Abstract
Background
Cell-free DNA (cfDNA) is free DNA found in circulating blood that originates from apoptosis or necrosis, and elevated cfDNA concentrations have been reported in cancers and other diseases.
Methods
In this study, the concentrations and fragment distributions of plasma cfDNA were preliminary investigated in elderly (n = 1) and paediatric (n = 1) patients with acute promyelocytic leukaemia (APL) treated with arsenic trioxide (ATO).
Results
A slight increase in cfDNA concentrations was observed in the APL patients compared with healthy controls. The change in plasma cfDNA concentrations corresponded to the change in plasma arsenic concentrations during ATO treatment. The fragment distribution pattern did not differ before and during treatment. Three ladder fragments were observed in part of the cfDNA in the second consolidation therapy in an elderly APL patient and the first consolidation therapy of a paediatric APL patient, while two fragments were observed in all other treatment periods. Moreover, APL-related gene mutations were successfully genotyped from plasma cfDNA by using polymerase chain reaction-based methods and these results are consistent with those from leukocytes.
Conclusion
This study is the first to report the concentrations and fragment patterns of cfDNA from APL patients treated with ATO. The results suggested that plasma cfDNA concentration in APL patients increased with ATO treatment and that cfDNA is released mainly via neutrophil extracellular traps (and/or necrosis) in addition to apoptosis. To confirm whether cfDNA concentrations and fragment patterns can be used as a biomarker for APL treated with ATO, further accumulative data are needed.
Keywords
Introduction
Circulating cell-free DNA (cfDNA) is free DNA in blood plasma that originates from apoptosis or necrosis of all cell types.1–3 Cell-free DNA has been reported to be relevant in cancer, diabetes, stroke, systemic lupus erythematosus, trauma, rheumatoid arthritis, and myocardial infarction.4,5 We previously reported elevated levels and three DNA ladder fragments of plasma cfDNA in cardiac disease patients. 6 In addition, we observed considerably higher plasma cfDNA levels in postmortem subjects than in living subjects. 7 Recently, plasma cfDNA has been used as a liquid biopsy, which enables early diagnosis, treatment guidance, prognostic information, and tumour monitoring with minimal invasion.6,8 Furthermore, cfDNA is a potential biomarker for haematological diseases.8,9
Patients with acute promyelocytic leukaemia (APL) have coagulopathy, and their bone marrow morphology shows microgranular APL and t(15; 17) chromosomal translocations. Intractable APL can be effectively treated with arsenic trioxide (ATO),10–12 and a recent study reported that ATO induces high rates of complete haematologic remission and molecular remission, followed by a long relapse-free survival. 13 We have previously reported the urinary arsenicals and 8-hydroxy-2′-deoxyguanosine concentrations of APL patients following ATO treatment.11,12 Previous studies reported cfDNA in patients with haematologic malignancy.14–16 However, to our knowledge, there are no reports on the plasma cfDNA concentration and fragment pattern of APL patients treated with ATO. Therefore, in this study, we preliminarily investigated the plasma cfDNA concentrations and fragment patterns of two APL patients, one elderly and one paediatric, in order to evaluate the potential of cfDNA as a new biomarker for APL treated with ATO.
Materials and methods
Plasma samples of APL patients
An elderly Japanese man aged 85 years and a Japanese girl aged 4 years were diagnosed as having APL.9,10 Their bone marrow morphology showed microgranular APL, and chromosomal translocation t (15; 17) and PML-RARA fusion gene were observed. They were treated with ATO (Trisenox®; Nippon Shinyaku, Kyoto, Japan) at Shimane University Hospital after giving informed consent. ATO was intravenously administered at a dose of 0.15 mg/kg/day for 6 weeks for the induction therapy. Two courses of consolidation therapy (0.15 mg/kg/day, 3 days a week for 5 weeks) were administered with a 6-week interval after a 4-week withdrawal period. Plasma samples from the older patient were collected during the two consolidation therapies while those of the paediatric patient were collected during the induction therapy. Plasma control specimens were obtained from healthy Japanese volunteers (n = 76; mean age ± standard deviation, 36.8 ± 10.9 years). Written informed consent was obtained from the elderly patient, from the parents of the paediatric patient, and from the healthy volunteers. This study was approved by the Human Ethics Committee of Shimane University School of Medicine.
Extraction of cfDNA
Plasma cfDNA was extracted from 1 mL plasma by using the Maxwell® RSC ccfDNA Plasma Kit (Promega Corp., Madison, WI) with the Maxwell® RSC Instrument (Promega Corp.) in accordance with our previous studies.6,7 The cfDNA (2 μL) was plated on Thermo Scientific μDrop™ plates and concentrations were determined with a Multiskan™ GO Microplate Spectrophotometer (Thermo Fisher Scientific Inc., Waltham, MA) by measuring spectrophotometric absorbance at 260 nm, 280 nm, and 320 nm.
