Abstract
Background
Measurement of antithyroglobulin antibodies (TgAb) is important in patients with differentiated thyroid carcinoma (DTC) with total thyroidectomy. These patients are monitored based on serum thyroglobulin (Tg) levels. TgAb is known to interfere with the measurement of Tg by immunoassay. This study evaluates a new methodology for the measurement of TgAb Alinity Abbott® and the concordance with other methods (first and second generation Advia Centaur Systems Siemens® and Phadia 250 Thermofisher®).
Methods
The technical characteristics of Alinity TgAb measurement methodology were analysed, with imprecision and repeatable studies. In order to assess concordance, a minimum of 69 and a maximum of 76 samples from patients with DTC and total thyroidectomy were processed in parallel by several TgAb measurement methodologies. Agreement rates were determined using kappa statistics. The correlation between the four methods was examined pairwise using McNemar test analysis.
Results
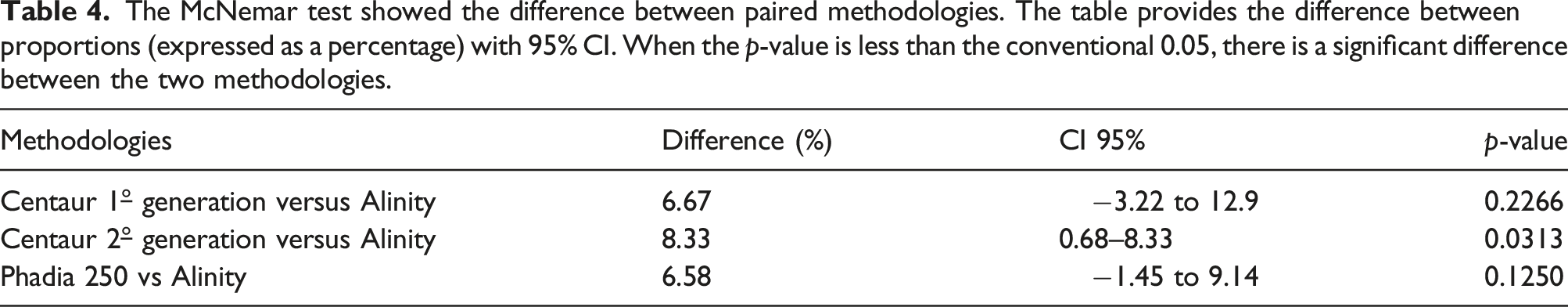
The coefficients of variation (standard deviation as a percentage of the mean % CV) for the Alinity Abbott kit reagent TgAb were within 10% included the functional sensitivity. On the other hand, the concordance analysis with the kappa index concluded substantial agreement. The McNemar test showed a significant difference between Alinity versus Centaur second generation (difference 8.33%, CI 95% 0.68–8.33, p = .0313).
Conclusion
The new methodology for the measurement of TgAb meets the imprecision standards while presenting an adequate concordance agreement with other methodologies available in laboratories. It is important to define the functional sensitivity when reporting results so that they are as reliable as possible.
Introduction
Thyroglobulin (Tg) is the most sensitive tumour marker for the follow-up of patients with differentiated thyroid carcinoma (DTC) after total thyroidectomy.1,2 However, its determination can be affected by different types of interference, among which the presence of antithyroglobulin (TgAb) antibodies could lead to an underestimation of its measurement by current chemiluminescence methods. 3
In patients with DTC treated and follow-up with Tg, TgAb bind to circulating Tg, decreasing its free concentration, thus providing falsely decreased results. On the one hand, it is essential to measure Tg and TgAb together; 4 if TgAb are positive could compromise the determination of Tg, and TgAb would be used as tumour markers in the follow-up of these patients.5,6 On the other hand, it is very important to define two different thresholds for TgAb: firstly, the interfering cut-off for the effects on Tg measurement and, secondly, positive cut-off for the diagnosis of autoimmune thyroid disease. 7 Interfering cut-off needs to calculate the functional sensitivity of these assays, since this concentration is considered critical for the interpretation of the result.
The prevalence of the presence of these TgAb in patients with DTC has been extensively studied by different authors, which some estimate around 20–30%.4,5,8
Guidelines require that TgAb should be measured in all specimens submitted for serum Tg testing because the qualitative status of TgAb (positive or negative) determines the risk of interference with Tg assay and serial quantitative TgAb measurements can be used as a surrogate tumour marker.1,9
Different assays for the measurement of TgAb are marketed, with a non-negligible variability. Some authors confirm that the degree of correlation of TgAb assays is low and the conversion of results from different methods is unreliable.6,8,10 Therefore, TgAb should be measured by the same assay during the follow-up of DTC patients. 7 Additionally, the laboratory must verify the data supplied by the manufacturer. For this reason, a comparison and interchangeability study must be carried out.
