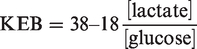Abstract
Background
Simultaneous cytological and metabolic investigation of the pleural effusion provides clinically relevant information about the type and intensity of immune response in the pleural cavity.
Methods
We investigated 1329 pleural effusions from patients with different pathological changes in the pleural cavity. Evaluated parameters were differential cell count of neutrophils, eosinophils, lymphocytes and monocytes, and values of the coefficient of energy balance.
Results
We found the lowest numbers of cells and the highest coefficient of energy balance values in patients with heart failure and sepsis; relatively high frequency of eosinophils and slightly decreased coefficient of energy balance values in patients with pneumothorax and haemothorax; the predominance of lymphocytes and low coefficient of energy balance values in patients with tuberculous pleuritis; the predominance of neutrophils and variable coefficient of energy balance values in patients after chest surgery; the highest presence of neutrophils and very low coefficient of energy balance values in patients with chest empyema and the predominance of lymphocytes and normal to low coefficient of energy balance values in patients with pleural malignancy.
Conclusions
Our findings in patients with heart failure and sepsis suggest the absence of inflammation in the pleural cavity. We observed the manifestation of tissue repair in patients with pneumothorax and haemothorax. Patients with tuberculous pleuritis were predominantly characterized by T cell-driven immune response and oxidative burst of macrophages. We found different intensities of immune responses to the chest surgery. The typical finding in patients with empyema was oxidative burst of neutrophils. In patients with pleural malignancy, weak cytotoxic inflammation predominates together with the intensive inflammation characterized by oxidative burst of macrophages.
Keywords
Introduction
An important part of the routine pleural effusion examination is its cytomorphological analysis.1–5 In addition to the detection of tumour cells, microbial agents, haemorrhage and tissue destruction, one can also evaluate the presence of immunocompetent cells.
Determination of a particular type of local immune response in the pleural cavity is possible by the assessment of both cellular and functional parameters. Activated immunocompetent cells consume a lot of energy.6–12 We consider the parameters of glucose metabolism as a suitable tool for the evaluation of the immune system activation in the pleural effusion. Many authors determined the concentrations of glucose or lactate in the pleural effusions.1–4,13–16 However, clinical value of these parameters is limited. The concentrations of glucose in the pleural effusions are dependent on the concentrations of glucose in the blood. Concentration of lactate in the pleural effusion is not based solely on the extent of anaerobic metabolism in the pleural space but also on the supply of energy substrate (glucose). Therefore, we derived an equation for the identification of the theoretical average number of molecules of adenosine triphosphate (ATP) produced from one molecule of glucose under conditions found in the extravascular compartment. We designed this parameter as the coefficient of energy balance (KEB)17,18
[glucose] = molar concentration of glucose in the pleural effusion (mmol/L)
[lactate] = molar concentration of lactate in the pleural effusion (mmol/L).
We initially used this equation exclusively for the analysis of the cerebrospinal fluid. Later, KEB application was extended to investigate other extravascular body fluids, including pleural effusions. 19
Two energy models of the pleural compartment 19
Oxygen is dissolved in the pleural effusion under physiological conditions, enabling predominantly aerobic metabolism in the pleural compartment. This is associated with a relatively high production of ATP, which is expressed as a high KEB value (Figure 1).
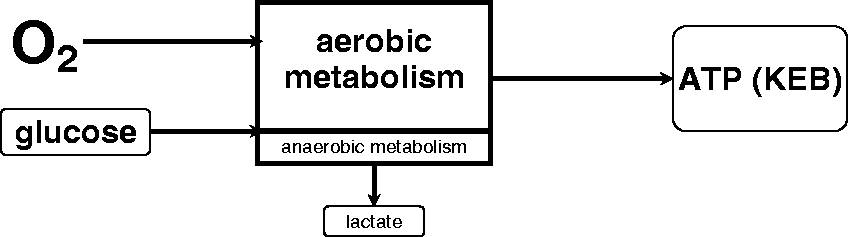
The first model of energy relationships in the pleural effusion – physiological response.
Pathological changes in the pleural compartment are usually associated with the inflammatory reaction (Figure 2). There is a great demand for activated immunocompetent cells in the pleural effusion for energy. Therefore, they consume higher amounts of glucose along with more oxygen utilization. This leads to the decreased level of oxygen in pleural effusion and results in anaerobic metabolism with an overproduction of lactate. Anaerobic metabolism is energetically less efficient, resulting in decreased ATP production. This is reflected in decreased KEB values.
