Abstract
Background
The dilution or adulteration of urine is a serious problem in drugs of abuse testing. Tests to identify adulteration are currently available. This study investigated the ability of the CEDIA® sample check to detect adulteration.
Methods
Eight different drugs of abuse were added to a urine sample obtained from a healthy, drug-free subject: 2-ethylidene-1,5-dimethyl-3,3-diphenylpyrrolidine (EDDP), 3,4-methylenedioxyamphetamine, benzoylecgonine, D-amphetamine sulphate, ethyl-D-glucuronide, morphine sulphate, oxazepam, (-)-11-nor-9-carboxy-Δ9-tetrahydrocannabinol. Urine samples were diluted to yield three samples of drugs of abuse concentrations close to general cut-offs as used in methadone treatment centres, by health authorities for psychological tests and in traffic medicine. Aspirin, citric acid, CrO3, H2O2, soap, sodium metaborate, vitamin C were added in three, HCl and NaOH in one, and NaN3 in two concentrations. All samples were measured with commercially available immunological assays shortly after sample preparation and 24 h later. All samples were further analysed with the CEDIA® sample check reaction which may identify adulteration.
Results
Oxidizing reagents (H2O2 or CrO3) are most effective in interfering in the measurement of benzoylecgonine, EDDP, ethyl-D-glucuronide and morphine sulphate. The measurement of (-)-11-nor-9-carboxy-Δ9-tetrahydrocannabinol is affected by many adulterants. Adulteration with HCl and NaOH was identified with the sample check reaction. NaN3 generated false negative results for a number of drugs of abuse.
Conclusions
Urine samples with drugs of abuse concentrations above cut-offs can be successfully tampered with adulterants in a way which cannot be detected with the CEDIA® sample check assay.
Introduction
Urine tests for drugs of abuse (DOA) are frequently performed in many settings and locations such as methadone treatment centres, prisons and laboratories involved in workplace testing. 1 Clinical and institutional staff cannot rely on patient self-reporting. Patients who have consumed illicit drugs may fear consequences such as loss of occupation in workplace testing, loss of privileges in prisons and/or many other adverse complications. Drug users may therefore tamper with urine samples submitted for DOA testing.2–6
DOA testing is commonly performed on clinical chemistry autoanalysers using immunoassay. Reagents which are designed to alter the urine matrix or destroy drug molecules in a way that provokes false-negative results in DOA analyses are widely available for sale on the internet.7–9 In the simplest case, an immunoassay incorporating an enzyme/substrate reaction may be inhibited by the addition of interfering substances to the urine sample. Therefore, the diagnostic industry is forced to develop procedures for detecting these adulteration methods. One example for such a monitoring assay is the CEDIA® sample check assay. 10 This assay is based on two inactive fragments of the β-galactosidase which have been genetically engineered. They can spontaneously re-associate to form an active enzyme with capacity to cleave a substrate, generating a colour change that can be determined spectrophotometrically.10,11 An adulterant interfering with the generation of an active enzyme complex or destroying the substrate or the product will provoke a reduction in assay signal.
However, the question remains to what extent these monitoring assays are able to detect sample adulteration. 12 There may be concentrations of adulterants that would be sufficient to produce a false-negative result in immunological tests for DOA, but cannot be detected with monitoring assays such as CEDIA® sample check.
It is common practice in many laboratories for samples which have tested positive for DOA by immunoassay to be confirmed by gas chromatography-mass spectrometry (GC-MS) or liquid chromatography-mass spectrometry (LC-MS). However, samples which have given negative results in immunoassays may not be investigated further if the CEDIA® sample check does not give any indication of adulteration. It is, therefore, essential to guarantee that immunoassay-based screening tests are not susceptible to commonly used adulterating agents.
In this study, we present data on the selective disturbance of immunological assays in DOA testing by adulterants.
Materials and methods
DOA analysis and reporting
Assay methods and cut-offs.
