Abstract
Selecting appropriate laboratory tests based on available evidence is central to improve clinical effectiveness and impacting on patient outcome. Although long studied, there is no mutual agreement upon pleural fluid (PF) management in the laboratory context. Given the experienced confusion about the real contribution of laboratory investigations to guide clinical interpretation, in this update, we tried to identify useful tests for the PF analysis, aiming to unravel critical points and to define a common line in requesting modalities and practical management. We performed a careful literature review and a deepened study on available guidelines to finalize an evidence-based test selection, intended for clinicians’ use to streamline PF management. The following tests depicted the basic PF profile routinely needed: (1) abbreviated Light’s criteria (PF/serum total protein ratio and PF/serum lactate dehydrogenase ratio) and (2) cell count with differential analysis of haematological cells. This profile fulfils the primary goal to determine the PF nature and discriminate between exudative and transudative effusions. In specific circumstances, clinicians may consider additional tests as follows: the albumin serum to PF gradient, which reduces exudate misclassification rate by Light’s criteria in patients with cardiac failure assuming diuretics; PF triglycerides, in differentiating chylothorax from pseudochylothorax; PF glucose, for identification of parapneumonic effusions and other causes of effusion, such as rheumatoid arthritis and malignancy; PF pH, in suspected infectious pleuritis and to give indications for pleural drainage; and PF adenosine deaminase, for a rapid detection of tuberculous effusion.
Background
Pleural fluid (PF) is a plasma ultrafiltrate, in a steady balance between filtration and reabsorption, providing lubrification between the pleural membranes. In medical laboratories, it represents a precious sample taken by an invasive procedure (i.e. thoracentesis), and therefore, caution should be pursued when handling and exploring this biological material with the aim to define its nature and achieving a final diagnosis for proper patient management. 1 Historically, pleural effusions have been classified as of exudative or transudative nature. Pleural fluid examination gained interest through time, starting 50 years ago with the proposal of so-called Light’s criteria 2 until the more recent release of guidelines and recommendations.3–8 Nevertheless, confusion still exists about the real utility of different tests for investigating the nature of PF, usually proposed as a simple list of examinations with a summary of pros and cons supporting (or not) the use, but without any definitive clarification about which exams clinicians should request to achieve correct diagnoses, with the aim of pursuing both appropriateness and effective clinical management. Given this unclear situation, which did not permit to the laboratories to standardize their service and to assure cost-effectiveness, we decided to define, based on existing scientific evidence, the best practice for PF laboratory investigation, including the optimal cut-offs to be applied for the recommended tests.
Pre-analytical phase: A frequently neglected aspect
Contrary to the normal practice, it is nowadays more and more evident that it is essential to pay special attention to the aspects related to sample collection and consider possible potential interferents in the PF examinations. Avoiding pre-analytical errors is particularly important given the usual unrepeatability of this fluid collection.
Selection of tubes for PF collection and sample handling
As per all laboratory analyses, special care should be taken when selecting the container for PF collection. Cell count and differential cell analysis should be directly performed on samples collected with EDTAK2 or K3 anticoagulants. No anticoagulants or additives are necessary for most biochemistry tests, although heparinized tubes may be used, except for glucose, for which PF sample must be collected in tubes containing a glycolysis inhibitor to avoid in vitro glucose consumption, especially in heavily cellular samples. 9 Before biochemical analyses, centrifugation is required to eliminate cell debris and correctly carry out investigations, including PF inspection (see below). For pH measurement, the PF sample must be collected in air-tight non-ventilated syringes and transported to the laboratory or point-of-care blood-gas station rapidly and, ideally, ice packed. Sterile tubes for adenosine deaminase (ADA) analysis are recommended. Finally, it should be remembered that lactate dehydrogenase (LDH) activity is very sensitive to cold and is lost if the PF (and serum) samples are stored frozen. Therefore, if not immediately assayed, PF specimens should be stored at room temperature, at which no loss of LDH activity occurs for at least 3 days. 10
Macroscopic PF inspection
Macroscopic appearance/odour of pleural fluids and possible underlying conditions.

Interferences
As for blood samples, sample quality and integrity should be considered in PF analysis. Lactate dehydrogenase activity is present at high levels in cytoplasm of many cells. This implies that, if cellular damage is present, for example, after a traumatic PF withdrawal, LDH activity may significantly increase. Consequently, LDH results can be misinterpreted, causing a diagnostic misclassification when Light’s criteria are employed (see below). A strategy, such as automatic photometric determination of interference indices, to evaluate sample quality before analysis would therefore be desirable even for the PF samples. A major limitation of this approach is however a lack of interference thresholds for this fluid. 13 Therefore, until specific investigations are conducted to define these thresholds, visual inspection by laboratory professionals is at minimum recommended to identify possible interference problems.