Mutation analysis
FLT3-ITD mutation, GATA2 rs2713604, JAK2 rs10974944, WT1 rs16754, KRAS, and NRAS genotypes in APL patients.
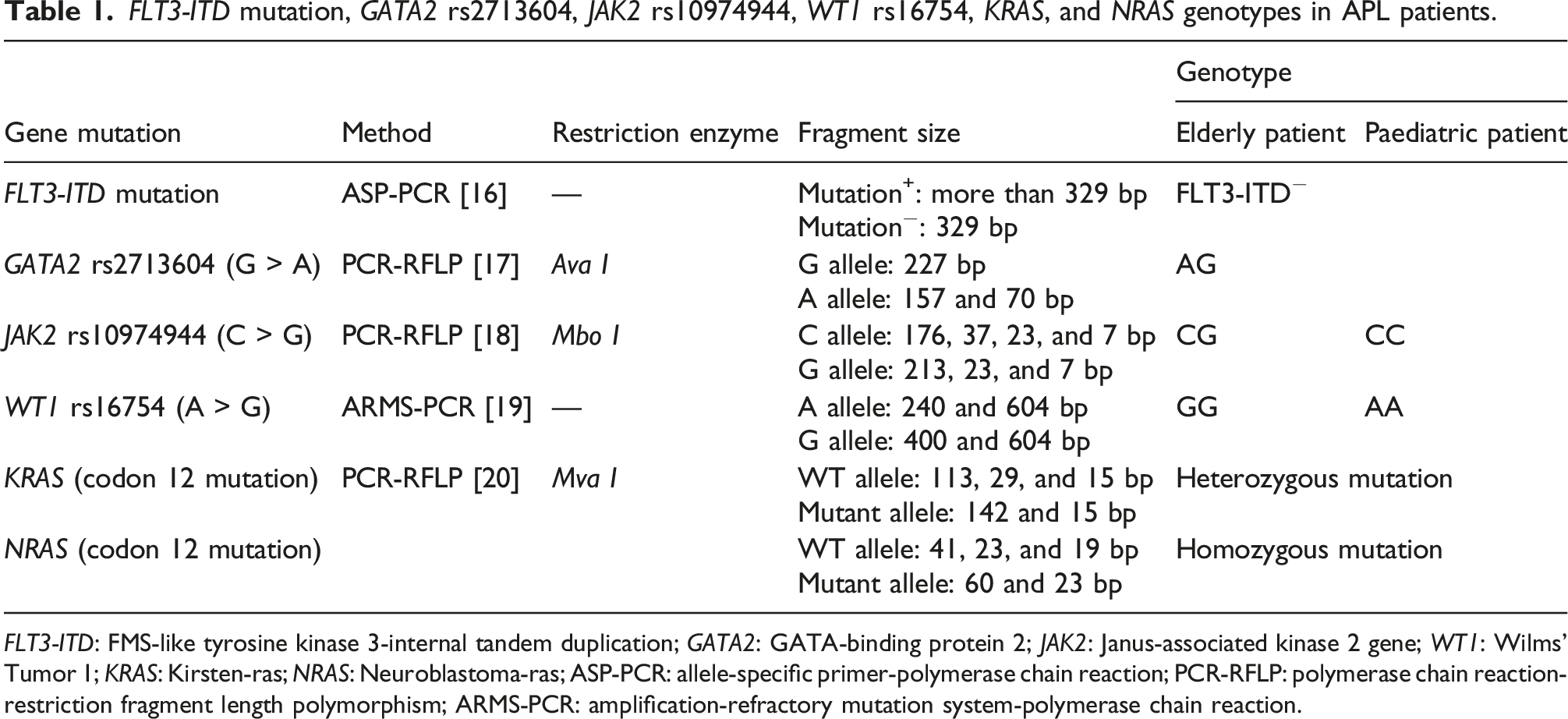
FLT3-ITD: FMS-like tyrosine kinase 3-internal tandem duplication; GATA2: GATA-binding protein 2; JAK2: Janus-associated kinase 2 gene; WT1: Wilms' Tumor 1; KRAS: Kirsten-ras; NRAS: Neuroblastoma-ras; ASP-PCR: allele-specific primer-polymerase chain reaction; PCR-RFLP: polymerase chain reaction-restriction fragment length polymorphism; ARMS-PCR: amplification-refractory mutation system-polymerase chain reaction.
Microchip electrophoresis
Plasma cfDNA was electrophoresed using an MCE-202 MultiNA automated microchip-based electrophoresis system equipped with MultiNA Viewer software (Shimadzu Corp., Kyoto, Japan) in accordance with our previous studies.6,7 Cell-free DNA samples were run with separation buffer reagents and DNA marker reagent (DNA-1000 kit; Shimadzu), and a 100-bp DNA ladder (Takara Bio, Inc., Shiga, Japan) was used as a marker. Amplified cfDNA samples for gene mutations were run with the separation buffer reagents and DNA marker reagent (DNA-500 kit; Shimadzu), and 25 bp DNA Step Ladder (Promega Corp.) was used as a marker.