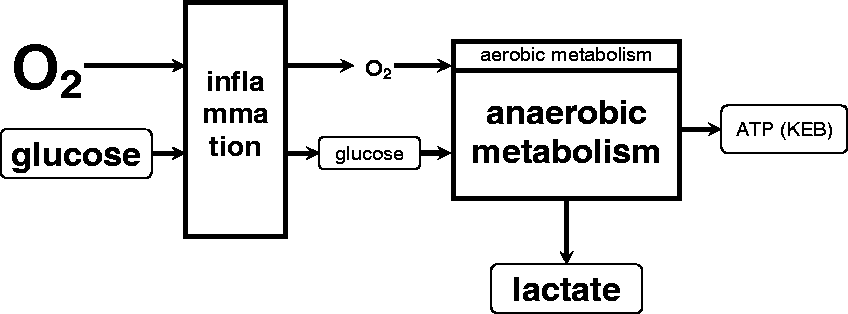
The second model of energy relationships in the CSF – local inflammatory response.
Material and methods
We investigated in total 1329 samples of the pleural effusions taken from 178 patients with heart failure, 58 patients with systemic sepsis, 88 patients with pneumothorax and haemothorax, 24 patients with tuberculous pleuritis, 252 patients who underwent chest surgery, 276 patients with chest empyema and 453 patients with malignancy with the involvement of pleura.
The pleural effusion samples were taken via pleural cavity drainage into a test tube without anticolagulants and immediately transported to our laboratory. The number of cells was determined, and microscopic smear using cyto-centrifuge method was prepared immediately after receiving the sample. Another aliquot of the sample was centrifuged, and the molar concentrations of glucose and lactate were determined. The rest of the supernatant was stored at +4°C to +8°C.
The total number of elements in the pleural effusion was calculated under the optical microscope using a Fuchs-Rosenthal chamber. Permanent cytological smears were stained using Hemacolor (Merck Co., Germany). Microscopic analyses were performed using Olympus BX40 microscope to determine cellular composition of pleural effusions.
We measured the molar concentrations of glucose using the hexokinase method and molar concentrations of lactate using the lactate-oxidase and peroxidase method on a Cobas 6000 analyzer (Roche Diagnostics, Czech Republic). The KEB values were calculated for all samples.
The concentrations of lactate dehydrogenase catalytic activities (LD) in the pleural effusions were measured using the IFCC method on a Cobas 6000 analyzer (Roche Diagnostics, Czech Republic).
The mass concentrations of total protein in the pleural effusions were measured using Biureth method on a Cobas 6000 analyzer (Roche Diagnostics, Czech Republic).
Numbers of nucleated cells and relative frequencies of neutrophils, eosinophils, lymphocytes and monocytes in the pleural effusions and the KEB values in the particular groups of patients are presented as a median and the first and the third quartile. Patients with heart failure were used as a comparative group. In addition, we have tested that KEB values distinguish between chest empyemas and other causes of pleural effusions. Statistical analysis was performed using the Mann-Whitney U test (p < 0.01, or p < 0.001, were considered as significant) via STATISTICA 13.3 software (StatSoft Inc., USA).
Based on the values of KEB, we stratified our patients in all patient subgroups as follows:
KEB values in the range from 38.0 to 28.0: predominant aerobic metabolism in the pleural effusion17–19 KEB values in the range from 28.0 to 20.0: slight increase in the anaerobic metabolism in the pleural effusion17–19 KEB values in the range from 20.0 to 10.0: moderate increase of anaerobic metabolism in the pleural effusion.17–19 KEB values <10.0: strong anaerobic metabolism in the pleural effusion.17–19
Table 2 shows the numbers and per cent of findings divided in consideration with the KEB values and predominance of the type of immunocompetent cells in pleural effusions.
Light’s criteria are the gold standard to differentiate transudates from exudates5,20 and have evolved over time. Therefore, their usage is not uniform. We compared specificities, sensitivities and diagnostic efficiency
Results
Pleural effusions of our patients with heart failure are represented by a low number of nucleated cells, predominance of lymphocytes and aerobic metabolism with predominantly high KEB values. Decreased KEB values are rare (Tables 1 and 2). Absence of significant pathological findings led us to using these patients as a comparative group.
