Abstract
Background
Currently, there are no national guidelines for antenatal drug testing. At Colchester Hospital, we use a strategy of screen-only using point-of-care testing to detect illicit drug use in pregnancy. To determine the suitability of this approach, we have compared the results of urine analysis by point-of-care testing with another NHS specialist clinical toxicology service that uses confirmation mass spectrometry.
Methods
A total of 482 anonymized random urine specimens from antenatal clinics were tested for six drug classes: amphetamine, benzodiazepines, buprenorphine, cocaine, methadone and opiates using the Alere™ Drug Screen Urine Test Cup. The manufacturer’s claims for positive cut-off and result stability were verified using spiked blank urine. Confirmatory testing was performed using ultra-performance liquid chromatography tandem mass spectrometry (UPLC-MS/MS) for detection of 26 individual drugs.
Results
Of 473 urine samples with adequate volume for point-of-care screening, 4.4% tested positive: 19 opiate and 2 cocaine. Concordance between point-of-care screening and UPLC-MS/MS confirmation was 97.9% for all drugs and 78.9% for opiates. Using spiked urine, only positive results for opiates were stable when read up to the manufacturer’s recommended time of 60 min.
Conclusions
The key advantages of using point-of-care devices to detect drug use in pregnancy are that is convenient and cheap. However, the clinical utility of point-of-care testing is limited by its poor sensitivity. Best practice is to confirm results using a more specific and sensitive method. As a result of this study, we are now reviewing our own procedures to consider introducing routine confirmation by mass spectrometry.
Introduction
The use of illicit drugs during pregnancy is associated with a plethora of adverse maternal and neonatal effects, including preterm birth, placental abruption, stillbirth, growth restriction, seizures, neonatal abstinence syndrome (NAS) and increased infant mortality. 1 It is crucial to identify pregnancies at risk of illicit drug exposure to enable appropriate and timely treatment for the neonate.
At Colchester Hospital, antenatal protocols involve the offer of point
POC devices employ competitive immunoassay-based methods and are useful for illicit drug screening due to their ease of use and rapid generation of results. Major limitations include their significantly lower sensitivity compared with mass spectrometric methods, their susceptibility to interference due to cross-reactivity of other drugs and metabolites, particularly prescription drugs, 2 and many immunoassays do not distinguish between individual drugs within a class.3,4 Gas- or ultra-performance liquid-chromatography coupled to mass spectrometry (GCMS and UPLC-MS/MS) are the gold standard methods for drug testing due to their vastly improved sensitivity and specificity compared with immunoassay techniques. 5
Study design
In the context of drug screening in pregnancy, methods with high sensitivity are required to reduce the risk of false
Methods
Chemicals and solutions
All solvents and water were purchased from Fisher Scientific® (Leicestershire, UK) and were LC-MS/MS grade. Certified reference material (CRM) for amphetamine, oxazepam, buprenorphine, benzoylecgonine (BZE), methadone and morphine were purchased from Cerillant® Sigma-Aldrich (Texas, USA) and LGC® (Luckenwalde, Germany).
Urine specimens
A total of 482 anonymized urine specimens were collected into plain containers from community and hospital antenatal clinics and from maternity triage over a period of five days and stored at 4°C. After POC drug screening, the urine specimens were stored at –20°C up to a maximum of three weeks.
POC testing
POC urine drug screening was performed within 48 h of urine specimen collection for 400 specimens and within 72 h for 82 specimens using the Alere™ Drug Screen Urine Test Cup (Abingdon, UK, product code QD1-162), which is the device in routine use within the antenatal department at Colchester Hospital. This qualitative lateral flow chromatographic competitive binding immunoassay tests for the presence of six classes of drug: amphetamine, benzodiazepines, buprenorphine, cocaine, methadone and opiates. 6 The stated positive cut-offs (ng/mL) for each drug class are presented in Table 1. Results should be read after 5 min left at room temperature and are claimed to be stable for up to 60 min. 6 Positive drug results above the cut-off concentration are indicated by the absence of a coloured line in the specific test line region of the strip. An internal control is included to confirm that enough sample volume has been added.
Concentrations of standards used for validating the Alere™ Drug Screen Urine Test Cup cut-offs.
