Abstract
Background
To determine delta check limits for immunoglobulins and complements in outpatients and inpatients based on patient data and biological variation due to the lack of relevant studies.
Methods
Patient data for IgA, IgG, IgM, IgE, C3, and C4 from 1 January 2022 to 31 December 2023 was collected from laboratory information system (LIS) in our clinical laboratory of Wuhan Union Hospital, which includes both outpatients and inpatients. The delta difference (DD), delta percent change (DPC), and reference change value (RCV) were calculated based on patient data and biological variation.
Results
For DDs, there are significant differences between outpatients and inpatients in C4, IgE, IgG, and IgM. For DPCs, the corresponding analytes which are significantly different are C3, C4, IgE, IgG, and IgM. Two sources of CVI to calculate the RCV of IgA, IgG, IgM, C3, and C4 were applied in this study, which revealed that two kinds of RCVs based on different biological variation databases are similar to each other, but both were smaller than delta check limits based on patient data, except for C4.
Conclusions
The delta check is a useful tool to monitor potential errors which may occur in total testing process. We hope our findings could be helpful for future studies focused on delta checks in immunological analytes.
Introduction
Nosanchuk and Gottman introduced the delta check concept in 1974 as a quality control tool for identifying misidentified specimens in clinical laboratories. 1 Delta check measures the difference between consecutive patient test results for effective and direct internal quality control of pre- and post-analytical errors in patient data monitoring.2,3 A change in the interval of results greater than a predefined limit may indicate a potentially overlooked error in the test results of the previous or current specimen. Common causes of errors in laboratory testing include issues related to specimen collection, analysis, and reporting. 4 Delta check methods, such as delta difference (DD), delta percent change (DPC), rate difference (RD), and rate percent change (RPC), are frequently employed based on the distribution of delta values in the patient population. 5 The delta check is a crucial tool in improving laboratory efficiency, serving as a key component of the automated report verification process. 6
Immunoglobulins (Igs) are a group of globulins with antibody activity that comprise approximately 20% of total plasma proteins and are widely distributed in the blood, tissue fluids, and exocrine fluids where they can specifically bind antigens and activate immune responses. 7 Five classes of Ig have been identified in the body, IgA, IgG, IgM, IgE, and IgD, while IgA, IgG, IgM, and IgE are commonly measured in clinical practice.7,8 Complements, a protein that is activated and enzymatically active, with 90% originating from the liver, show increased levels in cases of tissue damage and inflammation. 9 Its functions encompass the neutralization of viruses, enhancement of phagocytosis, and modulation of the immune system. C3 and C4 stand as the predominant complements in the immune system and are the most plentiful in serum, frequently utilized in clinical tests, and bear a close relation to immunity. 10 In recent years, research on delta check limits has primarily concentrated on chemical markers, neglecting the role of immunoglobulins and complements. With the expansion of tests offered by modern large clinical laboratories and the increase in the number of specimens handled, the rate of sample confounding also increases. Delta check is a potentially effective tool for the early identification of sample mix-up errors. Therefore, it is also necessary to use delta check limits for immunoglobulins and complements in immunological tests to monitor potential errors at any stages of specimen analysis.
Materials and methods
Methods
Statistical analysis
All data analysis was performed by Excel (Microsoft, Redmond, WA, USA; 2016 version) and SPSS (IBM SPSS Statistical for Windows, version 25.0). The Kolmogorov-Smirnov test was used to test the normality of the data. The
Delta check limits derived from patient data
Delta check limits derived from patient data of our laboratory were presented as delta difference (DD) and delta percent change (DPC). Means or medians were used to describe the statistical characteristics of DD and DPC based on the distribution of data.
DDs and DPCs for each analyte were calculated by the equations below: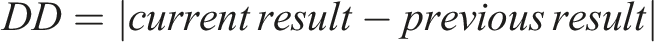
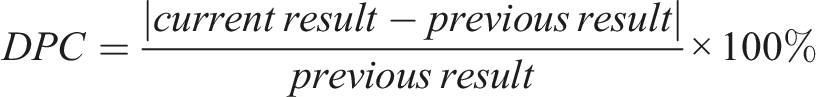
Delta check limits derived from biological variation (reference change values, RCVs)
RCVs for each analyte were calculated by the equation below: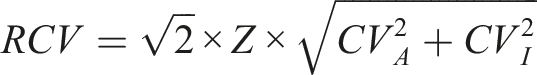
Results
The general characteristics of outpatients and inpatients.
