Abstract
Background
Faecal calprotectin has been identified as a useful biochemical marker in the differentiation of inflammatory bowel disease and irritable bowel syndrome. Typically, patients send faecal specimens in a pot for manual extraction by the laboratory. During the Severe Acute Respiratory Syndrome Coronavirus 2 (SARS CoV-2) pandemic, the routine laboratory service was temporarily suspended due to the potential increased risk to staff. In this study we investigated the possibility of patients collecting samples directly into the faecal extraction tubes.
Method
Patients submitted paired faecal samples for calprotectin analysis using a standard faecal container (current practice) and followed instructions for faecal collection using the BÜHLMANN CALEX® Cap device. Samples were returned to the laboratory immediately after collection. Laboratory staff manually extracted the calprotectin from the faecal samples using the CALEX® Cap prior to analysis of both extracts on the Cobas c702.
Results
91 paired faecal samples were included in the study. Clinical correlation was found to be 70% with numerical correlation showing a positive bias for the patient-collected CALEX® Cap sample when compared to the laboratory-extracted faecal sample around the clinical decision points 100–250 μg calprotectin/g faeces.
Conclusion
The study shows that collection of a faecal sample using the CALEX® Cap works well and is a good alternative to using standard containers. The correlation gives rise to the possibility that faecal calprotectin is not stable when collected into standard collection containers. Prior to further roll-out of this process, questions surrounding the current cut-offs would need to be addressed.
Introduction
Calprotectin is a cytosolic protein of neutrophils, which is released during an inflammatory response. Faecal calprotectin (f-cal) has been identified as a useful biochemical marker in the differentiation of inflammatory bowel disease (IBD) and irritable bowel syndrome (IBS), with the former group of patients yielding elevated results as a consequence of the activation of neutrophils during the manifestation of the disease. 1
In 2013, NICE published diagnostics guidance, DG11 which recommends that f-cal testing is used in adults with recent onset lower GI symptoms, without risk factors for colorectal cancer, to help to distinguish between IBS and IBD and prevent further unnecessary invasive investigation of IBS patients. 2
In August 2017, a Consensus paper was published endorsing a nationally agreed algorithm for use in Primary Care, based upon initial work done in the Yorkshire and Humber region by Turvill et al. 3 The algorithm recommends a very specific threshold of 100 μg calprotectin/g faeces (μg/g) to rule out organic bowel disease. Numerous studies have supported this algorithm and it has been widely adopted across the country, allowing colonoscopy resource to be targeted at those with persistent and elevated levels of faecal calprotectin. There is currently no international standardisation of f-cal assays.
Typically for f-cal analysis, patients send faecal samples in a standard faecal container to the laboratory and staff use the BÜHLMANN CALEX® Cap device to extract and prepare the sample for analysis.
In March 2020, the SARS_Cov-2 pandemic had a significant impact on the ability of the laboratory to manually process all f-cal samples. Additionally, publications at the time suggested that SARS_Cov-2 viral RNA was detectable in faecal material thereby potentially exposing laboratory staff to an increased risk during the processing of these samples. 4 The BÜHLMANN CALEX® Cap devices currently used by the laboratory to extract f-cal were therefore sent to patients to collect samples directly into the extraction device which would circumvent the potentially aerosol-generating extraction process and ease the burden on the laboratory.
The aim of this study was to compare the results obtained by the two collection methods; patients collecting faeces directly in to CALEX® Cap device and sending samples in standard faecal containers for the laboratory to extract.
Materials and methods
Reagents and consumables
The BÜHLMANN CALEX® Cap device contains an extraction buffer for use with the BÜHLMANN fCal turbo assay designed for the extraction of faecal calprotectin from faecal material. It was used as directed by the manufacturer. 5
The BÜHLMANN fCal turbo test is a particle enhanced turbidimetric immunoassay (PETIA) and allows quantification of calprotectin in faecal extracts on clinical chemistry analysers.
Method
Between the 25th May and 26th June 2020, secondary care clinicians across 4 different NHS Trusts covered by the Berkshire and Surrey Pathology Services network (BSPS) were asked to provide a list of patients who required faecal calprotectin measurement on the basis of clinical need. In total, 200 patients (70 male, 130 female) were sent a collection pack, comprising of: i) covering letter, ii) Pre-printed request form, iii) a CALEX® Cap collection device, iv) a standard faecal container and v) Instructions for each device. CALEX® Cap devices were stored at 2–8°C prior to use in the laboratory and prior to dispatch to patients. The age range of the patients was 5–92 years old. Patients were instructed to collect both samples from the same bowel motion; to write the date and time of collection on the tubes and to drop the completed collection pack at their GP practice as soon as possible following collection. Sample packs were transported to the laboratory via the regular pathology transport.
