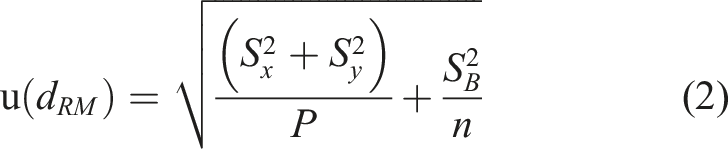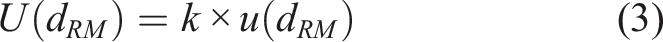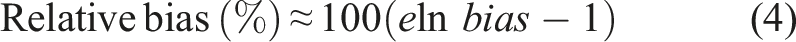Abstract
Background
Recalibration using serum pools assigned by higher-order reference methods had been demonstrated to be effective in improving the agreement among insulin immunoassays. To promote the application of serum pools in insulin harmonization, this study analyzed serum pools’ commutability between insulin immunoassays, and their short- and long-term stability at different temperatures. The agreement between commonly used immunoassays was also evaluated.
Methods
Insulin in 69 individual serum samples, 10 serum pools, and three EQA samples (lyophilized powder of serum pools) were detected by six widely used immunoassays. The commutability of serum pools and EQA samples was evaluated according to the IFCC-recommended approach. Serum pools’ stability at different temperatures was investigated by placing them at various temperatures for varying lengths of time. Individual serum samples’ results were analyzed using the Bland–Altman and Passing and Bablok regression analyses.
Results
Serum pools were commutable among most assays, the EQA samples—lyophilized serum pools—were non-commutable among most assays. Serum pools can be stably stored at −20°C and −80°C for at least one year, but can only be stably stored at room temperature for twenty-four hours. Significant relative differences were observed among assays. Recalibration using serum pools can only improve the assays’ agreement at middle and high insulin levels, but not at low levels.
Conclusions
Serum pools were commutable and stable for insulin measurement and can be used in insulin harmonization. The existing EQA materials were non-commutable between most assays, and other EQA materials, such as serum pools, should be studied.
Introduction
The prevalence of diabetes mellitus has grown substantially during the past decades and is projected to keep increasing in the future.1,2 In type 1 diabetes, lower insulin levels are observed along with absolute insulin deficiency. In the early stage of type 2 diabetes mellitus, insulin resistance is characterized by compensatory secretion of insulin, nevertheless, as the function of the islet beta cells fails, the secretion of insulin also decreases. As a result, serum insulin correlates with both the type and the duration of diabetes and was a routinely used biomarker to distinguish type 1 and type 2 diabetes when patients were diagnosed with diabetes. For patients with type 2 diabetes, insulin was also used to estimate insulin resistance before they were treated with any exogenous insulin. For patients treated with exogenous insulin, serum C-peptide is usually used for residual beta cell function. At present, insulin is commonly measured by various immunoassays. However, the widely used immunoassays have not been harmonized and standardized across manufacturers and clinical laboratories, and measurement results from different medical settings are not comparable. 3 As a result, it is difficult to incorporate insulin detection results across tests and laboratories and combine data from numerous investigations to develop evidence-based criteria and clinical guidelines. 4
In 2004, the American Diabetes Association, the National Institute of Diabetes and Digestive and Kidney Diseases, the European Association for the Study of Diabetes, the Centers for Disease Control, and the International Federation of Clinical Chemistry (IFCC) established the Insulin Standardization Workgroup to improve the agreement among different laboratories for insulin measurement. The group reported in 2007: 5 (1) most routine assays had negligible cross-reactivity from proinsulin and cleaved intermediate products of proinsulin conversion to insulin; (2) laboratories and manufacturers should use the SI unit pmol/L to report insulin concentrations; (3) agreement among various immunoassays was unsatisfactory for insulin; and (4) the isotope dilution–liquid chromatography/tandem mass spectrometry (ID-MS) measurement procedure could be used as the higher-order reference measurement procedure to calibrate routine assays. In 2009, studies conducted by W.Greg Miller, Linda M. Thienpont, and their colleagues demonstrated that using individual or serum pools assigned by ID-MS methods to recalibrate routine methods could improve the agreement among different immunoassays.6,7 Therefore, the serum pools and individual serums assigned by higher-order reference methods were recommended as the candidate secondary reference materials for insulin harmonization and standardization. However, no studies about the commutability and stability of insulin in serum pools—which are both crucial for their application in standardization or harmonization—were reported. In this study, we assessed the commutability of serum pools and the existing EQA materials (lyophilized serum pools) for insulin measurement. And the stability of insulin in serum pools was also studied. At the same time, we also evaluated the agreement between commonly used insulin immunoassays.