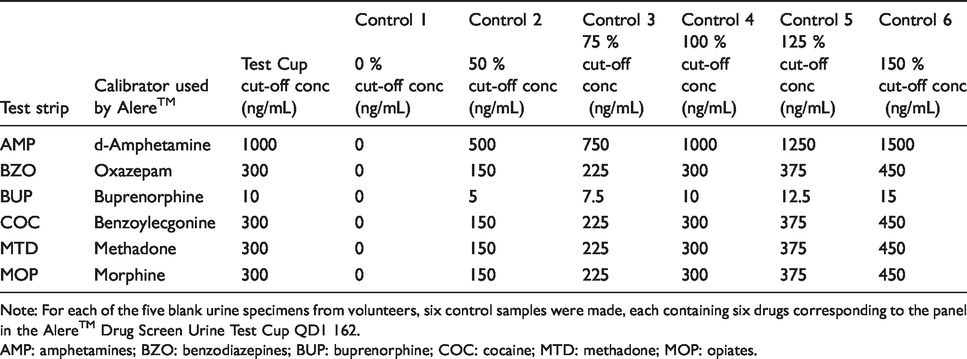
Note: For each of the five blank urine specimens from volunteers, six control samples were made, each containing six drugs corresponding to the panel in the Alere™ Drug Screen Urine Test Cup QD1 162.
AMP: amphetamines; BZO: benzodiazepines; BUP: buprenorphine; COC: cocaine; MTD: methadone; MOP: opiates.
POC verification studies
To confirm the quoted analytical specificity of the POC device for the six drug classes in the kit insert (Table 1), drug-free urine (blank) was collected from five different laboratory volunteers and spiked with CRM at concentrations of 0, 50, 75, 100, 125 and 150% of the positive cut-off values. 6 To confirm stability claims, results were read at the following time intervals post activation of the kit: 2, 3, 4, 5, 10 and 60 min.
Confirmatory (UPLC-MS/MS) testing
For confirmatory drug testing, a previously published method with liquid–liquid extraction and UPLC-MS/MS detection and with multiple reaction monitoring was used. 7 The method was validated according to internationally recognized guidelines.8,9 Briefly, illicit drugs were extracted from 400 µL of urine using a single reagent containing acetonitrile, ethyl acetate and 0.5 M ammonium carbonate buffer (pH 8). After mixing and centrifugation, the upper drug-containing layer was dried under a stream of nitrogen and reconstituted in 100 µL of loading buffer. The UPLC-MS/MS system comprised a Waters ACQUITY® UPLC system and Xevo TQD detector with positive electrospray ionization (Waters, Co., Milford, MA, USA). Chromatographic separation was performed using a Waters ACQUITY® UPLC HSS C18 1.8 µm 2.1 × 150 mm column and HSS C18 1.8 µm 2.1 × 5 mm VanGaurd pre column maintained at 30°C using 5 mM ammonium formate with 0.025% (v/v) formic acid (solvent A), and 0.025% (v/v) formic acid in 100% methanol (solvent B) at a flow rate of 290 µL/min with a gradient of 15% B increasing to 95% B for the first 3.50 min, back to 15% B over next 2 min, and held at 15% B for 0.5 min (total run time 5.5 min). Sample injection volumes were 15 µL. A list of the 26 drugs and metabolites and the positive cut-offs used are summarized in Table 2.
Drugs, street highs and metabolites measured by UPLC-MS/MS and the cut-offs in use at the time of analysis.
