Abstract
Introduction
Anti-thyroid stimulating hormone receptor antibody (TRAb) stability is stated as 7h at 20–25°C in the Roche Elecsys assay kit insert. The purpose of this study was to determine TRAb stability in whole blood and serum to assess the suitability of samples for reflective and weekly batch testing (with a single freeze-thaw cycle).
Methods
In the first study, blood from n = 5 volunteers was used to assess: (1) stability in whole blood at room temperature up to 24h, and (2) stability in serum at 4–8°C up to 72h. In the second study, n = 21 patient samples were analysed in serum stored at 4–8°C for two and five days post-preliminary analysis.
Results
There was a statistically significant decrease in TRAb concentration caused by the single freeze-thaw cycle of −8.9% ± 5.2% (p = 0.03). TRAb concentration decreased in whole blood between 0 and 24h by −16.5% ±9.2%, and declined in serum over time by −11.6% ±6.6% (at 12h). In the patient samples, serum TRAb concentration decreased over time by −4.6% ± 2.5% at day two and −6.5% ± 4.0% at day five.
Conclusion
TRAb concentration decreases over time in both whole blood at room temperature and serum samples stored at 4–8°C. A single freeze-thaw cycle also has a statistically significant effect on TRAb concentration.
Introduction
There was a change to stability information provided for the Roche TRAb assay from 3 days at 2–8°C (2019-01 v14.0) to 7 h at 20–25°C (2020-02 v1.0). It was not clear if this referred to whole blood or serum stability. The purpose of this study was to determine TRAb stability in whole blood and serum for the eligibility of TRAb for reflective testing and batch testing (including a single freeze-thaw cycle [FTC]). No previous published studies could be found documenting TRAb stability.
Methods
Study 1: Volunteer samples
In the initial study, n =10 volunteers were bled, but only n = 5 had a TRAb concentration above the assay limit of detection (LOD). We assessed: (1) stability in whole blood in a primary tube stored at room temperature (RT) for up to 24h, and (2) stability in serum in a Sarstedt False Bottom Tube (SFBT) stored at 4–8°C for up to 72h, and (3) the effect of a single FTC. One 7.5 mL and four 2.6 mL tubes (Sarstedt S-Monovette® Serum gel) were drawn from each volunteer during a single venepuncture in compliance with the EFLM-COLABIOCLI recommendations. 1 Blood in the 7.5 mL tube was allowed to clot in an upright position (median time 47 min) prior to centrifugation (4500 rpm, 7 min, RT). Serum was then separated into seven SFBT aliquots to test 4–8°C serum stability. The 2.6 mL tubes were retained at RT to test whole blood stability; they were then centrifuged under identical conditions and serum transferred to a SFBT. The SFBTs containing serum were stored frozen at −20°C prior to analysis for 13 days (other than the non-frozen 0h sample). SFBTs were thawed, serum homogenised and samples analysed in singlicate due to consumable constraints.
Study 2. Patient samples
We expanded the study to look at patient samples with TRAb concentration >1.53 IU/L using n = 21 residual patient specimens (n = 12 with known Graves’ disease (GD)). The median delay in time from blood sampling to laboratory receipt was 5 h (range 3 h–24h). Serum was frozen in SFBT on the day of receipt and baseline TRAb concentration was determined within 7 days of blood draw (d0 result). Samples were then stored at 4–8°C with repeat analysis on d2 and d5. Time points were selected as substantial instability in serum TRAb was seen at 24–72 h in study 1, and many laboratories retain primary samples for 3–7 days, so d5 stability may be informative.
TRAb assay
TRAb was determined using the Elecsys Anti-TSHR assay, a competitive immunoassay using porcine TSH receptor (cobas e411, Roche Diagnostics Ltd, Burgess Hill, UK). The LOD is 0.8 IU/L and limit of quantitation (LOQ) 1.1 IU/L. Statistical analyses were performed using the Analyse-It v4.51 add-in package for Microsoft Excel (Analyse-It Software Ltd, Leeds, UK). Inter-assay analytical % coefficient of variation (%CVa) calculated from quality control (QC) material (Lot no. 479113 and 479114; concentrations 5 and 17 IU/L) was ≤4% during the study.
There is no defined percentage maximum permissible instability (MPI%) for TRAb based on biological variation data. The intraindividual biological variation (%CVi) is reportedly 4.8%. 2 The unidirectional % reference change value (%RCV) (calculated as: 2(Z)1/2 x (%CVa2 + %CVi2)1/2 using a one-tailed Z-score = 1.65 for p < 0.05); was 14.6%. This was used as the acceptable MPI%. The upper limits of TRAb in healthy individuals and patients with thyroid disease without diagnosis of GD are 1.22 IU/L and 1.53 IU/L, respectively (97.5th percentiles). Roche reports an optimal cutoff of 1.75 IU/L for diagnosis of GD.
