Abstract
Multiple myeloma is a haematological cancer caused by malignant plasma cells in the bone marrow that can result in organ dysfunction and death. Recent novel treatments have contributed to improved survival rates, including monoclonal antibody therapies that target the CD38 protein on the surface of plasma cells. Anti-CD38 therapies are IgG kappa monoclonal antibodies that are given in doses high enough for the drug to be visible on serum protein electrophoresis as a small paraprotein. We present a case where isatuximab, the most recent anti-CD38 monoclonal antibody to be approved for treatment of myeloma, obscured the patient’s paraprotein on gel immunofixation, so that complete remission could not be demonstrated. This was resolved using the isatuximab Hydrashift assay. The interference on gel immunofixation was unexpected because isatuximab migrated in a position distinct from the patient’s paraprotein on capillary zone electrophoresis. We demonstrate the surprising finding that isatuximab migrates in a different position on gel electrophoresis compared to capillary zone electrophoresis. It is vital that laboratories are aware of the possible interference on electrophoresis from anti-CD38 monoclonal antibody therapies, and are able to recognise these drugs on protein electrophoresis. The difference in isatuximab’s electrophoretic mobility on capillary and gel protein electrophoresis makes this particularly challenging. Laboratories should have a strategy for alternative analyses in the event that the drugs interfere with assessment of the patient’s paraprotein.
Introduction
Multiple myeloma is a haematological cancer that accounts for 2% of all cancer cases in the UK (2017 data). 1 It is caused by malignant plasma cells, which are end-differentiated B cells that secrete antibody. Symptoms include fatigue, bone pain, pathological fractures, recurrent infection and renal impairment. Myeloma is incurable but survival rates are improving. Monoclonal antibody therapies targeting CD38, a cell surface protein found on plasma cells, emerged as a novel therapy for multiple myeloma in 2015 and have significantly improved the depth of response to treatment. Daratumumab (DarzalexTM, Janssen Biotech) was approved by NICE in 2018, and more recently, isatuximab (SarclisaTM, Sanofi) has also been approved for treatment of myeloma.
Serum protein electrophoresis is an important tool in the diagnosis and monitoring of myeloma. Laboratory scientists are trained to recognise normal and abnormal electrophoretic patterns and are looking for abnormal peaks or bands that may represent a monoclonal protein, suggesting the presence of a B-cell malignancy such as myeloma. Patients diagnosed with myeloma can then be monitored using the concentration of the monoclonal protein. The emergence of a new abnormal peak on protein electrophoresis could indicate relapse or lack of response to treatment. Complete remission may be defined as the absence of the paraprotein on serum and urine immunofixation. 2
Anti-CD38 monoclonal antibody therapies are IgG kappa antibodies that are given in doses high enough to achieve a serum concentration of approximately 0.5–2 g/L and can therefore be detected on serum protein electrophoresis. 3 We present a case involving a myeloma patient where isatuximab prevented demonstration of complete remission. We describe how this was resolved using an isatuximab Hydrashift assay, and demonstrate that isatuximab migrates differently on capillary zone electrophoresis (CZE) compared to gel electrophoresis.
Method
Serum samples were analysed by protein electrophoresis (Capillarys Flex 2 and Phoresis software, Sebia, UK; smoothing = 0, variable view) and serum immunofixation (Hydrasys, Sebia). A serum sample was analysed by isatuximab Hydrashift assay (Sebia) using a modification of the manufacturer’s instructions, with samples run 1/3 for the IgG lane and neat for all other lanes to increase sensitivity.
Residual patient samples that showed no abnormality on CZE were spiked with isatuximab (3 g/L) and analysed by CZE and by gel immunofixation.
Case summary
A 65-year-old man was diagnosed with myeloma in 2012. He showed a partial response to induction treatment (bortezomib/adriamycin/dexamethasone), but then progressed. Disease stabilised with second-line treatment (cyclophosphamide/lenalidomide/dexamethasone). He showed a very good partial response following third-line treatment with DT-PACE (dexamethasone/thalidomide/cisplatin/Adriamycin/cyclophosphamide/etoposide) and autologous stem cell transplant in 2013. He had a sustained remission until 2020, when he relapsed and was started on isatuximab in combination with pomalidomide and dexamethasone, to which he responded well.