The results of the analysis of the pleural effusions in seven groups of patients.
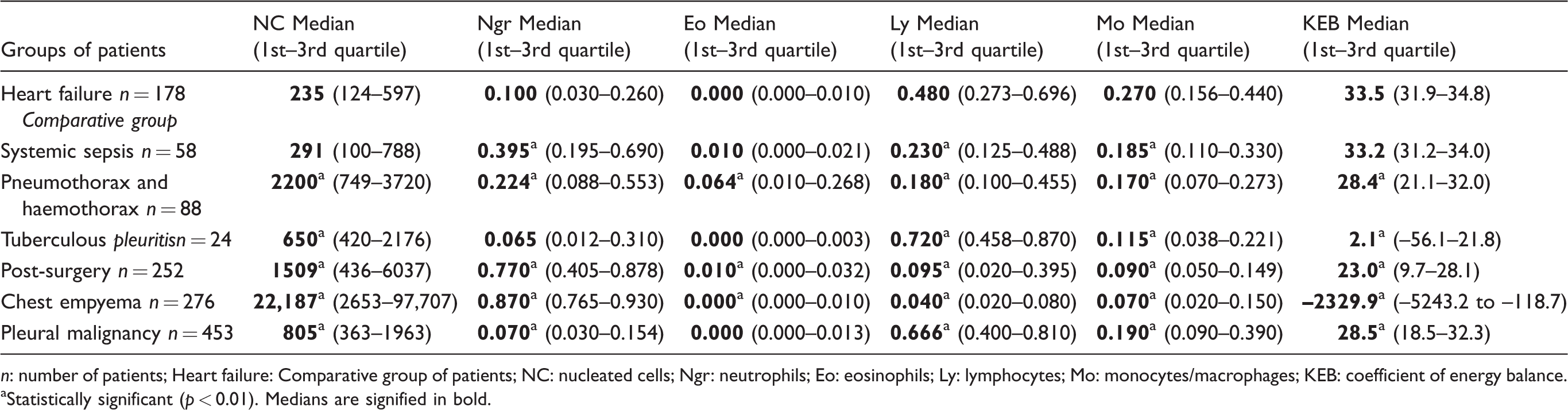
n: number of patients; Heart failure: Comparative group of patients; NC: nucleated cells; Ngr: neutrophils; Eo: eosinophils; Ly: lymphocytes; Mo: monocytes/macrophages; KEB: coefficient of energy balance.
aStatistically significant (p < 0.01). Medians are signified in bold.
Distribution of patients in accordance with predominant type of immunocompetent cells in pleural effusions and KEB values.
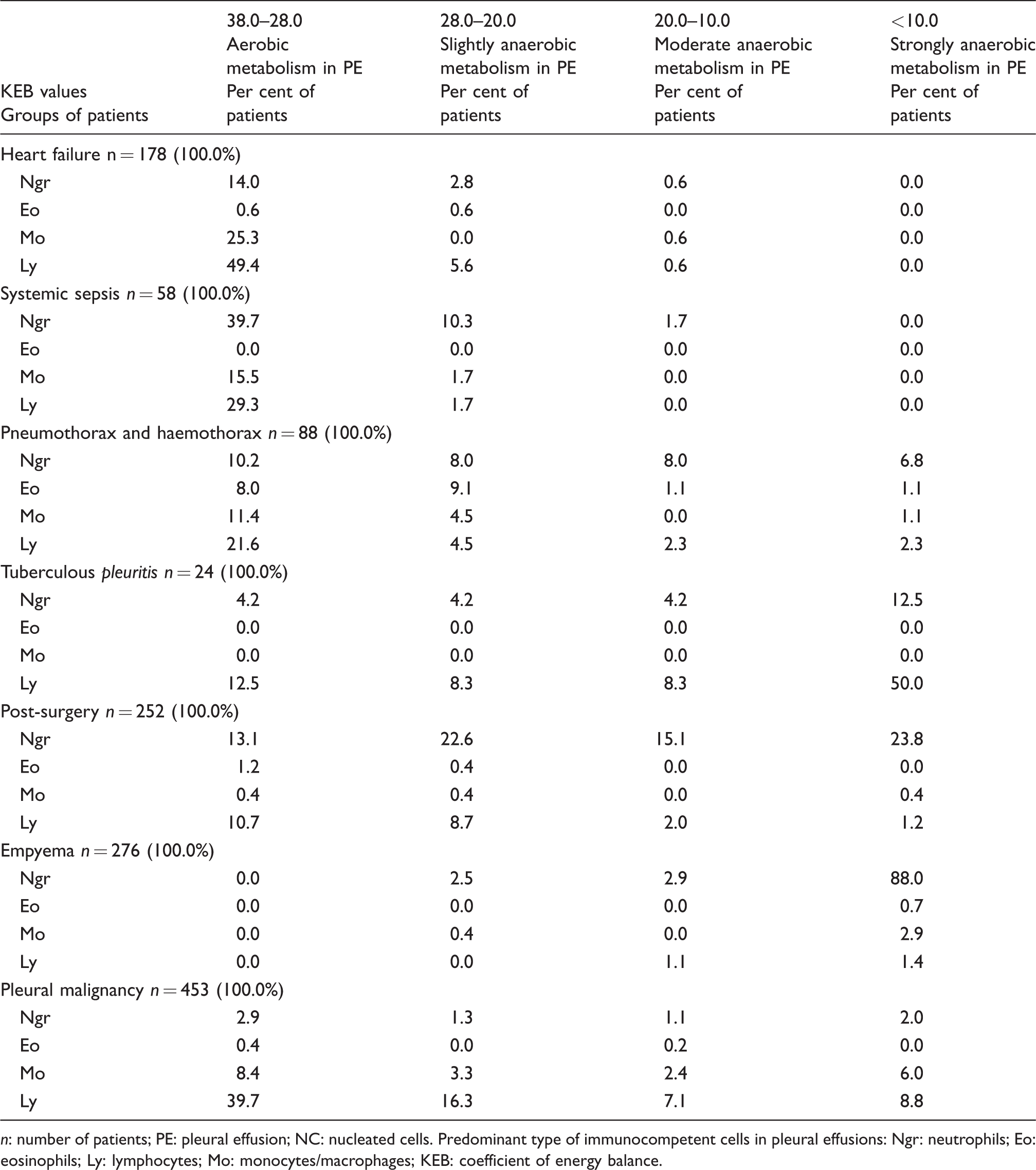
n: number of patients; PE: pleural effusion; NC: nucleated cells. Predominant type of immunocompetent cells in pleural effusions: Ngr: neutrophils; Eo: eosinophils; Ly: lymphocytes; Mo: monocytes/macrophages; KEB: coefficient of energy balance.
Pleural effusions of patients with systemic sepsis also show low number of nucleated cells and predominance of high KEB values. We observed a statistically significant increase in frequencies of neutrophils and a decrease of lymphocytes and monocytes/macrophages compared with patients with heart failure (Tables 1 and 2).
We found acceptable diagnostic efficiencies of low concentrations of lactate and total protein in pleural effusions in the subgroups of patients with heart failure and systemic sepsis. In evaluation of the high KEB values (>28.0), diagnostic efficiencies in both subgroups were improved (Table 3).
Specificities, sensitivities and diagnostic efficiencies of some traditional Light’s criteria parameters and KEB values in selected groups of patients.