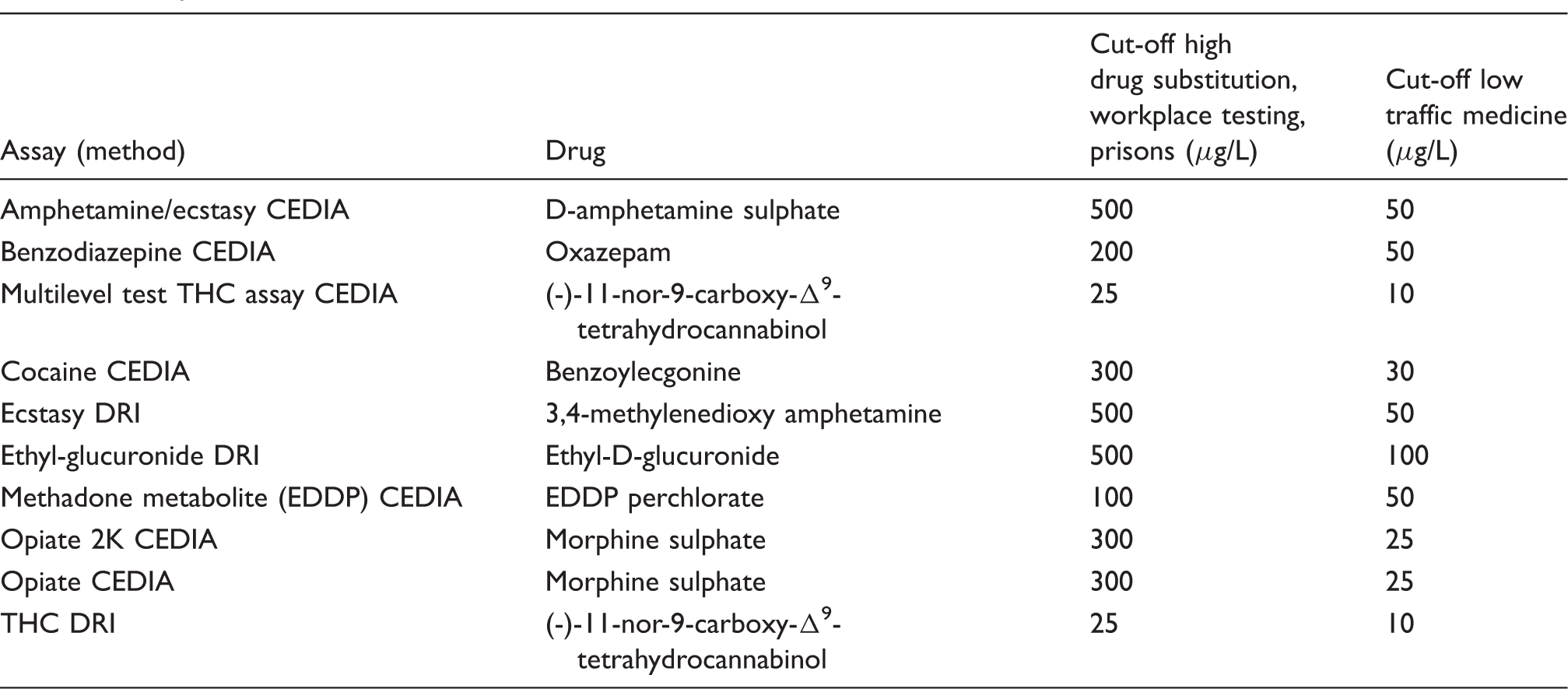
2K: cut-off concentration of 2000 ng/mL; CEDIA: cloned enzyme donor immunoassay; DRI: Diagnostic Reagents Inc.; EDDP: 2-ethylidine-1,5-dimethyl-3,3-diphenylpyrrolidine; THC: (-)-11-nor-9-carboxy-Δ9-tetrahydrocannabinol.