Results
Study 1: Volunteer samples
TRAb was above the LOD in five specimens; with concentrations ranging from 0.85 to 1.36 IU/L. There was a statistically significant decrease in TRAb concentration caused by the single FTC with an average percentage deviation (PD% [± 95% CI]) of −8.9% ± 5.2% (p = 0.03, one-tailed t-test). TRAb concentration declined in whole blood over time (at 12h the average PD% [± 95% CI] was −12.9% ± 6.8%) (Figure 1(a)). There was also a fall in TRAb concentration in serum over time (Figure 1(b)), with an average PD% (± 95% CI) of −11.6% ± 6.6% at 12h. Percentage deviation (PD%) of TRAb concentration over time. The average PD% is shown by a solid black line with the two bordered small dashed lines either side representing the 95% confidence intervals. The horizontal dotted black line corresponds to the percentage maximum permissible instability (MPI%). The grey lines correspond to the PD% for each individual blood sample. a. Whole blood stability (n=5) and b. Serum stability (n=5). The concentration of TRAb in the initial non-frozen serum is shown to the right of each grey line. c. Serum stability in patient samples (n=21). The concentration of TRAb in the initial non-frozen serum is indicated by the colour of the dashed line: Dark grey dashed line (1.65 to 3.52 IU/L, n=7); dark grey solid line (4.74 to 13.26 IU/L, n=7); light grey solid line (14.11 to 31.02 IU/L, n=7). The * indicates the two samples with a concentration change from >1.53 IU/L to <1.53 IU/L following two days storage at 4–8 °C. The post freeze-thaw stability equations are displayed on each graph. PD%, percentage deviation from the non-frozen 0h sample (±95% CI), calculated as (concentration - baseline concentration / baseline concentration) × 100. MPI%, percentage maximum permissible instability, used to illustrate a significant change and determined as the unidirectional % reference change value for TRAb.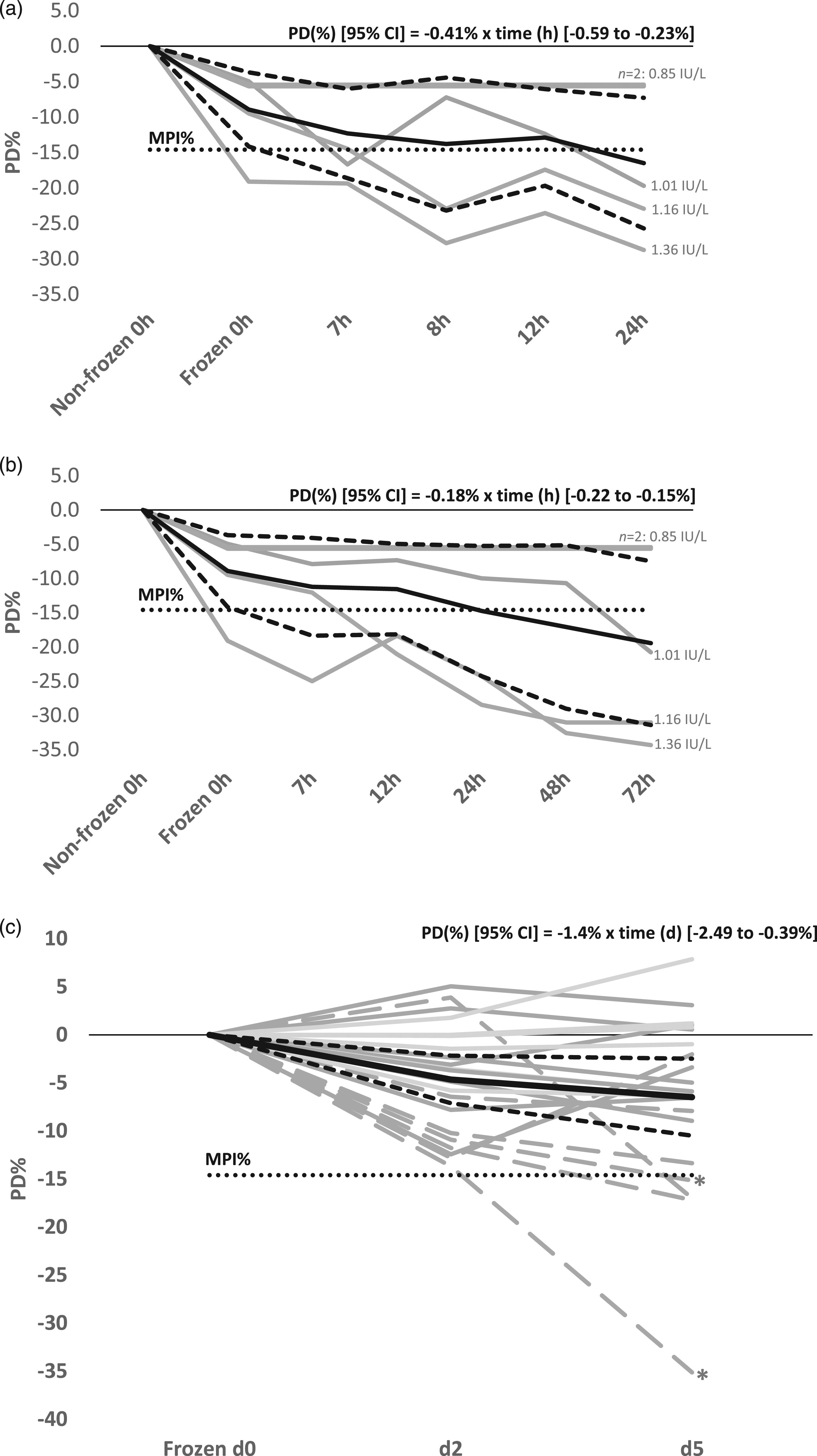
Study 2. Patient samples
Patient specimens (n = 21) had baseline TRAb concentrations of 1.65–31.02 IU/L (median 7.26 IU/L). The average PD% (±95% CI) was −4.6% ± 2.5% at d2 and -6.5% ± 4.0% at d5. The greatest decline in concentration was seen in samples with lower TRAb concentration (1.65–5.31 IU/L; n = 9), with an average PD% (±95% CI) of −9.1% ± 3.5% at d2 (Figure 1(c)). Results from two patients would have changed from >1.53 IU/L to <1.53 IU/L following 2 days storage of serum at 4–8°C.
Conclusion
