Abstract
Background
The novel daratumumab immunotherapy is a human IgG1 kappa antibody targeted against CD38, which is almost universally expressed on myeloma plasma cells. Daratumumab has efficacy in clinical trials for the treatment of multiple myeloma; however, it complicates laboratory monitoring of the serological response to treatment, as it is detected by serum electrophoresis and/or immunofixation.
Methods
Laboratory reports of electrophoresis patterns serially performed in a single laboratory of six patients with relapsed multiple myeloma receiving daratumumab therapy as part a clinical trial were reviewed retrospectively.
Results
Post administration of daratumumab therapy, an additional band was visible by serum electrophoresis, migrating to the mid-gamma region, which was confirmed as IgG kappa by immunofixation. In five out of the six patients, this band was quantified at <2.0 g/L. For one patient, this band co-migrated with the patient’s disease paraprotein band, so both bands were quantified together. The appearance of an apparent second paraprotein band while receiving treatment for multiple myeloma can cause anxiety for patients, confusion for healthcare workers and may also underestimate complete remission rates.
Conclusions
The clinical laboratory must be aware of the interference of daratumumab in serum electrophoresis. Effective communication between clinicians and the laboratory is essential for the production of clinically valuable, non-misleading reports for these patients.
Introduction
Multiple myeloma (MM) is a malignancy of plasma cells resulting in uncontrolled proliferation of a plasma cell clone. In a majority of MM, these cells produce large amounts of intact monoclonal immunoglobulin, termed M-(for monoclonal) protein or paraprotein, with around 15% producing clonal free light chains only. MM is currently an incurable disease, with most patients following a cycle of remission after treatment, followed by serial relapse at later stages or on permanent/maintenance therapy. Current treatment options include supportive care (bisphosphonates, corticosteroids, pain relief), to older chemotherapeutic agents (cyclophosphamide, melphalan, anthracyclines and bendamustine) and newer biological agents, such as proteosome-inhibitors (bortezomib, carfilzomib, ixazomib), immunomodulatory agents (thalidomide, lenalidomide, pomalidomide) and histone-deacetylase-inhibitors (panobinostat) with most younger/fitter patients undergoing autologous stem cell transplant. Several new therapies for MM have been developed, including monoclonals, such as elotuzumab (anti-CS1/SLAMF7), daratumumab and isatuximab, which are both anti-CD38 immunotherapies.
Daratumumab is a monoclonal of IgG1 kappa immunoglobulin class, which targets CD38-expressing cells present in patients with MM. Daratumumab has been used in phase III monotherapy combination comparative trials, where it has been shown to be effective at improving progression-free survival. 1 However, daratumumab has the potential to complicate serological assessment in the clinical laboratory, introducing possible confusion for patients and clinicians.
The clinical laboratory is essential in the longitudinal management of patients with MM including assessment of end-organ damage/function, monitoring myelosuppressive and metabolic consequence of treatment, and MM itself, provision of transfusion products, as well as being fundamental to monitoring of remission status and response to treatment, through serum protein electrophoresis (SPE), immunofixation (IF) and quantification of serum and urine light chains. It has been well documented that once a patient has commenced on daratumumab, it is not possible to obtain a reliable antibody screen when cross-matching blood products due to false-positive Coomb’s test, as daratumumab binds to low concentrations of CD38 on the surface of red cells giving a false-positive result. 2 However, enzyme treatment in vitro together with selection Kell-negative cells can obviate this effect. More recently, it has been shown that daratumumab is demonstrable by serum protein electrophoresis and can be detected as a discrete peak on a capillary electropherogram. Sheldon et al. 3 showed the presence of a discrete, quantifiable peak on capillary zone electrophoresis, in four patients taking daratumumab. This peak was resolved from the patients’ original paraprotein, and always migrated to the same position on the X axis of the electrophoretic trace.