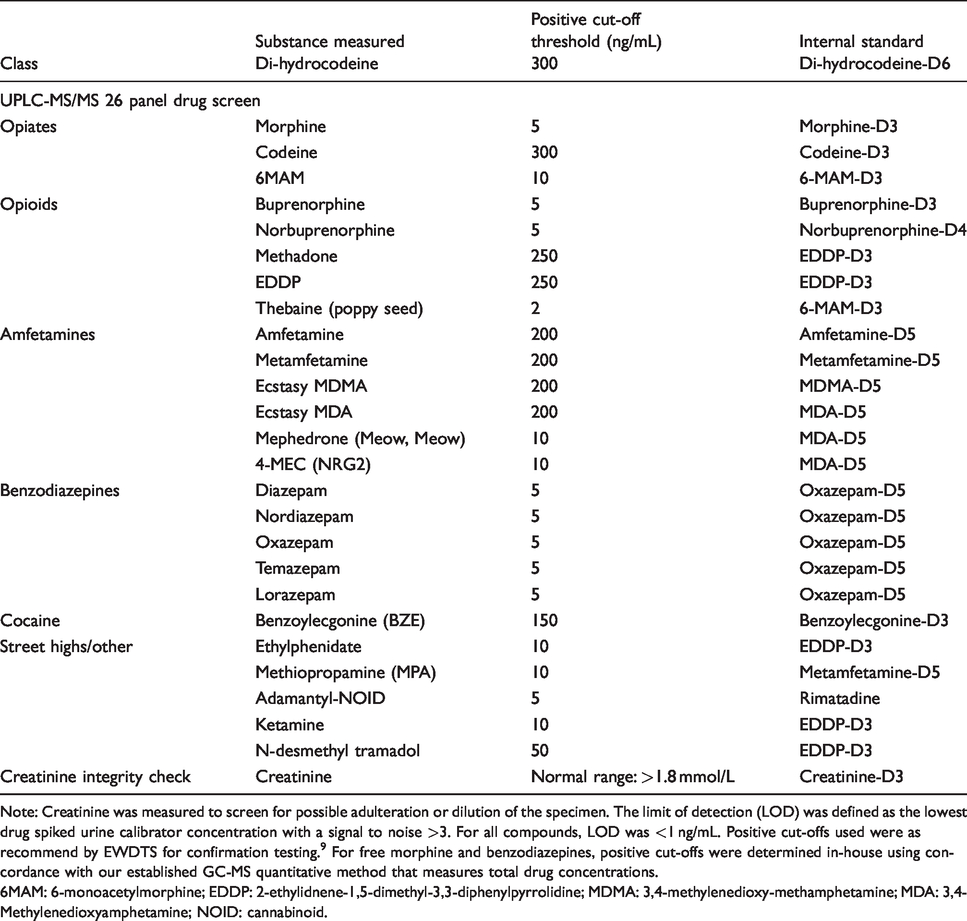
Note: Creatinine was measured to screen for possible adulteration or dilution of the specimen. The limit of detection (LOD) was defined as the lowest drug spiked urine calibrator concentration with a signal to noise >3. For all compounds, LOD was <1 ng/mL. Positive cut-offs used were as recommend by EWDTS for confirmation testing. 9 For free morphine and benzodiazepines, positive cut-offs were determined in-house using concordance with our established GC-MS quantitative method that measures total drug concentrations.
6MAM: 6-monoacetylmorphine; EDDP: 2-ethylidnene-1,5-dimethyl-3,3-diphenylpyrrolidine; MDMA: 3,4-methylenedioxy-methamphetamine; MDA: 3,4-Methylenedioxyamphetamine; NOID: cannabinoid.
Ethical permission
Ethical permission was sought and obtained from the Eastern Research Ethics Committee under reference number 15/EE/0407. Collection of urine specimens without consent was approved due to the deliberate break in the link between patient identifiable information and the specimens, through transfer of the specimen to a new, unlabelled container.
Results
Urine screening results
During the study period of one working week, 482 anonymized urine specimens were collected for POC drugs of abuse screening. Specimens were received from the following locations: hospital antenatal clinics (51.2%), community antenatal appointments (35.4%) and maternity triage (12.9%). The location was not stated on 0.4% of specimens. The mean gestational age was 29.9 weeks (range 6 to 42 weeks).
