Abstract
The introduction of high sensitivity measurement of cardiac troponin T (hs cTnT) and cardiac troponin I (hs cTnI) has given the laboratory the ability to measure very low levels of cardiac troponin. The limit of detection of these assays is well below the 99th percentile. These low levels can also be measured with small values of imprecision. A range of algorithms combining presentation measurement with repeat sample intervals of as little as one to 2 hours have been developed. These are able to predict with acceptable accuracy the diagnosis that would be achieved with continued repeat sampling out to six to 12 hours from presentation. In this article, we review the evidence for the diagnostic accuracy of these approaches and the practical aspects of implementation into routine clinical practice.
Keywords
Introduction
There are two criteria which define a high sensitivity cardiac troponin (hs-cTn) assay. First, the 10% coefficient of variation (CV) must be at or below the 99th percentile of the method. In practice, the 10% CV is often well below the 99th percentile. Second, the assay must be able to measure or quantify troponin, above the limit of detection (LOD), in 50% or more of the reference population, in both males and females. 1 The reference population has a profound effect on the value of the 99th percentile. 99th percentiles are progressively lower as the criteria to exclude occult cardiac disease become more stringent. 2 In addition, the ability to detect troponin in 50% of the reference population values can be compromised, particularly in females as troponin concentrations in healthy female controls are lower than males. 3
The introduction of hs cTn should be seen as evolution rather than revolution, as progressive improvement of analytical sensitivity and reduction of imprecision is a characteristic of immunoassay development. There has been variation in the pace of development between troponin assays. The change from the fourth-generation cardiac troponin T (cTnT) assay to the fifth generation hs-cTn assay was marked by a significant improvement in assay sensitivity. This increase in analytic sensitivity was less marked with troponin I assays, as compared to troponin T, generally, since earlier generation troponin I assays were already quite sensitive 4 and the impact of the shift to hs-cTn less dramatic. The clinical impact of hs-cTn arises from two assay features: first, the ability to measure very low values of cTn; second, to measure these values with very low imprecision. The ability to measure very low values means that the earliest phase of troponin release can be measured reliably. The ability to measure with low imprecision means that serial measurements at very low cTn values across short time intervals becomes possible. It is therefore possible accurately to reflect the release curve of troponin much earlier. These two features have resulted in the development of rapid diagnostic strategies for early detection of myocardial infarction based on single and rapid serial troponin measurement,5,6 updating previous strategies based on rapid serial measurement of creatine kinase and its MB fraction. 7
Rapid Diagnostic Algorithms for Confirmation and Exclusion of Myocardial Infarction
The definition of myocardial infarction (MI) has always required a cTn value that exceeds the 99th percentile and undergoes a significant rise. Rapid algorithms are not primarily diagnostic but predictive. They are based on the findings that early troponin measurements can be used to predict subsequent values taken three to six hours later. There is an inference that very low value troponin, such as below LOD, can rule out MI as the likelihood of a subsequent troponin rise to beyond 99th percentile is very remote. High values rule in MI. Values remaining stable at a value below the 99th over one to two hours are likely to remain so and rule out MI. Therefore, these algorithms categorise patients into rule-in, rule-out and intermediate. It is important to understand how hs-cTn assays can be used in this context. There are two components to rapid diagnostic algorithms: admission measurement, and serial measurement using a short repeat sampling interval (one to three hours).
On presentation, high-sensitive troponin value interpretation and inference is predicated on three characteristics of hs-cTn and the pathophysiology of coronary atherosclerosis and plaque rupture. First, a rise in troponin exceeding the 99th percentile that might previously have been borderline or not detected using a non-high sensitivity assay, can now be reliably measured. 8 Second, very low troponin values are associated with a very low probability of underlying cardiac disease and even atherosclerosis in the absence of overt cardiac disease.9–12 This translates into a low risk that the patient admitted with chest pain and a very low troponin is likely to have underlying ischaemic heart disease as the cause. Rule-out thresholds are close to or at the limit of detection of the assay, hence around the fifth percentile. Finally, and often overlooked is prior probability of disease. The prevalence of MI in chest pain populations is in the range 3–20%. When prior probability is low, diagnostic sensitivities of 95% will translate into impressive negative predictive values of >99%.
Serial measurement is contingent on low short-term intra-individual biological variation, and the ability to perform measurements with low degrees of analytical imprecision. Short-term biological variation of cTn is very low 13 so serial measurement over short time-frames allows early detection of change to confirm or exclude evolving myocardial injury.
Summary of studies of rapid diagnosis. (RCT, Randomised controlled trial. NPV, Negative Predictive Value. hs cTn, High Sensitivity Cardiac Troponin. hs-cTnT, High Sensitivity.
