Abstract
Background
Delayed time from collection to centrifugation may cause erroneously high lactate levels in vitro (from continued blood cell metabolism under anaerobic conditions in the collection tube) if not collected in appropriate collection devices, consequently increasing the risk for inappropriate patient care or harm. We undertook a study to determine the turnaround time for lactate testing in a tertiary care setting and also performed short- and long-term lactate stability studies in blood collected in sodium fluoride/potassium oxalate (NaF/KOx) collection tubes.
Methods
The hospital lab information system was mined for 6 months to determine patient samples that may have exceeded the time from collection-to-receival in lab of 15-min. Lactate stability was evaluated in unspun NaF/KOx collection tubes at 15 min intervals for to 2 h; and separately at 2, 6, 12, 24, and 48-h post-collection.
Results
A total of 8,929 plasma samples were collected in 6 months, and 1/3 were not received in the lab within 15 min from collection. In NaF/KOx additive, lactate levels had minor increases over 2 h, and incremental increases at an average rate of 0.0035 mmol/L/h over 48 h with maximum increase of 9.8% at 48 h. However, the average change across all time points were within local allowable performance goals (at ≤4 mmol/L ± 0.5 mmol/L; at >4 mmol/L ± 12%).
Conclusion
A small proportion of lactate specimens may experience delay in processing. Although lactate levels may incrementally increase over 48-h at room temperature in unspun NaF/KOx collection tubes, the changes may not be clinically impactful.
Introduction
The metabolism of glucose in cells during anaerobic respiration results in the production of intermediary lactate, adenosine triphosphate (ATP) and water. The ATP is subsequently used for energy, which helps drive cellular function. 1 Uncorrected hyperlactatemia results in metabolic acidosis, a severe life-threatening medical condition. Lactate levels are used clinically to monitor critically ill patients, particularly those with sepsis, septic shock, tissue perfusion perturbation and metabolic derangements.1–6 Thus, providing timely, accurate and precise lactate test results is important for all laboratory and clinical stakeholders.
Delayed time from collection to centrifugation results in erroneously high lactate levels in vitro in blood (from continued blood cell metabolism under anaerobic conditions in the collection tube) if not collected in an appropriate glycolic inhibitor additive, consequently increasing the risk for inappropriate patient care or harm. 7 The ideal time from collection to centrifugation for lactate remains a matter of debate, with clinical laboratories striving for 15–30 min. 7 Point-of-Care Testing (POCT) devices, such as blood gas instruments for example, eliminate the need for centrifugation and the results are often available quickly at the bedside of the patient. 8 However, not all institutions have POCT devices. Furthermore, testing of lactate in a plasma matrix using a core laboratory instrument remains a common and preferred practice due to better quality management oversight of the testing process. Blood collection devices containing lithium heparin or sodium fluoride/potassium oxalate (NaF/KOx) additives are commonly used and are usually delivered to the laboratory at room temperature or on ice, and are usually centrifuged within 15–30 min from collection.9–11 However, this practice is not standardized, and some laboratories have even extended the allowed turnaround time (TAT) interval to 45 min or 1 h, and some even longer, without having the appropriate evidence to support this practice. Furthermore, many large tertiary care centres are also reference sites, performing referred-in work from remote laboratories or clinical stations that may not have the appropriate pre-analytical equipment or technical oversight to process blood specimens (e.g. centrifugation of whole blood to isolate plasma for send out testing), thus furthering the delay for specimen processing and testing. Unlike NaF/KOx, lithium heparin does not inhibit metabolism of cells, which results in increased production of lactate in vitro over time. A recent 6-year retrospective study evaluated lactate results from NaF/KOx versus heparinized whole blood and determined that NaF/KOx lactate were lower than heparinized lactate by 20%, and the stability was evaluated to be acceptable within clinical decision-making limits for 120 min for Na/KOx plasma samples. 12 Since most manufacturers of hospital-based laboratory instruments have validated the use of the NaF/KOx additive in their assays, use of NaF/KOx fortified collection tubes for labile tests, such as lactate, is increasing. However, the long-term stability of lactate in uncentrifuged whole blood collected in NaF/KOx additive is weak.