All samples were received in the laboratory within 3 days of sample collection and upon receipt, the faecal samples collected into standard faecal containers were extracted by competent laboratory staff using the CALEX® Cap device according to manufacturer instructions. The samples collected directly into the CALEX® devices by patients were centrifuged for 10 min at 3000 × g upon receipt and the supernatant transferred to a clear tube prior to analysis.
F-cal extracts were analysed on the Cobas 8000 c702 module in a UKAS-accredited laboratory using the BÜHLMANN fCal turbo assay and run in line with manufacturer’s instructions. The manufacturer quotes the limit of quantitation of the assay to be 23.7 μg/g with an analytical range of 9.13–13,339 μg/g. Samples with a concentration greater than 2000 μg/g were diluted automatically 1:10 by the analyser.
All internal quality control and external quality assessment samples run for the duration of the study were found to be within acceptable values. Inter-assay coefficients of variation were 6.1% and 2.9% at calprotectin concentrations of 85 μg/g and 276 μg/g, respectively. Each pair of samples (lab and patient-extracted CALEX® Cap devices) were analysed on the same analytical run, according to manufacturer’s instructions. All samples were analysed within 3 days of sample collection.
Data analysis
The mean, median and interquartile range (IQR) was determined for each dataset using Microsoft Excel. The Mann-Whitney U was used to determine if there was a significant difference between the data. Cohens-κ was used to determine clinical correlation using Analyse-it (Analyse-It Software Ltd, Leeds, UK, Software version: Method Validation edition) on a Windows 10 platform.
Results
Descriptive statistics comparing the differing extraction processes (results below analytical range have been excluded from mean and median calculations – N = 76).

Numerical concordance
A Mann–Whitney U test was used to compare the numerical values of the patient-extracted CALEX® Cap and the laboratory-extracted values. No significant difference was found between the two sets of data overall and when excluding those results less than the limit of detection of the assay (p = 0.054). Figure 1 shows the spread of results obtained: The interquartile range of the lab-extracted faecal calprotectin and the patient-extracted faecal calprotectin across all samples. The median is shown as a horizontal line and the mean depicted as an ‘x’. Data has been truncated such that all results >1000 have been set as 1000 and all results < Limit of Detection have been set at the LOD (9 μg/g).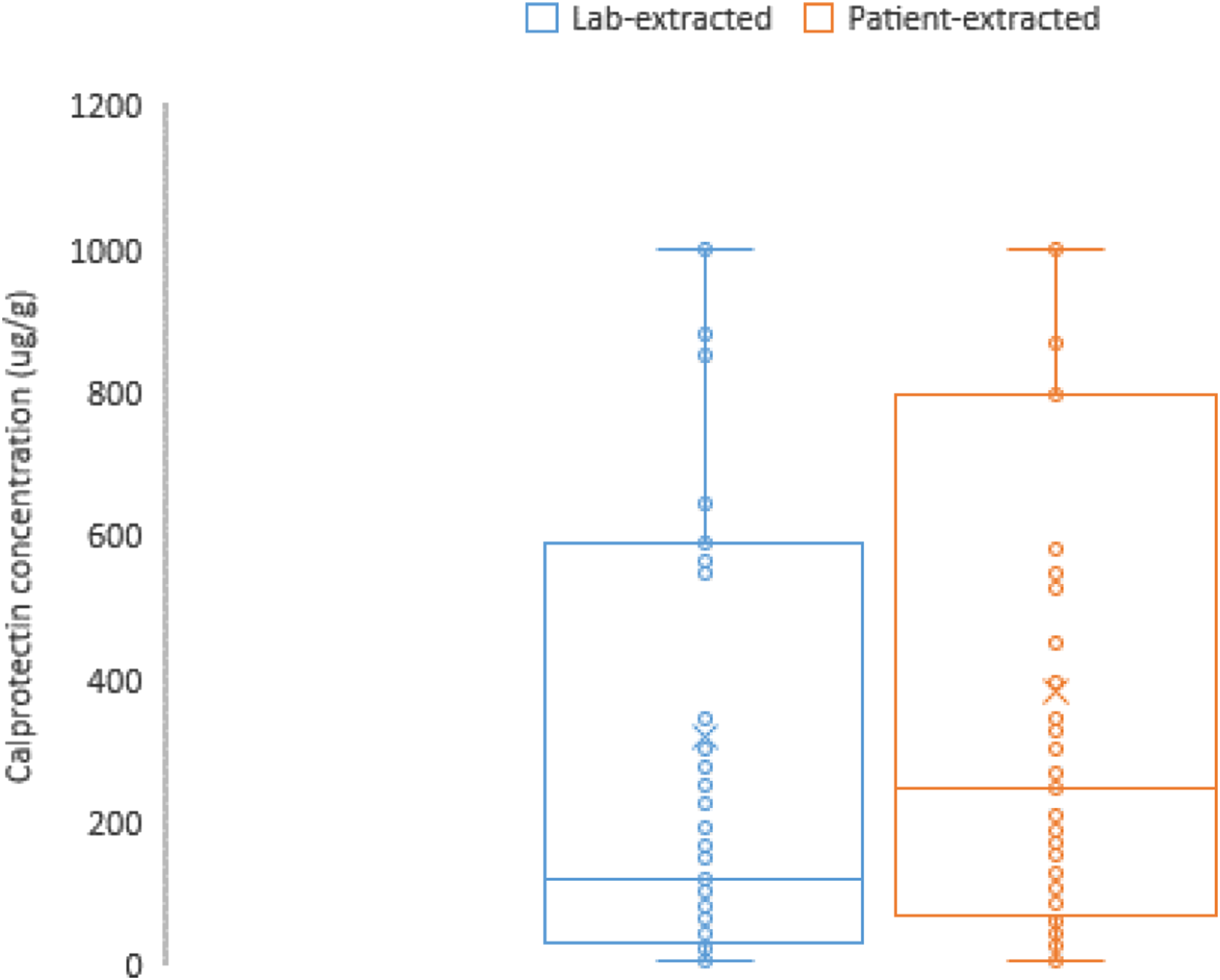
Figure 2 shows the numerical correlation of this data. When evaluating all data points (2a), correlation was found to be R2 = 0.70. Figures 2(b) and (c) show the correlation at the clinical thresholds of <100 μg/g and between 100 and 250 μg/g respectively, with correlation of the latter group found to be particularly poor with R2 = 0.08. Regression plot of patient-extracted samples versus laboratory-extracted samples. 