TRAb concentration appears to decline over time in both whole blood at RT and serum stored at 4–8°C. This suggests TRAb stability differs from some autoantibodies, which increase in serum over time. 3 The fall in concentration may be due to structural changes or degradation of TRAb so that it no longer interacts with the porcine TSH receptor in the Roche immunoassay. A single FTC also resulted in a statistically significant fall in concentration; however, this may be time-dependent as samples were stored for 13 days at −20°C. Higgins et al. 4 reported a similar fall in anti-cardiolipin antibody concentration with a single FTC (average %PD -17.6%). In contrast, Männisto et al. 5 found no statistically significant change in anti-thyroid peroxidase antibody for up to 6 days in serum stored at 4°C (n = 8), and no effect was seen following a single FTC.
A larger study using patient samples from individuals with GD, including testing of stability following freezing, is required. In addition, immunoassays available on different platforms should be assessed as the detection of TRAb in stored samples may differ depending on assay components.
The initial part of this study was conducted using healthy volunteers, who by virtue of selection criteria had low quantitative results (many below the LOD), making it difficult to extrapolate findings to samples from clinically relevant patients. In the second part of the study, we assessed longer-term serum stability in patient specimens for reflective testing purposes, however, ethical approval would be needed to expand the study to look at pre-centrifugation stability. Stability in serum up to 5 days (4–8°C) had wide inter-individual variability, but instability remained within quality specifications for 17/21 specimens.
TRAb stability studies are hindered by a lack of assay standardisation and high variability in TRAb concentration in GD. TRAb assays are often interpreted in a semi-quantitative manner, 6 and due to the inter-individual variability, low sample numbers, and singlicate analysis, we focussed our interpretation on the average PD% results rather than 95% CI limits. In addition, two of the volunteer samples had concentrations between the LOD and LOQ, however, the functional sensitivity of the Roche assay is reportedly <1.1 IU/L.6-7 If a delay in centrifugation is combined with a FTC, delay in freezing of serum or its analysis, and assay imprecision; pre-analytical error due to TRAb instability could be clinically significant. This is particularly pertinent at the low-end of the measuring range for GD diagnosis or detection of relapse.
Until extensive studies are undertaken to verify TRAb instability, we aim to separate whole blood within 12h and either freeze serum within 12h for batch analysis, or analyse samples on the day that blood is drawn.
Footnotes
Acknowledgements
We would like to thank the volunteers that kindly donated blood samples for the study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Guarantor
SH.
Contributorship
SH oversaw data analysis and interpretation, and wrote the first draft of the report.
JC and RI contributed to the concept of the study and carried out laboratory sample analysis.
RI performed venous blood sampling of volunteers. All authors approved the final version.