The purpose of this study is to verify the technical specifications of a new methodology for the measurement of TgAb in Alinity systems (Abbott® Laboratory), especially imprecision and cut-off. In addition, the interchangeability and concordance with other previously implemented methodologies (TgAb first and second generation Advia Centaur® Siemens and Phadia 250 Thermofisher®) will be assessed.
Material and methods
Samples
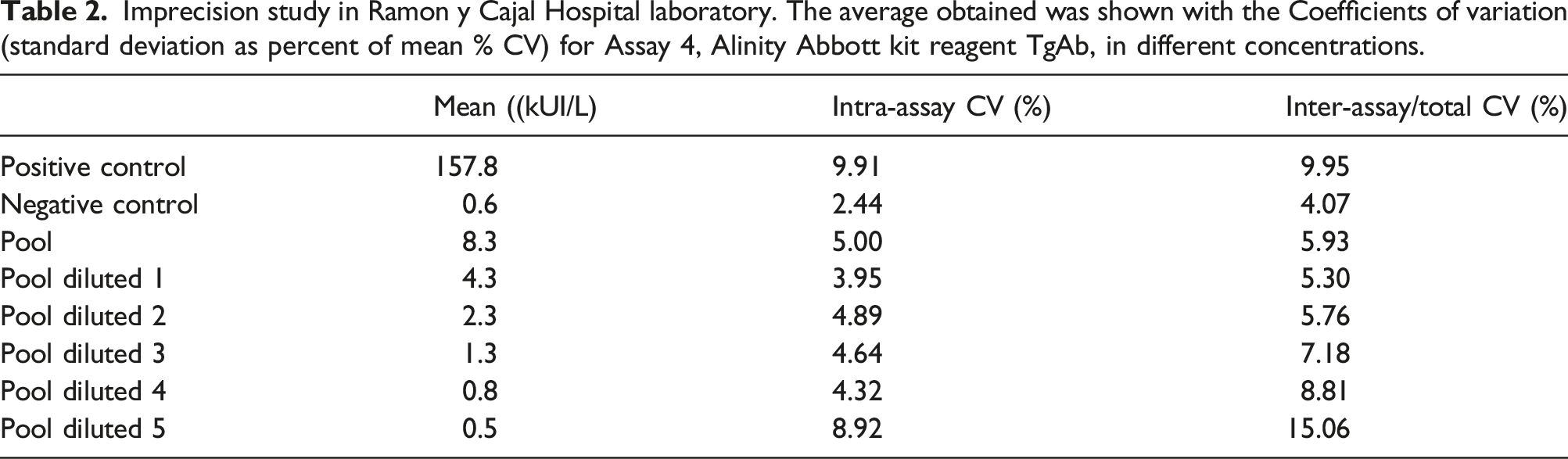
In order to verify the technical specifications of the new Alinity Abbot kit reagent TgAb, assay imprecision and functional sensitivity are studied. An imprecision study was carried out, 11 processing in triplicate for 5 days, two controls supplied by Abbott, positive control (mean 159.8 kUI/L) and negative control (mean 0.5 kUI/L). To complete the measurement range and to assess imprecision, a pool of samples with a concentration of 8.3 kUI/L was prepared. According to the manufacturer’s instructions and from this concentration, different dilutions were made with Calibrator A (TgAb-free), to obtain solutions with concentrations close to the cut-off of the assay (4.11 kUI/L). Furthermore, all these concentrations were measured in triplicate for 5 days to obtain their coefficient of variation and corresponding mean.