Notably, the study conducted by W. Greg Miller and Linda M. Thienpont et al. 7 found the recalibration with serum pools failed to improve the agreement between insulin immunoassays for low-level (<60pmol/L defined by isotope dilution liquid phase tandem mass spectrometry) samples. One possible explanation was that only three serum pools were used to recalibrate the results of routine assays in their study, so the regression line used to draw the new calibration curve might be wrongly extrapolated to concentrations below the three serum pools. As a result, inappropriate conclusions might have been made at low levels. To verify this hypothesis, 10 serum pools with concentrations extended to the low concentrations were utilized to recalibrate the results of individual serum samples in this study.
Materials and methods
Specimens
The 69 individual serum samples included in this study were collected from the leftover serum samples in Beijing Hospital between August 2020 to January 2021, and these samples were stored at 4°C for no more than 24 hours after collection. Those individual samples were collected from the health examination population. These samples were frozen at −80°C. Serum samples with hemolysis, icterus, or lipemia were considered deviant and were excluded herein. The Ethics Committee of Beijing Hospital approved this study and exempted the need for obtaining informed consent (approval number 2018BJYYEC-019-01). For the preparation of the serum pools, extra-individual serum samples were first grouped based on their preliminary results from Beijing Hospital (the measurement system was Siemens ADVIA centaur XP). Samples with similar insulin levels were then thawed, pooled overnight at 4°C on the magnetic stirring equipment, and filtered using a vacuum pump (0.45 μm and 0.22 μm filter membranes, respectively). As a result, 10 serum pools were prepared, aliquoted, and stored at −80°C until they were shipped to laboratories on dry ice. The three EQA samples of lyophilized serum pools were provided by the Chinese National Center of Clinical Laboratories.
Analytical methods
According to our previous study, 8 measurement systems from Roche Cobas e801 (Basel, Switzerland), Abbott I2000 (Chicago, USA), Siemens ADVIA Centaur XP (Berlin, Germany), Beckman Dxi800 (Brea, California, USA), Mindray CL8000 (Shenzhen, China), and Snibe Maglumi X8 (Shenzhen, China) were used by most laboratories in China for insulin detection. Six immunoassays from those manufacturers were included in this study and all assays claimed that their calibrators traced to the same WHO international reference reagents (WHO IRR 66/304). Detailed information on those six assays including manufacturer, instrument, reagents, calibrator, and measurement principle was provided in Supplemental Table 1. Before the study, the imprecision (Total CV%) of those assays has been validated by measuring three different levels of serum samples twice a day for three consecutive days (Supplemental Table 1).
Measurement protocol
Laboratories were instructed to measure individual serum samples, serum pools, and lyophilized serum pools in random order as specified in the study protocol. All measurements were based on laboratories’ usual procedures and product calibrators. Each sample was measured three times in three separate runs and each laboratory was requested to submit three repeated values for each sample.
The commutability assessment procedure
The commutability of serum pools and lyophilized serum pools for insulin measurement was evaluated based on the difference in bias analysis recommended by IFCC.9,10 IFCC recommends assessing the difference in bias and associated uncertainty to determine the commutability of processed samples. Briefly, the uncertainty of bias of assays was calculated according to the following equation (1), (2), and (3). The BRM is the mean bias of the processed samples such as serum pools and EQA materials; the BCS is the mean bias of clinical samples (individual serum samples in this study). Sx represents the standard deviation of assay X. SY represents the standard deviation of assay Y. P is the number of tests repeated (p = 3). SB is the standard deviation of the biases between assays X and Y, and n is the number of clinical samples. k is the expansion factor and k = 2. The U (dRM) is the expanded uncertainty of dRM. The commutability evaluation limit C is usually fixed and based on the medical requirements of the testing. The Insulin Standardization Workgroup recommended a total allowance error (TEa) of ±32% for a single measurement for insulin. In this study, 1/2 of the TEa of ±32% was set as the commutability evaluation limit C (16%). When the dRM ± U (dRM) was within, exceeding, or overlapping 0 ± C, the commutability of processed samples was considered commutable, non-commutable, and inconclusive, respectively.
