Abstract
Background
The need to detect new psychoactive substances in biological samples is of crucial interest. In this paper, the specificity of a benchtop immunoanalyzer commercialized by Randox was evaluated on real patient samples.
Method
The Evidence Investigator was assessed to screen for NPS on 80 serum and urine samples coming from patients admitted to the emergency department. Targeted NPS were included in various categories such as synthetic cannabinoids, opioids and benzodiazepines. Results were compared with a chromatographic technique coupled with mass spectrometry.
Results
No NPS was detected by the reference technique. Concerning immunoanalysis, some piperazines were positive, caused by the presence of medicine containing this chemical structure. Clonazepam and fentanyl derivatives were confirmed in some cases, but sometimes the positivity was explained by other opiates or benzodiazepines, which also explained two samples falsely positive for etizolam.
Conclusions
The Randox Evidence Investigator was rapid and easy to use. It can be used as a first intention but always followed by a more specific technique in order to detect false positive result.
Introduction
New psychoactive substances (NPS) are defined as ‘substances of abuse, either in a pure form or a preparation, that are not controlled by the 1961 Single Convention on Narcotic Drugs or the 1971 Convention on Psychotropic Substances, but which may pose a public health threat’. 1 To date, 950 NPS have been reported all over the world. 2 Depending on their pharmacological effect, NPS can be classified in various categories including, by order of frequency: stimulants, synthetic cannabinoid receptor agonists (SCRA), hallucinogens, opioids, sedatives/hypnotics and, finally, dissociatives. Some substances (5%) are not yet assigned in a category. 3 These various pharmacological effects can naturally lead to a wide range of symptoms when patients present themselves at emergency departments for intoxication. For example, tachycardia, nausea and agitation are typical signs of intoxication due to SCRA intoxications. 4 Similarities with phenethylamines, piperazines and cathinones intoxication are observed. Indeed, these stimulant compounds can cause a sympathomimetic toxidrome including tachycardia, agitation and seizure.5,6 Next to the same sympathomimetic toxidrome, adverse effects due to tryptamines consumption can also include altered perceptions, ‘bad trips’ and psychosis. 7 Such as older opioids, new synthetic opioids lead to the triad of symptoms consisting of decrease of consciousness, myosis and respiratory depression. 8 Finally, designer benzodiazepines share the toxicity of the pharmaceutical ones, with lethargy and drowsiness being common adverse effects. 9
Even if treatment of intoxication is most of time symptomatic, identification of the incriminated substance is of major interest, notably for epidemiological purpose and public health.
Analytical methods dedicated to NPS detection are reviewed elsewhere.10–13 Almost one thousand of compounds (to which metabolites must be added) have to be screened for, potentially presents at low concentration, which makes the task particularly challenging. Chromatographic techniques based on mass spectrometry, and especially high-resolution devices, are the gold standard. 14 However, these devices are prohibitively expensive, analysis is often time consuming, and requires highly specialized staff. Consequently, they cannot be available in all healthcare facilities and turnaround time is rarely compatible with clinical management. Development of automatic techniques that could circumvent these drawbacks could be interesting. In this context, we have evaluated the specificity of the Evidence Investigator commercialized by the Randox Laboratories (Crumlin, United Kingdom). This evaluation was performed on real samples.
Material and methods
Patient samples
Thirty-nine serum samples and 41 urine samples were analysed by both techniques. These samples were taken from patients admitted to the emergency unit of the University Hospital of Liege (Belgium). These patients were suspected to be under the influence of psychotropic substances (drugs of abuse or medicine), so blood and urine analysis were part of their medical care. Our study was approved by the institutional ethics committee considering that we analysed residual samples and did not request for new ones.
