Abstract
Background
Sapropterin has been approved as a treatment option for individuals with Phenylketonuria in the United Kingdom. Individuals are assessed as responsive to Sapropterin by a ≥30% reduction in Phenylalanine (Phe) concentrations using dried blood spot (DBS) specimens. DBS quality is critical for accurate and precise measurement of Phe. Currently, UK national guidelines for DBS specimen acceptance do not exist for patient-collected DBS specimens. We adopted evidence-based guidelines for specimen acceptance criteria and retrospectively assessed the impact of introducing these guidelines on specimen rejection rates. Methods: Laboratories were invited to audit the quality of DBS specimens routinely received for Phe monitoring using: (1) existing acceptance/rejection criteria and (2) proposed national guidelines.
Results
Ten laboratories audited 2111 specimens from 1094 individuals. Using existing local guidelines, the median rejection rate was 4.0% (IQR 1.5–7.2%). This increased to 21.9% (IQR 10.0–33.0%) using the proposed guidelines. Where reason(s) for rejection were provided (n = 299); 211/299 (70.6%) of DBS specimens were too small or multi-spotted. 380 individuals had more than one sample evaluated; 231/380 (60.8%) provided specimens of acceptable quality, 37/380 (9.7%) consistently provided poor-quality DBS specimens.
Conclusions
There is significant variability in the quality of patient-collected DBS specimens. If unacceptable specimens are not rejected, imprecise/inaccurate results will be used in clinical decision making. Using annual workload data for England, this equates to 12,410 incorrect results. Individuals and parents/carers should receive ongoing training in blood collection technique to ensure use of evidence-based acceptability criteria does not cause undue distress from increased sample rejection rates.
Introduction
Phenylketonuria (PKU) is an inherited metabolic disorder (IMD) caused by deficiency in the enzyme phenylalanine hydroxylase (OMIM #261600). Traditionally treatment comprises a low protein diet and phenylalanine (Phe) free amino acid substitute with monitoring of dried blood spot (DBS) Phe and tyrosine concentrations. 1 Individuals collect blood from a finger prick onto a filter paper collection device and mail the specimen to the laboratory. In 2021, the UK National Institute for Health and Care Excellence (NICE) recommended Sapropterin-dihydrochloride for treatment of PKU. 2 Sapropterin is a synthetic form of tetrahydrobiopterin, the cofactor for phenylalanine hydroxylase. Responsiveness to Sapropterin is defined, using nationally agreed protocols, as ≥30% reduction in DBS Phe concentrations compared to baseline. 3
DBS specimens are currently collected onto filter paper collection devices (e.g. PerkinElmer-226 or Whatman-903). 4 The application of blood to the filter paper is non-volumetric and it is standard practice to assume that a 3.2 mm diameter sub-punch equates to 3.1 μL of whole blood. The importance of good quality DBS specimens to minimise sources of pre-analytical variation in the measurement of analytes has been reported.4,5 Significantly lower Phe concentrations are seen in DBS specimens <8 mm in diameter (up to 20%) and compressed specimens (up to 35%), whilst multilayered specimens result in a positive bias (up to 15%). 4 It has been recommended that only DBS ≥8 mm but <14 mm should be accepted for newborn screening and that insufficient, compressed or multi-spotted specimens should also be rejected.4,5
We have derived a set of evidence-based guidelines for DBS specimen acceptability to be adopted by UK metabolic biochemistry laboratories and assessed the impact of the introduction of the guidelines into routine clinical practice.
Methods
In November 2021, laboratories in the UK Metabolic Biochemistry Network (MetBioNet) (n = 17) were invited to retrospectively audit at least 200 DBS specimens received for Phe monitoring in the preceding 3 months. DBS specimen quality was assessed against two sets of criteria: (1) each laboratory’s existing specimen acceptance policy, where one existed (2) guidelines proposed by representatives of MetBioNet and the UK Newborn Screening Laboratory Network (UKNSLN) based on published work: • One evenly saturated circle of blood greater ≥8 mm in diameter, formed from a single hanging drop of blood, which has completely soaked through the filter paper. • Expired filter paper cards must not be used. • Blood spots must not overlap, be multi-spotted or multilayered, damaged, contaminated, or be compressed during drying. • Drying must be complete before specimen is placed in glassine envelope. • Samples must arrive in the laboratory within 14 days of collection.