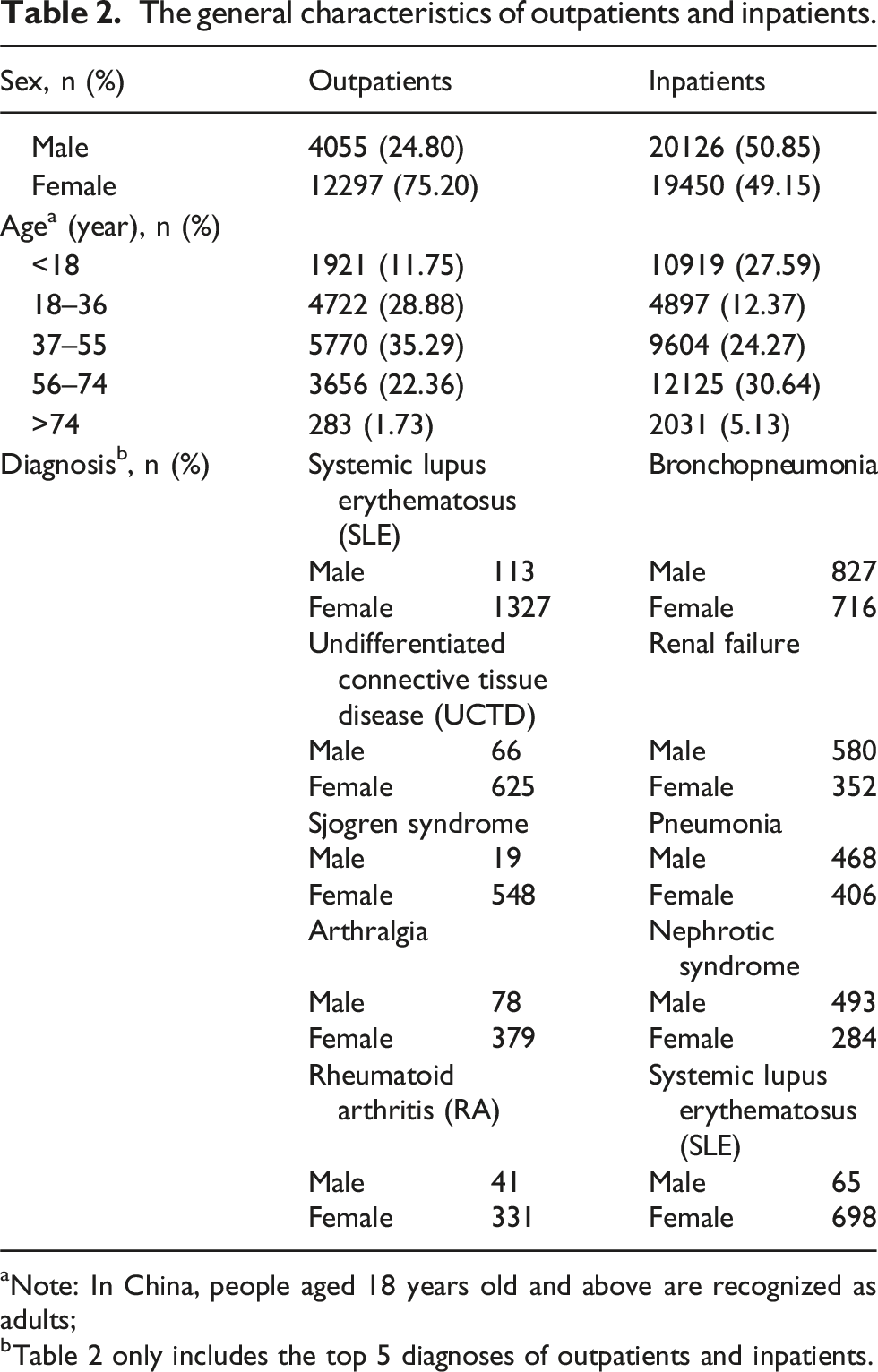
aNote: In China, people aged 18 years old and above are recognized as adults;
bTable 2 only includes the top 5 diagnoses of outpatients and inpatients.