Total arsenic analysis
The total arsenic concentration in plasma was determined by high-performance liquid chromatography (LC10A Series, Shimadzu) coupled with inductively coupled plasma mass spectrometry (HP-4500; Hewlett-Packard, Avondale, PA) in accordance with our previous reports.11,12 Plasma was filtered and analyzed without dilution. The concentrations of total arsenic in plasma are expressed as μg/mL.
Statistical analysis
The differences in cfDNA concentrations between the control group and APL patients were analyzed with Dunnett’s test. In addition, the correlation between arsenic and the cfDNA concentration was analyzed with Spearman’s rank correlation test. These analyses were performed by using Bell Curve for Excel (Social Survey Research Information Co., Ltd., Tokyo, Japan).
Results and discussion
cfDNA concentrations
Cell-free DNA levels in the cerebrospinal fluid from leptomeningeal metastasis patients with haematologic malignancies have been reported to be about 10 times higher than those of the controls.
15
We previously reported elevated cfDNA concentrations in patients with cardiac disease (9-fold) compared with control subjects: among cardiac diseases, cfDNA concentrations in myocardial infarction and cardiac angina were significantly elevated.
6
The cfDNA concentrations of our two APL patients treated with ATO were compared at all time points with those of the control subjects (Figure 1). Although the difference was small, the cfDNA concentration in the APL patients was significantly higher than that of the control subjects (2-fold). Box plots of the plasma cfDNA concentrations in plasma from healthy control subjects and APL patients. *p < .05 and **p < .01 when compared with the control group using Dunnett’s test.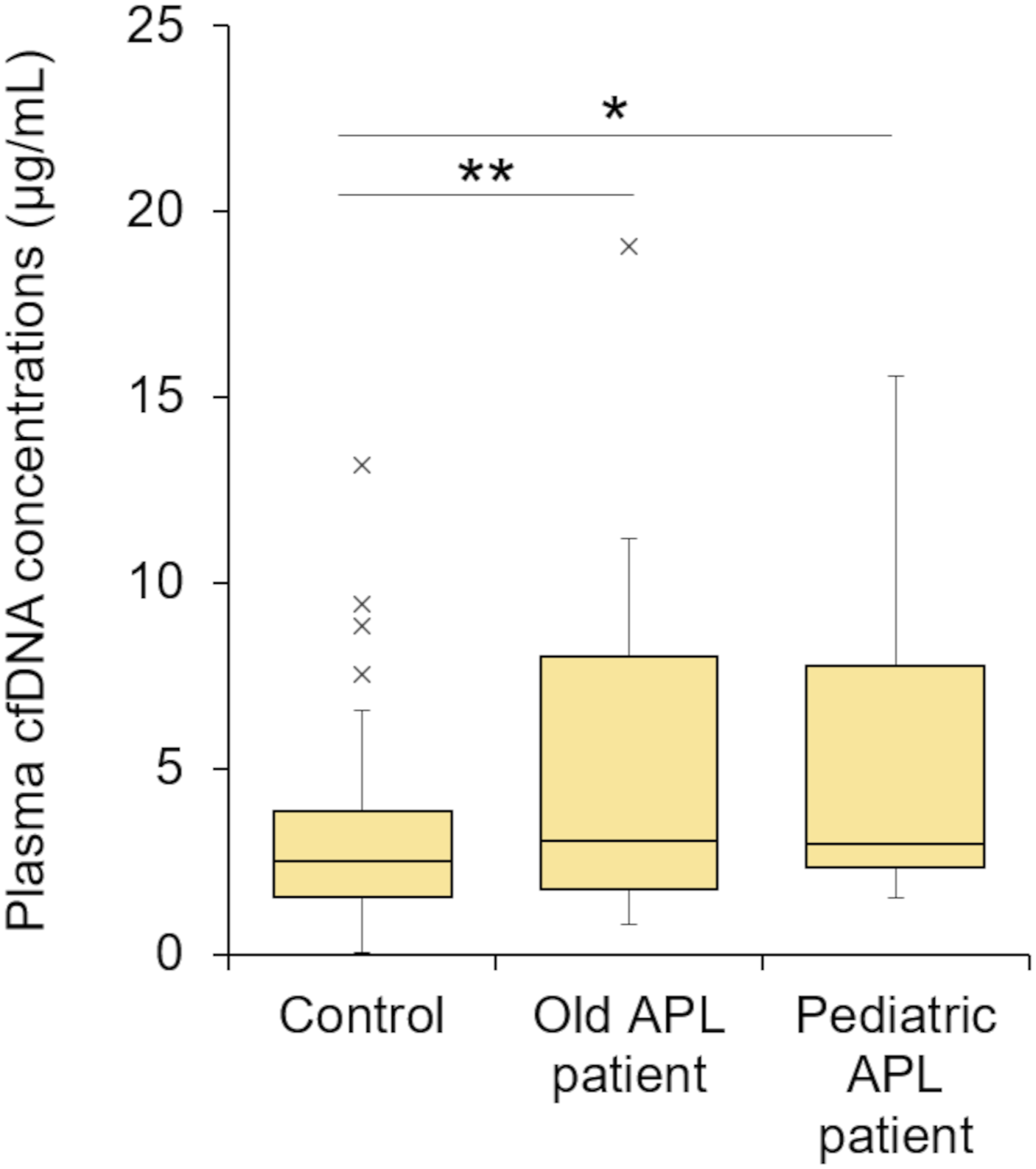
Previous work showed elevated plasma cfDNA levels during the first 3 weeks of induction therapy, with the highest levels observed at week 1 in acute lymphoblastic leukaemia patients.