Immunoanalysis
Immunoanalysis was performed by Biochip Array Technology 15 on the Evidence Investigator semi-automated immunoanalyzer commercialized by Randox. This technology allows the simultaneous detection of related compounds using the same reagents, calibrators and controls. These are applied on a solid plate containing nine biochips – one biochip for one sample – and every single biochip is composed of several discrete tests regions. Each test region contains antibodies able to recognize a new psychoactive substance. Finally, competitive chemiluminescent assays are used to detect the compounds: the compound of interest that is eventually present in the patient sample competes with the labelled compound present in the reagent for the antibody.
Two kits were tested on all samples: NPS I (11 related compounds/test regions) and NPS II (14 tests regions).
New psychoactive substances I contains antibody directed against JWH-018, UR-144, AB-PINACA, AB-CHMINACA, mephedrone, alpha-PVP, benzylpiperazines, mescaline, salvinorin and phenylpiperazines.
New psychoactive substances II can detect furanylfentanyl, acetylfentanyl, carfentanil, sufentanil, ocfentanyl, AH-7921, MT-45, U-47700, W-19, etizolam, clonazepam, mitragynine, naloxone and buprenorphine metabolite.
Analytical cut-offs.
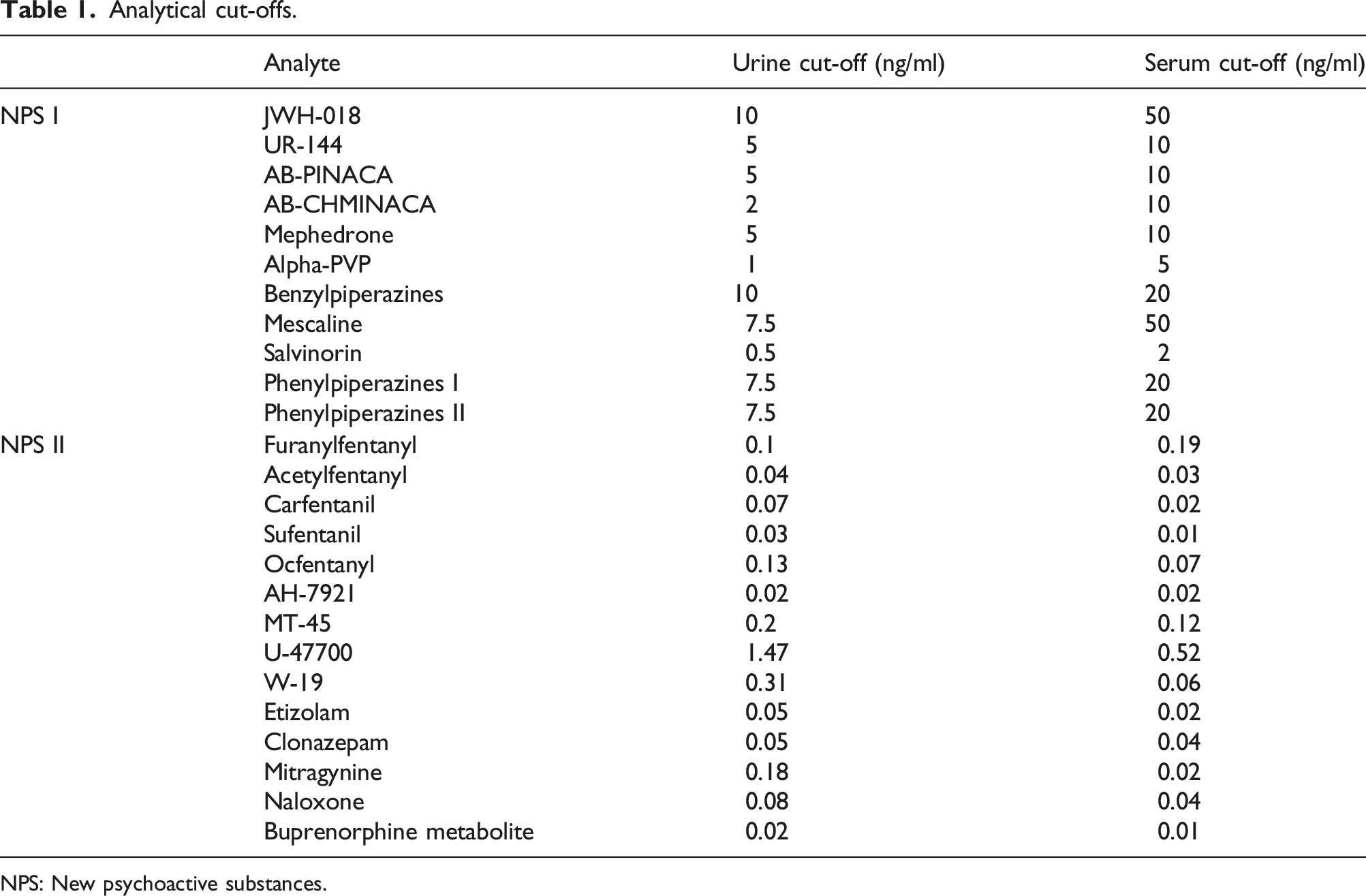
NPS: New psychoactive substances.
