Abstract
Background
Spuriously high results using the Abbott Architect enzymatic creatinine assay were noted to be particularly associated with very small sample volumes. This led us to query the effect of under-filling lithium heparin tubes on the measured enzymatic creatinine result.
Methods
Blood was provided by 5 laboratory personnel and then decanted into 5 x1.2 mL Sarstedt S-Monovette tubes, giving final blood volumes of 200, 400, 600, 800 and 1200 μL. Plasma was analysed using Abbott Architect Jaffe, enzymatic creatinine, Beckman Coulter (AU500) enzymatic creatinine and Roche (Cobas c702) enzymatic creatinine assays. Saline was also added to Sarstedt 1.2 mL and Teklab 2 mL tubes and analysed using the Abbott Jaffe and enzymatic creatinine methods.
Results
Increasing degrees of under-fill were associated with greater over-estimation of creatinine using the Abbott enzymatic assay, but no difference was noted using Jaffe methodology on the same platform or enzymatic assays provided by Roche or Beckman. On average, creatinine was 40.6% (+27.7 μmol/L) higher when only 200 μL of blood was present in the tube. Small volumes of saline added to lithium heparin tubes measured significant creatinine concentrations using the Abbott enzymatic method.
Conclusions
Lithium heparin directly interferes in the Abbott Architect enzymatic creatinine assay. Under-filling lithium heparin tubes can lead to clinically significant over-estimation of creatinine results by this assay. Users of this assay should be aware of the potential for spurious results in small sample volumes collected into lithium heparin tubes and implement robust procedures for identifying and reporting results on these samples.
Introduction
Lithium heparin is a commonly used additive for blood collection tubes and is the primary tube type used by the acute areas of adult hospitals as well as the neonatal unit and throughout the paediatric hospital in our Board. Interference in clinical chemistry tests from increased concentrations of lithium heparin in blood in under-filled tubes has been reported previously 1 but not for creatinine. Numerous publications have addressed the lack of specificity of creatinine analysis using Jaffe methodology and heralded the use of enzymatic methods. 2 These are now the method of choice recommended by clinical guidelines. However, enzymatic methods are not without problems 3 ; interference from haemoglobin, bilirubin and glucose still occurs, for example, albeit to a lesser extent than with the Jaffe methodology. We report for the first time the potential for significant over-estimation of creatinine by the Abbott Architect enzymatic creatinine assay in under-filled lithium heparin tubes.
Method
Five volunteers from laboratory personnel (3 female and 2 male) each provided 5 mLs of blood which was collected into a syringe and decanted into 5 1.2 mL lithium heparin tubes (Sarstedt S-Monovette) to give varying degrees of under-fill from 800 to 200 μL. One tube was filled to the line for each patient (1.2 mL) to represent the baseline value for that individual. Tubes were inverted several times to achieve thorough mixing with the anticoagulant as per manufacturer’s instructions. Plasma was separated, anonymised and stored at −20°C prior to analysis. Samples were defrosted, centrifuged and analysed in a single batch prior to transport to external laboratories. All laboratories analysed the samples in a single batch to minimise analytical variation. Creatinine was measured via 4 different kits: Abbott alkaline picrate kinetic Jaffe, Abbott enzymatic creatinine (Abbott Architect C16000), Beckman enzymatic creatinine (Beckman Coulter AU5800) and Roche enzymatic creatinine (Roche Cobas c702). All of the enzymatic methods use creatininase, creatinase and sarcosine oxidase in a series of reactions resulting in the production of hydrogen peroxide. In addition to the investigations on whole blood, different volumes of saline were added to 1.2 mL Sarstedt tubes (75/150 μL) alongside 2 mL Teklab tubes (100/200 μL) (Teklab, UK) and analysed on the Abbott enzymatic and Jaffe methods in order to demonstrate that the interference was independent of other analytes in the blood and the type of tube used. All samples were analysed according to manufacturer’s instructions, and there was no evidence of haemolysis.