Therefore, the aim of the present study is to retrospectively examine lactate TAT in a large tertiary care centre, and to prospectively evaluate lactate stability in NaF/KOx whole blood at room temperature over a short term (120 min) and long term (48 h) period of time, to reflect scenarios encountered in both acute care hospitals and referred-in/reference site laboratories.
Material and methods
The work was conducted as a laboratory quality improvement project without use of actual or personal information from patients and was deemed exempt from institutional research ethics board review.
Specimen collection to receipt turnaround time
To determine TAT of lactate received in our laboratory, which is part of a large tertiary acute care centre, we reviewed the hospital lab information system (LIS; Cerner Millennium) for all lactate orders. All core laboratory lactate orders (excluding blood gas lactate conducted by point of care testing) were reviewed for 6 months period of time, from 1 May 2019 to 31 October 2019. Data was stratified based on time of collection to receival in lab for centrifugation, and was based on the time interval of ≤15 min or >15 min (based on institutional practice), and further stratified according to 15-min intervals for up to 2 h.
Lactate stability
Extra whole blood specimens collected in Becton Dickinson (BD) Vacutainers 13 × 75 mm, 4.0 mL grey-top (BD Cat# 368921) containing 2.5 mg/mL sodium fluoride/2.0 mg/mL potassium oxalate (NaF/KOx) were received in the laboratory from in-patient clinical areas with varying acuity of patients were anonymized and used in this study. Approximately 0.5 mL aliquots of whole blood from 15 different patient samples (representing 15 biological replicates) were transferred in eight labelled Roche Diagnostics 13 × 75 mm standard round bottom clear aliquot tubes (baseline, 15, 30, 45, 60, 75, 90 and 120 min), capped, and the whole blood stored untouched, at room temperature (21–23°C), under standard fluorescent lighting conditions. The first aliquot was centrifuged and plasma tested immediately, while the remaining seven were tested at their respective time intervals. After their respective time period, each whole blood sample was then centrifuged and the plasma was immediately analysed for lactate in single technical measurements. In a separate experiment, additional 10 samples (representing 10 biological replicates) were evaluated at baseline (<15 min from collection), 2, 6, 12, 24 and 48-h post-collection and were handled in the same manner as above. Lactate was measured using the Roche L-Lactate (LAC) colorimetric enzymatic (lactate oxidase/peroxidase) method on the Roche c702 (Roche Diagnostics Corp., Indianapolis, IN, USA) and all samples were analysed as per the manufacturer’s instructions for use. The analytical measurement range (AMR) of the lactate assay is 0.2–15.5 mmol/L and contains a single reference interval of 0.5–2.2 mmol/L. Three levels of commercially prepared, matrix-appropriate, quality controls are performed daily, with long-term stable performance of the assay as follows: Level 1 (mean of 1.51 mmol/L) is 1.6%, Level 2 (mean of 3.56 mmol/L) is 1.3% and Level 3 (mean of 5.94 mmol/L) is 1.4%. The achieved precision of the lactate assay is well within the institution’s precision goal of ±4.0% across the AMR. The allowable performance limits and acceptability of results between time points were assessed in accordance with the Accreditation Canada Diagnostics/Institute of Quality Management in Healthcare Canada and the Royal College of Pathologists of Australia (at ≤4 mmol/L ± 0.5 mmol/L; at >4 mmol/L ± 12%). The first lactate result (the 15-min time period) was set as the baseline result and the mean absolute and mean percent differences from baseline (±SD error bars) are represented in the figures. Changes over time were also evaluated for statistically significant differences using ANOVA with repeated measure and Geisser–Greenhouse correction method, a p < .05 was considered to be statistically significant.