Analytical interferences can also be an issue. Almost all studies measuring albumin gradient (see below) used non-specific albumin assays based on the bromocresol green principle. 6 As recently demonstrated, the use of non-specific methods for the measurements of serum albumin may however have a high negative clinical impact. 14 It is unclear if this lack of method selectivity may significantly influence albumin gradient results and their interpretation. 7
Test selection
Light’s criteria and albumin gradient
Studies from which current guidelines are drawn focus on PF testing and concentrate on the management of this peculiar sample.3–5 First in line is the distinction between transudates and exudates and their different pathophysiology. Transudates origin from a gap between hydrostatic and osmotic pressure, frequent causes of which are cardiac failure and cirrhosis. Exudates derive from an increased capillary permeability of the pleural membranes, generally related to inflammation, infection, or malignancy. Light’s criteria were conceived for PF discrimination in exudate or transudate, originally requiring measurement of LDH and total proteins, both in PF and serum, and calculation of PF to serum ratios as well as the interpretation of LDH absolute value in PF. 2 In their initial study, Light and co-workers 2 found both elevated sensitivity [99.0%, 95% confidence interval (CI): 94.7%–99.9%] and specificity (97.9%, CI: 88.7%–99.9%) for these criteria. However, all following authors failed to obtain the same optimal results for several reasons, including the fact that Light’s proposed cut-offs cannot be easily extrapolated in other settings or populations and that the use of an absolute LDH cut-off is controversial,11,15,16 since enzyme results are strictly dependent from the employed laboratory method (while ratios are method independent). 10 Considering Light’s criteria, one important concept should be emphasized: a panel testing is useful only when each included test helps identifying a different subset of the evaluated population; if tests of the panel provide overlapping information, it should be recommended to perform only the more accurate one. Heffner et al. 17 had the merit to show that the LDH absolute value in PF can be removed from Light’s criteria because, as expected, it is highly correlated with LDH ratio (r = 0.84) and does not add anything of significance to the discriminative power of the criteria. They proposed the introduction of ‘abbreviated Light’s criteria’, which include only PF/serum total protein and PF/serum LDH ratios, as the best biochemical approach to diagnose exudates, avoiding duplication of data relative to reporting LDH absolute value in PF, as the diagnostic accuracy of these abbreviated criteria when both ratios exceed the related cut-offs remains unchanged when compared to the classical Light’s criteria.
The main limit of Light’s criteria, both classic and abbreviated, is the misclassification of transudates as exudates, because they lack in specificity.11,18,19 This is true mainly for patients with cardiac failure treated with diuretics, causing a great loss of fluids with no effect on serum proteins, which in turn increase inside the pleural cavity mimicking the exudate range. 18 To try to solve this issue, and generally bypass the dichotomic classification of transudates and exudates, Heffner et al. proposed to apply a Bayesian approach in which they suggest calculating continuous likelihood ratios to deduce the post-test probability of an effusion being of a certain aetiology, starting from a clinically established pre-test probability. 20 Although interesting, this approach is however of difficult application in clinical practice. 19 To overcome the problem of PF misclassification in cardiac failure patients, the calculation of serum to effusion albumin gradient (SEAG) was therefore proposed. In the original study, using a SEAG >12 g/L to indicate a transudate gave a specificity of 100%. 21 As often happens for a newly proposed test, the SEAG specificity decreased to 92% in a further validation study, which however confirmed that the use of SEAG was able to reduce the misclassification rate in patients receiving diuretics. 22 In a further study, the improvement was not statistically significant. 23 Nevertheless, Light 24 himself more recently supported the use of SEAG.
In conclusion, even if some authors advanced criticisms based upon the uselessness of LDH ratio calculation because of the presumed independency of PF and serum enzyme values,25,26 Light’s criteria remain the first-level approach as biochemical investigation of PF.3–5 As the ‘abbreviated Light’s criteria’ (PF/serum total protein ratio >0.5 and PF/serum LDH ratio >0.6, respectively) maintain the same diagnostic accuracy, they should be preferred. SEAG calculation should be limited to evaluate pleural effusions in patients with cardiac failure assuming diuretics.
Automated cell count
Differential cell count parameters in the pleural fluid and corresponding decision thresholds for specific clinical conditions.