Of the 482 urine specimens, nine had insufficient volume for POC testing. All of the remaining 473 samples were analysed by both POC and UPLC-MS/MS. In total, 21 (4.4%) tested positive by POC for at least one drug class; 19 (4.0%) specimens were opiate positive and 2 (0.4%) cocaine positive. Overall concordance with UPLC-MS/MS urine drug analysis was 97.9%. Concordance for cocaine was 100% with UPLC-MS/MS urine concentrations for BZE of 1957 ng/mL and 1512 ng/mL (positive cut-off threshold 100 ng/mL). Concordance between the 19 opiate-positive POC results and UPLC-MS/MS opiates (morphine, codeine, dihydrocodeine [DHC], heroin-specific metabolite 6-MAM and thebaine) was 78.9%. Fifteen were confirmed true positives: five morphine positive, four morphine plus codeine positive, three DHC positive, two codeine positive and one morphine plus thebaine positive. One of the morphine-positive samples contained a trace of 6-monoacetyl morphine (6MAM) by UPLC-MS/MS, i.e. a 6MAM peak was visible on the UPLC-MS/MS spectrum at a concentration above the limit of quantitation (2 ng/mL) but below the positive cut-off of 10 ng/mL. The remaining four samples were false positives (negative by UPLC-MS/MS); one of these contained a trace of morphine and another a trace of morphine and codeine. UPLC-MS/MS analysis detected a further six urine samples positive for morphine (false negatives by POC). Using the POC device, four of these produced a faint coloured line for the opiate test but in accordance with the kit insert were interpreted as negative. 6 No other drugs in the 26-drug panel were detected.