Results
Lactate turnaround time
Our LIS data mining revealed that in a 6-month period, 8,929 patient specimens were received in the core lab for lactate testing, of which 69% of the specimens made it to the core lab within 15 min of collection (Figure 1). Approximately 30% of specimens arrived in the lab after 15 min, which represents a significant proportion of samples not meeting internal policy for rapid turnaround. About 82% of all specimens made it to the laboratory within 30 min from collection, 88% within 45 min, 91% within 60 min and 98% within 2 h. Therefore ∼2% of specimens exceeded 2 h. Six-month lab information system audit of patient samples from time of collection to receival in lab for centrifugation. Results are stratified based on 15 min intervals until maximum time of 2 h from collection to analysis. N = 8929.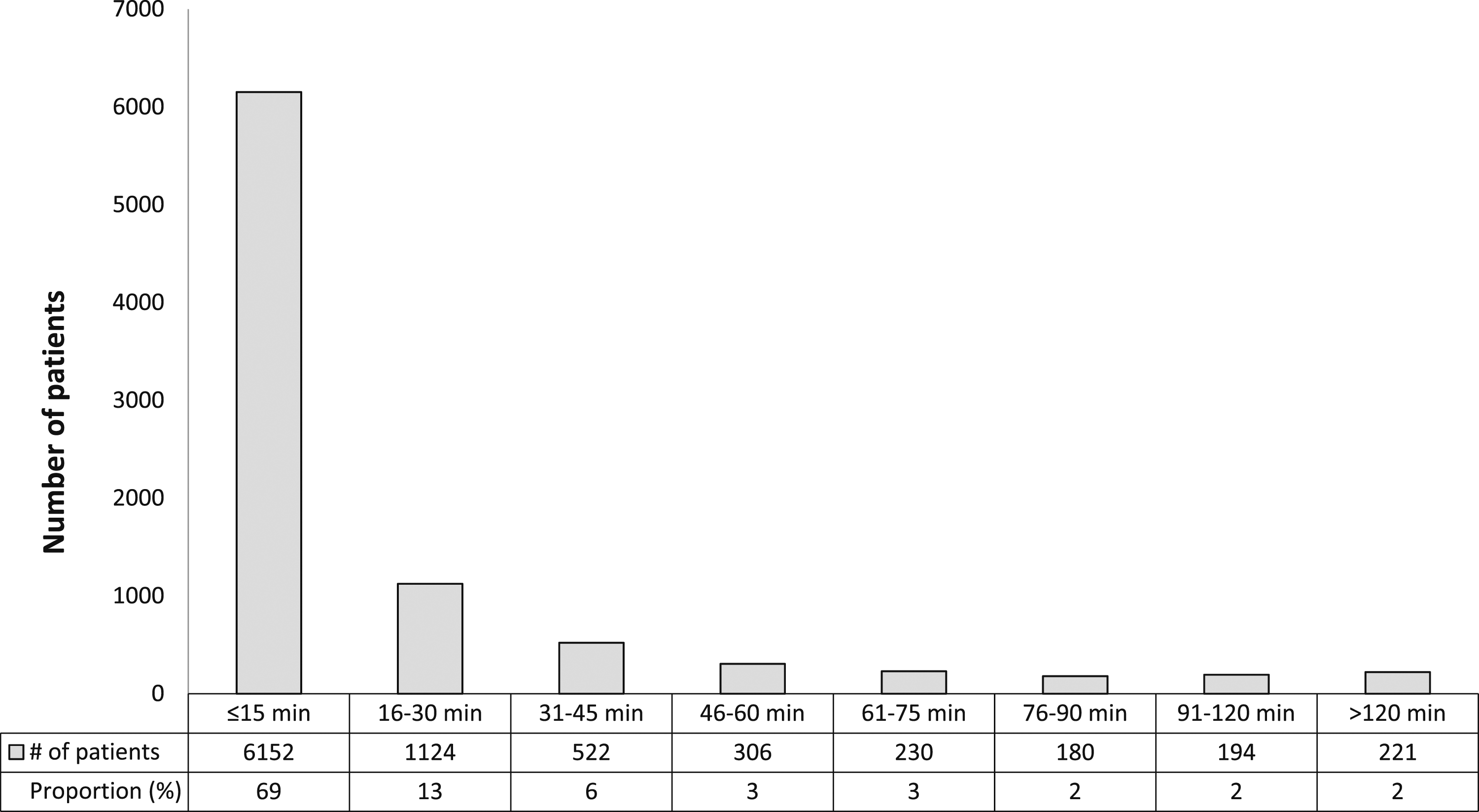
Lactate stability
Our study demonstrates that lactate is very stable in whole blood (uncentrifuged tubes) at room temperature for 120 min when collected in NaF/KOx collection tubes (Figure 2). From 15 to 75 min, lactate levels did not change significantly (percent differences ranged from −0.61% to +0.49%). At 