The frequency of serial measurement of immunoglobulins and complements for outpatients and inpatients.
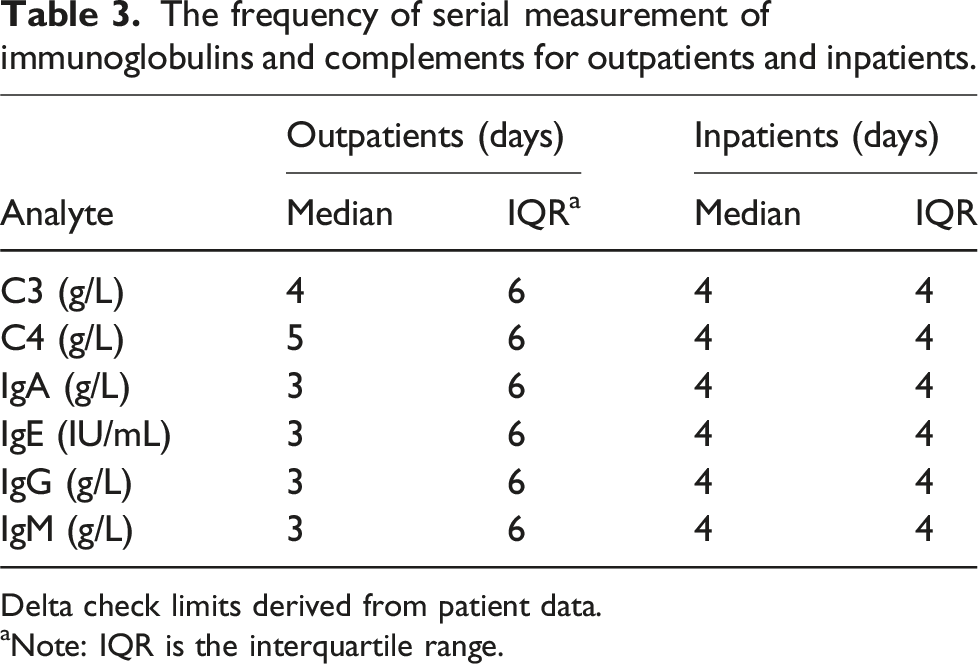
Delta check limits derived from patient data.
aNote: IQR is the interquartile range.
Delta check limits for outpatients and inpatients based on patient data.