Data were collected using a standardised audit proforma and analysed in Microsoft Excel 2016. For each specimen reviewed, an anonymised patient identifier was applied, and data captured on whether the specimen was accepted/rejected and, where possible, the reason for rejection. Patient demographics were not collected. Rejection rates were calculated for each laboratory against each set of criteria. Where more than one specimen from an individual patient was included, the consistency of the quality of the DBS was categorised as always good, always poor, or a mixture. Laboratories were also asked to provide data on assay performance so that the contribution of analytical imprecision could be assessed. Rates of rejection by laboratory are summarised as median (interquartile range).
Results
Ten of the 17 laboratories submitted data on 2111 specimens from 1094 individual individuals. The median rejection rate using current local laboratory practice was 4.0% (range 1.5–7.2%), increasing to 21.9% (range 10.0–33.0%) with the proposed guidelines (Figure 1). Where a rejection reason was provided (N = 299); 211/299 (70.6%) were small or multi-spotted specimens. 380 individuals collected more than one specimen; 37/380 (9.7%) consistently provided poor-quality specimens. 112/380 (29.5%) had a mixture of acceptable and unacceptable specimens (Figure 1). Effect of proposed new guidelines on assessment of blood spot quality amongst laboratories and patients (a) comparison of rejection rates, current versus new guidelines between laboratories and (b) consistency of blood spot quality amongst patients with more than one blood spot.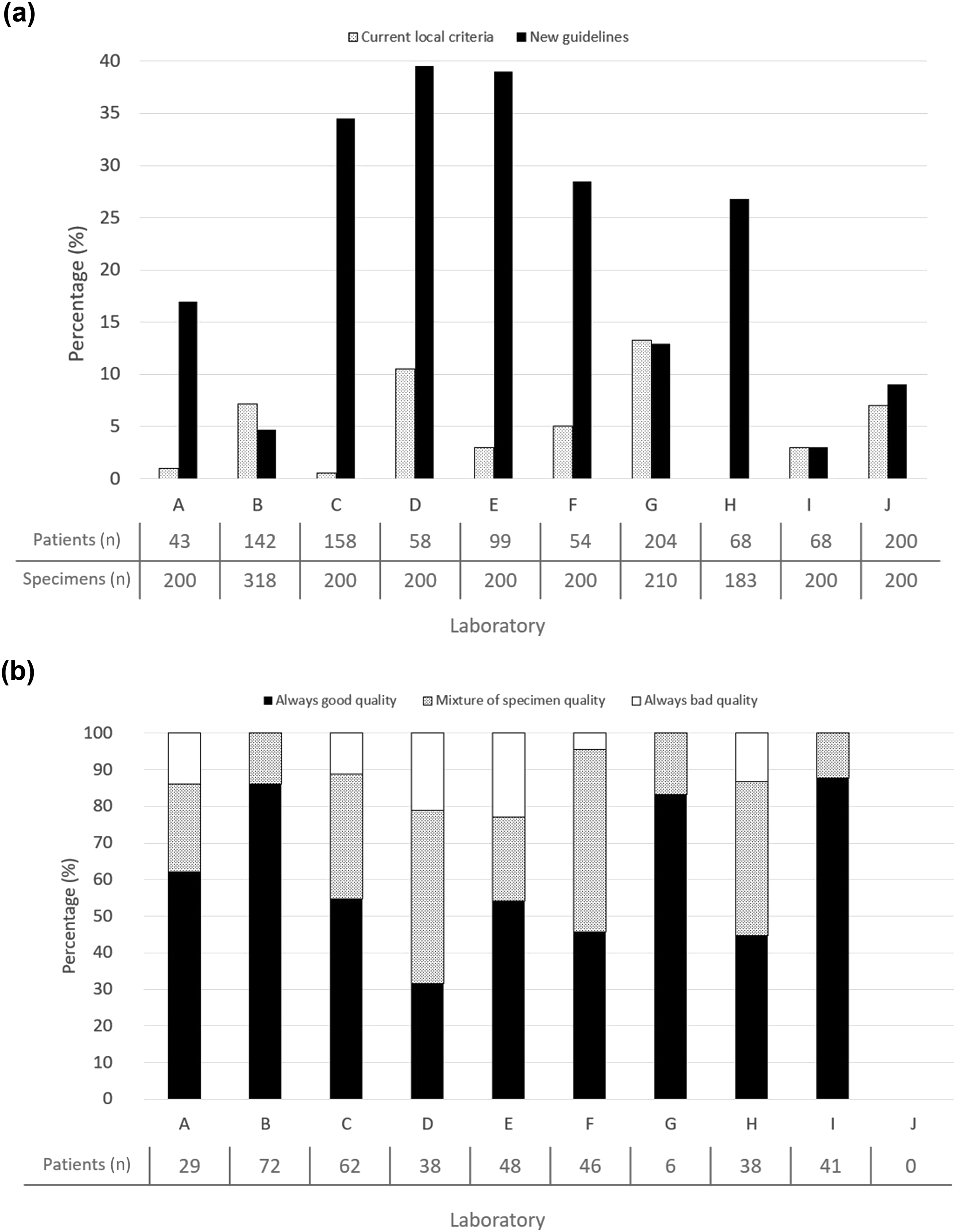
Inter-batch imprecision data was supplied by 8/17 laboratories; with the coefficients of variation (CV%) ranging from 4.5 to 10%.
Discussion
The accurate and precise measurement of Phe concentration in DBS specimens is essential for the accurate monitoring of individuals with PKU and has come to the fore recently with the requirement to assess Sapropterin responsiveness. The effect of DBS quality on the measured concentration of Phe and tyrosine has been documented in the literature.4,5