Short- and long-term stability experiments
Serum pools’ short- and long-term stability at various temperatures were defined by storing them at different temperatures (−80, −20, 4°C, and room temperature) for varying periods of time. After that, serum pools that were always stored in liquid nitrogen were measured alongside those samples that were stored at different temperatures for varying periods of time. In each storage condition, three aliquots of each serum pool were included and each aliquot was measured twice, and six repeated results were collected for each serum pool. At last, the mean of six replicate values of serum pools that underwent storage condition alternation and serum pools that did not were calculated and compared to define serum pools’ stability under different storage conditions.
Recalibration
The 10 serum pools were used to recalibrate the results of individual serum samples and the target values for those recalibration materials were defined as the overall means of all assays. The ordinary linear regression equation between the target values and results from each assay was used to draw the new calibration curves. The results of individual serums were then recalibrated using the new calibration curve. Assays’ agreement before and after recalibration was compared and analyzed.
Statistical analysis
The units of results from those assays were first transformed into SI unit pmol/L before further statistical analysis (The unit conversion coefficient of 6.0 was used to convert U/ml to pmol/L 11 ). The overall mean of all assays for individual serum samples were set as the target values of individual serum samples. To assess the agreement between those assays, the Bland–Altman analysis and the Passing and Bablok regression analysis were carried out between the target values of individual serum samples and individual serum samples’ results from each assay. The Spearman’s correlation coefficient was used to describe the correlation among those assays. Based on the biological variability, the Insulin Standardization Workgroup recommended a total allowance error (TEa) of ±32% for a single measurement for insulin, 5 and 1/3 of the TEa (10%) was used to assess the stability in this study. The samples were considered to be stable if the measurement results of samples with storage conditions alternations were within 100 ± 10% of the results of samples without storage condition change.
All calculations were completed in Microsoft Excel 2016 (Microsoft Corporation, Redmond, Washington, USA) and MedCalc statistical software 18.11.6-64-bit (Mariakerke, Belgium).
Results
Commutability assessment
Serum pools’ commutability between those six assays was analyzed in 15 assay pairs. Despite the relative difference of most individual serum samples presented approximately constant scatter widths for most assay pairs, some individual serum samples at the low level showed inconsistent relative differences from other