16
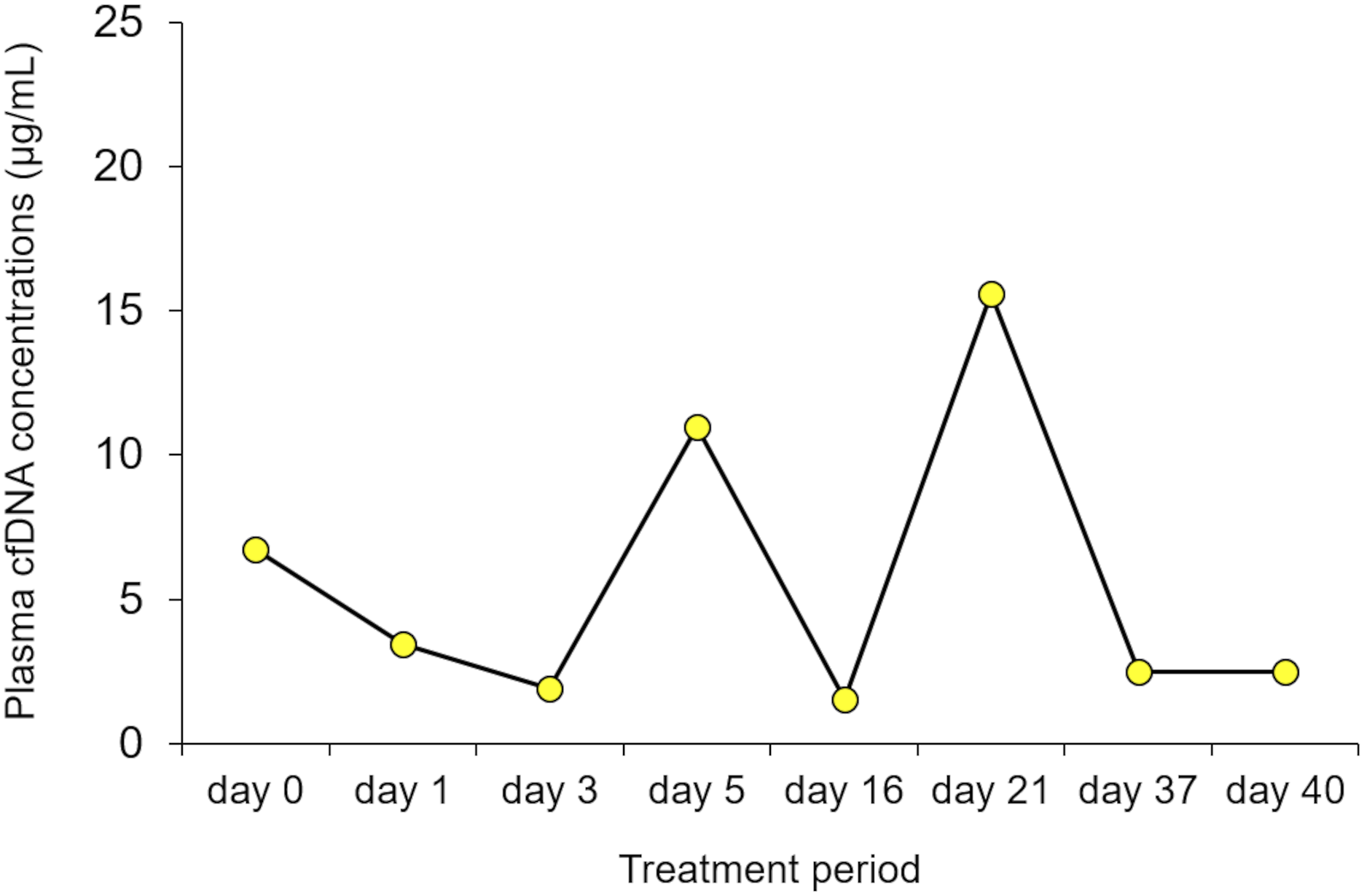
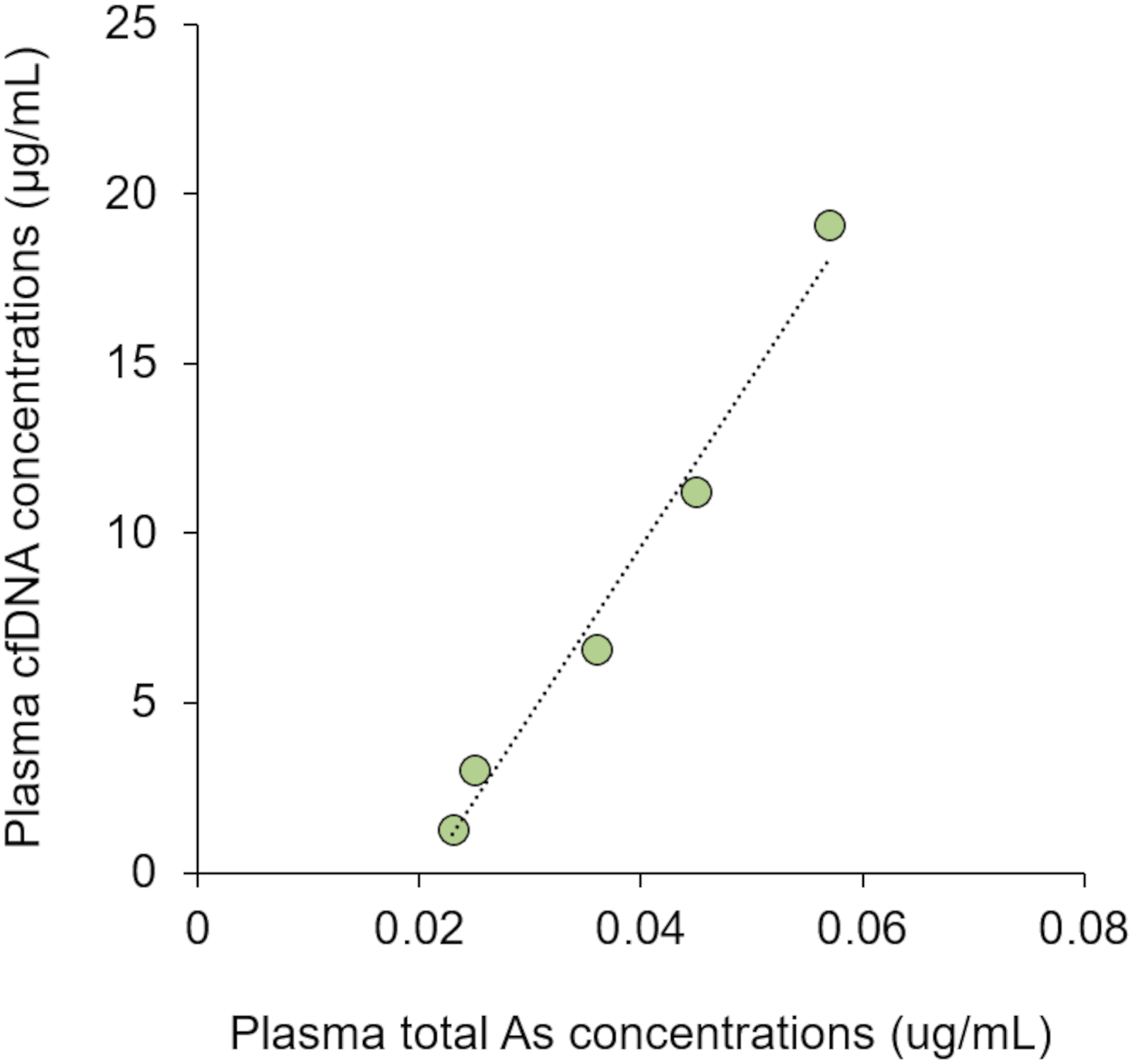
In this study, time course changes in plasma cfDNA levels in APL patients during the ATO therapy were investigated (Figures 2 and 3). The plasma cfDNA concentration in the elderly APL patient increased with treatment duration during the first consolidation therapy, and the plasma cfDNA concentration fluctuated during the second consolidation therapy (Figure 2). Similar to the second consolidation therapy of the elderly patient, the plasma cfDNA concentration in the paediatric APL patient fluctuated during the induction therapy (Figure 3). The discrepancy between the results shown in Figure 2(a) and those in Figures 2(b) and 3 is related to the plasma total arsenic concentration, which showed a tendency similar to that of the cfDNA concentration (data not shown). Although no correlations were observed between plasma total arsenic and cfDNA concentrations in the second consolidation therapy in the elderly patient