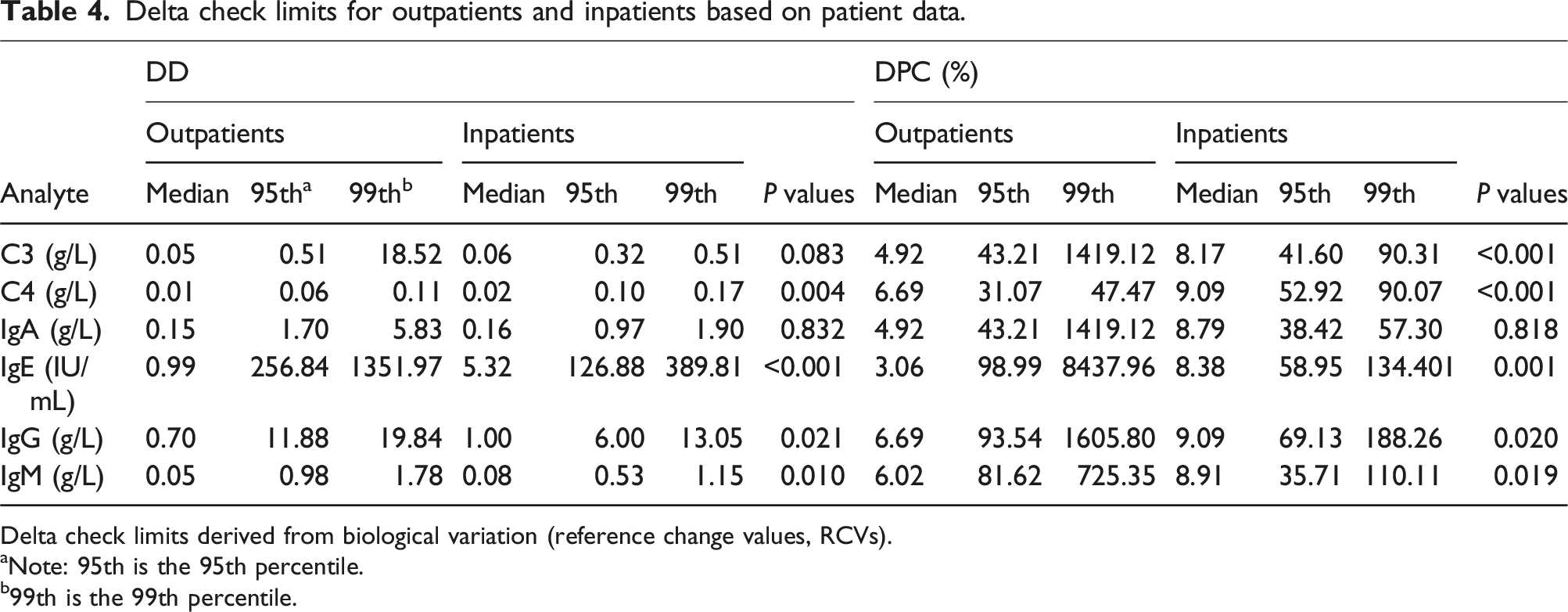
Delta check limits derived from biological variation (reference change values, RCVs).
aNote: 95th is the 95th percentile.
b99th is the 99th percentile.
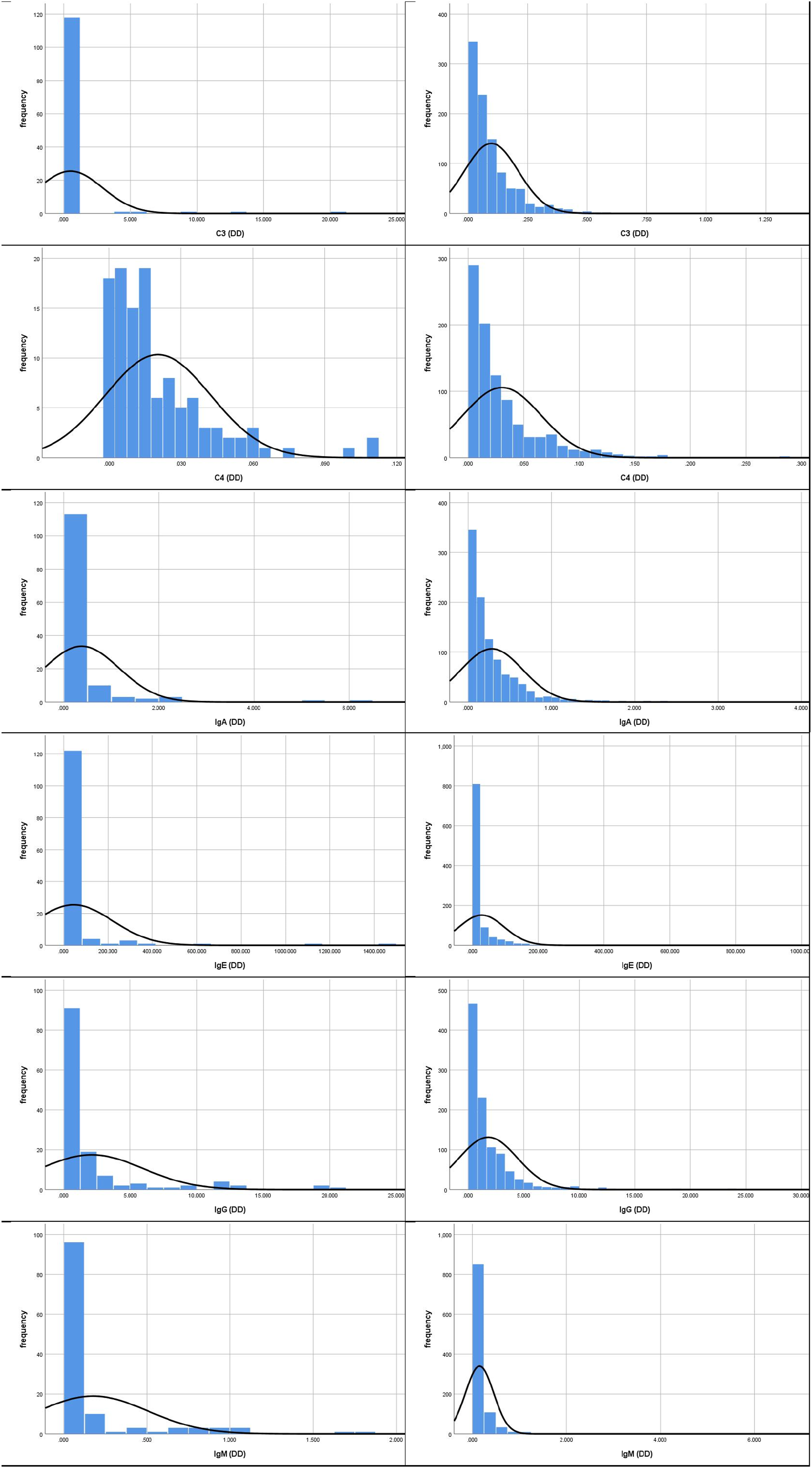
The frequency distribution histogram of DD values for outpatients and inpatients (left: outpatients; right: inpatients).