Serum and urine were analysed with the same reagents. As recommended by Randox, sera are diluted four times prior to analysis. The kits can be used for whole blood samples which have to be centrifuged before analysis as indicated in the instructions for use coming from Randox.
First, assay buffer is added to 25 µl of sample before a 30-min incubation shaking. Then conjugate is added before a second identical incubation cycle. Six washing cycles are then performed before addition of a mix of luminol and peroxide and final detection of chemiluminescence in the instrument. Calibration curves corresponding to nine standard levels were approved by the aim of two levels of quality controls.
Chromatographic analysis
Standards and chemicals
Prazepam, used as internal standard, was purchased from Certa (Braine-l’Alleud, Belgium). All solvents were LC–MS or HPLC grade and supplied by J.T. Baker (Phillipsburg, USA). For mobile phases preparation, sodium dihydrogen phosphate monohydrate was purchased from J.T. Baker, ammonium formate was supplied by Fisher Chemical (Merelbeke, Belgium) and finally formic acid was purchased from Biosolve (Dieuze, France).
Sample preparation
Serum and urine were submitted to the same liquid–liquid extraction. After addition of an internal standard (prazepam 10 mg/L) and basification (Na2CO3 1 M), 5 mL of a mix of diethyl ether/dichloromethane/n-hexane/n-amylic alcohol (50/30/20/0.5:V/V) were added to 1 mL of the sample. After mixing for 15 min and centrifugation, the organic layer was evaporated to dryness and reconstituted in the mobile phase. Extracts were injected on UHPLC-TOF-MS.
The apparatus was an Eksigent LC 100 XL combined with a Triple TOF 4600 from Sciex (Framingham, USA) and the screening method was developed by Sciex [inspired from Ref. 16]. Injections of 10 µL of the samples were done on a Kinetex C18 column, 2.6 mm, 100 Å, 50 × 3.00 mm (Phenomenex, Torrance, USA). A gradient was applied at 30°C, with mobile phase A consisting of 10 mM ammonium formate while mobile phase B was a mix of acetonitrile and methanol with 0.1% formic acid (50/50). A constant flow of 0.4 mL/min was applied using the following gradient: the initial condition of 98% of mobile phase A was held during 1 min. Then, the gradient linearly decreased to 0% of A in 10 min, held for 3 min. Finally, the gradient returned to initial conditions and was maintained for 2.5 min prior to the next injection. The TOF was equipped with a DuoSpray Ion source working in positive electrospray ionization mode. Source conditions were as follows: ion source gas 1 40 psi, ion source gas 2 60 psi, curtain gas 30 psi, source temperature 500°C and ion spray voltage floating 5500 V. The mass acquisition method was a TOF survey scan from 50 to 1100 Da (cycle time 1 s, accumulation time 0.150 s), combined with a second experiment of product ion scan with an information dependent acquisition method on a maximum of 20 candidates per cycle. Data were acquired with Analyst 1.7.1 software and finally processed with the PeakView software 2.2, including a comparison to a library purchased by Sciex and regularly updated inhouse.