Presence of red blood cells in PF is mainly related to traumatic withdrawal. A strategy has been proposed to differentiate between this condition and the presence of a true haemorrhagic effusion utilizing a haematocrit measurement on the PF sample and on a paired blood sample. A PF haematocrit 50% higher than the corresponding value on blood would suggest a haemorrhagic effusion. 11
Additional tests useful in specific situations
PF Triglycerides
Interpretative criteria for selected biochemical tests on pleural fluid.

PF pH and glucose
The biochemical tests with a better performance for detecting an infective aetiology of a pleural effusion are glucose and pH measured in PF.3,30 In these conditions, both test values decrease because of the inflammatory cell and infectious agent metabolisms. A PF acidosis, that is, a pH value <7.30, shows a good accuracy. Furthermore, when the pH value goes below 7.20, it may indicate the need for draining a parapneumonic effusion. 30 A PF glucose concentration <60 mg/dL (3.4 mmol/L) may indicate various clinical scenarios, including parapneumonic effusions, but even malignancy or rheumatoid arthritis. 28 This test is therefore not very specific but helpful if considered together with clinical data.8,11 Some authors have restricted its use only to the investigation of suspected rheumatoid effusion, as the only condition with undetectable glucose concentrations in PF was rheumatoid empyema.16,31 In addition to preservation of PF samples for assay by using tubes containing a glycolysis inhibitor, glucose measurements on PF should be performed after at least 8 h of fasting to permit the PF/plasma equilibration.
PF ADA
A marked increase in PF ADA levels has long been proposed as a diagnostic marker of extrapulmonary tuberculosis. 32 Nevertheless, only recently the scientific evidence has supported its introduction in the related guidelines.33,34 In a meta-analysis, which included 174 studies, the determination of ADA in PF showed sensitivity and specificity of 0.92 (CI: 0.90–0.93) and 0.90 (CI: 0.88–0.91), respectively, for tuberculous pleurisy, with positive and negative likelihood ratios of 8.92 and 0.09 at an ADA decision level of 40 U/L. 35
Evidence gaps
The role of amylase measurement in PF remains greatly controversial. Basically, there is no evidence of the utility of this test, since amylase (or better pancreatic lipase) 10 can be easily measured on serum samples to identify the presence of a pancreatic injury, avoiding thoracentesis and its possible complications, while pancreatic fistulas are better detected by other clinical and instrumental means. 36 Accordingly, CLSI guideline and other authoritative works do not recommend routinely performing amylase in PF.3,6 Epithelial tumours of the lung and ovary may produce considerable amylase activity (usually salivary-type) and give rise to hyperamylasemia. 10 PF may also contain the enzyme as a result of the presence of the tumour. It remains however unclear which added value the measurement of amylase in PF may provide in these conditions. 37
The NT-proBNP determination in PF has been proposed as an alternative to SEAG for detecting transudates related to cardiac failure, which are potentially misclassified as exudates by using Light’s criteria. 38 However, if a cardiac origin of pleural effusion is suspected and drainage is not necessary, the cardiac impairment can be detected through B-type natriuretic peptide measurement in blood, avoiding thoracentesis, also considering that blood and PF concentrations of this analyte are strictly related. 7
Areas for further research
Although a plenty of publications have referred to diagnostic accuracy of Light’s criteria and/or SEAG for distinguishing exudates from transudates, few studies have investigated other combined ratios or grandients.26,39 For instance, the albumin PF/serum ratio could be considered in further research for the main diagnostic purpose, either singly or in combination with LDH ratio as a replacement of total protein ratio. Study protocols should be performed aiming to evaluate the accuracy in distinguishing between exudates and transudates of estimates containing the measurement of albumin instead of the measurement of total proteins both as simple PF/serum albumin ratio and ‘modified’ Light’s criteria, that is, PF/serum albumin and PF/serum LDH ratios. These studies should be carefully designed to establish optimal diagnostic thresholds, without forgetting the impact of the assay selectivity in measuring albumin on serum and PF and the need to use accurate methods.7,40 Another area of investigation can be the definition of the interference thresholds by haemolysis on the LDH assay on PF at which the misclassification between exudates and transudates by using Light’s criteria becomes clinically relevant; this should be obtained by a systematic evaluation of the haemolysis index detected automatically by chemistry platforms.13,41 More in general, studies about PF must explicitly report pre-analytical and analytical details. 7 Finally, external quality assessment exercises should be routinely organized by scheme providers to assure the adequate quality of PF and other body fluid measurements. As Chubb and Williams anticipated few years ago, 7 in an era of new regulatory frameworks, the lack of measuring systems specifically validated and marketed for use with body fluids other than serum and urine may become an issue difficult to solve. 42
Conclusions
Recommended steps for pleural fluid analysis.