samples (Figure 1 A, C, E, G, I, and K). According to the IFCC guideline, in this case, it is inappropriate to assess low-level (<120 pmol/L) samples’ commutability. Therefore, only the commutability of serum pools with concentrations >120 pmol/L and lyophilized serum pools with concentrations >120 pmol/L were assessed (5 serum pools and 2 lyophilized serum). The Ln (concentration) was used for statistical analysis and the criterion (16%) was converted to an absolute number (±0.148) by using equation (4). The Bland–Altman analysis plots of all assays. The X-axis refers to the mean of all assays, the Y-axis means the relative difference between assays. Code A, C, E, G, I, and K refer to the Bland–Altman analysis plots between different assays before recalibration, and codes B, D, F, H, G, and L refer to results after recalibration.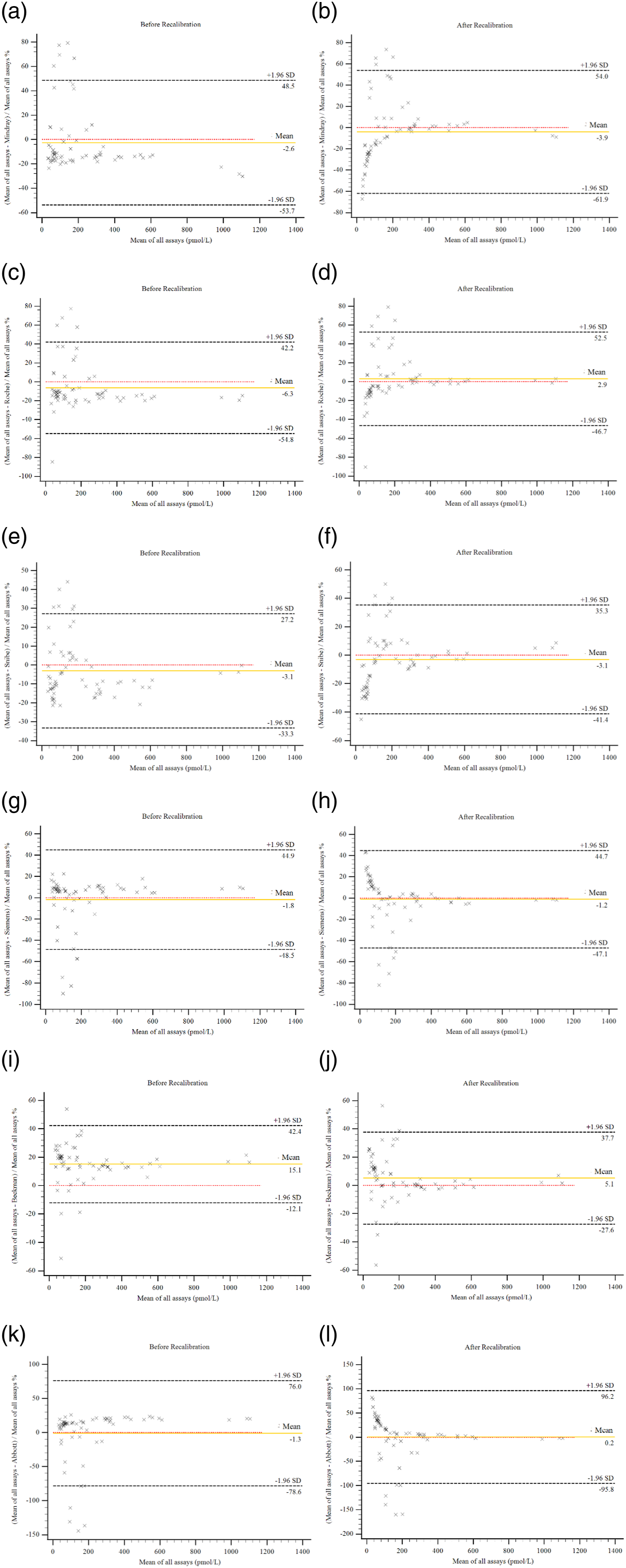
For serum pools, no non-commutability results were found across all assay pairs, and only some inconclusive results were found for serum pools 1, 3, and 5. Serum pools 2 and 4 were commutable between all assays (Figure 2). Notably, compared to serum pools, the EQA samples—the lyophilized powder of serum pools—were only commutable on 3 of 15 assay pairs (Figure 2). Commutability results of serum pools and EQA samples based on the IFCC approach. The mean bias of clinical samples (individual serum samples) is presented by the solid black line. The assessment limits were shown by the dashed lines. The EQA samples and serum pools were shown in squares and triangles, respectively. The x-axis is the mean concentration of the relevant two assays.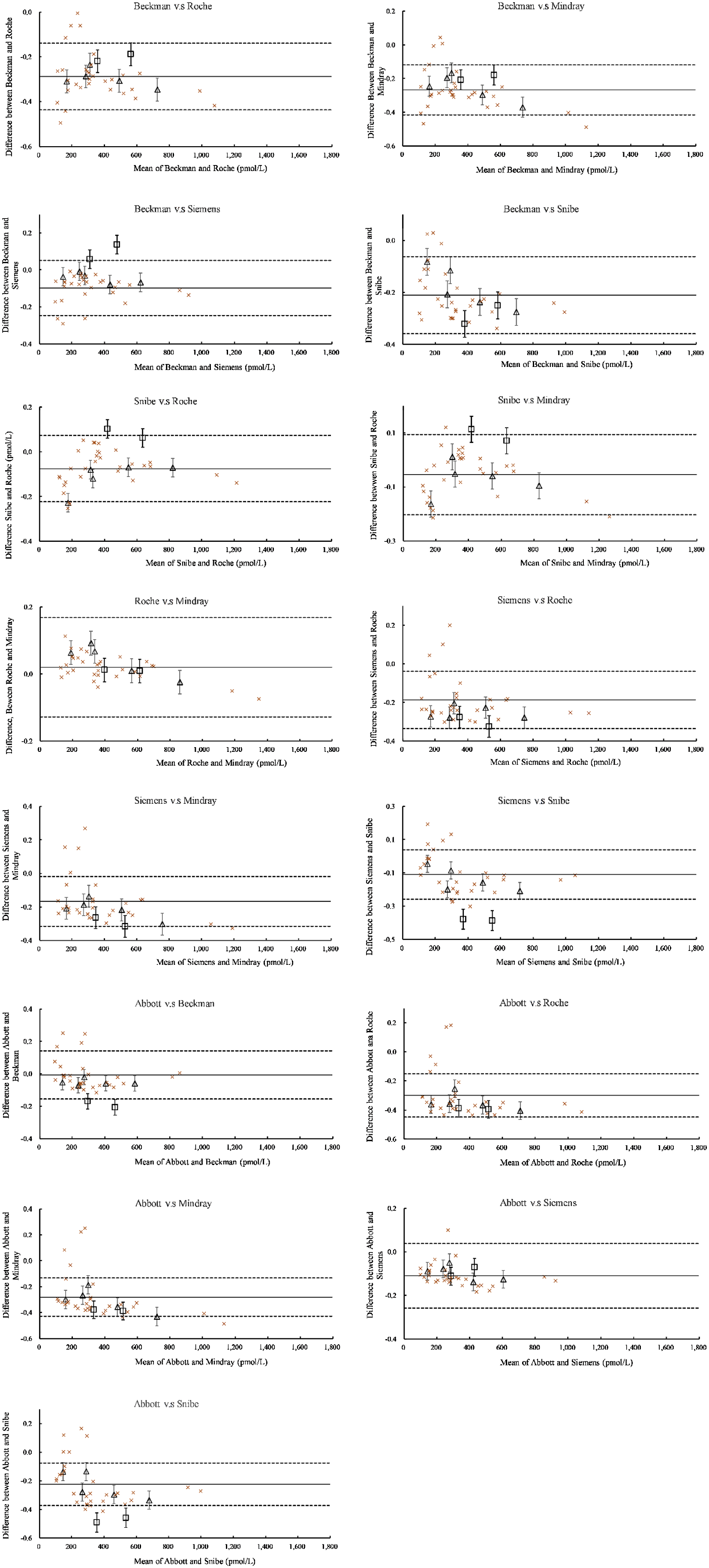