Delta check limits for outpatients and inpatients based on biological variation.
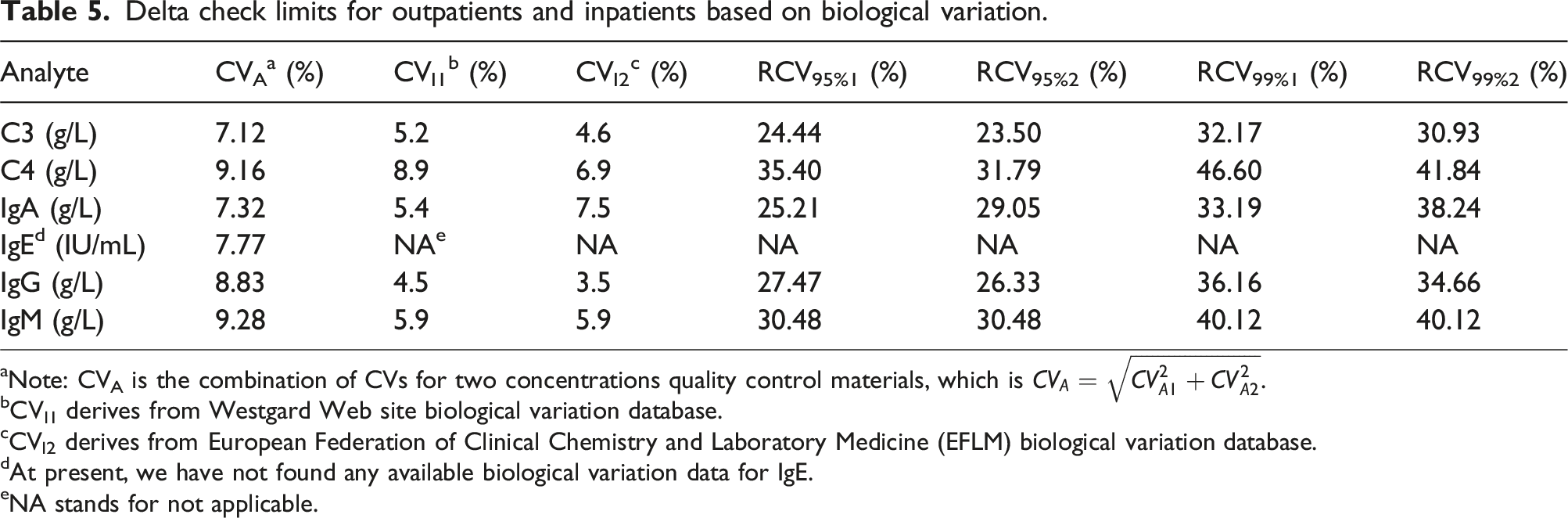
aNote: CVA is the combination of CVs for two concentrations quality control materials, which is
bCVI1 derives from Westgard Web site biological variation database.
