry analyses in a laboratory setting, significant delays could ensue before specimens arrive to the laboratory. This study evaluated the stability of eight chemistry analytes up to 4 h post-collection.
Methods
Blood was collected by venipuncture from 20 healthy volunteers and tested at times 0, 30, 60, 90, 120 and 240 h. Approximately 100 µl of blood was dispensed into a CHEM 8+Cartridge and processed on a model 300 i-STAT 1 Analyzer (Abbott Point of Care Inc.) and ANOVA was used to assess statistical significant difference from the initial time point.
Results
While the manufacturer recommends testing within 30 min of collection, no significant variation was observed for most analytes with time points extending up to 4 h. In contrast, glucose concentrations decreased significantly (P < 0.0001) over time at an average rate of 0.0032 mmol/L per min.
Conclusions
This study provides supporting data suggesting that delays up to 4 h can be tolerated, giving ample time for collection and transport of specimens to the clinical laboratory. For glucose, POC testing could still be used, taking into account the collection time and the average rate of decrease.
Introduction
Ebola virus disease (EVD) is a deadly disease caused by infection with Ebola virus, which is transmitted through contact with blood or body fluids. 1 As of December 2014, the EVD in West Africa has caused 17,908 cases and 6373 deaths, and has been declared a public health emergency of international concern.1–5 Hospitals worldwide have been developing protocols for the management of a suspect EVD, which includes the collection of blood for Ebola virus diagnostics and chemistry analyses.2–5
Patients with EVD present with a severe gastroenteritis leading to dehydration and electrolyte abnormalities and as such, routine chemistry analysis is essential for patient management.2,3 However, there are challenges in terms of safety and biocontainment for laboratory diagnostics since blood from a patient with EVD is highly infectious. While some hospitals are equipped to use rapid diagnostic testing at the patient bedside (i.e. point-of-care [POC] testing), the Public Agency of Canada (PHAC) and the Centers for Disease Control and Prevention (CDC) have released interim biosafety guidelines suggesting that specimen processing should only be carried out by trained personnel in a biological safety cabinet (BSC) in a minimum containment level 2 (CL2) laboratory with enhanced precautions.4,5 Clinical microbiology laboratories are CL2 compliant, and with enhanced personal protective equipment (PPE); this environment is suitable for the processing of specimens from patients under investigation for EVD.4,5 The challenge for POC chemistry testing in a laboratory setting is with the additional precautions required during collection and transport of these specimens; significant delays could ensue before specimens arrive to the laboratory. 5 This study evaluated the stability of eight chemistry analytes up to 4 h post-collection.
Materials and methods
Sample collection and processing
Blood was collected by venipuncture from 20 healthy volunteers and placed in BD Vacutainer lithium heparin tubes (Becton Dickinson, Franklin, NJ) and held at room temperature until use. At times 0, 30, 60, 90, 120, and 240 h, the tubes were mixed by inversion and approximately 100 µL of blood was dispensed into an i-STAT CHEM 8+Cartridge (Abbott Point of Care Inc., Abbott Park, IL) using an i-STAT dispensing tip. Each cartridge was placed on a model 300 i-STAT 1 Analyzer and values plotted over time. Results of sodium (Na), potassium (K), chloride (Cl), urea, ionized calcium (iCa) and total carbon dioxide (TCO2) were expressed in millimoles per litre (mmol/L) and for creatinine, as micromoles per litre (µmol/L). The average values ± standard deviation (S.D.) for each analyte were normalised by subtracting the value of the initial time point (Figure 1).
Analyte stability when normalized to the initial time point. Analytes included: (a) sodium; (b) potassium; (c) chloride; (d) creatinine; (e) urea; (f) ionized calcium; (g) TCO2; (h) glucose.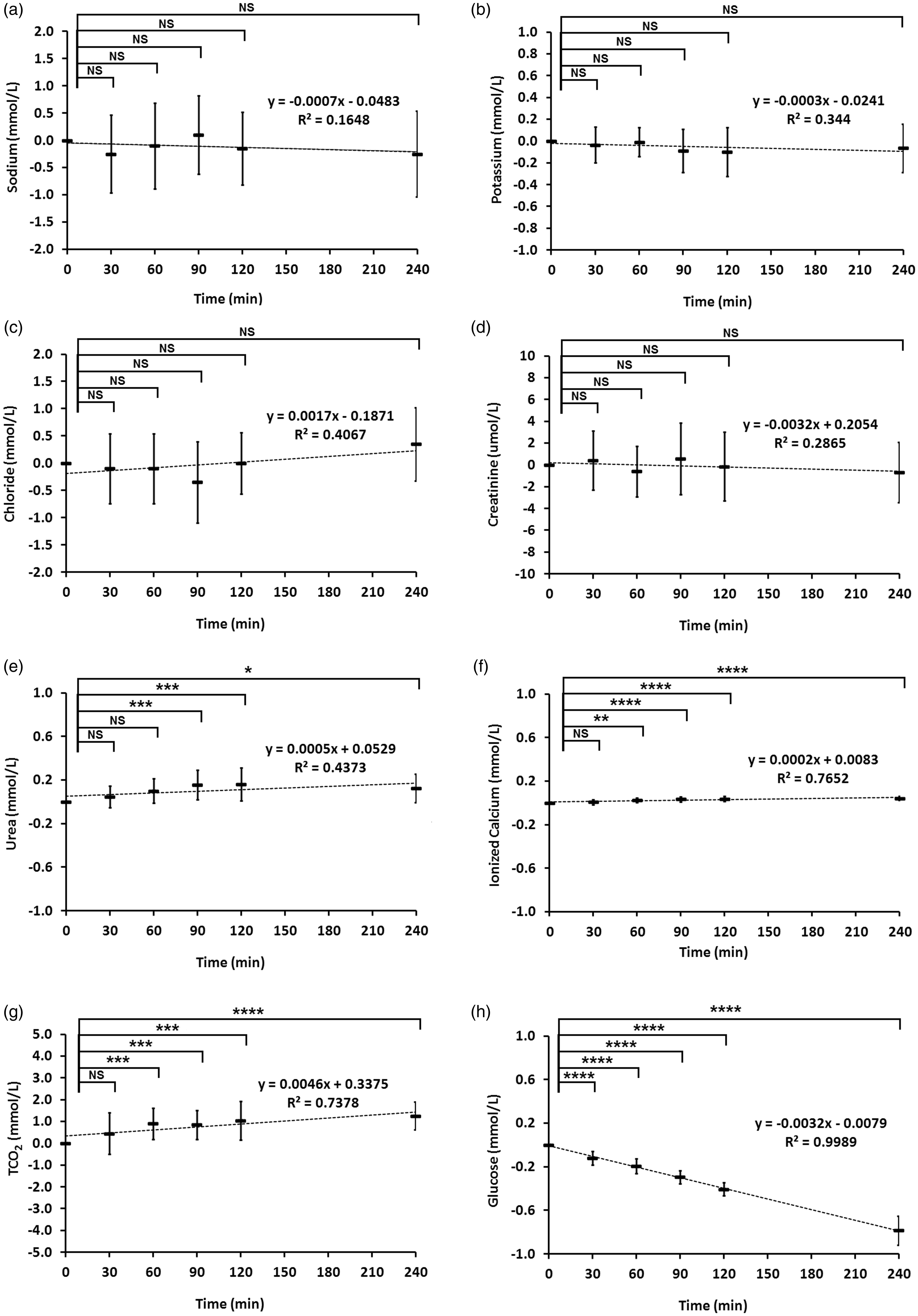
Statistical analyses
For statistical analyses, analysis of variance (ANOVA) was performed using Prism 6 software (GraphPad Software Inc.) version 6.05 where P values of <0.05 were considered statistically significant.