This audit demonstrated that on average 21.9% of DBS specimens would have been rejected if the new evidence-based acceptance criteria had been in place. In England it is estimated from the number of patients known to be on diet (collated by Wood et al.) 6 and the recommended frequency of testing, 1 that approximately 62,048 samples were analysed in 2021. Extrapolating the data from this audit, in 2021 12,410 results were potentially inaccurate leading to incorrect clinical decisions on titrating dietary restriction. Some laboratories had a substantial number of patients who produced specimens of inconsistent size and quality, which makes the interpretation of trends in results unreliable. The degree to which the audit predicts the outcome of introducing new criteria will vary between laboratories. While each laboratory audited a similar number of specimens, laboratories providing their service to a small number of individuals inevitably included multiple specimens from more individuals. The inclusion of multiple specimens from a single individual allowed assessment of the intra-individual consistency of collecting DBS specimens. Based on our findings, nearly 70% of individuals require re-education on DBS specimen collection to achieve accurate and precise Phe results.
There was marked variation in current practice; one laboratory had no local rejection criteria while others use more stringent criteria similar to the national newborn blood spot screening guidelines, reflecting the lack of evidence-based best practice guidelines for DBS monitoring. Adoption of national criteria for DBS specimen acceptance will support UK metabolic biochemistry laboratories to maintain consistency in DBS acceptance/rejection and ensure more accurate and precise measurement of Phe and tyrosine whilst emerging quantitative blood spot collection technologies are being validated. 7 National guidelines on DBS quality can also be applied to other IMD patient groups requiring biochemical monitoring, for example, maple syrup urine disease.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethics approval was not sought as this was an audit of routine clinical and laboratory practice using existing specimens. Patient consent was not formally asked for from those individuals data included in this audit. Patient identifiable information was not used or anonymised at each stage of the data collection process.
Guarantor
SH.
Contributorship
SLH, NC, RSC, SJM contributed to design of the study, collection of data, analysis of data, preparation, and review of the manuscript. GH, PG, HA, RB, CC and HK, contributed to collection of data and review of the manuscript.