Adulteration agents and reference materials
In our study, we used adulterants that were previously reported to cause false-negative results in DOA testing.13–17 Sources of adulterants were: aspirin (Bayer, Leverkusen, Germany), sodium metaborate (Fluka Chemie, Buchs, Switzerland), soap (Pril) (Henkel, Düsseldorf, Germany), H2O2 (Merck Schuchardt OHG, Hohenbrunn, Germany), HCl (Merck), NaOH (Merck), CrO3 (Merck) and vitamin C (Das Gesunde Plus) obtained from the DM-Drogeriemarkt (Karlsruhe, Germany). For citric acid, the juice of one lemon was filtered. Reference materials for EDDP, 3,4-methylenedioxyamphetamine, benzoylecgonine, D-amphetamine sulphate and Ethyl-D-glucuronide were provided by LGC Standards (Wesel, Germany). Oxazepam, morphine sulphate and THC were obtained from Sigma-Aldrich (Schnelldorf, Germany). All drugs were obtained in solid form as free amines or acids and used to prepare stock solutions. The concentrations of these stock solutions were checked with our immunological assays using commercial calibrators that were obtained in solvent, ready to use form. All CEDIA (cloned enzyme donor immunoassay) and DRI (Diagnostic Reagents Inc.) product line assays for the analysis of DOA and CEDIA® sample check assay were obtained from Thermo Fisher Scientific (Darmstadt, Germany).
Urine sample spiking
Concentrations of DOA in urine samples.

EDDP: 2-ethylidine-1,5-dimethyl-3,3-diphenylpyrrolidine.
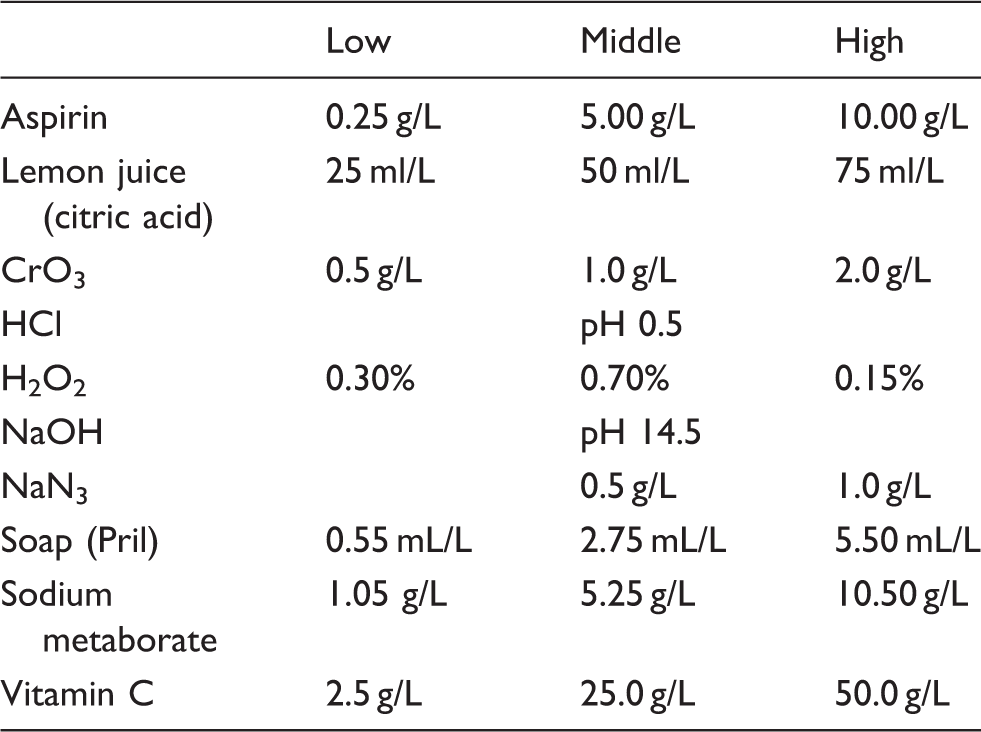
Concentrations of adulteration reagents in urine samples.