90 min, the average lactate levels increased by 3.1% (range −2.3% to 10.5%, p < .001) and by 120 min it increased by 4.1% (Range 0.8% to 8.1%, p < .01). Although, the 90 and 120 minutes lactate values were statistically significant relative to baseline, the magnitude of the lactate changes were near the internal precision limits of the assay (±4.0%) and it would not be considered a clinically significant change. Lactate stability over time for up to 120 min, N = 15. (a) Mean absolute change from baseline and (b) Individual lactate levels over time. Allowable performance limit: For <4 mmol/L = ±0.5 mmol/L; ≥4 mmol/L = ±12%. Whole blood specimens were stored at room temperature for their respected time before centrifugation and immediate analysis. * = p < .001 relative to baseline.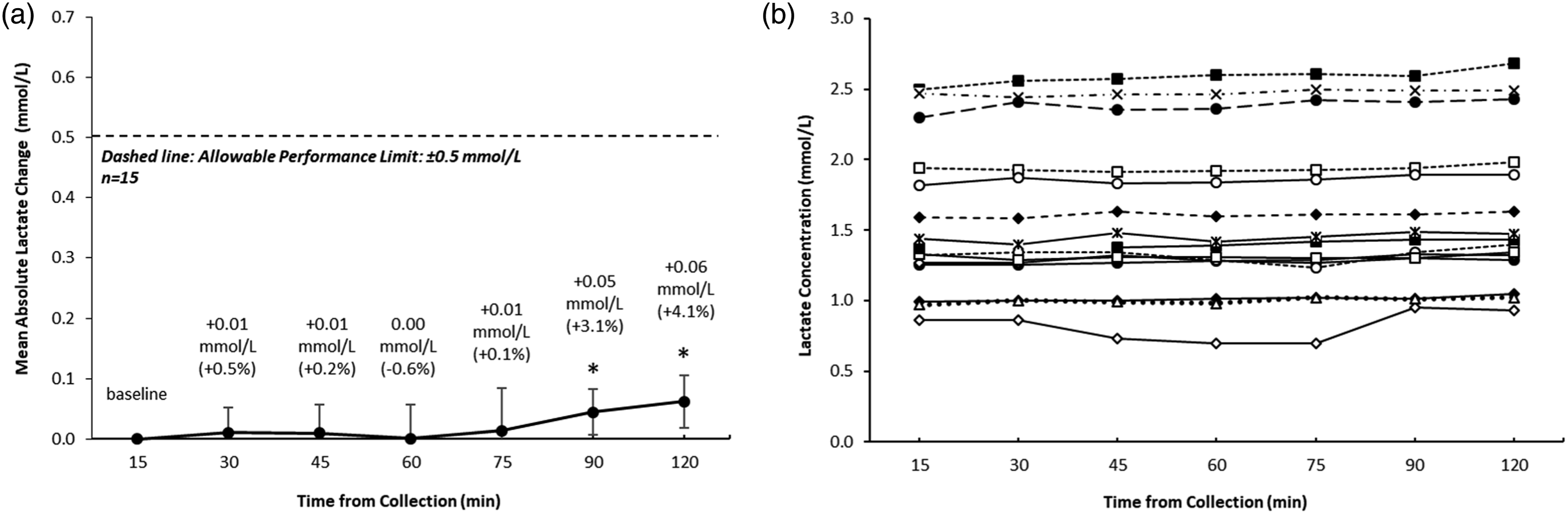
We further determined that lactate is reasonably stable in NaF/KOx tubes for up to 48 h post-collection (Figure 3). There was an appreciable, time-dependent average increase in lactate from baseline to 48 h at a rate of 0.0035 mmol/L/h. The 6 (+4.7%), 12 (+8.0%), 24 (+6.8%) and 48 (+9.8%) hour time points were statistically significant (p < .05) compared