Approximative limit of detection of the method.
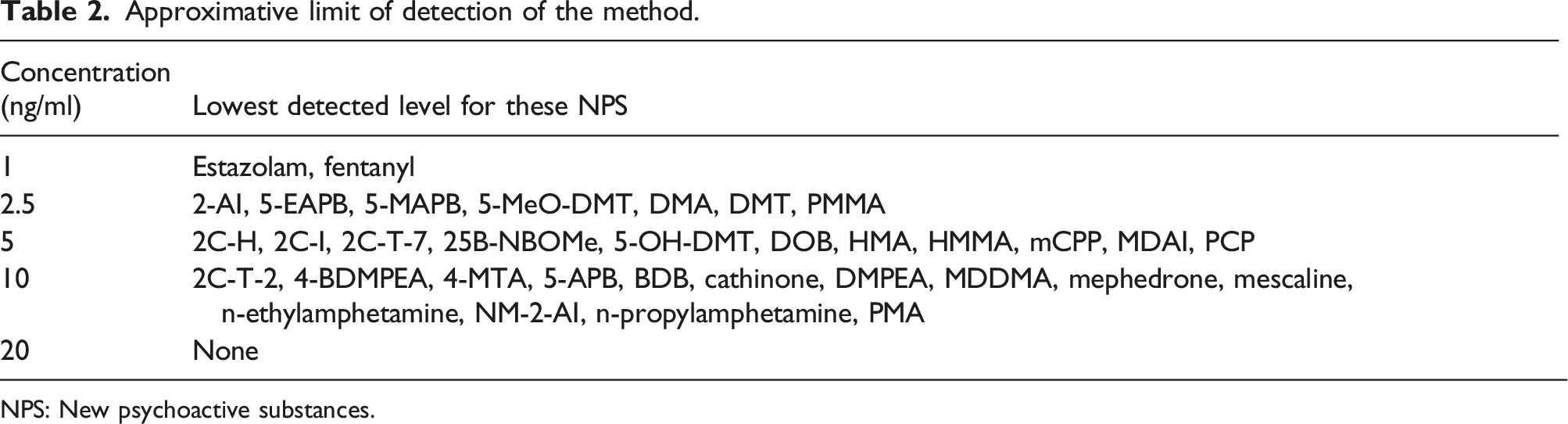
NPS: New psychoactive substances.
Results and discussion
Randox Evidence Investigator was really easy to use compared to UHPLC-TOF-MS. Sample preparation was clearly faster for immunoanalysis compared to chromatography and requires a significantly lower sample volume. Another benefit of this technology is that results were quickly obtained, without the need of a difficult interpretation, nor the use of a as complicated as powerful software. However, discrepancies between both techniques were observed and they are discussed in details below. Considering its well-known specificity, UHPLC-TOF-MS was considered as the reference method.
Serum samples
Thirteen out of the 39 serum samples led to a positive result for
No NPS was found by UHPLC-TOF-MS in serum samples, except for ketamine in one of them, and 1-(m-chlorophenyl)piperazine (mCPP) in two of them. This compound is used as an NPS but is also the main metabolite of trazodone, an antidepressant frequently prescribed in Belgium. Indeed, trazodone was chromatographically confirmed with mCPP in both samples.
Twenty-five samples did not contain any psychoactive substances at all.