Results
A total of 75 urine samples were spiked with eight DOA in concentrations as outlined in Table 2. Reagents that are frequently used for adulteration were added in various concentrations (see Table 3). Concentrations of DOA were chosen to capture both cut-offs. The two following different cut-offs below were used to discriminate between ‘positive’ and ‘negative’ for DOA in our study (see Table 1):
Cut-off 1 is used in many methadone treatment centres and prisons. Cut-off 2 is used by the German traffic medicine authorities.
18
The different samples were analysed for DOA in immunological assays on a clinical chemistry analyser and the CEDIA® sample check was carried out on every sample. The sample check reaction indicates ‘evidence for adulteration’ if the signal of the measurement is lower than 80% of the original determination. For our tests, the adulterant concentrations were chosen based on concentrations that are reported elsewhere.13–17
In our study, the following adulterants at given concentrations were effective in interfering with the sample check reaction: HCl: <pH 1, NaOH: >pH 14, CrO3: >1.0 g/L, H2O2: >0.7%, soap: >0.5 mL/L and vitamin C: >50.0 g/L. Aspirin, boric acid, citric acid and NaN3 had no effect on sample check reaction at the concentration tested (Table 3).
Urine samples were further investigated in immunological assays for DOA. Analytical data of the DOA measurements were presented on a two-dimensional figure (the exemplar of benzodiazepine is shown [Figure 1]). In this representation, the concentrations of DOA are plotted on the ordinate and the two cut-offs in horizontal bars. The concentrations of adulterants are plotted on the abscissa in which a vertical bar indicates the limit where the sample check reaction is lowered by more than 20%. The value 20% was taken from the manufacturer’s instructions. The horizontal and the vertical bars separate four different quadrants (Figure 1 and Table 4). Data points in quadrant 3 (Q3) represent a successful tampering with adulteration reagents. These data points are ‘negative’ for DOA, although the original DOA concentration was above cut-off. All the other quadrants represent either samples positive for DOA (Q1 and Q2) or samples negative for DOA, but positive for sample check which would indicate adulteration of this sample (Q4). With the help of this two-dimensional array, we have analysed the data of the determinations of DOA in the presence of various adulteration reagents. Data are summarized in Table 5.
Interpretation of analytical data of DOA in a two-dimensional array. Data from drugs of abuse (DOA) determinations were plotted with the drug concentration on the ordinate and concentration of adulteration reagents on the abscissa. For explanation, see text and Table 4. Data analysis with a two-dimensional array. DOA: drugs of abuse. Treatments resulting in false-negative results.