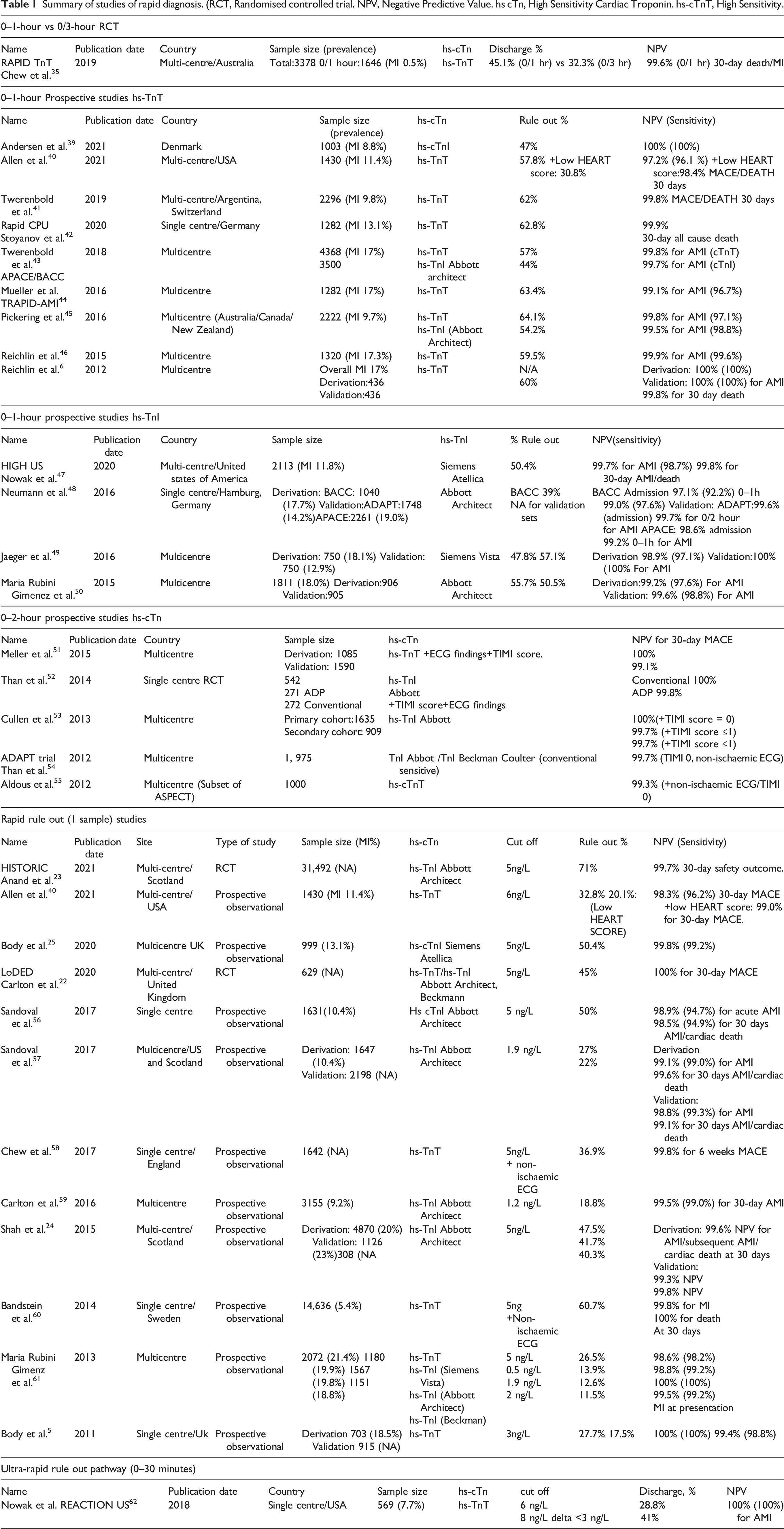
Strategies for implementation of hs-cTn for rapid diagnosis can be divided into three types, although these are not mutually exclusive. Admission measurement, often referred to as ‘one and done’ was originally proposed by Body for single-sample rule out. 5 This strategy has the strongest evidence base from meta-analysis of large-scale observational studies 21 and, most importantly, from two randomised controlled trials (RCT).22,23 When combined with risk scoring, the use of a single low troponin value excludes up to 50% of chest plain patients without MI depending on the series.24–26 A refinement proposed as part of the ESC guidelines is to use a single high hs cTn cut off to rule in MI. However, the diagnostic efficiency of this strategy is questionable. 27
The second strategy is to utilise serial measurement, with the first sample measured on presentation and the second measured one to 2 hours from admission, combined with calculation of the change (delta) between values. A value remaining below an optimised decision threshold (typically not the 99th percentile) combined with a low delta value allows rapid rule out MI. A high delta value rules in MI. In practice, admission measurement with rule out (and rule in) can then be followed by serial testing at one to 2 hours. Intermediate values (not ruled out or ruled in) require further testing. The third option is a composite approach using admission measurement followed by serial measurement at 3 hours but using the 99th percentile. 28 Here, rule out MI is either on admission measurement alone or both values remain below the 99th percentile with a low delta value. A high delta with a rise to a value exceeding the 99th percentile rules in MI.