Results
Analysis of samples from the patient after starting treatment with isatuximab showed the presence of a ‘zone’ in the fast gamma region in addition to the patient’s IgG kappa monoclonal protein in the mid-gamma region on CZE (Figure 1). The paraprotein concentration decreased until at 6 months after starting isatuximab treatment, it was no longer clearly visible as a discrete band on serum protein electrophoresis. Further investigation by gel immunofixation found that isatuximab co-migrated with the patient’s paraprotein (Figure 1(c)), so complete remission could not be confirmed. Six months later, his sample was tested using the isatuximab Hydrashift assay, which confirmed that his paraprotein was no longer detectable on serum immunofixation (Figure 1). Interference of isatuximab on gel immunofixation resolved using Hydrashift assay. Comparison of samples taken before (a) and 3 months after (b) isatuximab treatment, analysed by CZE. The patient’s paraprotein (star) runs in the mid-gamma region and isatuximab was detected as a small ‘zone’ (arrow) in the fast gamma region. Four months after treatment, the patient’s paraprotein was no longer clearly visible on CZE (c) but analysis by immunofixation (d) showed a single diffuse IgG kappa band in the mid-gamma region and no band in the fast gamma region. (e) Analysis of a sample taken 11 months after isatuximab treatment by Hydrashift assay. Anti-isatuximab antibody is added to the IgG and kappa lanes of the immunofixation gel and binds to any isatuximab in the sample, causing isatuximab (arrow 1) to migrate in the alpha region in these two lanes (arrow 2). No paraprotein was visible in the gamma region, confirming complete remission.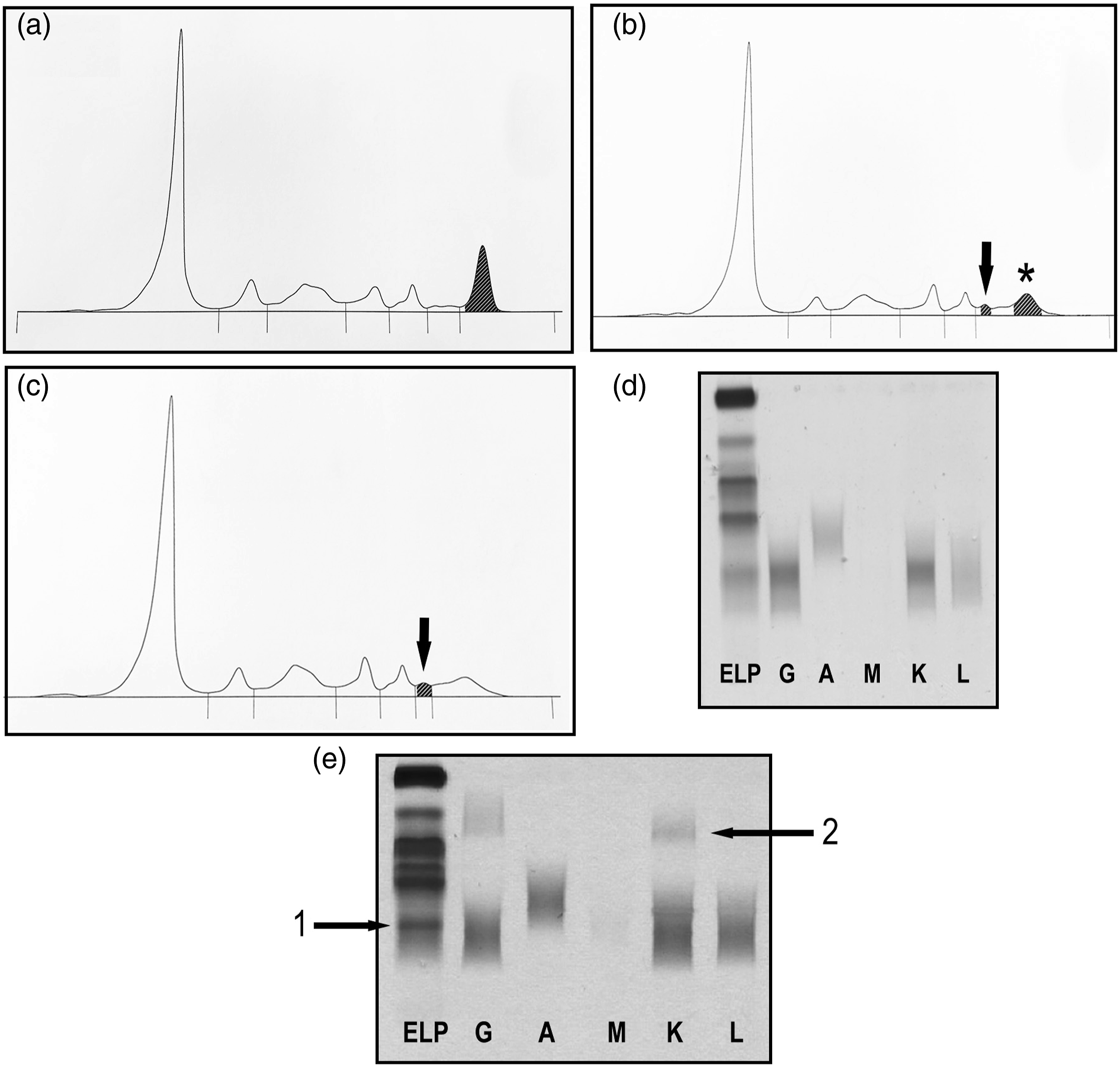
To further confirm that isatuximab migrated in a different position on CZE compared to gel electrophoresis, we selected a patient sample with a normal electrophoretic pattern and a patient sample with an IgG lambda paraprotein that ran with similar electrophoretic mobility to isatuximab on CZE. We spiked the samples with 3 g/L isatuximab and showed that isatuximab migrated in the fast gamma on CZE but the mid-gamma on gel immunofixation. Isatuximab migrated anodal to (to the left of) the IgG lambda paraprotein on CZE but cathodal to (below) the IgG lambda paraprotein on gel electrophoresis (Figure 2). Appearance of isatuximab on protein electrophoresis. (a) A normal serum sample was spiked with 3 g/L isatuximab, resulting in a peak running in the fast gamma region on CZE (black arrow). (b) The spiked sample was analysed by gel immunofixation and showed an IgG kappa band in the mid-gamma region (black arrow). A patient sample containing a small IgG lambda paraprotein (grey arrow) was spiked with 3 g/L isatuximab (black arrow) and analysed by CZE (c) or gel immunofixation (d). Isatuximab appeared as a peak anodal to (left of) the IgG lambda paraprotein on CZE, but as a band cathodal to (below) the IgG lambda paraprotein on gel immunofixation.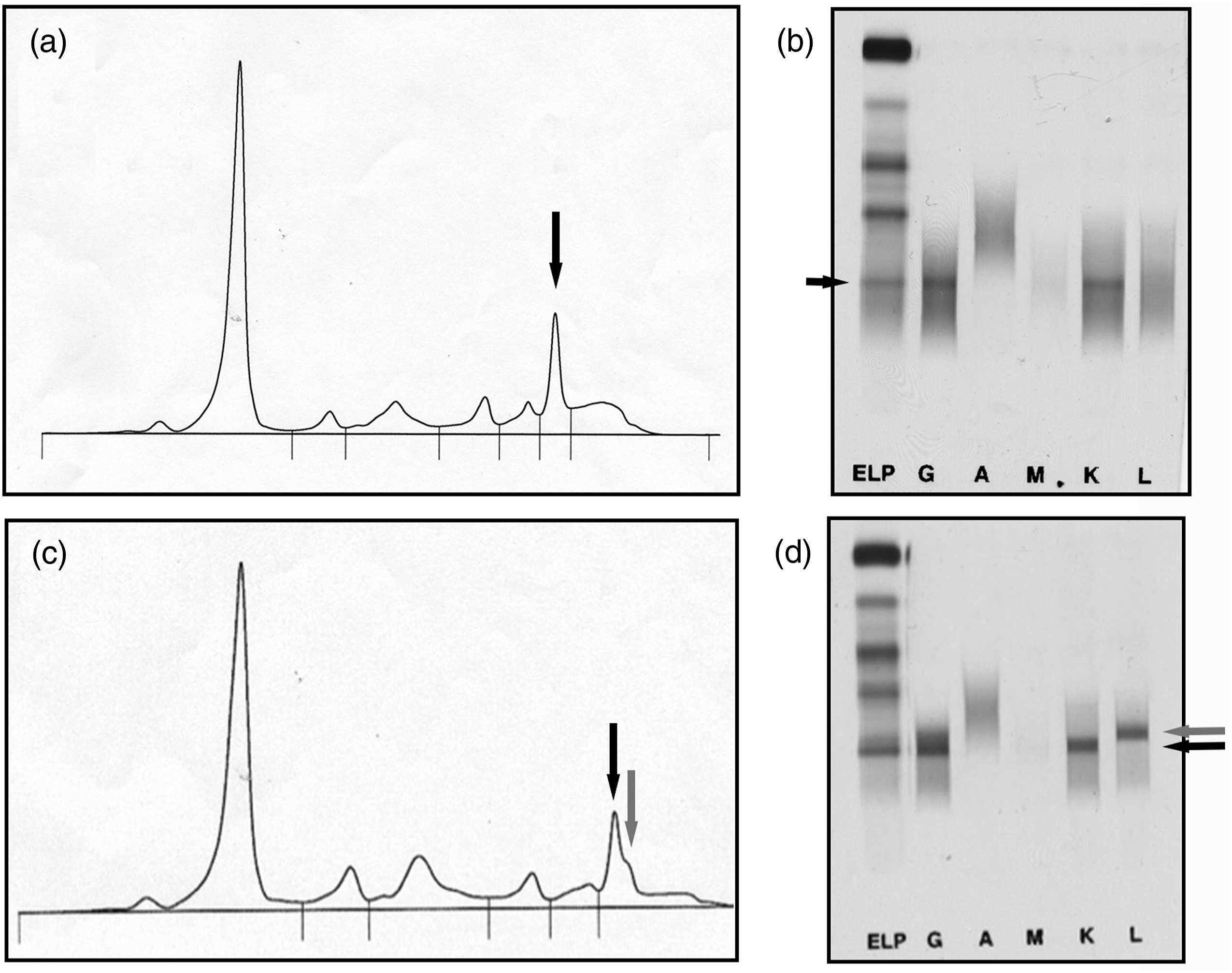