The short- and long-term stability of serum pools
Results showed that insulin in serum pools could be stably stored at 4°C and −20°C for at least 16 days, but it could only be stably stored at room temperature for no longer than 4 hours (Supplemental Table 2, Figure 3A). For long-term stability, insulin in serum pools could be stably stored at −20°C and −80°C for at least one year (Figure 3B, Supplemental Table 3). The short- and long-term stability of insulin in serum pools. The X-axis refers to the storage time, and h, D, and M refer to hours, days, and months, respectively. Y-axis means the percentage of results stored at relevant conditions relative to results from samples stored in liquid nitrogen. Code A refers to the short-term stability at −20, 4°C, and room temperature. 4°C-PS 1 means the stability of serum pool 1 at 4°C, RT-PS 2 means the stability of serum pool 2 at room temperature, and so forth. Code B refers to the long-term stability of insulin in serum pools at −20°C and −80°C, and −20-PS 1 means the stability of serum pool 1 at −20°C, −80-PS 2 means the stability of serum pool 2 at −80°C, and so forth.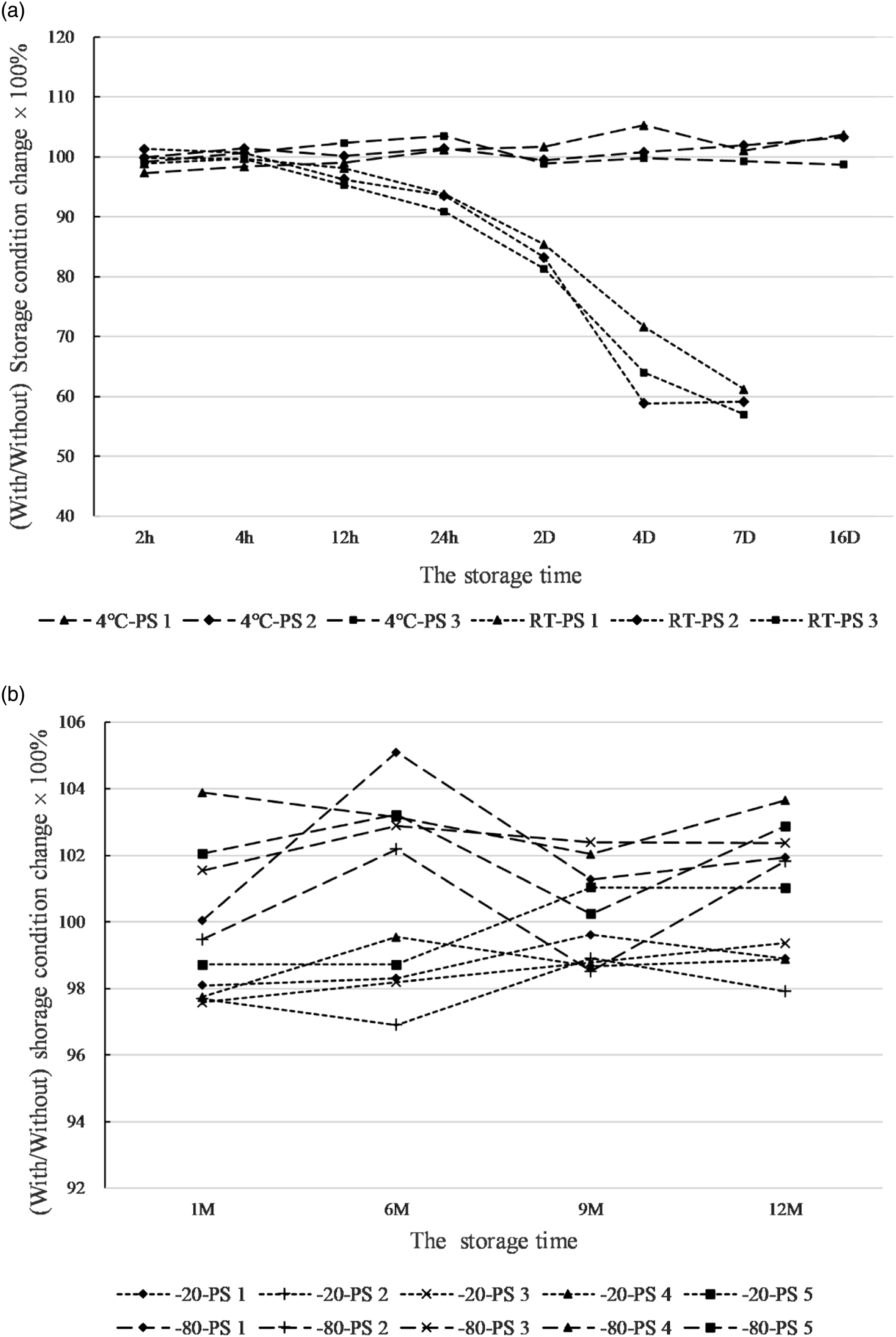
The agreement among assays
The Passing and Bablok and Bland–Altman analysis results before and after recalibration.