2A = total data, 2B = data <100 μg/g, 2C = data at the clinical cut off of between 100 and 250 μg/g.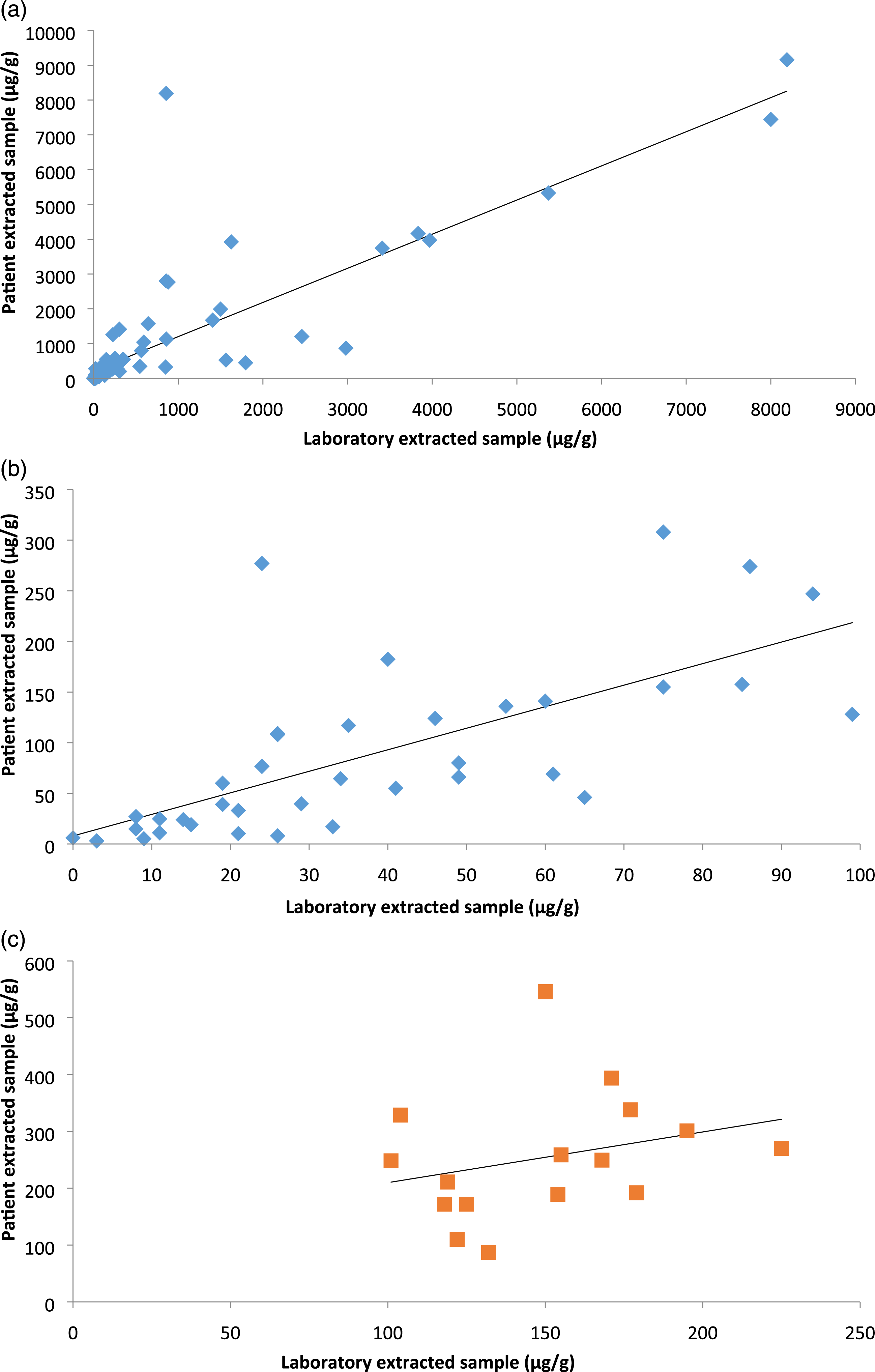
Concordance of categorisation
Concordance of categorisation of the differing extraction processes.
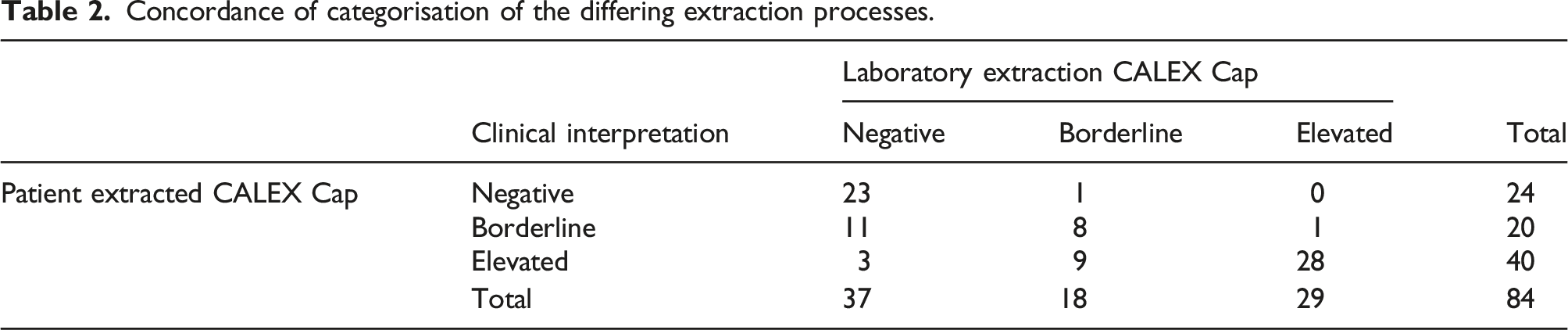
Reviewing each clinical decision threshold separately, the data shows a 77% concordance between the patient-collected CALEX® Cap device and the lab-extracted CALEX® Cap device at a f-cal concentration of <100 μg/g. Statistical analysis of this concordance by Cohens Kappa suggests that concordance has substantial agreement (0.624). Similarly, 84% clinical concordance was found at f-cal concentrations between 101 and 250 μg/g interpreted as a fair agreement by Cohens Kappa (0.252). 98% clinical concordance was found above 250 μg/g with a Cohens-Kappa of 0.686, signifying substantial agreement.
Of the discordant samples, the majority of the patient-extracted samples (22/25) gave faecal calprotectin results which fell into the higher clinical category of results when compared with the laboratory-extracted sample. There were 6 samples which were found to be highly discordant. Five patients were found to have elevated patient-extracted calprotectin results but negative laboratory-extracted calprotectin. One patient was found to have a positive lab-extracted calprotectin whist having a negative patient-extracted sample.