cCVI2 derives from European Federation of Clinical Chemistry and Laboratory Medicine (EFLM) biological variation database.
dAt present, we have not found any available biological variation data for IgE.
eNA stands for not applicable.
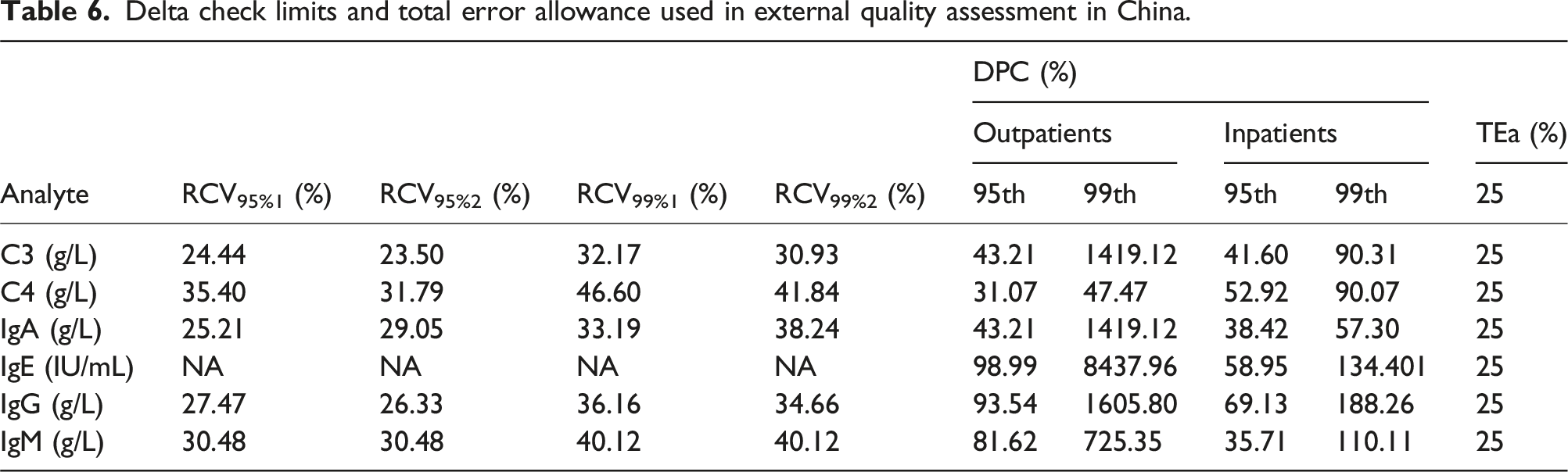
Delta check limits and total error allowance used in external quality assessment in China.
Discussion
Immunoglobulins (Igs) are important functional proteins that are produced by the body in response to infection, the importance of different classes of Ig varying in diseases. Complements are important molecules in non-specific immunity and are mainly present in the serum and tissue fluids of the organism. The production of complement involves a variety of tissues, with the liver being the main tissue involved. Depending on the half-life of Ig, which ranges from 3 to 6 weeks, continuous monitoring of Ig levels is required in hospitalized patients, and it is generally recommended that measurements be repeated after 28 days. 7 However, in this study, we find that many outpatients and inpatients have repeat measurements of immunoglobulins and complements within 7 days. The time interval for delta checks chosen for this study is 7 days and there is no definitive conclusion about the time interval for now.