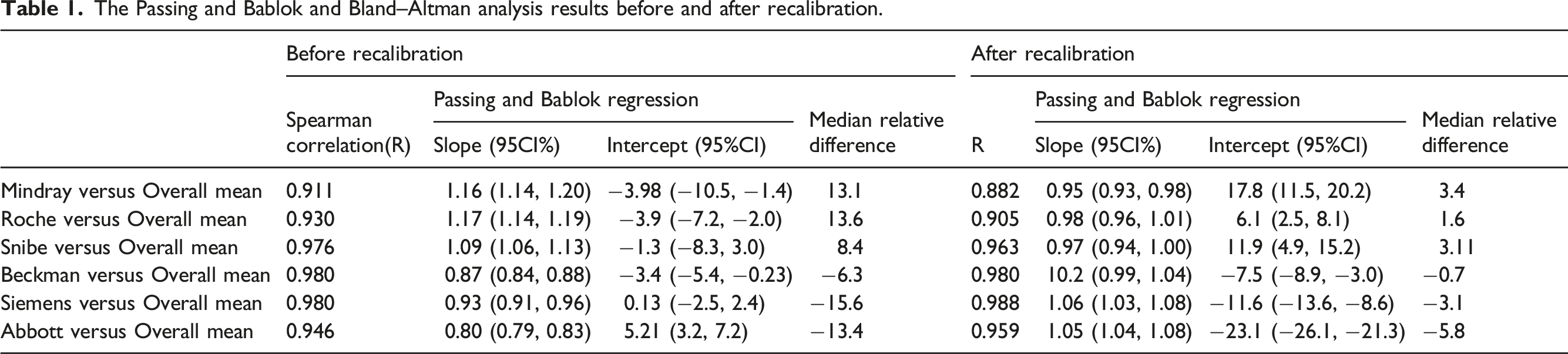
In the Bland–Altman plots, most individual serum samples showed consistent relative differences, indicating their differences were proportionate to the concentration. (Figure 1A, C, E, G, I, and K). Since the distribution of the relative differences of some low-level samples is visibly at odds with that of the majority of the samples (Figure 1A, C, E, G, I, and K), it is more accurate to use the median of the relative difference rather than the mean to depict the distribution of relative difference of most samples (Table 1). Despite all median relative differences falling within the range of overall mean ± 15%, the relative difference of many samples at the low concentrations exceeded the total allowance error of ±32%.
Recalibration
After the recalibration using the 10 serum pools, the Bland–Altman analysis showed much smaller medians of relative difference indicating the agreement among assays was significantly improved (Figure 1B, D, F, H, G, and L). However, the Passing and Bablok regression lines’ intercept became even much bigger for all assays (Table 1, Supplemental Figure 1), implying the assays' agreement at low levels might be even worse.
After the recalibration, the mean and median relative differences were indeed closer to the zero line since the relative differences of samples were more symmetrically distributed on either side of the zero line. However, the distribution of relative difference of samples at low concentrations becomes even broader on either side of the zero line, and the assays agreement at low concentrations actually became worse.
Discussion
Accurate detection of insulin plays a critical role in the diagnosis and treatment of diabetes mellitus.7,12 However, according to the report in 2007, 5 the agreement and consistency among insulin immunoassays did not meet the required desirable performance criterion recommended by the Insulin Standardization Workgroup.
Accurate measurement of insulin at low concentrations is clinically important in the screening of type 1 diabetes and the dysfunction of islet cells in late-type 2 diabetes and is also helpful in judging the causes of hypoglycemia. 13 However, in this study, we found that the agreement among laboratories for low-level samples is highly variable and recalibration with serum pools did not improve the agreement. Therefore, the sample-specific interferences might play a critical role in the inconsistency among insulin immunoassays for those low-level samples. Before recalibration, the Bland–Altman plots (Figure 1A, C, E, G, I, and K) showed that most samples presented approximately constant relative difference, but a few samples (10%–15%) at the low concentrations displayed obviously inconsistent relative differences. One possible explanation is that some interfering or cross-reacting substance in those samples might generate different levels of responses among assays. For instance, the heterophile antibody (e.g., human anti-mouse antibody) may result in either a false increase or decrease in measurement results according to the instructions of those manufacturers. Notably, for those samples with discordant relative differences, the results from Siemens and Abbott were much bigger than those from Roche, Mindray, and Snibe. The results of Abbott and Siemen were much bigger than the target values and were easy to exceed the upper limit. Instead, Mindray, Snibe, and Roche were easier to exceed the lower limitation since their results are much smaller than the mean. Interestingly, Siemens and Abbott were based on a similar measurement principle, and Roche, Mindray, and Snibe were based on another similar principle (Supplemental Table S1). Therefore, the interfering or cross-reacting substances contained in those samples might have different levels of responses to assays with different measurement principles. On the other hand, our study also demonstrated that the recalibration using a wide range of serum pools that extended to the low concentrations could not improve the assays agreement at low concentrations which verified the hypothesis of previous study. 7
Although recalibration using serum pools cannot improve the assays agreement at low concentrations, it was effective in reducing the constant relative difference over most of the measuring range and was suggested to be used as the second reference material in insulin standardization and harmonization.6,7 However, no studies about the commutability and stability of serum pools were reported for insulin. In this study, the commutability of serum pools for insulin detection was assessed based on the approach recommended by IFCC. Unfortunately, since the difference among assays at the low concentration is neither constant nor proportional to the concentration, the widths of the relative difference at the low level were too wide to assess the commutability; therefore, only the commutability at high levels (>120pmol/L) was assessed. Results showed the serum pools were commutable between