POC verification results
The results of adding CRM spiked urine at different concentrations to the POC testing device for all six test strips are presented in Figure 1. Results were read after the 5 min incubation as stated in the kit insert. For cocaine, methadone and benzodiazepine test strips, at the quoted analytical specificity of 300 ng/mL, only two samples tested positive for BZE and four for methadone and oxazepam. Increasing the concentration to 450 ng/mL (+50%), four samples tested positive for cocaine (BZE) and benzodiazepine (oxazepam) and all five samples for methadone. All five urine samples tested positive for buprenorphine and morphine at the quoted cut-off concentration or higher.
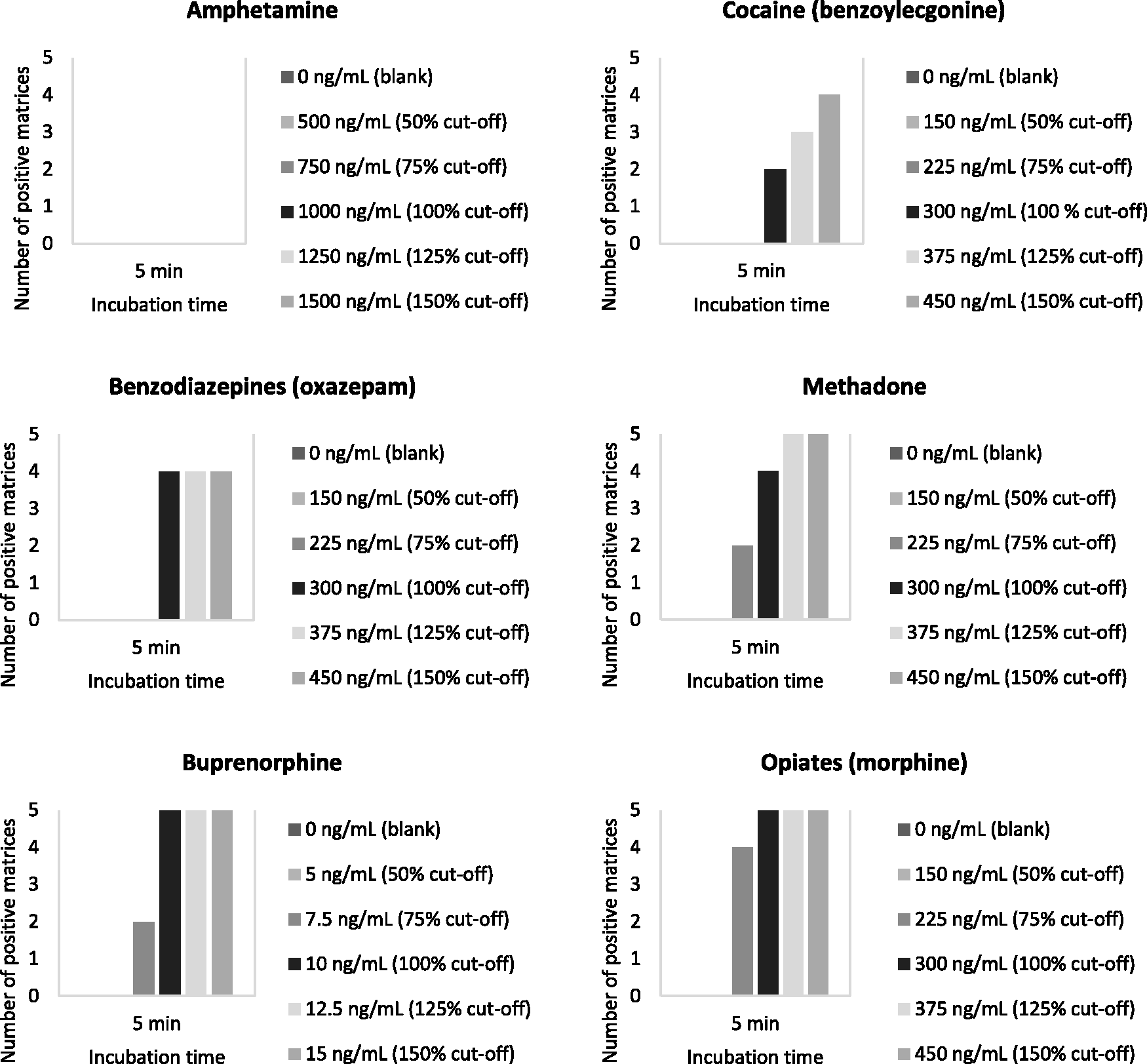
Verification of analytical specificity of the Alere™ Drug Screen Test Cup using drug-free urine from five volunteers spiked with drug at concentrations below and above the cut-off concentration quoted in the manufacturer’s kit insert.
The effect of incubation time on the stability of results using the POC device with spiked urine samples is shown in Figure 2. For cocaine (BZE), methadone and benzodiazepines (oxazepam), the number of samples testing positive was greater after just 2 min compared with the recommended 5 min. Compared with 5 min, the number of positive results at the maximum recommended reading time of 60 min decreased for all drugs tested other than morphine.
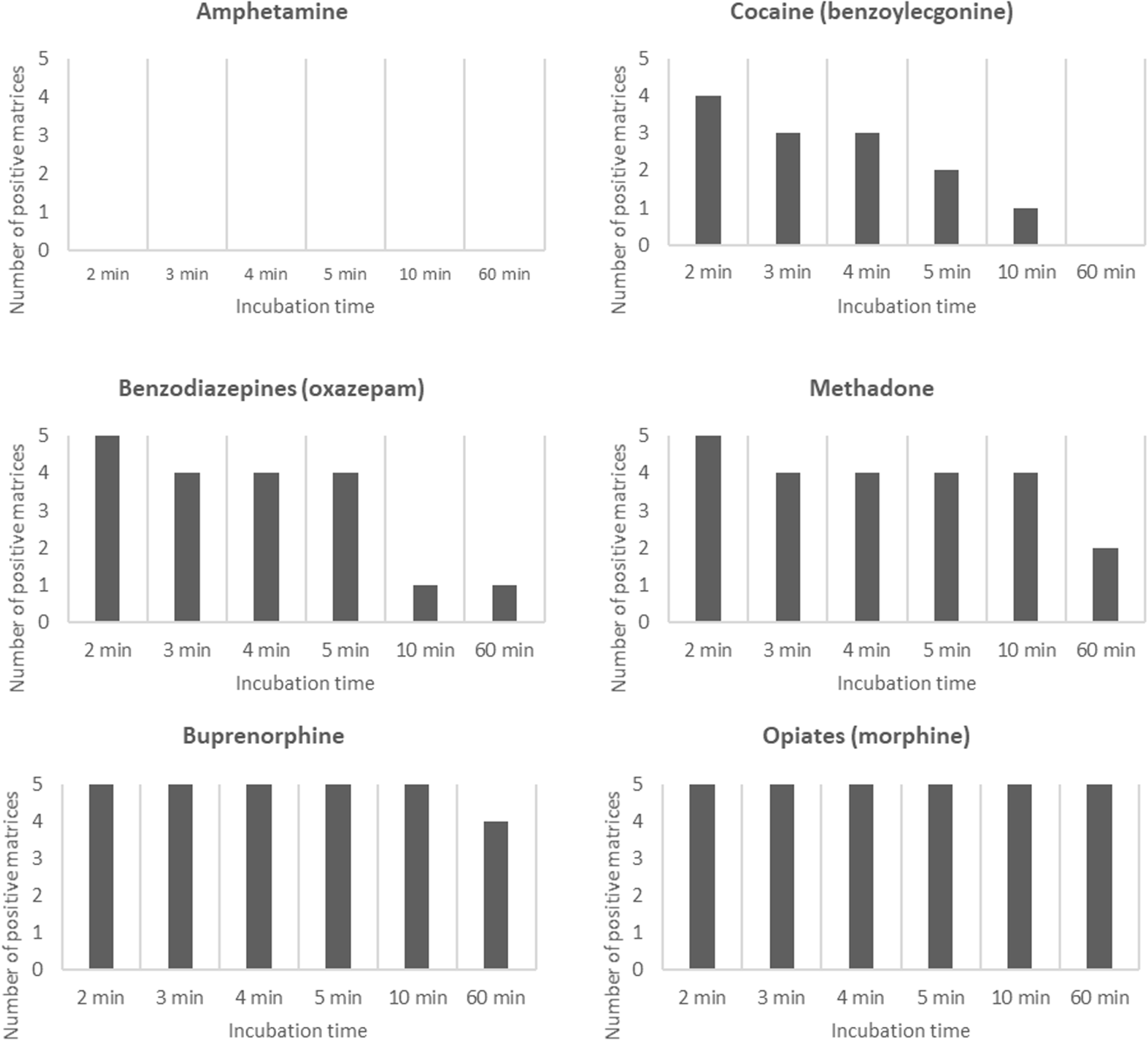