This extra peak may impact upon assessment of remission status according to the International Myeloma Working Group Uniform Response Criteria. In these criteria, complete remission (CR) is defined as ‘Negative immunofixation on the serum and urine, and disappearance of any soft tissue plasmacytomas and < 5% plasma cells in bone marrow’. 4 Reporting of any ‘paraprotein’ band, even an artefact of their therapy, will potentially prevent a patient meeting this definition, resulting in inaccurate response assessment, potentially underestimating efficacy of trial combinations based on daratumumab. In addition, if the new daratumumab band is misinterpreted, by an interested patient, or clinician, it potentially causes unnecessary stress and anxiety.
A retrospective examination of the reporting of SPE results in MM patients taking daratumumab as part of a clinical trial was performed in a tertiary cancer centre with a large Clinical Trials Unit and Specialist Haemato-oncology Department. This was with the aim of improving reporting practices for these patients to accurately clarify remission status, ensure that we do not misascribe progression of MM, to maximize precision of response-assessments, avoid unnecessary stress for the patient and provide added value for the clinical teams.
Methods
Six patients diagnosed with MM and receiving daratumumab monotherapy as part of an extended access peri-licence clinical trial were identified. SPE and IF scans and laboratory reports pre- and postdaratumumab therapy were reviewed. SPE and IF were performed on the Hydrasys 2 Scan (Sebia UK, Ltd). Densitometry was carried out using Phoresis software (Sebia UK, Ltd), with paraprotein bands quantified using the perpendicular drop method. To gain an understanding of the utility of the electrophoresis reports produced by the laboratory while these patients were being treated with daratumumab, a questionnaire was sent to a Consultant Haematologist specializing in MM.
Results
Review of SPE gels showed that post daratumumab therapy, an additional band was visible on the first sample post-treatment (approximately four weeks), migrating to the mid-gamma region. This band was confirmed in all cases as IgG kappa by IF and quantified as a paraprotein band. Figure 1 shows IF gels for the cases examined. For five of the six patients (cases 2–6) in Figure 1, this band was quantified discretely from the original paraprotein band. For one patient, the second band co-migrated with the patient’s myeloma paraprotein, and hence the two bands were quantified together (case 1). At the time of reporting, the laboratories were not aware that these patients were being treated with daratumumab, as it was not mandated within trial. This led to the reports being potentially unclear to the clinicians as evidenced by the questionnaire results, highlighting that some reports were clear that there were second bands but others not, and that it was ‘difficult to assess patients who had IgG Kappa secretory disease’.
