Abstract
Background
SARS-CoV-2 serological testing has seen extensive academic and clinical use from investigating correlates of immunity to seroprevalence, convalescent plasma and vaccine trials. Interpretation of these studies will depend on robust validation of the longitudinal sensitivities of these assays, especially in the context of mild disease which makes up the majority of the Coronavirus Disease 2019 (COVID-19) caseload.
Methods
Hospital staff (n = 94) returning to work following polymerase chain reaction confirmed COVID-19 were offered antibody testing to assist with laboratory verification. Initial specimens were collected at median 29 days post-symptom onset and run on the Roche, Abbott, Siemens and DiaSorin platforms. Re-sampling occurred at median 142 days from a subset of the initial cohort (n = 62) that had volunteered to provide further serum samples to assist in longitudinal sensitivity analysis. Samples that were not run across all four platforms were excluded from analysis.
Results
Comparative sensitivity analysis was conducted on 89/94 of the initial specimens and 55/62 of the repeat specimens. Sensitivity at initial sampling ranged from 78 to 87% across platforms. At re-sampling, sensitivities were: 100% (Roche), 45% (Abbott), 100% (Siemens), and 80% (DiaSorin). Paired analysis using the longitudinal cohort (n = 55) demonstrated stable or increasing median assay values on three platforms, with a clear reduction seen only on the Abbott platform (4.78 to 1.34) with corresponding sensitivity drop-off (81.8% to 45.4%).
Conclusion
The Abbott assay demonstrated sensitivity drop-off and decrease in median assay signal below detection threshold at four to five months. This has implications on the interpretation and design of future studies.
Keywords
Introduction
SARS-CoV-2 serological tests have seen growing interest in the immunological responses following infection with concern that antibodies may wane rapidly, especially after mild illness.1,2 This has wider implications with up to 80% of those with Coronavirus Disease 2019 (COVID-19) experiencing mild to moderate disease. 3
We present a longitudinal sensitivity analysis of four different SARS-CoV-2 serological assays in convalescent healthcare workers following predominantly mild COVID-19.
Methods
Serum specimens
Unvaccinated staff following polymerase chain reaction confirmed COVID-19 at Manchester University NHS Foundation Trust (MFT) were offered antibody testing and consented in line with UK National External Quality Assessment Services (NEQAS) to assist in verification and quality control. This was undertaken in collaboration with Public Health England and UK NEQAS, with both receiving an aliquot of serum samples for their use. Ethical approval requirements were waived by Medical Directors at MFT and R&I on the basis of UK NEQAS compliance for laboratory test verification.
At initial sampling, 94 volunteers were bled at median 29 days post-symptom onset (interquartile range, IQR 24–32 days). Of these, 92 were classed as mild (not requiring hospital admission), 1 as moderate (≥1 night in hospital) and 1 as severe (high dependency or intensive care).
To validate sensitivity later in the convalescent phase, 62 of the initial 94 staff members volunteered to be re-bled at median 142 days post-symptom onset (IQR 136–151 days). Of these, 60 were classed as mild, 1 as moderate and 1 as severe.
Assays
Specimens were analysed on four SARS-CoV-2 serological chemiluminescent/electrochemiluminescent immunoassays intended for qualitative reporting only: Roche Cobas e 801 total IgM/IgG nucleocapsid antibody assay (MFT, threshold for positivity ≥1.0 COI). Abbott SARS-CoV2 Architect IgG nucleocapsid antibody assay (Pennine Acute Hospitals NHS Trust, threshold for positivity ≥1.40 S/C). Siemens SARS-CoV-2 Centaur total IgM/IgG spike S1 RBD antibody assay (Salford Royal NHS Foundation Trust, threshold for positivity ≥1.0 Index). DiaSorin LIAISON IgG S1 and S2 spike assay (Cumberland Infirmary, threshold for positivity >12 (equivocal) (Note: Equivocal threshold used as classification for positivity) and ≥15 (positive) AU/mL). (Note: Equivocal threshold used as classification for positivity).