Discussion
To date, there is nothing in the peer-reviewed literature, other than a single conference abstract to assess the impact on calprotectin results when patients collect samples directly in to the CALEX® device compared to being collected in to a faecal pot and extracted in the laboratory. 6
Our results show a positive bias in samples collected directly in to devices compared to those collected in to faecal pots. This suggests that faecal degradation is occurring whilst samples are in transit to the laboratory when collected in to faecal pots. Temperature is largely considered a major factor in the degradation process, 7 however, pH has also been shown to be important.8,9 The effect of pH on the samples would be largely negated by collection directly into a buffer within a collection device.
Substantial agreement was found in the clinical concordance of the two extraction strategies at the current clinical decision thresholds and this alternative collection process has the potential to replace the current laboratory-based extraction process. The potential impact on currently derived clinical thresholds would need to be considered. A limited amount of data is currently available on the stability of faecal calprotectin. Data from the manufacturer suggests that faecal calprotectin is stable for up to 7 days at room temperature following collection into a CALEX® Cap extraction device. 5 Roseth et al. concluded that calprotectin was stable for 7 days at room temperature, although the authors noted a transient decrease in concentration at 48 h which had reverted to original concentration by day 7. Graphical representation of the data suggested marked differences in the stability of individual samples. 1 A more recent study suggests the samples remain unchanged for 3 days, followed by a decrease of 28% at day 7 for samples stored at RT. 10 Alternative publications suggest that stability is only preserved when samples are stored at 4°C and highlight the importance of refrigerating samples prior to dispatch to the laboratory.9,11–13
F-cal assays require the extraction of faecal material into an extraction buffer prior to analysis. Studies assessing stability of samples before extraction and once in the extraction buffer is limited. One study by a Spanish group looked at this particular aspect with results suggesting a good correlation between samples defined as being taken ‘recently’ and those stored at room temperature for 4 days. 14
A recently published study suggested excellent clinical correlation between manually extracted samples and the CALEX® Cap device when used by laboratory staff. 15 Upon receipt of the faecal material, samples were manually extracted using a traditional weighted-in method and were also extracted via the CALEX® Cap device. Correlation of the data (both analytically and clinically) gave excellent result and the authors concluded that the CALEX® Cap was a suitable alternative to a manual extraction process and provides comparable results if samples are transferred at same time. However, in this study, samples were transferred in to the buffer at the same time, so the impact on sample degradation (or lack thereof) that might occur if a patient collects the sample in to the device in their home was not assessed. Instead this study assessed the difference in manual versus CALEX® tube collections methods and demonstrated no significant variation from this pre-analytical change. This supports the outcome of our study in that f-cal is unstable until transferred in to the collection device and that these differences are unlikely to be due to pre-analytical processing differences but are most likely due to degradation of the calprotectin in faeces.
Patients are now becoming more familiar with self-collection of samples using extraction devices and find the collection method acceptable to use, 16 especially following the implementation of faecal immunochemical testing as part of the National Bowel Cancer Screening Programme and the NICE DG30 guidelines into cancer. Some manufacturers are also exploring the use of single home collection devices for both FIT and f-cal. 17
A limitation of this study is that samples were collected by patients themselves at home and there is no way to verify if the samples were collected according to the manufacturer’s instructions or that both samples were collected at the same time. Also, the cohort of patients selected may have been highly motivated to submit a calprotectin sample and the uptake may not be reflective of the general population.
When considering this study, questions have arisen about the stability of faecal calprotectin within standard faecal containers. The majority of the UK have well-embedded IBD pathways based on clear clinical cut offs as defined by NICE guidelines DG11. 2 No recommendations were made in relation to the use of a specific f-cal assay. There is no international standardisation of f-cal assays leading to different assays giving different results. The consequences of implementation of an alternative collection procedure that gives rise to the considerable positive biases seen here could lead to increased numbers of colonoscopy referrals. This data suggests that 13 patients may have been placed on an alternative clinical pathway had the patient-extracted CALEX® result been used for routine treatment decisions.
Should the findings of this study be replicated by other studies, further work would be needed to ensure appropriate clinical decision limits were established for such devices prior to implementation with collaboration with both primary and secondary care teams to ensure that colonoscopy services are not overwhelmed with referrals.
Footnotes
Acknowledgements
We would like to thank Alpha Laboratories, Eastleigh, UK for supplying the kits and reagent for this project.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
None required.
Guarantor
HB.
Contributorship
HB - Project design, data interpretation and writing the manuscript. JO - Contributed to project design and sample logistics; data collation and interpretation. GPJ - Sample analysis and logistics. SOD - Data interpretation and review of manuscript. MB - Project design, logistics and pre-analytical preparation. SCB - Project design, data interpretation, contribution to manuscript and review.