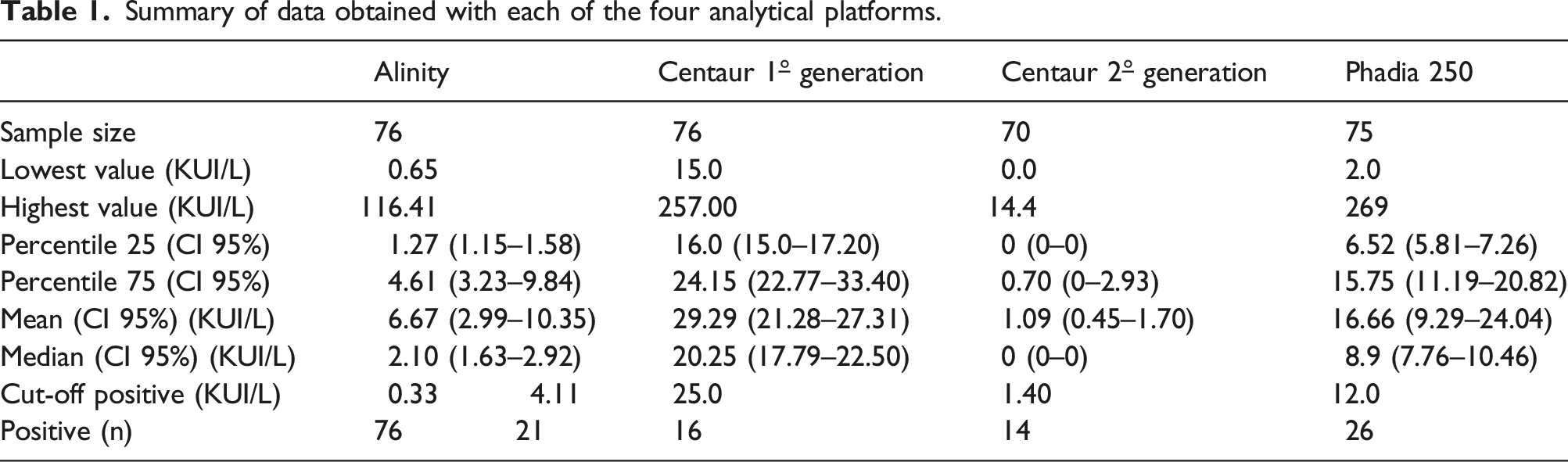
For the concordance and interchangeability study, serum samples were collected from patients with a diagnosis of DTC, who had been previously undergone a Total Thyroidectomy (TT), with undetectable Tg levels, and who had been routinely measured both parameters, Tg and TgAb. The samples were selected between January and June 2021 and processed in parallel by the four assays indicated in Laboratory Methods (Assay 1, 2, 3 y 4). All four TgAb assays were standardized against the WHO reference serum 65/93 and performed according to the manufacturer’s instructions. All samples that do not meet these criteria will be excluded. In addition, according to the consensus document of the SEQC (Spanish Society of Clinical Chemistry), 12 a minimum of 40 samples is recommended as long as the measurement range to be studied is covered. Therefore, a minimum of 70 samples and a maximum of 76 samples were selected, depending on the sample volume available.
Laboratory methods
TgAb assays: The methods for the measurement of TgAb were defined as follows:
Assay 1. First generation reagent TgAb in Advia Centaur XP Siemens® equipment based on a chemiluminescent competitive immunoassay. The ADVIA Centaur TgAb assay is a competitive immunoassay using direct chemiluminescent technology. Autoantibody against thyroglobulin in the patient sample competes with polyclonal human TgAb bound to polyclonal goat anti-human antibody covalently coupled to paramagnetic particles in the solid phase for a limited amount of acridinium ester-labelled human thyroglobulin in the Lite Reagent. 13
Assay 2. Second generation reagent TgAb in Advia Centaur XP Siemens® kit based on a chemiluminescent competitive immunoassay. The ADVIA Centaur aTgII assay is a 1-step, fully automated, dual analyte capture immunoassay using acridinium ester chemiluminescent technology for the detection of autoantibodies to thyroglobulin. This assay uses human thyroglobulin in the lite reagent and solid phase. In the Lite reagent, Tg is labelled with acridinium ester. In the solid phase, Tg is labelled with biotinyl and bound to streptavidin-coated paramagnetic particles. The TgAb from the patient sample binds to the Tg in the Lite reagent and the solid phase, thereby forming a bridge. 14
Assay 3. Phadia 250 Termofisher® kit reagent TgAb, corresponding to a sandwich fluorescent enzyme immunoassay. TgAb are coated with human thyroglobulin antigen. If present in the patient’s sample, antibodies to Tg are attached to the specific antigen. After removal of the unbound antibodies, enzyme-labelled antibodies directed against human IgG antibodies (EliAIgG Conjugate) are added to form antibody-conjugate complexes. 15
Assay 4. Alinity Abbott® kit reagent TgAb with chemiluminescent immunoassay technology for microparticle chemiluminescent immunoassay. This assay is a 2-step immunoassay for the quantitative determination of IgG class antithyroglobulin (TgAb) autoantibodies in human serum and plasma using chemiluminescent microparticle immunoassay (CMIA) technology. The sample, Tg-coated paramagnetic microparticles and assay diluent are combined and incubated. The TgAb present in the sample binds to the Tg-coated microparticles. The mixture is washed. Acridinium-labelled human IgG antibody conjugate is added to create a reaction mixture and incubated. After a wash cycle, the preactivator and activator solutions are added. 16
Tg assay: The measurement of serum thyroglobulin was performed on Roche® Cobas e6000 electrochemiluminescence, ultrasensitive method assay. Undetectable Tg levels are considered <0.1 ng/mL.