Results
Figure 1(a) to (d) shows no significant variation in Na, K, Cl and creatinine with time points extending up to 4 h. While statistical differences were noted for urea, iCa and TCO2 (Figure 1(e) to (g)), no values differed more than 5% from the initial time point (Table S1, available online), and as such, these would not be considered clinically significant. In contrast, glucose concentrations decreased significantly (P < 0.0001) over time, and after 90, 120 and 240 min, the average decreased would be considered clinically significant at 5.7%, 7.8% and 15.1%, respectively (Table S1). The plot of concentration and time displayed high correlation (R2 of 0.9989), and the slope decreased at an average rate of 0.0032 mmol/L per minute (Figure 1(h)). This trend was consistent for all specimens (Table S2, available online).
Discussion
This study supports the off-label use of the detection of eight analytes from the CHEM 8+cartridge on an i-STAT instrument up with processing delays up to 4 h. The results of this study should not be extrapolated to other instruments or analytes. It should be noted that the i-STAT instrument has Food and Drug Administration clearance for intended use in critical care patients and has been used in patients under investigation for EVD. 6 EVD patients will be critically ill with multiple electrolyte abnormalities and ideally, hyponatremic, hypokalemic and hypocalcemic specimens should be tested to ensure stability.2,3 This study was limited to healthy volunteers with results falling within reference ranges. Future studies will evaluate the stability of chemistry analytes in patients with abnormal or critical values.
Alternatives to POC testing include virus inactivation in the clinical specimen or processing on closed automated analysers. While virus inactivation methods like heat have been shown to have little effect on electrolytes, it is detrimental for enzymatic reactions and other diagnostic tests. 7 While Ebola specimens can be processed on some closed automated analysers, many laboratory directors have been uncomfortable with the potential risk these specimens pose to health care workers. 5 In addition, commercial vendors have no guarantees of the efficacy of their decontamination procedures, and it is unclear whether these vendors will continue to provide service to instruments used to process a specimen containing Ebola. 4 The advantage of POC instruments is that they offer a balanced solution between rapid results and staff safety, and they can be placed in a class II BSC, thus ensuring compliance with the biosafety guidelines when processing bloods from patients under investigation for EVD.4,5 Coupled with blood smears for malaria and other POC instruments for liver function tests and cell blood counts, POC testing for chemistry analytes plays an important role in the management of patients under investigation for EVD.2–5 By centralizing the testing to the microbiology section within the laboratory, our laboratory can provide testing for patients under investigation for EVD with only a limited the number of technologists who require maintenance of competence in donning and doffing of PPE, and simple laboratory tests like POC testing.
In sum, this study provides supporting data suggesting that delays up to 4 h can be tolerated, giving ample time for collection and transport of specimens to the clinical laboratory. For glucose, POC testing could still be used, taking into account the collection time and the average rate of decrease at 0.0032 mmol/L per minute.
Footnotes
Acknowledgements
We would like to thank our phlebotomy staff for their assistance with specimen collection. We would also like to thank the Department of Pathology and Laboratory Medicine at NSHA for their ongoing support.
Declaration of conflicting interests
None.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethics approval
Not applicable.
Guarantor
JL.
Contributorship
JL, AL, BN and TH researched the literature and contributed to writing drafts and final manuscript. RM, CJ, and CR processed the specimens and contributed to writing. CH, JM, and JL coordinated the project from design to completion. Statistical analyses were performed by AL and JL. All authors reviewed and approved the final version of the manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