Verification of the 60 minute stability of the Alere™ Drug Screen Test Cup using drug-free urine from five volunteers spiked with drug at the cut-off concentration.
The specificity of the POC testing device for different opiates was assessed using blank urine spiked with either morphine or 6MAM at their respective cut-off concentrations (Figure 3). The long-term stability was assessed up to 48 h at room temperature. The POC testing device detected both morphine and 6MAM at 75% of the cut-off concentration at the recommended 5 min incubation period. At or above the cut-off concentration, results were stable up to 60 min for both opiates and up to 48 h for 6MAM.
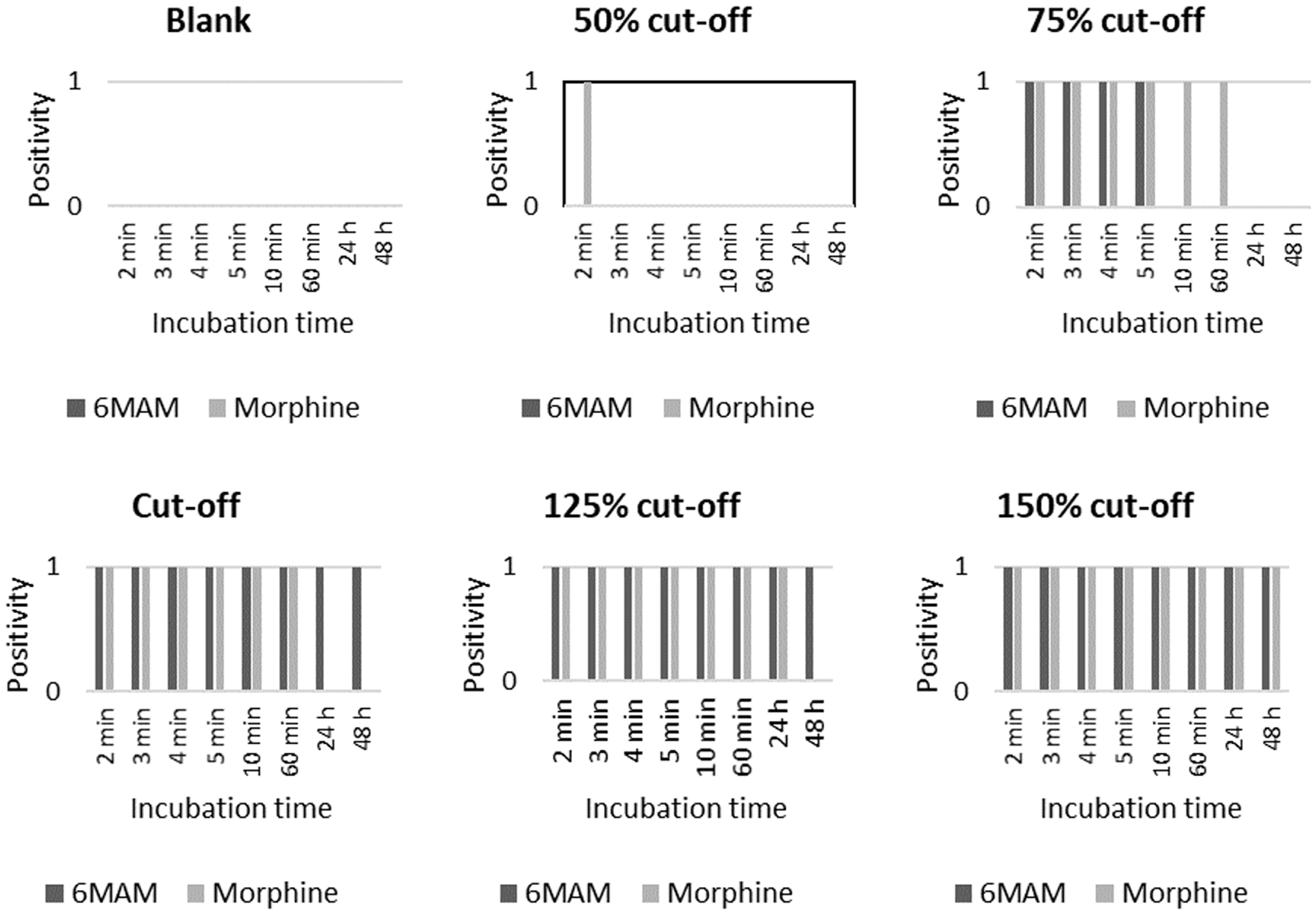
Assessment of the analytical specificity and long-term stability of the Alere™ Drug Screen Test Cup for morphine vs. the heroin metabolite 6-monoacetyl morphine (6MAM).
Discussion
Importance of timing in POC testing
Screening for the presence of drugs using a POC device is often carried out by midwives working in a busy, time-pressured environment and it is not always possible to adhere to the manufacturer’s recommended reading time for POC results. This study verified the manufacturer’s stability claims for morphine but for all other drugs the positive rate declined at 60 min. The decline in positivity was particularly stark for cocaine and benzodiazepines (Figure 2), highlighting the importance of adhering to the recommended incubation time to avoid false negatives.
Significance of sensitivity