All assay kits were verified and put into routine clinical use as per local protocols, with the initial samples contributing to verification. All aliquots were separated promptly and frozen at −80°C until analysis.
Statistical software
Analysis was conducted using Microsoft Excel 2016.
Results
Due to sampling errors, a small number of samples were not analysed on all four platforms and therefore excluded. One sample was taken only 13 days post-symptom onset and excluded. Comparative sensitivity analysis was thus performed on 89/94 of the initial cohort and 55/62 of the re-sampled cohort. The median days post-onset for these cohorts were 29 and 142, respectively. Initial sensitivity ranged from 78 to 87% across all platforms and was similar when considering only staff from whom a repeat sample was obtained (Table 1). Sensitivity at re-sampling increased to 100% for the Roche and Siemens assays remained similar for the DiaSorin assay, and were markedly lower for the Abbott assay (Table 1).
SARS-CoV-2 antibody immunoassay sensitivity following predominantly mild illness.
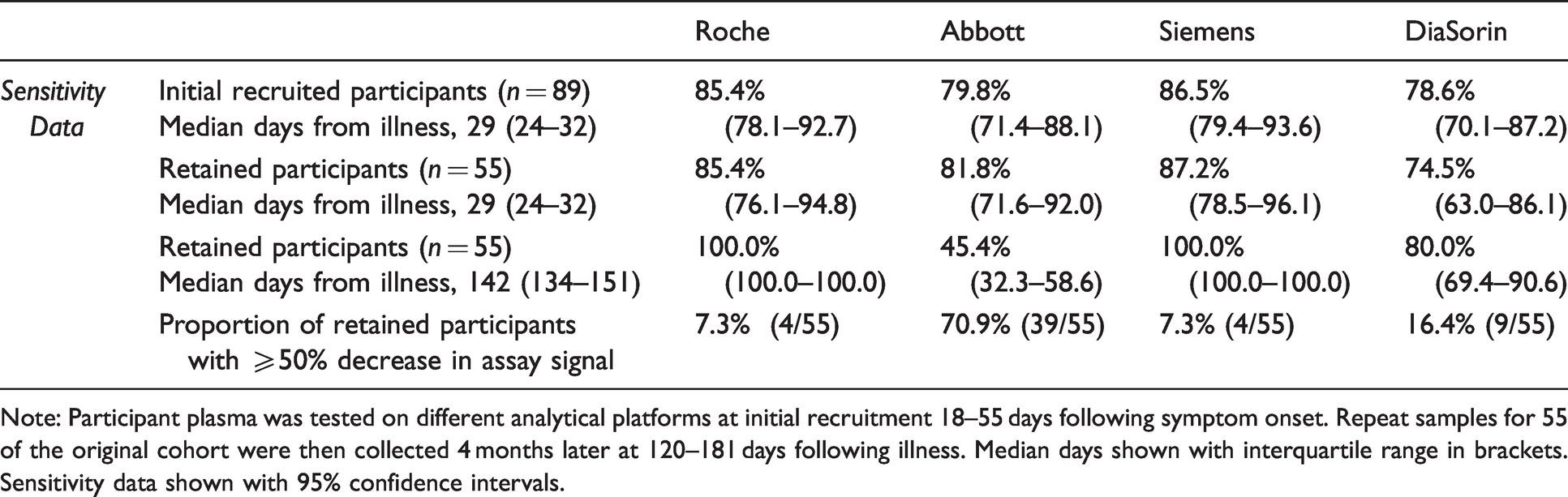
Note: Participant plasma was tested on different analytical platforms at initial recruitment 18–55 days following symptom onset. Repeat samples for 55 of the original cohort were then collected 4 months later at 120–181 days following illness. Median days shown with interquartile range in brackets. Sensitivity data shown with 95% confidence intervals.
Comparison of paired serum samples from the longitudinal cohort (Figure 1) showed an interval increase in the median values for the Roche (15.3 and 24.6) and Siemens (3.0 and 7.1) assays, a similar overall signal for the DiaSorin assay (30.0 and 32.6), and a clear reduction for the Abbott assay (4.78 and 1.34). Regardless, for each assay, there were some participants with large decreases in assay signal, although this was most pronounced for the Abbott (Table 1.)