The others were chosen in order to check if drugs of abuse or common (mostly psychotropic) drugs could lead to a false positive result when tested with the immunoassay. These samples were positive for at least one of the following substances: alprazolam, 7-amino-flunitrazepam, amisulpride, amphetamine, benzoylecgonine (cocaine metabolite), bisoprolol, cetirizine, citalopram, clotiapine, domperidone, hydroxyzine, levetiracetam, lorazepam, lormetazepam, MDMA, methadone, methylphenidate, mirtazapine, morphine, paroxetine, pipamperone, prothipendyl, quetiapine, risperidone and metabolite, tramadol, sulpiride, venlafaxine and metabolite, zuclopenthixol and zolpidem.
Urine samples
Urine results.

Fifteen urine samples led to a positive result for
Fifteen urine samples tested positive for
Ten urine samples tested positive for at least one
- one result was four times above the cut-off (for carfentanyl) and lots of compounds were detected (opiates indicating an heroin consumption, methadone, cocaine, clonazepam, pipamperone, mirtazapine, trazodone); - another was 8 times above the cut-off for acetylfentanyl and chromatographic analysis revealed other opiates (tramadol, pholcodine, morphine); - one sample was slightly above the cut-off for carfentanyl (0.34 for a 0.25 cut-off) and tramadol was detected in the sample; - a sample containing tramadol gave a result of 1.87 for the acetylfentanyl test (cut-off = 1); - a sample containing tilidine gave a result of 0.26 for the carfentanyl test (cut-off = 0.25); - finally, a sample that did not contained any opiates gave a result slightly higher than the cut-off for carfentanyl (0.27 > 0.25).
For fentanyl derivatives, we assume that false positive results can be explained by a cut-off that should probably be raised, or by cross-reactivity with other opiates. Clinical impact of this false positive result is low, as the same antidote can be administrated for all opiates intoxication. Fentanyl derivatives are highly potent compounds exerting an effect at low dose and low biological concentration. Our chromatographic detection limit for fentanyl is 1 ng/mL (but no lowest concentration was tested), whereas it is included between 0.3 ng/mL and 1.3 ng/mL depending of the derivative with the immunoanalyzer. True positive samples (not detected by chromatography) cannot be excluded, but patient file did not support the hypothesis of NPS consumption. Moreover, in their study evaluating Biochip Array Technology, Larabi et coll. 18 found three patient samples tested positive for fentanyl derivatives, and the concentration was higher than 1 ng/mL, sometimes largely.
Immunoassay detected
Finally,
Unfortunately, our population did not include any real NPS consumer according to UHPLC analysis, which represents the main gap of our study. Another study involving NPS consumers should be done in order to complete the present one and fully evaluate the instrument and check if the immunoanalyzer is able to detect NPS when they are present and therefore also test the sensitivity of the method.
If the lack of specificity can firstly be seen as a disadvantage, it is sometimes a benefit to detect more compounds than predicted. For example, cross-reactivity can be useful to detect a whole family, or metabolites of a totally metabolized compound. Moreover, a positive result is often the proof of consumption, even if it does not concern the compound that gives a positive result (for example: clonazepam test positive due to another benzodiazepine compound). In any cases, chromatographic analysis must be undertaken in order to confirm the result and specify the compound that gives rise to the positivity. Immunoanalysis can be done as a first intention (by night or during the weekend for example) before the chromatographic confirmation which is often slower to implement.
Finally, limitations of the tests are addressed and continuously improved by the company commercializing the kits.
Conclusions
As it is clearly easier to use than chromatographic techniques, immunoanalysis for drugs of abuse can probably be used as a first intention. A positive result can alert the clinician and help with the diagnosis and patient management. However, it should not exist on its own in a healthcare facility, considering the risks of hasty conclusion.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Our study was approved by the institutional ethics committee considering that we analysed residual samples and did not request for new ones.
Guarantor
MD.
Contributorship
M. D. and C. C. conceived the study and were involved in results interpretation. P. P. and A. W. carried out samples analysis and data interpretation. N. G. and R. B. helped in sample analysis and interpretation. M. D. discussed with the Ethics Committee, researched the literature and wrote the manuscript. All authors reviewed and approved the final version of the manuscript.