Results
Abbott Jaffe, Beckman enzymatic and Roche enzymatic methods did not demonstrate interference from lithium heparin tubes; the analytical variation in creatinine result between different sample volumes did not deviate from that expected from routine use (mean %CV for the 5 patients for each method was as follows: Roche 1.29, Beckman 1.28 and Abbott Jaffe .94). However, the Abbott enzymatic method (%CV at 57.7 μmol/L, 2.6) demonstrated substantially higher creatinine results as the volume of blood in the tube decreased (Figure 1). Mean %CV for the 5 patients for this method was 14.72. Baseline values varied from 54.4 to 79.9 μmol/L. On average, results were 17.8% (12.1 μmol/L) higher when the tube was half-filled with whole blood; this increased to 40.6% when there was only 200 μL of blood. This represented a mean error of +27.66 μmol/L (21.5–41.9 μmol/L) when there was only 200 μL of blood in the tube. This degree of interference was independent of the baseline creatinine concentration: when 100 μL of saline was added to the 1.2 mL tube, a creatinine result of 45 μmol/L was generated by the Abbott enzymatic method (undetectable by Jaffe). A similar interference with the creatinine assay was seen when saline was added to Teklab tubes, although a smaller volume of saline was required to achieve a similar creatinine result (Table 1). (a) Difference in creatinine between a full Sarstedt S-Monovette 1.2 mL lithium heparin tube and one containing only 200 μL of blood. Central line for each method indicates mean change in creatinine and outer lines represent the range. Dots represent individual volunteers. (b) Relationship between the measured creatinine (Abbott enzymatic) and the amount of whole blood in a Sarstedt S-Monovette 1.2 mL lithium heparin tube from 5 people. Different symbols indicate individual volunteers. Creatinine concentrations measured in different volumes of saline using the Abbott enzymatic method. Teklab tubes (2 mL) contain 16 IU lithium heparin/mL blood. Sarstedt 1.2 mL tubes contain 35 IU lithium heparin/mL blood. *Predicted concentration of lithium heparin is based on complete dissolution of lithium heparin into the liquid.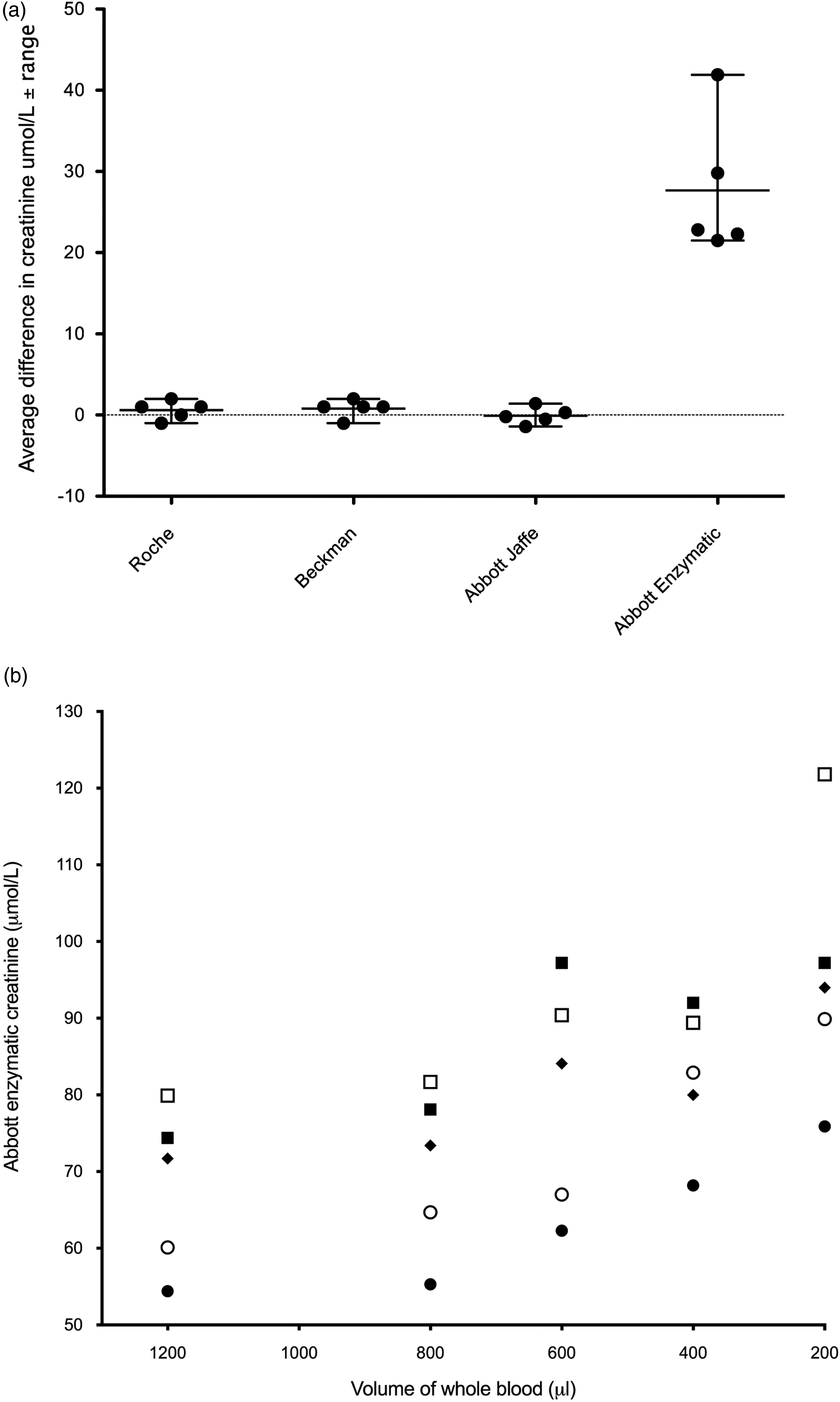
Discussion
This study has demonstrated for the first time a positive interference in the Abbott enzymatic creatinine assay caused by excess lithium heparin, which could have a detrimental impact on the patient. This interference was not apparent in other enzymatic assays provided by 2 other major manufacturers, Beckman Coulter and Roche Diagnostics (Figure 1). Interference from heparin has been observed in a variety of tests 4 and increases in creatine kinase (CK) and gamma-glutamyl transferase (GGT) have been previously noted on the Beckman UniCel DxC system using under-filled tubes. 1 In the case of the Beckman CK assay, the mechanism of interference has been attributed to the precipitation of β lipoproteins. This interference could be overcome through the addition of imidazole to stabilise the pH (Beckman Coulter AU series CK). 1 A heparin agonist such as protamine sulphate resolved interference in GGT observed in Roche Hitachi Modular D and P systems that was attributed to interactions between heparin and an IgM paraprotein 5 ; however, it is not clear what the mechanism is for the interference in the Abbott enzymatic creatinine assay. The wavelengths employed to determine the creatinine concentration are very similar for both Roche (700/546 nm) and Abbott (700/548 nm) enzymatic methods, suggesting that spectral interference from heparin is not the issue. The measurement of a clinically relevant creatinine concentration in saline that had been mixed with the lithium heparin anticoagulant suggests that the mode of interference is a direct reaction of lithium or heparin with a component reagent in the reaction pathway. Although the chemistries of the reactions are broadly similar between the 3 enzymatic methods, they do differ in the ingredient that reacts alongside 4-aminoantipyrine with hydrogen peroxide. Abbott employs ESPMT (N-ethyl-N-sulfopropyl-m-toluidine) as a chromogen, as do some other enzymatic creatinine kits, for example, Erba Mannheim, ABX Pentra and Genzyme diagnostics.