All of the proposed strategies consider the question of diagnosis or exclusion of non-ST elevation myocardial infarction (NSTEMI). The problem is that patients present with chest pain and only a minority have ischaemic heart disease (8%, angina, 9% MI).
29
Another important complicating factor is competing causes of acute myocyte injury leading to troponin elevation; non-ischaemic causes are collectively more prevalent than NSTEMI.
30
They also do not address the problem of chronic myocardial injury. Practical strategies for use of hs cTn measurement in routine clinical practice need to consider all of these factors. One example of such an approach is illustrated in Figure 1. An objective method of describing chest pain for the chest pain score in HEART.
Practical aspects of implementation
The published analytical performance of the existing hs cTn methods means that in theory all should reach the analytical goals required for measurement close to the limit of detection of the assay and serial measurements over short time intervals. However, local verification of analytical performance is desirable, as not all instrumentation may necessarily achieve what is theoretically possible. 31 This can be remedied but usually requires close working with the manufacturer.
Presentation sample draw and single-sample rule out of low-risk individuals should be standard practice as it is strongly evidence-based and will allow immediate discharge of a significant proportion of chest pain patients. Patients not in this group, irrespective of the first troponin level, require serial testing over one to 3 hours to determine which of three categories they fall into. Where all cTn values are below the 99th percentile without a significant delta, myocardial injury is excluded. Discharge is then dictated by clinical suspicion of unstable angina or exclusion of serious non-ischaemic pathology. and the patient can be discharged. A significant delta value indicates acute myocardial injury (which will include MI) and requires hospital admission and assessment. The higher the troponin and the larger the delta value, the greater the probability the patient will have sustained an NSTEMI. Those with cTn above the 99th percentile but with minimal change require further assessment that the likelihood of acute injury is low but the troponin elevation requires explanation and possibly investigation. The use of the term ‘troponitis’ is entirely inappropriate. 32
The challenge with short interval serial measurement is the turnaround time that is possible with central laboratory analysis. Very few laboratories can provide detailed information on turnaround time measured from blood draw to provision of results. Most simply detail time from sample receipt in laboratory to result availability. Although laboratories aim for a 60-minute turnaround time, there is likely variation in total turnaround time and this is unlikely to be achieved consistently irrespective of timing of troponin sampling. This challenges the concept of the 0/1-hour ESC algorithm. Results for the first sample may not be available before the time for the second sample draw. The ESC guidelines 16 recommend (section 3.3.4.3) the second sample should be taken at 1 hour in the absence of the presentation sample result. This will have two consequences. First, two samples will be taken on all patients, which increases the cost and reduces the convenience of presentation sampling. Second, it is appropriate to await the results of the second sample, even if the presentation sample supports discharge, thus delaying discharge.
Pragmatically, the use of a 0–2 hour strategy, or a combined strategy based on 0–3 hour sampling would seem to be a solution. The UK Advanced Access Collaborative has sponsored a series of studies aimed at the implementation of rapid diagnosis based on hs-cTn. These case studies are available to read via the NEQAS cardiac biomarker website (https://www.ukneqas-cm.org.uk/). The results of these studies suggest that implementation of presentation sampling is usually straightforward but serial measurements is more complex and a 0–2 hour strategy was easier to implement. It is unlikely that a 0–1 strategy can be successfully implemented without the provision of hs-cTn by point of care testing (POCT).
Does rapid diagnosis make a difference?
There is evidence from randomised controlled trials (RCT)s of POCT that rapid diagnosis provision of cTn results can improve patient flow but the findings are not consistent and depend on local pathways.33,34The RCT of presentation sampling of cTn demonstrated safety but did not result in significant changes in length of stay. 22 The single RCT of 0–1 hour testing versus conventional testing did show a reduction in length of stay but the comparison was not with diagnosis based on the 99th percentile 35 and there are concerns about long-term outcomes in those who had rapid diagnosis and discharge. 36 Additionally, rapid diagnosis in this study did not appear cost-effective, 37 similar to findings with rapid diagnosis based on POCT. 38 Comparison of the modified 0–3 hour algorithm with usual care also demonstrated reduction in length of stay. 23 There was therefore evidence that provision of rapid diagnosis will reduce length of stay. In addition, COVID 19 has emphasised risks of prolonged ER or hospital study. A reduction of potential exposure to Covid-19, or other infections, provides a powerful incentive for accelerated safe discharge.
In conclusion, rapid diagnostic pathways based on hs-cTn should be part of the repertoire of all UK laboratories. Single-sample rule out on admission using hs-cTn is theoretically achievable with all hs-cTn methods currently available. The optimal serial testing strategy will depend on part on local circumstances. A RCT between strategies is likely to provide the most robust economic analysis of accelerated diagnostic pathways in practice.
Footnotes
Acknowledgements
Ms Susan Critichley and the Advance Access Collaborative; Dr Alan Reid of UK Cardiac NEQAS.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Guarantor
PC.
Contributorship
PC wrote the draft manuscript, AD compiled the evidence table, all three authors have approved the final version of the manuscript.