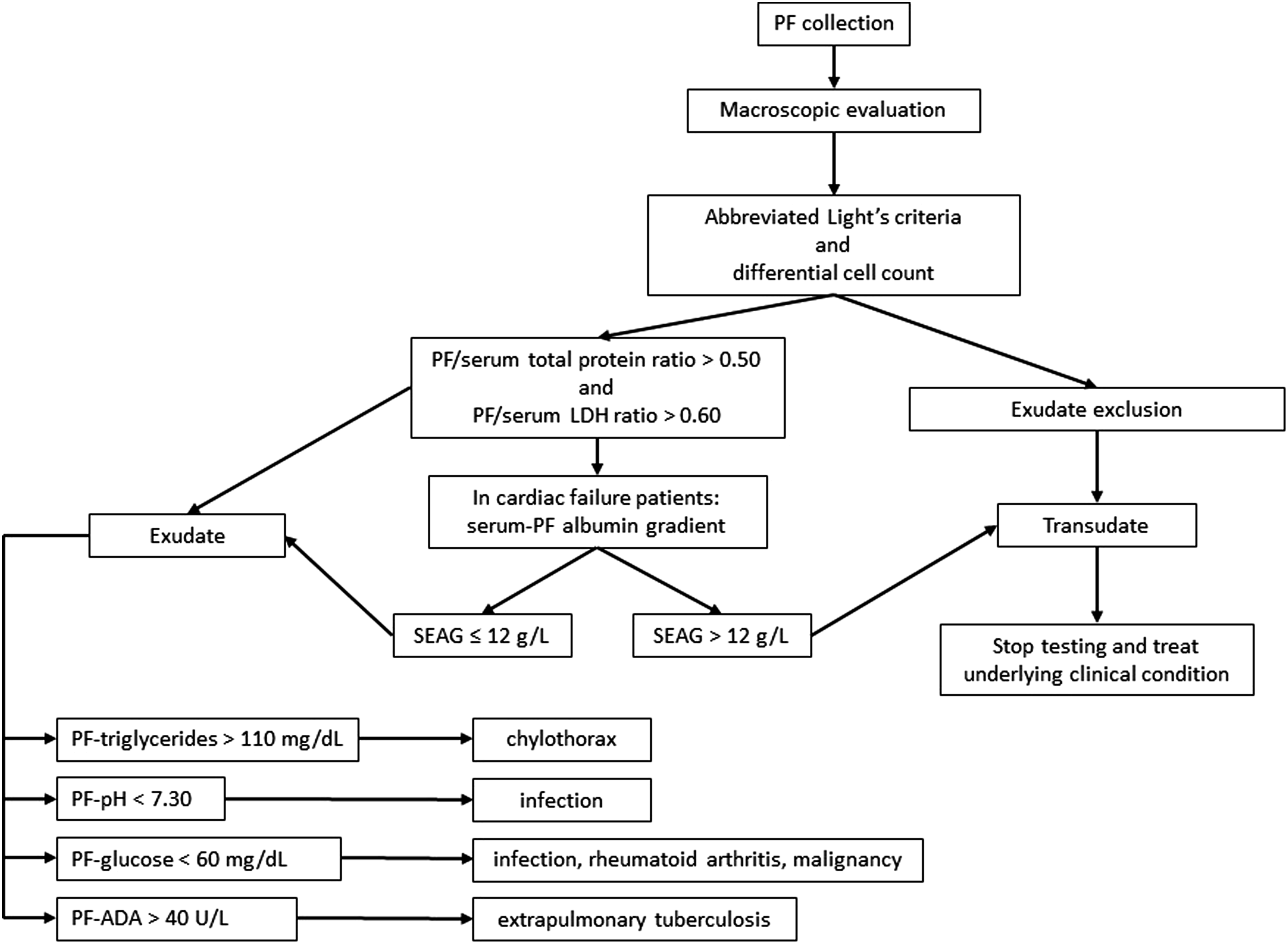
Flowchart describing pleural fluid (PF) management. LDH, lactate dehydrogenase; SEAG, serum–effusion albumin gradient; ADA, adenosine deaminase.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Guarantor
MP.
Contributorship
CA and LR researched literature. CA and MP conceived the study. CA and EA cured data analysis. CA wrote the first draft of the manuscript. EA, AD, and MP supervised the study and reviewed and edited the manuscript. All authors approved the final version of the manuscript.