This study used Sarstedt S-Monovette 1.2 mL tubes, which have a particularly high lithium heparin concentration of 35 IU/mL suggesting that cross reactivity could be exaggerated using this assay and tube combination. However, it will not be limited to this tube type. Investigations using saline demonstrated similar creatinine results were achievable using tubes from another manufacturer when the volume was altered to reach the same overall predicted lithium heparin concentration (Table 1). The degree of interference experienced in patient samples is likely to be variable and will depend not only on the extent of mixing of the sample prior to centrifugation but also what form the lithium heparin is delivered in, for example, spray coated tubes or beads. Heparin on the walls of the tube may have a smaller effect if the blood is not mixed thoroughly in accordance with manufacturer’s instructions. The larger 4.9 mL Sarstedt tubes used in our Health Board have a more typical lithium heparin concentration of 16 IU/mL; however, this is present on beads in the bottom of the tube, and the total concentration of lithium heparin in the tube is 78.4 IU (c.f. 42 in 1.2 mL tubes.) Although in this study, tubes were mixed sufficiently and could be thought to represent a ‘worst case scenario’, we have encountered examples of significant errors in patient samples. One of the most extreme was for a 20-day-old well baby who had a creatinine of 83 μmol/L on a small sample which repeated as 29 μmol/L when a sufficient volume of blood was collected. Examination of monthly patient results in our Board demonstrated the proportion of paediatric creatinine results above the age-adjusted reference interval rose from approximately 11.4% to 20.6% since the switch from Teklab to Sarstedt tubes (mostly 1.2 mL).
There is no doubt that excess lithium heparin causes spurious over-estimation of creatinine by the Abbott enzymatic creatinine assay and that any laboratories using this assay need to devise a robust procedure to identify and appropriately report results generated on under-filled lithium heparin tubes. If these errors are not identified, they have the potential to lead to unnecessary intervention which could cause harm to the patient. Rebleeding inpatients and re-call of out-patients to follow up on elevated results is a significant cause of concern for clinicians, parents and patients as well as a cost to the healthcare provider. As it is unrealistic to avoid lithium heparin samples for core chemistry testing in paediatric patients, it is imperative that assays are able to perform within the required specifications for clinical need. The potential for incorrect results in this context must be addressed.
Footnotes
Acknowledgements
We would like to thank staff from Clinical Biochemistry at the Royal infirmary of Edinburgh who provided the phlebotomy and volunteers who provided the blood samples.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not required.
Guarantor
MS.
Contributorship
MS conceived the idea, performed the analysis on the Abbott site and wrote the manuscript. HW prepared the figures. HH and KH arranged analysis on the alternative assays. All authors approved the final manuscript.