to baseline, but the 2 h was not significant. Even though the lactate increased over time, all of the incremental increases were within the allowable performance limit (<4 mmol/L = ±0.5 mmol/L; ≥4 mmol/L = ±12%) which would not be considered clinically significant. Lactate stability over time for up to 48 h, N = 10. (a) Mean absolute change from baseline and (b) Individual lactate levels over time. Allowable performance limit: For <4 mmol/L = ±0.5 mmol/L; ≥4 mmol/L = ±12%. Whole blood specimens were stored at room temperature for their respected time before centrifugation and immediate analysis. * = p < .05 relative to baseline.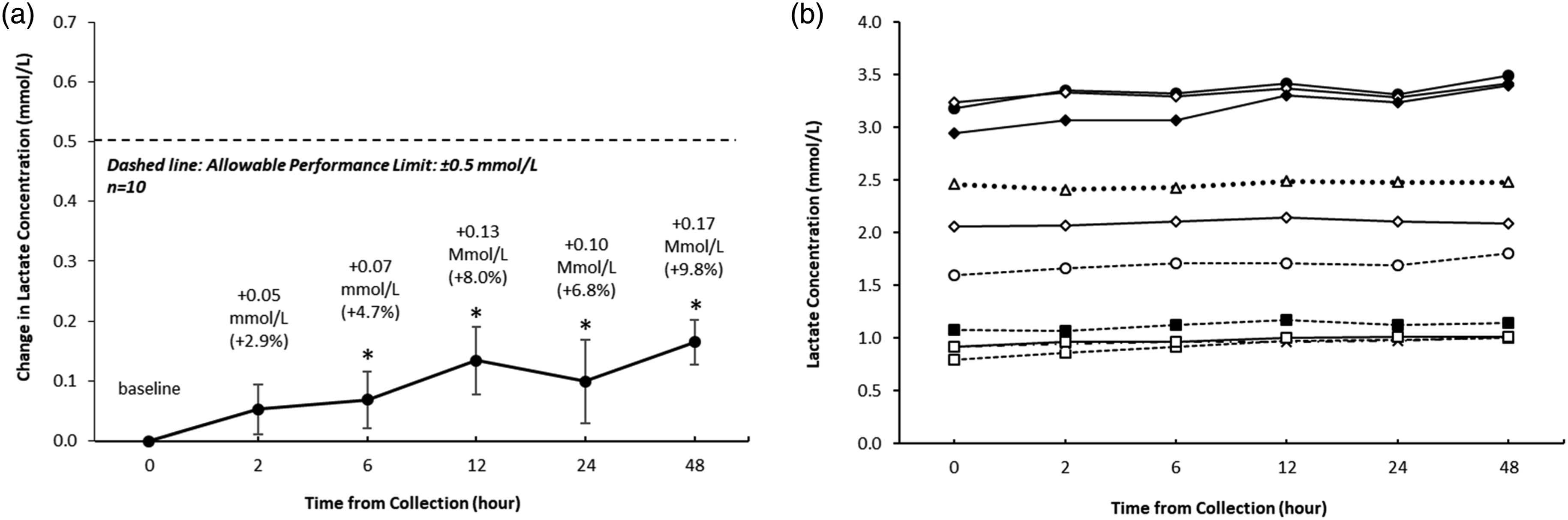
Discussion
Given the lack of standardization for pre-analytical processes, particularly from time of collection to centrifugation of time sensitive analytes, such as lactate, we sought to determine the proportion of patient samples that would experience delay in the pre-analytical processing. We also evaluated short- and long-term stability of lactate in whole blood that was collected in sodium fluoride/potassium oxalate (NaF/KOx) additive from hospitalized patients of a large tertiary acute care hospital.