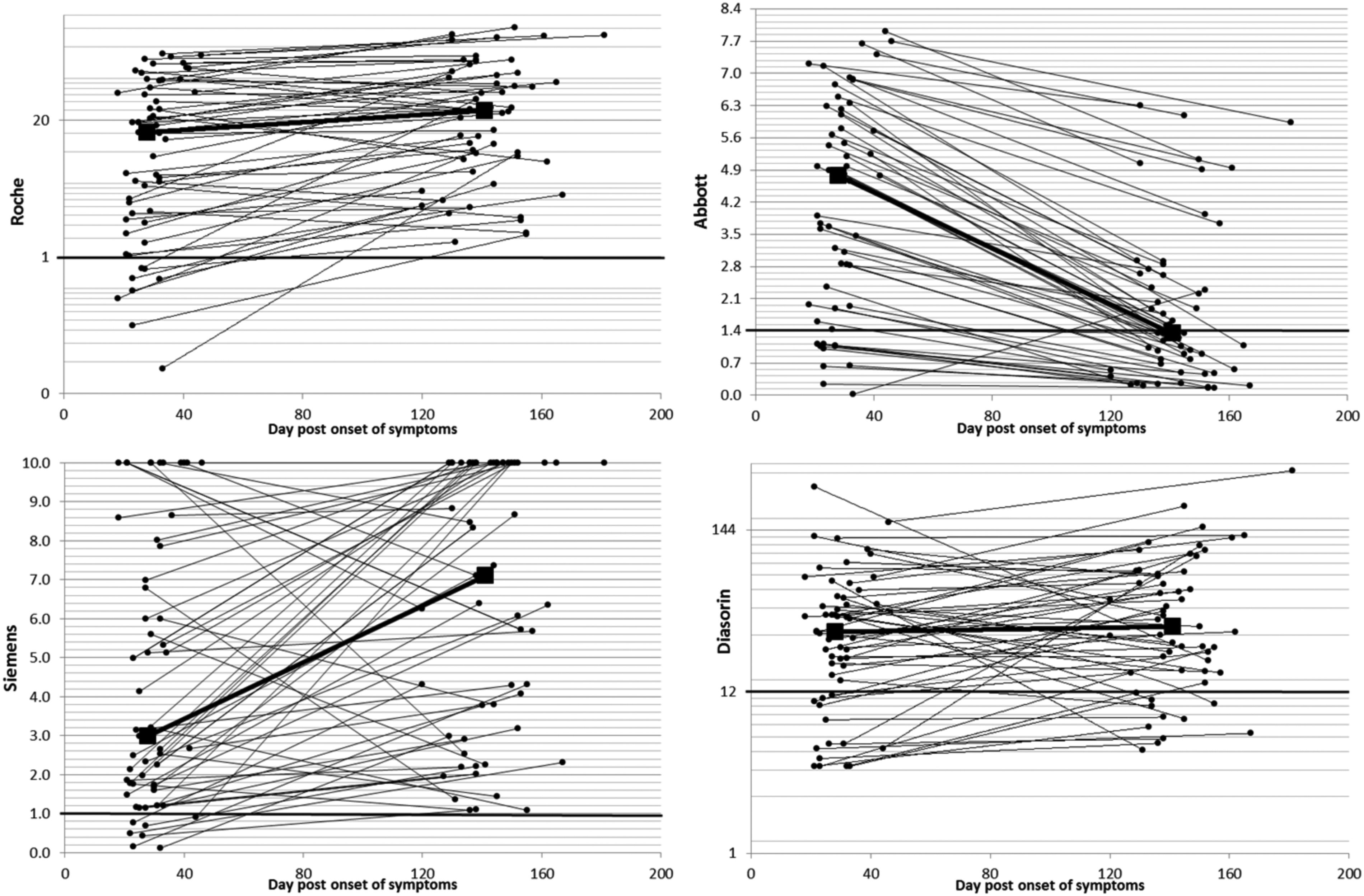
Comparison of paired values obtained at initial and repeat sampling by assay. Bold line denotes assay threshold for positivity. Median values are also given in by bold line and squares. Note logarithmic scale for Roche and DiaSorin assays. Mean assay signal ratios between first and last samples with 95% confidence intervals as follows: Roche 2.16 (0.79–3.52), Abbott 0.38 (−0.91 – 1.67), Siemens 1.82 (0.51–3.13), DiaSorin 1.29 (−0.05 – 2.62).