most assays and only got a few inconclusive results between some assays. On the other hand, the short- and long-term stability at different temperatures was also defined for insulin in serum pools. Insulin in serum pools could only be stably stored at room temperature for not more than 4 hours, whereas it could be stably stored at 4°C for at least 16 days. Therefore, samples should not be placed at room temperature for too long during the daily measurement and should be stored at 4°C when immediate testing is impossible. At −20°C and −80°C, insulin in serum pools could be stably stored for at least one year. In a word, our study demonstrated that the serum pool was suitable reference material for insulin measurement and could be used in insulin standardization and harmonization since (a) it was commutable between most assays; (b) it can be stably stored at −20°C and −80°C for a long time; and (c) recalibration with it could improve the agreement among insulin assays. However, harmonization/standardization—activities of tracing the results of routine measurements to the same standard or SI units—can only correct the constant bias over most of the measuring range. It cannot improve the agreement at low concentrations since the source of inconsistency at low concentration was not only from the valuation transfer and traceability system but also from other factors such as the sample-specific effects, antibodies, and calibration methods. Therefore, to improve the agreement at low concentrations, manufacturers and researchers need to conduct studies focusing on additional factors such as sample-specific interferences or the measurement principle.
In addition to the standardization and harmonization, the EQA program is also used to monitor the performance of clinical laboratories and improve the agreement among assays.14,15 However, for practical reasons (stability, levels of the analytes, cost, etc.), the EQA materials usually were processed samples whose commutability is unknown. In this case, without the commutability information, it is hard to improve the between-assays variations shown in the EQA program. Consequently, unnecessarily repeated tests may be required especially when patients transfer between hospitals. In this study, the commutability of the lyophilized powder of serum pools—the Chinese EQA materials for insulin measurement—was also assessed. Results showed the lyophilized powder of serum pools were only commutable on 3 of 15 assay pairs. One possible explanation is that some new matrix effects might be introduced into the sample during the production process. EQA samples based on other materials should be studied.
In conclusion, the commutability and stability of serum pools for insulin measurement were been defined in our study. Meanwhile, the EQA materials currently used in China for insulin measurement were found to be not commutable between most assays, and serum pools were a better choice for the insulin EQA program. On the other hand, we also demonstrated that the recalibration using a wide range of serum pools that extended to the low concentrations could not improve the assays' agreement at low concentrations and other studies should be conducted to improve the assays’ agreement at low concentrations. This study also provided reliable data about the difference among various insulin immunoassays, which may provide information for laboratories to choose measurement systems, manufacturers to elevate performance, and clinicians to better interpret measurement results of serum insulin and C-peptide.
Supplemental Material
Supplemental Material - Application of serum pools in insulin harmonization: Commutability and stability
Supplemental Material for Application of serum pools in insulin harmonization: Commutability and stability by Yuhang Deng, Chao Zhang, Jing Wang, Jie Zeng, Jiangtao Zhang, Tianjiao Zhang, Haijian Zhao, Miao Li, Yang Zhao, Wei Gan, Yan Shao, Hongyuan Yu, Weiyan Zhou and Chuanbao Zhang in Annals of Clinical Biochemistry
Footnotes
Acknowledgements
The authors gratefully acknowledge the colleagues at West China Hospital, Sichuan University Department of Laboratory Medicine; Beijing Aerospace General Hospital Department of Laboratory Medicine; Snibe Diagnostic Co. Ltd. Shenzhen China; and Mindray Co. Ltd. Shenzhen China for their kind assistance in testing samples during this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This study was approved by the Ethics Committee of Beijing Hospital, with an exemption from informed consent (approval No. 2016BJYYEC-121-03).
Guarantor
ZCB.
Contributorship
ZCB, ZWY and DYH conceived the study and gained funding. WJ, ZJ, ZC, ZJT, ZTJ and ZHJ provided the idea for the study. DYH was responsible for participant recruitment, data collection and data analysis. LM, ZY, GW, SY and YHY performed the testing from different measurement systems. DYH wrote the first draft of the manuscript and all authors reviewed and edited the article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.