Discussion
We have presented a case illustrating how isatuximab interfered with demonstration of complete remission by immunofixation. We have shown an example of when laboratories may need to use the isatuximab Hydrashift assay to enable assessment of the patient’s paraprotein. We have also demonstrated that isatuximab migrates in the fast gamma region on CZE but in the mid-gamma region on gel electrophoresis. This phenomenon is not seen with daratumumab, which migrates in the slow gamma region on both CZE and gel immunofixation, as we have reported previously. 4
The finding that isatuximab migrates in a different position on CZE compared to gel electrophoresis was a surprising finding. It has been shown previously, 3 but was not commented on explicitly. Differences in electrophoretic mobility between different electrophoresis systems have been reported previously. 5 The electrophoretic mobility of an individual protein in an electric field depends on its isoelectric point, the pH of the system buffer, and the matrix through which it is migrating. 6 Capillary zone electrophoresis typically uses a slightly higher pH buffer, for example, pH 9.3, compared to gel electrophoresis, as the system is designed to maximise endosmotic flow in the capillaries. In addition, the formulation of the immunofixation gels used is designed to maximise separation of proteins in the gamma region, which enhances the difference in migration of isatuximab. It remains to be determined which of these factors contribute to the difference in electrophoretic migration between CZE and gel electrophoresis.
Many drugs can cause interference on protein electrophoresis. Some drugs are detected on CZE as abnormal peaks due to their ability to absorb at approximately 200 nm wavelength, for example, antibiotics such as ceftriaxone and contrast agents such as OmnipaqueTM.7,8 They can be demonstrated as interference because they are not detected on gel electrophoresis, which uses protein-binding dyes such as acid violet. Such interference can be avoided altogether with better timing of when the blood sample is drawn. Monoclonal antibody therapies are a newer class of drugs that have the added confounding factor of being monoclonal proteins in their own right and can therefore cause interference on electrophoresis. 9 Whether or not these therapies are detected on CZE depends on a plasma concentration of at least 0.5–1 g/L being achieved, which is the limit of detection for CZE. 10 This has become of particular relevance with the introduction of the anti-CD38 monoclonal antibody therapies for treatment of myeloma, where a key part of patient monitoring is detection of monoclonal proteins by protein electrophoresis.