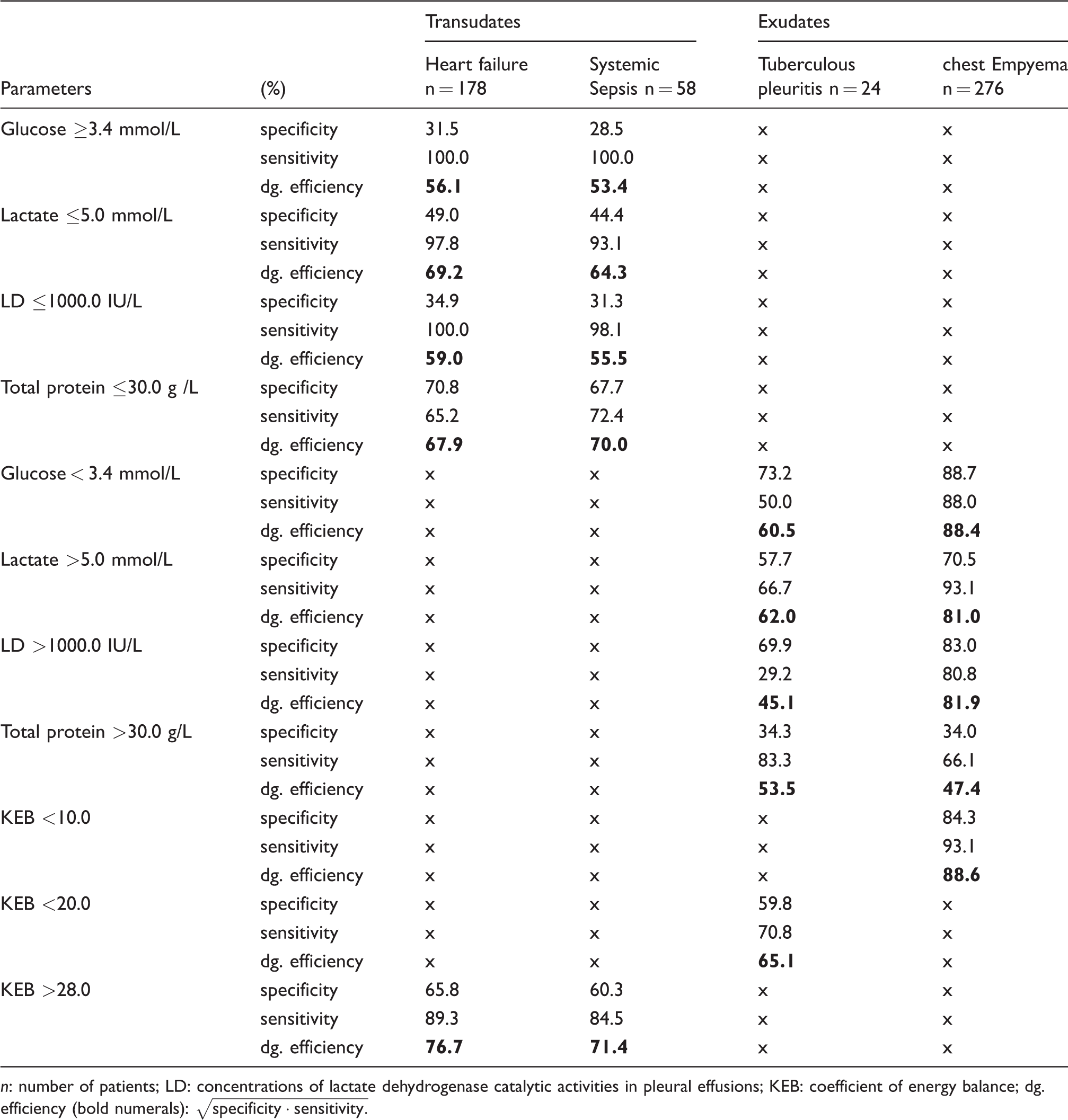
n: number of patients; LD: concentrations of lactate dehydrogenase catalytic activities in pleural effusions; KEB: coefficient of energy balance; dg. efficiency (bold numerals):
In pleural effusions of patients with pneumothorax and haemothorax, we found a statistically significant increase in the number of nucleated cells compared with patients with heart failure. Lymphocytes and monocytes/macrophages predominated in cases of aerobic metabolism with high KEB values. In contrast, increased frequencies of neutrophils were associated with a decrease of KEB values. We further observed a significant increase in the frequency of eosinophils (Tables 1 and 2).
In our small group of patients with tuberculous pleuritis, we found a significant increase in the number of nucleated cells in pleural effusions compared with patients with heart failure. Lymphocytes and a significant decrease of KEB values dominate. We further found several cases with a predominance of neutrophils, also with a trend to low KEB values (Tables 1 and 2). Acceptable diagnostic efficiencies were found in the evaluation of the low concentrations of glucose and the high concentrations of lactate in pleural effusions of these patients. We reached slightly better results in the evaluation of the KEB values <20.0 which define a middle to strong anaerobic metabolism in pleural cavity (Table 3).