Data collection and statistical analysis
All statistics analyses were performed using R commander v 2.6–2 y Medcalc v 11.4.2.0. The correlation between the four methods was examined pairwise using McNemar test analysis, where p < .05 was considered statistically significant. The agreement rates between four assays were determined by using the kappa statistics with 95% confidence intervals (CI) and with k values of <0, 0.01–0.2, 0.21–0.40, 0.41–0.60, 0.61–0.80 and 0.81–0.99, which represent less than chance agreement, slight, fair, moderate, substantial and excellent agreement, respectively. 17
Results
Summary of data obtained with each of the four analytical platforms.
Imprecision
Imprecision study in Ramon y Cajal Hospital laboratory. The average obtained was shown with the Coefficients of variation (standard deviation as percent of mean % CV) for Assay 4, Alinity Abbott kit reagent TgAb, in different concentrations.
Imprecision comparative results in manufacturer´s laboratory and Ramon y Cajal Hospital laboratory, using a control Positive provided by the manufacturer and a Panel 1 with concentration 4.67 kUI/L. Panel 1 consists of a pool of samples prepared by the manufacturing laboratory and whose concentration (4.67 kUI/L) is similar to that obtained in the Pool Diluted 1 prepared in our laboratory (4.30 kUI/L).

Concordance and interchangeability study
The concordance analysis was performed with the kappa index. The TgAb results obtained by each of the four methodologies were dichotomized into ‘positive’ and ‘negative’, according to the cut-off points established in clinical practice: • Assay 1 First generation reagent TgAb in Advia Centaur XP Siemens: 25 kUI/L. This is the Limit of Quantification calculated by Ramon y Cajal Hospital laboratory. Is defined as the lowest concentration that can be measured with a coefficient of variation less than or equal to 20%. • Assay 2 Second generation reagent TgAb in Advia Centaur XP Siemens: 1.4 kUI/L. This is the Limit of Quantification calculated by manufacturer laboratory. Is defined as the lowest concentration that can be measured with a coefficient of variation less than or equal to 20%. • Assay 3 Phadia 250 Termofisher kit reagent TgAb: 12 kUI/L. This is the Limit of Quantification calculated by manufacturer laboratory. Is defined as the lowest concentration that can be measured with a coefficient of variation less than or equal to 20%. • Assay 4 Alinity Abbott kit reagent TgAb. For this method, a concordance study was carried out using two cut-off points. Firstly, 4.11 kUI/L corresponds to the concentration within the 97.5% percentile of the population studied by manufacturer. Secondly, 0.33 kUI/Lis the limit of quantification calculated on more than 60 replicate samples and whose coefficient of variation does not exceed 20%. Kappa values have been calculated by using both cut-off points.
The McNemar test showed the difference between paired methodologies. The table provides the difference between proportions (expressed as a percentage) with 95% CI. When the p-value is less than the conventional 0.05, there is a significant difference between the two methodologies.
Discussion
The high prevalence of TgAb in patients with CDT (8) makes their measurement in the follow-up of these patients indisputable. Therefore, when changing the measurement methodology, it is necessary to conduct a study of both, imprecision and interchangeability, with respect to the technique in use. The Alinity reagent for the measurement of TgAb has an acceptable inaccuracy at all concentrations tested, including those close to the cut-off point indicated by the manufacturer (4.11 kUI/L). In a large number of cases, these cut-off points are optimized for the diagnosis of autoimmune thyroid disease, but not to detect interference in the measurement of thyroglobulin, and they are usually especially high. 5
Kappa index for the different methodologies and the different cut-off points in Alinity.

The McNemar test showed only one significant difference between Alinity versus Centaur second generation (difference 8.33%, IC95% 0.68–8.33, p = .0313)
Conclusion
Overall, despite the differences between the methodologies provided by various authors,10,18,19 the concordance of this new Alinity assay is acceptable as long as the cut-off point of 4.11 kUI/L is used. In view of the calculated data on the inaccuracy of this TgAb assay, further studies are considered to assess a possible lower functional sensitivity than that detailed by the manufacturer.
Our study had a number of limitations: A very limited number of samples; in order to reduce the degree of uncertainty of our results, it would be convenient to increase the sample size. On the other hand, our study showed concordance between the results of Alinity thyroglobulin antibody and the other methodologies, but could not ensure the presence of interference by thyroglobulin antibodies in the measurement of thyroglobulin, which would have required the measurement of this parameter by mass spectrometry.
Footnotes
Acknowledgements
The support of all the participants who have agreed to participate in this study is greatly appreciated.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The study was evaluated and approved by the Ethical committee of Hospital Ramón y Cajal (approval date: 14/06/2021 Acta 415).
Guarantor
AMGC.
Contributorship
All authors contributed to the study conception and design. The first draft of the manuscript was written by AMGC and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