Currently, there are several methods for setting delta check limits in clinical laboratories, each with its own advantages and disadvantages. The calculation of RCV which is based on biological variation and analytical imprecision is simple for laboratory personnel and numerous researchers have reported applying this methodology in their laboratories.2,3,14,15 However, according to our study, there are several issues with this method. First of all, not all analytes have available BV data for now, such as IgE in this article, of which the RCV will not be calculated due to the lack of relevant data. Secondly, the delta check limits set by RCVs appear to be more conservative, which may generate more false positive alarms in practical applications, resulting in alarm fatigue and waste of resources in clinical laboratories. Furthermore, the calculation of RCV only involves the biological variation and analytical imprecision of test items, which cannot estimate the impact of different patient types, such as inpatients and outpatients, whose RCV is the same. Meanwhile, due to the fact that internal quality control materials usually have more than one concentration level, often two or even three, multiple CVA may appear. As for which CVA to choose for RCV calculation, there is currently no definite conclusion. In our study, CVA is calculated by the equation
The advantage of using real patient test results for delta check limit setting is that it can set delta limits for each test item and is more in line with the actual situation of the clinical laboratories. However, the difficulty of this method lies in data statistics and analysis, which may require the participation of computer information technology personnel and cannot be independently completed by laboratory personnel, resulting in some additional costs. The sudden increases or decreases in the test results of patients may be accompanied by significant changes in the patient’s condition or fluctuations in laboratory performance. Therefore, in our study, DDs and DPCs are directly taken as absolute values without considering whether it is increasing or decreasing, which is the same as in some studies.4,16 Our approach can reduce the calculating pressure of the LIS and is practical for clinical laboratories with incomplete computer information system construction. In our study, we find that DDs of inpatients are significantly higher than those of outpatients for C4, IgE, IgG, and IgM. For DPCs, we get the same conclusion for C3, C4, IgE, IgG, and IgM. In practical clinical laboratory applications, the choice of DDs or DPCs can be determined based on the order of magnitude of the test results. For C3, C4, and IgM in this article, their test results are generally below 1 or 0.1 g/L, so DDs may be more appropriate for them. If applying DPCs for C3, C4, and IgM, even small patient result changes may cause huge DPC changes, which will mislead laboratory personnel to take unnecessary actions. For analytes with larger orders of magnitude of the test results, such as IgE and IgG in this article, DPCs may be more suitable.
One more issue we need to discuss is the index of individuality, which is calculated as
In recent years, autoverification has become increasingly important due to the progress in laboratory quality management and the development of information technology.15,21–24 We think delta checks may be more important in autoverification than in patient-based real-time quality control (PBRTQC),25,26 which involves some other patient-based quality controls, such as average of normal (AoN), moving average (MA), moving median (MM), and so on.27–29 The delta check limits obtained in this study can serve as a reference for the rules in autoverification in our laboratory. However, we would like to point out that autoverification is essentially the use of computer programs to mimic laboratory personnel in reviewing test reports, which can also identify errors that laboratory personnel may overlook, so as to improve the efficiency of workflow, turnaround time (TAT), and the performance of clinical laboratories. The problem is that when laboratory personnel review test results, it is itself a subjective process. For the same test results, some people believe that it can be directly reviewed, while others think that additional attention is needed. There are several causes for this disagreement: (1) Differences in knowledge and experience among laboratory personnel; (2) laboratory personnel’s attitude about their work; and (3) the expected clinical use of the measurands. Therefore, laboratory personnel should not excessively pursue higher pass rates when applying autoverification in their laboratories and autoverification should mainly focus on undisputed test results, leaving the controversial test results to laboratory personnel.
In summary, this study reported delta check limits for IgA, IgG, IgM, IgE, C3, and C4 based on patient and biological variation, as well as the index of individuality of these analytes, which could be applied in the quality control and autoverification procedure in our laboratory. The delta check is a useful tool to monitor potential errors which may occur in total testing process. We hope that the findings presented in this study can be helpful for future studies focused on delta checks in IgA, IgG, IgM, IgE, C3, and C4 and other immunological analytes.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Guarantor
Not applicable.
Contributorship
Xiangmei Gong and Shukang He wrote this article; Li Luo conducted the statistical analysis; Chunyu Ding, Xin Qin and Yilong Yuan conducted literature review; Pengcheng Cai and Lihua Yang reviewed this article. All authors contributed to the article and approved the submitted version.