In pleural effusions of our patients after chest surgery, we observed a significant increase in the number of nucleated cells with a predominance of neutrophils and variable KEB values which indicated different metabolic conditions. Predominance of lymphocytes in pleural effusions was usually accompanied by high or only slightly decreased KEB values. Common findings were relatively higher frequencies of eosinophils (Tables 1 and 2).
Pleural effusions of our patients with chest empyema are typically represented by very high numbers of nucleated cells, dominance of neutrophils and very low KEB values. Other findings were rare (Tables 1 and 2). We found that significantly decreased KEB values (P < 0.001) distinguish between chest empyemas and other causes of pleural effusions.
Further, we found similarly high diagnostic efficiency of the very low KEB values (<10.0) compared with low glucose concentrations in pleural effusions of these patients. Diagnostic efficiencies of the high concentrations of lactate and lactate dehydrogenase catalytic activities in pleural effusions were lower. Diagnostic efficiency of the high concentrations of total protein in pleural effusions was insignificant (Table 3).
In the relatively large group of patients with pleural malignancy, we found a significant increase in the number of nucleated cells with a predominance of lymphocytes in pleural effusions compared with patients with heart failure. Presence of monocytes/macrophages is significantly lower. Neutrophils were found sporadically. We observed a predominance of high or slightly decreased KEB values. Low KEB values were less frequent (Tables 1 and 2).
Discussion
Heart failure and systemic sepsis
The most common reason to develop a pleural effusion in patients with heart failure or sepsis is a systemic response, and the pleural cavity is not affected in majority of cases. 21 As a common consequence of this, the pleural effusion is characterized by low numbers of immunocompetent cells and high KEB values indicating predominantly aerobic metabolism in the pleural compartment. In accordance with Light’s criteria, pleural effusions are usually classified as transudates.1,4,5,13,20,21
We observed higher frequencies of neutrophils in pleural effusions from patients with sepsis when compared with patients with the heart failure (Table 1). We explain this phenomenon as the manifestation of increased systemic distribution of neutrophils during sepsis.23,24
Pneumothorax and haemothorax
Pneumothorax and haemothorax are caused by injury in the pleural cavity. In these effusions, there are elevated numbers of erythrophages, siderophages and haematoidin, suggesting a scavenging reaction in pleural cavity. 25 The relative numbers of neutrophils, lymphocytes and monocytes/macrophages are the same. We found statistically higher numbers of eosinophils in pleural effusions in this group of patients. Some authors explain this phenomenon as a result of chest trauma and the presence of either air or blood in the pleural cavity.1,3,4,26 We suggest that eosinophils are involved in the post-traumatic repair and injury healing. 27
In patients with pneumothorax and haemothorax, we observed an aerobic or slight anaerobic metabolism in the pleural effusions. We found more than 20% of cases had moderate or strong anaerobic metabolism in accordance with more intensive inflammatory response in pleural cavity (Table 2). The KEB values were subsequently increased, indicating resolution of the local inflammation. In contrast, further decreases of KEB values were usually associated with the progression of the local pathological process, most often, microbial infection.
Tuberculous pleuritis
Pleural effusions of patients with tuberculous pleuritis are usually characterized as exudates with predominance of lymphocytes or in the early phase, the presence of neutrophils and decreased concentrations of glucose.2–5,15,21 The cytological-energy findings in our small group of patients with the tuberculous pleuritis are similar. In patients with chest empyema, we observed significantly decreased KEB values in majority of patients which indicate anaerobic metabolism. Lymphocytes are prevailing cells in effusions of patients suffering from tuberculous pleuritis with the exception of patients in the very early stage of infection. The principal inflammatory response in these patients is Th1-driven immune reaction to Mycobacterium tuberculosis. Activated CD4+ Th1 T cells produce large amounts of interferon γ (IFN-γ) which is essential for the full activation of macrophages which are able to kill and eliminate phagocytosed pathogens.2,15,28 The explanation for this predominant anaerobic metabolism is an oxidative burst of activated macrophages consuming large amounts of oxygen. 18
Chest surgery
Moderate increases in the count of leucocytes with a predominance of neutrophils and slight increase of eosinophils were found in effusions of patients after chest surgery (Table 1).21,26 These findings together with the aerobic or slightly anaerobic KEB values suggest a local reaction to the tissue damage and bleeding into the pleural cavity.27,29–31 However, more profound changes with moderate or even strong anaerobic metabolism indicate a more intensive inflammatory response which was found in more than 40% of patients (Table 2).