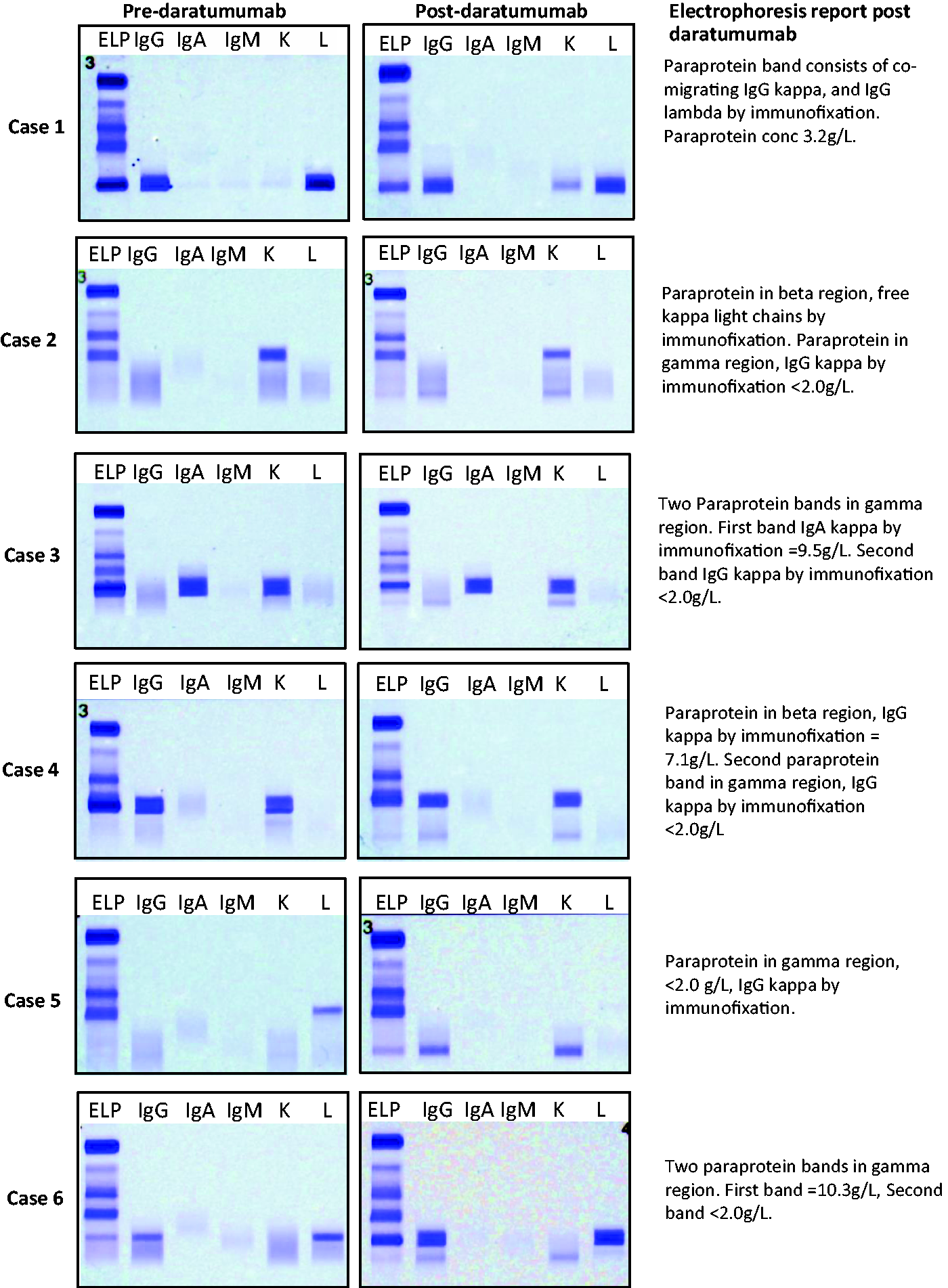
Immunofixation gels of six cases showing electrophoresis patterns observed pre- and post-daratumumab therapy. The postdaratumumab immunofixation gels show the appearance of an IgG kappa band in the mid-gamma region.
Discussion
Daratumumab can be detected on an SPE gel from at least one month of commencement. Unless the laboratory is aware of possible SPE/IF-interfering monoclonal immunotherapies, and when patients are starting and ceasing them, there is a risk that the appearance of a second band, caused by the presence of the monoclonal therapy, will be reported and quantified by the laboratory as a second paraprotein band. This may result in a patient failing to meet the IMWG criteria for CR, especially where daratumumab co-migrates with patients’ paraprotein. Commercially available assays such as Hydrashift 2/4 Daratumumab may be a useful tool in confirming daratumumab interference 5 and allowing a more accurate assessment of remission status in these patients.
Daratumumab was interim-NICE approved via the Cancer Drugs Fund in January 2018 as a fourth line monotherapy for MM and was approved as a combination therapy alongside bortezomib and dexamethasone for first relapse in March 2019, so we expect to see a marked increase in the number of patients on this therapy, especially as daratumumab is usually given until refractoriness. New immunotherapies are emerging such as isatuximab, which has already been shown to interfere in SPE. 6 Therefore, it is imperative when interpreting electrophoresis results to be mindful of these interferences, ensure the laboratory are aware of which patients are treated with monoclonals and to consider the possible impact on SPE reporting. Traces of daratumumab may stay in the circulation for six months post cessation. This may also potentially be the case for new immuno-conjugated monoclonals. Effective communication between pharmacy, clinical trials, haematology and the laboratory is essential for the development of clinically valuable, accurate reports for these patients.
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JC: clinical research with daratumumab, chair/speaker panel for Janssen/Johnson and Johnson.
Other authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Guarantor
ST.
Contributorship
ST conducted the study and drafted the article, JR performed the original electrophoresis, SW and TG identified suitable patients, SK and JC provided clinical input and PJM devised the project and provided a supervisory role. All authors reviewed the article and approved the final version of the article.