Women may deliberately abstain from taking drugs in the several days prior to their antenatal appointment meaning that drug concentrations in their urine are low. There are no recommended cut-offs specifically for antenatal drug screening; however, the European Workplace Drug Testing Society (EWDTS) recommend the following cut-offs for screening methods employed for urine drug testing: amphetamines group: 500 ng/mL, benzodiazepines: 200 ng/mL; buprenorphine: 5 ng/mL; cocaine metabolites: 150 ng/mL; methadone: 300 ng/mL and opiates (total): 300 ng/mL. 9 Examination of the positive cut-off concentrations for the drugs in the POC testing device panel (Table 1) indicates that amphetamines, benzodiazepines, buprenorphine and cocaine do not meet the EWDTS recommendations for analytical specificity.
Analytical performance of POC testing
Analysis of anonymized urine specimens for opiates gave a diagnostic sensitivity of 73.9% for the POC testing device compared with the gold standard method UPLC-MS/MS (17 true positives/[17 true positives + 6 false negatives]). Eleven specimens (52.4% of opiate-positive specimens) tested positive for morphine only; heroin use cannot be excluded in these patients since morphine has a longer urinary half-life than 6MAM. 5 The other opiates identified include prescription drugs (codeine and DHC) and the dietary opiate thebaine. This lack of specificity to the type of opiate may lead to mothers being falsely accused of illicit drug use, which would be circumvented by screening with UPLC-MS/MS.