The recommended dose for daratumumab and isatuximab is 1800 mg subcutaneous and 10 mg/kg intravenous, respectively. In our experience, this produces a small peak of 1–2 g/L in the slow gamma region for daratumumab, and a small peak of approximately 0.5–1g/L in the fast gamma for region isatuximab on CZE. Detection of these peaks in an individual patient’s sample will depend on the amount and electrophoretic pattern of the background immunoglobulins, particularly for isatuximab.
It is important that laboratories are familiar with the appearance of the anti-CD38 therapies on protein electrophoresis to avoid misinterpretation of results as a possible relapse of myeloma. It is uncommon for a patient to have an IgG kappa monoclonal protein that migrates in exactly the same position as daratumumab or isatuximab, so if a new small IgG kappa paraprotein is detected in a myeloma patient that migrates in a position consistent with one of the anti-CD38 monoclonal antibody therapies, the patient record should be checked to see if this patient has started treatment with the relevant drug. This could be further helped if clinical staff stated these drugs in the clinical details accompanying blood samples.
In the rare event that a patient does have an IgG kappa monoclonal protein that migrates in the same place as daratumumab or isatuximab, the presence of the patient’s paraprotein can be confirmed using the appropriate Hydrashift assay. In the case of isatuximab, running the protein electrophoresis by an alternative method (CZE vs gel electrophoresis) may help visualise the patient’s paraprotein. In the case we presented here, isatuximab obscured the patient’s paraprotein on immunofixation, preventing us from confirming complete remission. This was resolved using the Hydrashift assay.
In summary, it is vital that laboratory staff are aware that current myeloma treatment strategies include monoclonal antibody therapies that can be detected on serum protein electrophoresis. We hope that the images we have presented are helpful to laboratories in being able to recognise these drugs and assist in accurate interpretation of protein electrophoresis results to facilitate the best patient care.
Footnotes
Acknowledgements
We would like to thank Natalie Hopkins for assistance with reagents.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Written informed patient consent was given for this study to be published.
Guarantor
RW.
Contributorship
RW, MVC and AC designed and carried out the laboratory analysis. RW, SL and JM reviewed and interpreted the results. RW wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