Our data shows that an appreciable proportion of patient specimens (about 20%) may experience delay in processing (>30 min) with a small proportion of specimens exceeding 2 h of delay (2%), which may lead to erroneous lactate measurements if the appropriate blood collection additive is not properly selected. Furthermore, depending on individual institutional policies some patient specimens that arrive to the laboratory after the acceptability time (e.g. 30 min post collection) may be cancelled to avoid erroneous results from being resulted out. This may delay the results for clinical use, require recollection of patients, increases the cost of personnel time and consumables, and certainly may increase the risk for patient harm. 7
Several studies have examined stability of lactate in whole blood, plasma or serum, with either heparin or fluorinated collection tubes7,9,10,12–14; however, the conditions outlined in these studies failed to fully reflect the delayed timing conditions experienced by some remote/rural laboratories or nursing stations with pre-analytical constraints (e.g. not having centrifuges for rapid separation of plasma from cells). Some studies have shown that lactate was stable in heparinized tubes for 15–30 min post-collection (stored at room temperature or 4°C), 7 whereas others have demonstrated that lactate increased in heparinized tubes within the 15–30 min 13 and 120 min 12 post-collection, thus adding to variability of information. It is in general agreement that non-glycolytic inhibitor additives will result in erroneous increase in lactate over time, as such, glycolytic inhibitor additives are preferred for lactate testing. A recent study found that lactate was relatively stable in whole blood collected in NaF/KOx additive when stored at room temperature and centrifuged late at 48 h post collection. 14 Although their results are in line with our observations, the study did not evaluate lactate stability at incremental time points and the subjects used were normal healthy volunteers and not in-patients from a tertiary/quaternary care hospital. Another recent study evaluating retrospective lactate practices in a 6-year span concluded that lactate in NaF/KOx plasma was stable for 120 min, however limited information was provided beyond this time frame and the study has questioned the need for longer time points. 12 Lastly, our lactate results are also in line with other time-sensitive analytes collected in NaF/KOx tubes, such as glucose, where the delay from collection to centrifugation was acceptable beyond 2 h (4 to 48 h) as glycolysis was effectively inhibited.15,16
One important limitation of the present study is ∼40% of the samples studied over the 48 h had lactate levels in the abnormal range (greater than reference interval), and there were no samples in the extremely high range. Kinetics of lactate stability may change at extreme levels; this assessment was outside the scope of this study.
Conclusion
Overall, our study demonstrates that a significant proportion of patient specimens are not received in the laboratory within 15–30 min. Furthermore, our data supports the use of NaF/KOx for lactate measurements if delay in the pre-analytical processing is expected. We observed minor changes of lactate over 2 h and statistically significant incremental increases over 48 h; however, depending on local institutional allowable performance limits, the increases may not be clinically significant. Collecting blood in NaF/KOx additive would extend the specimen collection and acceptability criteria for lactate, which in turn, should lead to reduction of erroneous lactate results, decrease lactate cancellations and recollections, decrease unnecessary cost, and improve overall patient safety and care.
Footnotes
Acknowledgements
We thank the core lab and specimen receiving area staff at LHSC for their hard work with processing and testing specimens.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work was conducted as a laboratory quality improvement project without use of actual or personal information from patients and was deemed exempt from institutional research ethics board review.
Guarantor
Ivan Stevic.
Contributorship
IS drafted the manuscript. All authors have critically appraised and reviewed the manuscript and have approved the manuscript for publication. IS and VB conceptualized and designed the study. JB and RM performed patient analysis, provisioned materials and patients, and provided administrative support. IS and VB performed data analysis and all authors assisted with interpretation of data.