or the first consolidation therapy in the paediatric patient, significant positive correlations were observed between the plasma total arsenic concentration and cfDNA concentration during the first consolidation therapy in the elderly APL patient (Figure 4; Spearman’s rank correlation, r = 1.000, n = 5). Time course change in the plasma cfDNA concentrations of the elderly patient with acute promyelocytic leukaemia during the (a) first consolidation therapies and (b) second consolidation therapies with arsenic trioxide. Time course change in the plasma cfDNA concentrations of the paediatric patient with acute promyelocytic leukaemia during the induction stage of arsenic trioxide therapy. Relationship between plasma total arsenic and plasma cfDNA concentrations in the elderly acute promyelocytic leukaemia patient during the first consolidation therapy with arsenic trioxide.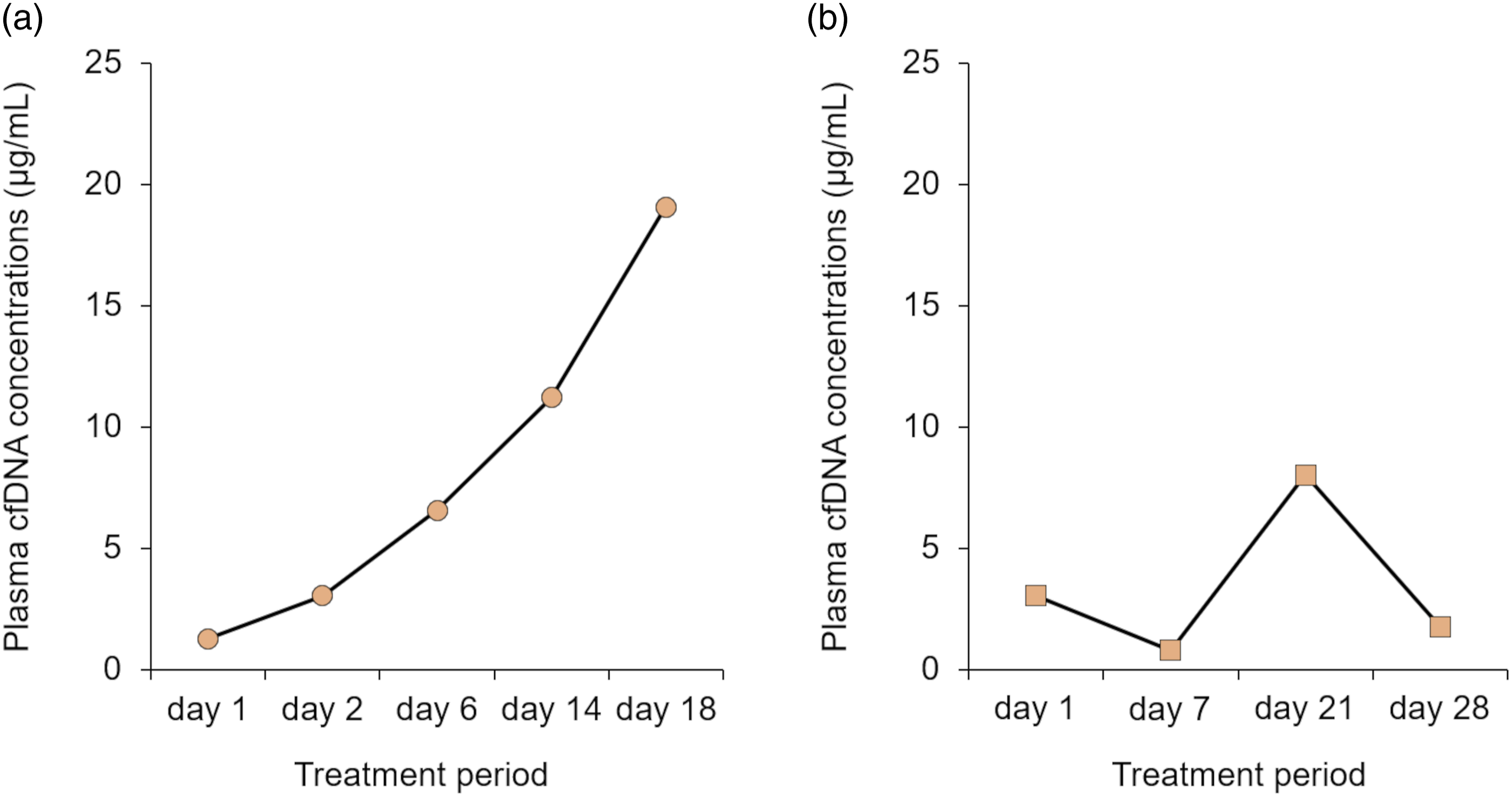
The formation of neutrophil extracellular traps (NETs) is a cell death pathway distinct from apoptosis that is called NETosis, and cfDNA is also presumed to be released via NETosis, in addition to apoptosis.22,23 Plasma cfDNA is used as a marker of NET formation. 24 We recently reported that cfDNA is chiefly released by neutrophils via NETosis, in addition to necrosis, in cardiac disease patients. 25 A previous in vitro study demonstrated that APL cells from patients undergo NETosis (ETosis) through autophagy induced by ATO treatment. 26 Disseminated intravascular coagulation (DIC) is highly prevalent in APL (up to 90% of cases satisfy the criteria for DIC), and APL-associated DIC is dominated by bleeding. 27 Although no studies have investigated the relationship between DIC and NETosis in APL, NETosis may provide additional ways of modifying the course of DIC in patients with acute leukaemia. 27
Fragment distribution of cfDNA
Evaluation of cfDNA fragmentation is a relatively new application, and cfDNA analysis has the potential to help identify cancers and cancer type.