The sensitivity for the other drugs in the POC panel in patient specimens could not be calculated due to low or absent numbers of positive specimens. Instead, spiked blank urine was used to verify the analytical sensitivity of the POC testing device. This showed good performance for morphine, methadone, benzodiazepines and buprenorphine but poor performance for cocaine; hence, false negative results may be obtained when the cocaine metabolite BZE is present at the cut-off concentration. Sensitivity and stability were similar for morphine and 6MAM (Figure 3).
The kit insert for the POC testing device quotes a cut-off of 1000 ng/mL for amphetamine, based on calibration with D-amphetamine. 6 Street amphetamine is usually a racemic mixture of D and L amphetamine; 10 hence, a D,L-amphetamine mixture was used in the spiking experiment to a target cut-off of 1000 ng/mL. No positive results were obtained at any concentration or time interval tested (Figures 1 and 2). The kit insert quotes a separate cut-off of 3000 ng/mL for D,L-amphetamine (compared with 500 ng/mL recommended by the EWDTS) which may explain why false-negative results were obtained in the spiking experiment.
The purpose of this study was to assess the suitability of the POC device that is currently employed for drugs of abuse screening at Colchester Hospital. A limitation of current practice is that this panel does not include cannabis despite the specific mention of this drug in NICE guidelines for antenatal care. 11 Moreover, the 26-drug LC-MS/MS panel that is used in the few cases where confirmatory testing is performed does not include cannabis, since its lipophilic nature means it cannot be measured under the same analytical conditions as the other drugs in the panel. Future work should include screening for the prevalence of cannabis use in the local antenatal population.
Should we screen with LC-MS/MS?
The variation in sensitivity observed for detecting the drugs in the POC panel raises the question of whether UPLC-MS/MS ought to be used as the screening method since its detection limits are significantly lower. However, there are currently no mass spectrometric facilities within the pathology network serving Colchester Hospital and samples are referred externally for toxicological analyses, increasing both turnaround time and expense. Moreover, for cannabis, a separate confirmatory method (gas chromatography-mass spectrometry) is required, further adding to turnaround time and expense. From a clinical perspective, a rapid turnaround time is desirable in the management of a neonate presenting with neonatal abstinence syndrome: since symptoms can take up to five days to present, 12 the child may begin withdrawal at home in an environment in which drugs are used. Knowledge of a positive urine drug test prior to discharge enables prompt instigation of treatment, which may not be possible if UPLC-MS/MS were used as the screening method.
Feedback from a query raised via the ACB Mailbase revealed that there is variation in practice within the UK regarding the methods used for antenatal and neonatal drugs testing, emphasizing a need for national guidance in this area.
Conclusion
This study sought to test the suitability of POC screening for the detection of intrauterine drug exposure and identified limitations in its sensitivity and specificity. These limitations can be overcome by UPLC-MS/MS drug testing, but whether this provides results in a clinically useful timeframe is questionable. Further work is required to examine the cost effectiveness of introducing UPLC-MS/MS screening and the impact on neonatal outcomes when intrauterine drug exposure is detected early.
Footnotes
Acknowledgements
RCD would like to thank Jo Jerrom and the midwifery team for their help in specimen collection and Professor Jonathan Berg and the team at Birmingham City Toxicology Laboratory for providing advice, training and reagents.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: This project was undertaken by RCD as part of a Master’s degree in Clinical Biochemistry at the University of Manchester. CS and LF have nothing to disclose.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was funded by a Scientific Scholarship from the Association of Clinical Biochemistry and Laboratory Medicine (ACB), and from the Research and Development department at Colchester Hospital University NHS Foundation Trust. Neither funding body were involved in conducting the research or writing the manuscript.
Ethical approval
Ethical approval was sought from the Eastern Research Ethics Committee under reference number 15/EE/0407, and obtained on 17 November 2015.
Guarantor
RCD.
Contributorship
CS conceived and supervised the project. RCD conducted the study. The article was written by RCD and proof-read and edited by CS and LTF.