a
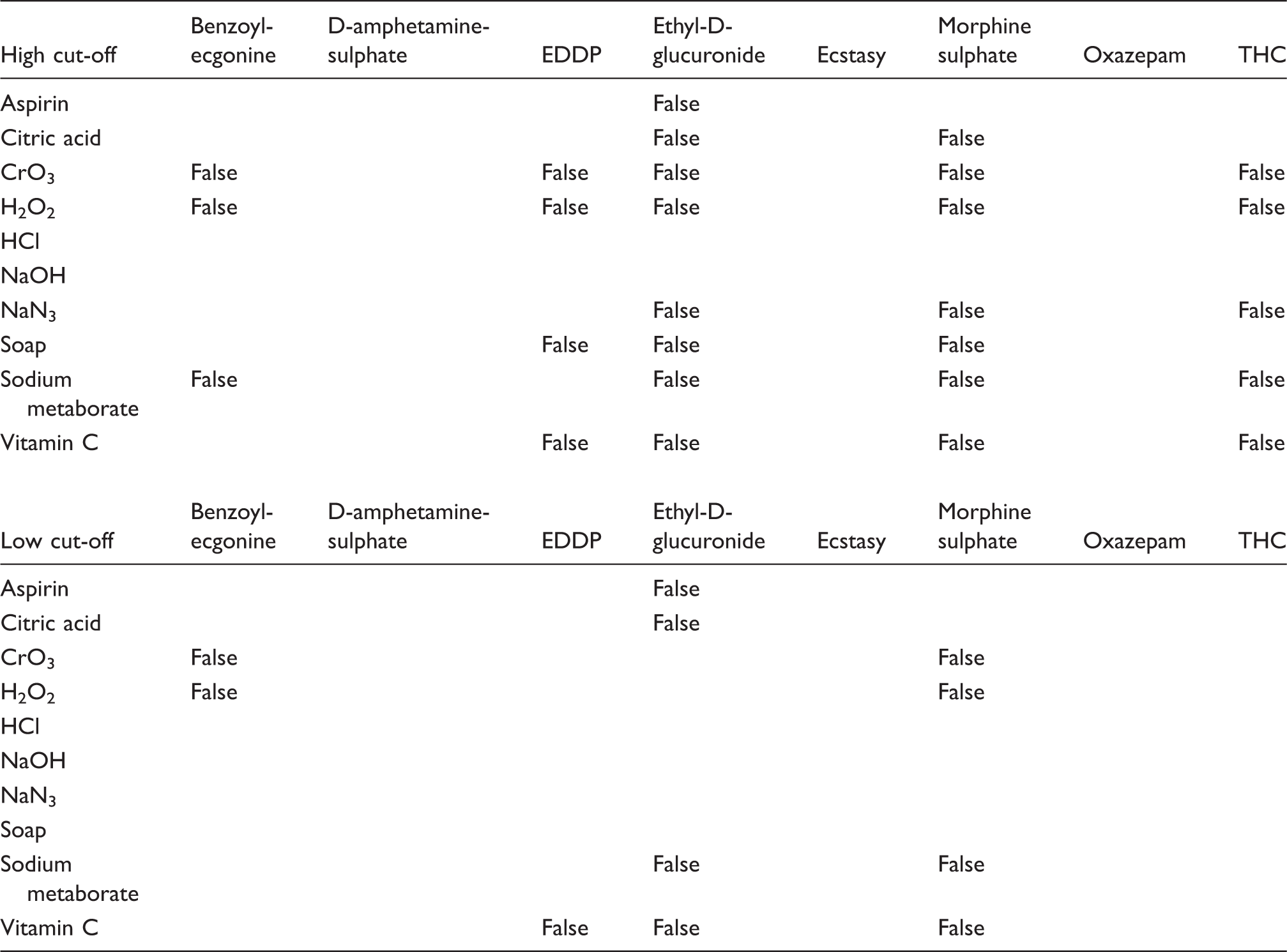
EDDP: 2-ethylidine-1,5-dimethyl-3,3-diphenylpyrrolidine; THC: (-)-11-nor-9-carboxy-Δ9-tetrahydrocannabinol. False-negative results (=false) represent samples that contain DOA in concentrations above cut-offs when adulteration reagents are added at concentrations which are not detected in the sample check reaction (Q3: see Figure 1). All other data were identified as being from samples only in Q1, Q2 or Q4 (see Figure 1).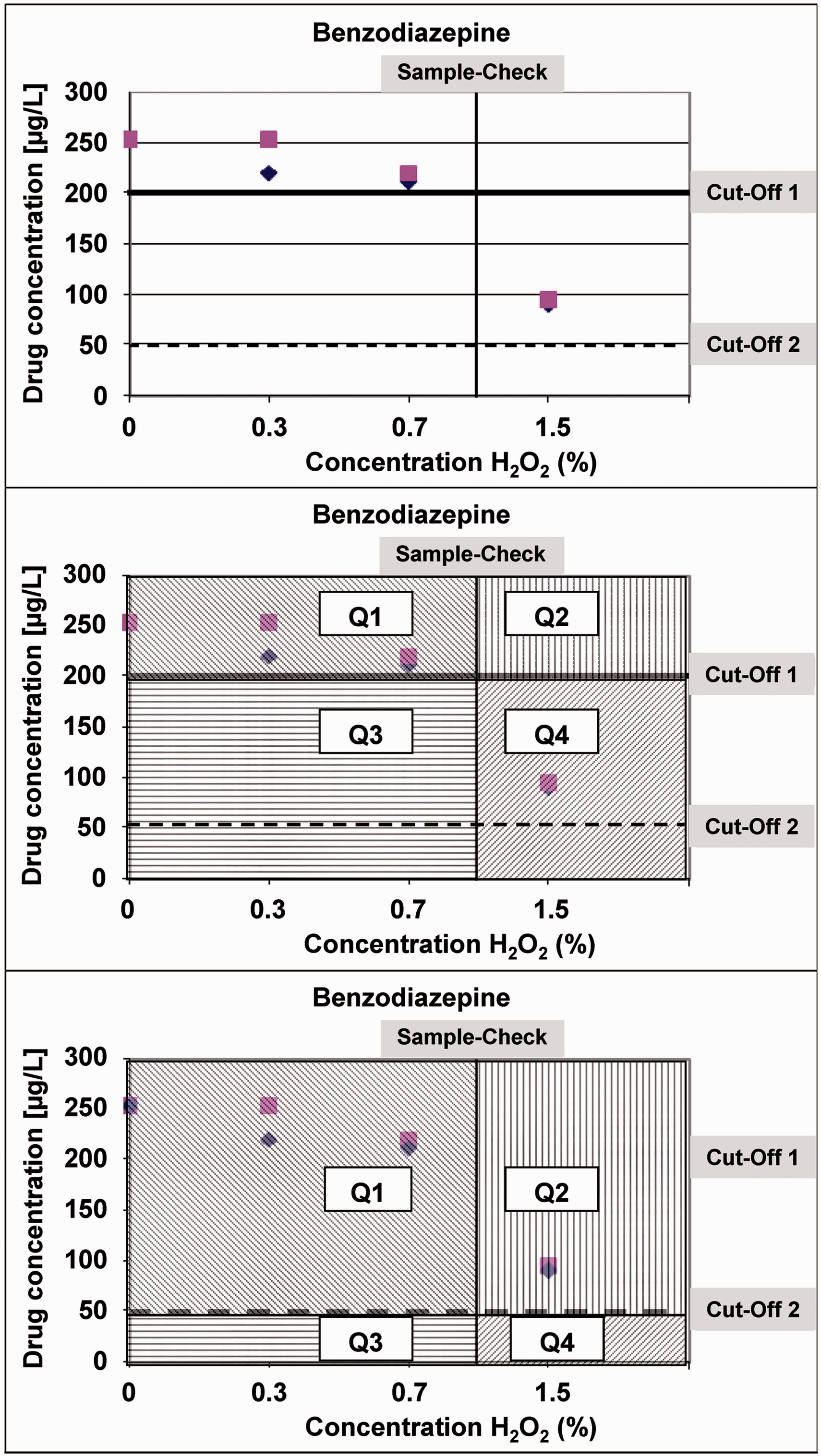

With regard to treatments that produce false-negative results, we found:
oxidizing reagents such as H2O2 or CrO3 are most effective in producing false-negative results in assays for benzoylecgonine, EDDP, ethyl-D-glucuronide and morphine sulphate in the cut-off 1 area and benzoylecgonine and morphine sulphate in the cut-off 2 area. The measurement of THC is tampered with many adulteration agents in cut-off 1; in contrast, there is no influence in quadrant 3 for cut-off 2. HCl and NaOH adulteration can be determined effectively with the sample check reaction. There is no signal in Q3 for D-amphetamine sulphate and Ecstasy for any of the adulterants tested. NaN3 can create false-negative results with cut-off 1 but not with cut-off 2.