28
We have previously reported the presence of three ladder fragments – 150–200 bp, 300–400 bp, and 500–600 bp – in plasma cfDNA in almost all cardiac disease patients and that the 150–200 bp/500–600 bp ratio was higher in myocardial infarction patients than in other cardiac disease patients. Here, we subjected plasma cfDNA from APL patients to microchip electrophoresis, and gel-like images are shown in Figure 5. Three ladder fragments – 150–200 bp, 300–400 bp, and 500–600 bp – were observed on day 7 and day 21 of the second consolidation therapy in the elderly patient (Figure 5(a)) and on day 3, day 5, day 21, and day 40 of the induction therapy in the paediatric patient (Figure 5(b)). In all other treatment periods, two fragments (150–200 bp and 300–400 bp) were observed, whereas 500–600 bp fragments were rarely observed (Figure 5). The bands detected on days 6 and 18 of the second consolidation therapy in the elderly patient were weak (Figure 5(a)). In contrast, the cfDNA concentrations of these samples were high (Figure 2(a)). This discrepancy may due to the fact that the cfDNA on days 6 and 18 was released mainly from NETosis (and/or necrosis), not by apoptosis. The three ladder fragments of cfDNA are characteristic of apoptotic cell death,
29
but apoptotic DNA fragments might be shorter (about 140–200 bp).
30
In contrast, long DNA fragments are released from neutrophils,
31
and necrotic cells generate mainly cfDNA fragments larger than 10,000 bp.
32
Present methods cannot observe fragments over 1,000 bp, and thus no cfDNA fragments released from NETosis (and/or necrosis) were observed. Gel-like image of plasma cfDNA from (a) elderly and (b) paediatric acute promyelocytic leukaemia patients treated with arsenic trioxide. Marker: 100-bp ladder marker.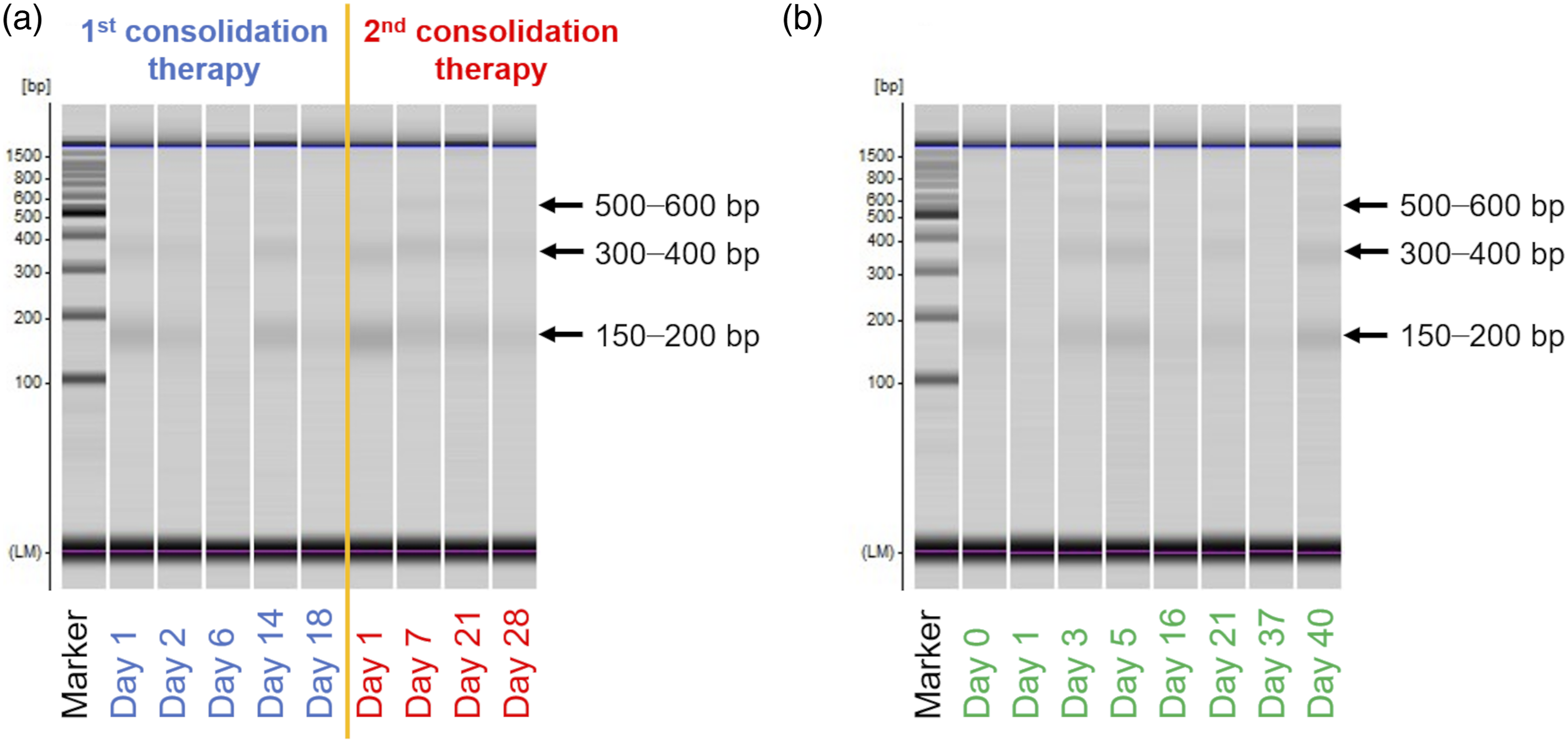
Plasma sample before treatment is available only for the paediatric patient, and two bands were observed in this sample (Figure 2(b)). There was no difference between the ladder pattern before and during treatment. As mentioned above, three ladder fragments were observed in part of the cfDNA in the second consolidation therapy in the elderly patient as well as the first consolidation therapy in the paediatric patient. This suggests that the fragment pattern of cfDNA does not reflect the treatment period or the cfDNA concentrations. This study is preliminary and thus further research with larger numbers of samples is needed to confirm whether cfDNA can in fact be used as a biomarker for APL treated with ATO.
Mutation analysis