Discussion
Validating the longitudinal sensitivity of SARS-CoV-2 antibody tests will be crucial for the design and interpretation of future studies. This is vital in the context of mild disease which makes up the majority of the population targeted by NHS England’s pillar 3 of testing.
In our population of convalescent staff with predominantly mild COVID-19, there was a relative increase in assay signal and sensitivity at re-sampling for the Roche, Siemens and DiaSorin kits.
This suggests antibodies may be present for as long as four months following mild COVID-19. Whether this implies persisting humoral immunity depends on the future characterization of serological correlates. Nevertheless, this contrasts with studies demonstrating detectable SARS-CoV-2 antibody concentrations falling rapidly in individuals following mild disease. The REACT-2 study, 4 notably, showed evidence of declining seroprevalence in the UK population over three to four months.
We note REACT-2 utilized different methodologies with point-of-care testing and random, non-sequential sampling versus our longitudinal cohort. Indeed, between rounds 1 and 3 of REACT-2, there were demonstrated decreases in the proportion of volunteers reporting screening symptoms or confirmed COVID-19 contacts (−20.0% and −26.4%, respectively).
Additionally, REACT-2 reported that this decrease in seroprevalence was not seen in healthcare workers (+3.45%, [−5.7 + 12.7]), a finding consistent with our analysis of convalescent hospital staff. It may be plausible that low-level exposure to SARS-CoV-2 in the hospital setting may contribute to persistent antibody concentrations seen in our cohort.
Interestingly, this was not seen on the Abbott platform which demonstrated declining assay signal with sensitivity of 45% at re-sampling. This relative decrease seen on the Abbott platform has been described in other longitudinal cohorts.5,6 Differences in assay format and design beyond the main assay target may account for this, as the Roche nucleocapsid assay did not show the same deterioration. The same samples were later re-assayed on another Abbott platform in an independent laboratory with similar results, although sample integrity may have been compromised with time. An equivocal range was not available for Abbott at the time of sampling. Utilization of the equivocal threshold now proposed by Abbott of 0.49 S/C mitigates this relative decrease in sensitivity but does not prevent it (93% on initial sampling, 78% on re-sampling).
Conclusion
Antibodies following predominantly mild COVID-19 infection appear to persist for as long as four to five months and remain above the positivity threshold by three of the four platforms analysed (Roche, Siemens, DiaSorin). A relative decrease in assay signal and sensitivity was seen only by the Abbott platform. These tests have been deployed for surveillance of exposure, so any drop-off in sensitivity has real-world implications in managing the pandemic.
Footnotes
Acknowledgements
We thank Ghazala Barlas, Angela Bedford, Melissa Blaylock, Emma Cowling, Siobhan Easton, Emily Gallimore, Caroline Guy, Alexia Harrison, Andrew Hodgkinson, Martin Lewis, Claire Manfredoni, Gary McCann, James Osborne, Darren Powell, Garren Scott, Laura Singh, Parmesher Singh and Lloyd McLeggon Watkinson for contributions to assay set-up and processing of samples.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by local NHS departmental budgets.
Ethical approval
Waived by local medical director and R&I on the basis of compliance with NEQAS/RCPath guidance for laboratory test verification purposes.
Guarantor
TG.
Contributorship
TG and CRE designed the study, obtained local approval, recruited volunteers and analysed initial data. CC, DD, AH and JS contributed essential reagents or tools, liaised with their individual local departments to process samples and contributed to further data analysis. MS drafted and wrote the manuscript. All authors provided critical revisions, reviewed the manuscript and approved the final version of the manuscript.