Discussion
Commercial immunological screening assays are used for testing of DOA in forensic toxicology, workplace drug testing, antidoping and clinical toxicology. They are widely applied in government institutions, including the army, department of Veterans Affairs, prisons and laboratories who are entrusted with DOA testing for clarification of road traffic offenses or drug workplace violations. These assays are comparatively cheap and can be easily installed and carried out on clinical chemical analysers.
Our data suggest that some of the adulterants studied can interfere in the measurement of immunological DOA assays resulting in false-negative results. Importantly, some of these interfering substances were not detected with the CEDIA® sample check in our assays in which we applied the common cut-off values.
For successful manipulation of a DOA test, the concentration of adulterants must be high enough to alter the signal to create a false-negative report but still be low enough so that the sample check reaction will stay above 80% of the original value. Where the urine DOA concentration is sufficiently high (giving a positive DOA result [Q1 and Q2]), the required adulterant concentration to generate a negative result would be sufficiently high as to be detected with the CEDIA® sample check (Q4). It must be recognized that DOA users commonly try to dilute her or his urine by drinking excessive amounts of water and so reducing the urine DOA concentration and therefore making the detection of adulteration more difficult, i.e. a smaller concentration of adulterant which cannot not be detected by CEDIA® sample check may be sufficient to give a false-negative DOA result. However, significant dilution of urine will be flagged by a urine creatinine concentration that falls below 1.768 mmol/L (= 0.2 g/L) Substance Abuse and Mental Health Services Administration (SAMHSA) cut-off limit for diluted urine. 19 It is unlikely, however, that dilution and adulteration will simultaneously meet the concentration ranges necessary for successful cheating.
Some DOA-measurements like D-amphetamine sulphate and Ecstasy were not affected by any of the adulterants studies, while others such as THC will not be impaired at lower cut-offs. Nonetheless, because of the many possibilities to falsify a test result, it is impossible to provide general instructions in DOA testing.
Some laboratories use additional assays to monitor adulterants beside the CEDIA sample check®. Examples are the ‘Trinder reaction’ that is very sensitive for H2O2, the iodine/starch reaction that can detect oxidizing reagents, or different dipstick test systems or methods of wet chemistry that can detect NaN3. All these measurements require simple and cheap reagents, and most of them can be installed on a clinical chemical autoanalyser. In addition, some laboratories make use of the fact that cut-off concentrations are much higher than the analytical limits of the method such as the LLOQ.20,21 This has been introduced into DOA testing to determine DOA in diluted urine. In addition, it is possible to report data from DOA measurements with semi-quantitative data (DOA concentration/g creatinine) thus indicating that DOA are still present in a urine sample, although the report is ‘negative’. With the help of these calculations, the danger of reporting false-negative results can be minimized.
Based on all these challenges, some laboratories completely avoid the use of immunoassay tests and use only methods such as GC-MS or LC-MS which have a much lower level of quantification and can even identify drug molecules that have been chemically altered. While they are more commonly used as a reference method to confirm a positive result from an immunoassay screening, they may also be used for screening itself. They have the added advantage of being able to identify ‘designer drugs’ or plant-based drugs for which immunoassays are not available. Nevertheless, immunoassay methods remain in common use because of lower cost and greater ease of automation.
In summary, our study indicates that urine samples for DOA testing can be adultered in a way which may not be detected with the CEDIA® sample check assay. The effect of some adulterants may be limited to specific drugs. If there is suspicion regarding possible adulteration of samples in the presence of negative DOA test results, further investigation should be undertaken.
Footnotes
Acknowledgement
The authors wish to thank Joseph Tilley for his helpful suggestions and for proofreading the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Guarantor
RK.
Contributorship
BM and BH: performance of measurements, data compiling and evaluation of data; RK: design of the study, interpretation of data and writing of manuscript; RW: interpretation of data, writing of article.