Recent studies reported gene mutations in FLT3-ITD, GATA, JAK2, WT1, KRAS, and NRAS in APL patients.33–35 In this study, gene mutations in these genes were assayed using cfDNA from APL patients (Table 1). Genotyping using cfDNA from APL patients was successful. Both patients were negative for the FLT3-ITD mutation and heterozygous for GATA rs273164. The elderly patient was heterozygous for JAK2 rs10974944 and the paediatric patient was homozygous for the wild-type. As for WT1 rs16754, the elderly patient was homozygous for the mutant allele and the paediatric patient was homozygous for the wild-type allele. Both patients were heterozygous for the KRAS mutation and homozygous for the NRAS mutation. To confirm that the genotypes obtained from cfDNA are consistent with those obtained from DNA in leukocytes, gene mutation analysis was also performed using DNA extracted from whole blood. All of the mutation analysis results for cfDNA were consistent with those of DNA obtained from leukocytes (data not shown).
Conclusion
In this study, plasma concentrations of cfDNA and its fragment distribution were preliminarily investigated in elderly and paediatric APL patients during ATO treatment. The cfDNA concentrations were significantly higher in APL patients than in controls. The change in plasma cfDNA concentration during ATO therapy corresponded to the change in the plasma total arsenic concentrations. No difference was observed in fragment distribution pattern before and during treatment. Three ladder fragments (150–200 bp, 300–400 bp, and 500–600 bp) were observed in part of the cfDNA in the second consolidation therapy in the elderly patient as well as the first consolidation therapy in the paediatric patient, while two fragments (150–200 bp and 300–400 bp) were observed in all other treatment periods. Moreover, the band densities did not correspond to the cfDNA concentrations. These results suggested that plasma cfDNA in APL patients may be released mainly via NETosis (and/or necrosis) in response to ATO treatment in addition to apoptosis. Using cfDNA, APL-related mutations could be genotyped, which were consistent with those of leucocytes. This study is the first to report the concentrations and fragment patterns of the cfDNA from APL patients treated with ATO. To confirm whether cfDNA concentrations and fragment patterns can be used as a biomarker for APL patients treated with ATO, further accumulative data are needed. Cell-free DNA analysis may become part of clinical practice. The results of this study provide fundamental information on the cfDNA of APL patients treated with ATO.
Footnotes
Acknowledgements
The authors would like to thank Dr. J. Tanaka and Dr. R. Kanai for assistance in collecting samples.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a Japan Society for the Promotion of Science Grant-in-Aid for Scientific Research (B) (grant number 21H03212).
Ethical approval
The study, including usage of plasma derived from patients and control subjects, was reviewed and approved by the Human Ethics Committee of Shimane University School of Medicine and was conducted in accordance with the principles of the Declaration of Helsinki.
Guarantor
JF.
Contributorship
JF performed the formal analysis and wrote the original draft. NN reviewed and edited the article. HT contributed to the sample collection. All the authors approved the final version of the article for submission.