Determination of the KEB values allowed us to monitor local inflammatory responses within the pleural cavity. A decrease in the KEB value indicates the progression of the inflammatory response as a consequence of usually microbial infection. On the other hand, increased KEB values indicate resolution of inflammation.
Chest empyema
Pleural effusions of the patients with chest empyema are defined as exudates with predominance of neutrophils and low concentrations of glucose.4,14 When compared with other groups of our patients, the results are significantly different. High numbers of neutrophils and very low KEB values in the pleural effusions indicate very strong anaerobic metabolism in the pleural cavity. The usual reason for this is a purulent inflammation with an oxidative burst of neutrophils exposed to extracellular bacteria.6–8,11
Pleural malignancy
The presence of the tumour cells found by the cytological analysis of the pleural effusion is an important method of pleural tumour detection.3–5,16,32 It is also valuable to evaluate immunocompetent cells in the pleural effusion. In accordance with Karkhanis and Joshi 4 and Garrido et al., 21 we found a predominance of lymphocytes in the effusions of patients with the malignancy, resembling cellular composition of patients with tuberculous pleuritis.1,3,4,13,21 The presence of tumour cells could elicit an oxidative burst of macrophages resulting in a local increase of anaerobic metabolism. 33 We found this phenomenon in approximately a quarter of all patients with pleural malignancy (Table 2). On the other hand, the relatively high KEB values in this group of patients indicate moderate local immune response in terms of cytotoxic or antibody-mediated inflammation.34–36
Conclusions
The cytological-energy analysis of the pleural effusions allows us to determine the type and intensity of local immune or inflammatory response in the pleural cavity.
The high or only slightly decreased KEB values in patients with heart failure or systemic sepsis indicate a local reaction reflecting the systemic changes. Higher frequencies of neutrophils in the pleural effusions from patients with systemic sepsis usually are associated with an increased systemic distribution of neutrophils.
Variable distribution of immunocompetent cells and a high or slightly decreased KEB values in patients with pneumothorax and haemothorax indicate a local scavenging reaction and tissue repair in the pleural cavity. A profound decrease of KEB values usually indicates progression of the inflammatory response in the pleural cavity. The most frequent reason for this is microbial infection.
The predominance of lymphocytes and low KEB values in patients with tuberculous pleuritis is typical for Th1-driven immune response against M. tuberculosis and oxidative burst of macrophages.
The predominance of neutrophils and slight or moderate decrease of KEB values in patients after chest surgery reflects a local reaction of innate immunity cells to the tissue damage and bleeding into the pleural cavity. Very low KEB values in approximately 40% of these cases indicate oxidative burst of neutrophils and potential purulent inflammation.
A significant predominance of neutrophils in the pleural effusions with very low KEB values indicates purulent inflammation and is typical for patients with chest empyemas.
Predominance of lymphocytes and relatively high KEB values are seen in patients with malignancy of pleura. This is associated with a weak cytotoxic reaction and antibody-mediated inflammatory response in the pleural cavity. The high variability of the KEB values in this group of patients can indicate a unique pattern which can include very intensive inflammation in the term of an oxidative burst of macrophages.
Footnotes
Acknowledgements
We thank Gregory Jeffrey Evans for critical proofreading of the article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Internal Grant of the Krajská zdravotní, a.s. in Ústí nad Labem, Czech Republic ‘IGA-KZ-2017–1-5’ and by Charles University in Prague, Faculty of Medicine in Hradec Králové, Czech Republic, project ‘PROGRES Q40/10’.
Ethical approval
This study was approved by the local Ethics Committee of the Masaryk Hospital Ústí nad Labem (reference number: 261/1).
Guarantor
PK.
Contributorship
PK conceived of the study, carried out the cytological-energy investigation of the pleural effusions and drafted the article. VM carried out the clinical study. IM performed the statistical analysis. MČ carried out the cytological study. IS carried out the clinical study. JKra carried out the clinical study. OK carried out the clinical study. IM-W carried out the clinical study. JKu carried out the clinical study. EH carried out the cytological-energy investigation of the pleural effusions. JKre participated in the design of the study and corrected final text. All authors reviewed and edited the article and approved its final version.