Abstract
Background
This study aimed to establish anti-Mullerian hormone age-specific reference intervals and determine the correlation between the anti-Mullerian hormone concentration and age, body mass index and concentrations of follicle-stimulating hormones and luteinizing hormone in healthy Chinese girls.
Methods
Serum anti-Mullerian hormone concentrations of 1702 healthy girls (0−12 years), recruited between March 2018 and December 2019, were determined using the Beckman Access 2 automated chemiluminescence immunoassay. Single-year-specific medians of anti-Mullerian hormone and effects of age, body mass index, follicle-stimulating hormone and luteinizing hormone on anti-Mullerian hormone concentration were analysed.
Results
The anti-Mullerian hormone median level continued increasing from birth, reached its peak at age 9 at 4.45 ng/mL (interquartile range [IQR] 2.58–6.90) and then gradually decreased. At age 12, the median reached 1.98 ng/mL (IQR 1.05–3.46). Age-specific reference intervals for anti-Mullerian hormone were established in healthy Chinese girls aged 0–12 years. Anti-Mullerian hormone concentrations showed a moderately positive correlation with age (r = 0.33, P < 0.001). In contrast, follicle-stimulating hormone (r = –0.29, P < 0.001) concentrations were weakly negatively correlated with the serum anti-Mullerian hormone concentration.
Conclusion
We established single-year-specific reference intervals for anti-Mullerian hormone in Chinese girls using the Beckman chemiluminescent platform. This reference range can help clinicians accurately understand anti-Mullerian hormone secretion in healthy girls and promote its clinical use.
Introduction
The anti-Müllerian hormone (AMH) is a 140-kilodalton dimeric glycoprotein hormone belonging to the transforming growth factor-beta superfamily. 1 AMH is expressed by ovarian granulosa cells and mainly persists in preantral and early antral follicles.2–4 In girls, AMH is extremely low after birth, and then it gradually increases during infancy and childhood. 5 The serum AMH concentrations show strong and positive correlations with the antral follicle count, and it declines steadily with advancing age. 6 AMH is therefore considered an accurate marker in the evaluation of the ovarian follicular reserve.7,8 In addition, AMH concentrations vary to a less extent across different menstrual cycles as well as within one menstrual cycle compared with other ovarian-activity hormones, such as the follicle-stimulating hormone (FSH).9,10 The serum concentration of AMH is used to evaluate premature ovarian insufficiency 11 in girls with Turner Syndrome who are suffering from an accelerated loss of follicles and who have extremely low AMH concentrations. 12 On the contrary, the serum concentration of AMH is two to three times higher in adolescents with polycystic ovary syndrome (PCOS) than in healthy adolescents. 13 In addition, studies have demonstrated that the determination of AMH concentrations assists in assessing the postchemotherapy ovarian function of girls with cancer who were treated with gonadotoxic chemotherapy. 14 Furthermore, AMH is an important indicator of precocious puberty and delays in the onset of puberty in girls.15,16
There is a need for a precise and reliable, fully automated AMH assay. In the past, the Gen II enzyme-linked immunosorbent assay (ELISA) AMH assays were time consuming and would be more prone to the interoperator’s bias within the assay itself. 17 Beckman Coulter has developed a fully automated chemiluminescence immunoassay for detecting AMH concentrations. This fully automated AMH assay that provides more reproducible and accurate results is a more sensitive and specific chemiluminescence method for detecting AMH than the Gen II ELISA method. 18 The automatic platform provides the technical support for the detection and extensive application of AMH.
Accurate clinical diagnoses for AMH-related diseases by clinicians have relied on AMH reference intervals. In recent years, AMH reference intervals have been established in adult females in western and Asian populations, which are now widely applied in clinics. 19 However, so far, age-specific reference ranges for AMH concentrations based on large populations of healthy girls are lacking, thus limiting their extensive clinical applicability in girls. Hence, it is necessary to establish AMH reference intervals and analyse the changes in the serum concentrations of AMH in healthy girls from birth to puberty. The aim of this study was to establish AMH single-year-specific reference intervals in a population of healthy Chinese girls aged 0–12 years using the Beckman Coulter Access 2 immunoassay. Furthermore, we evaluated the correlation between AMH concentration and age, body mass index (BMI), FSH, luteinizing hormone (LH) and the LH/FSH ratio in healthy girls.
Materials and methods
Study population
From March 2018 to December 2019, the study population was recruited from the Eugenic Genetics Laboratory of Wuhan Children’s Hospital and five randomly selected schools in China. According to CLSI C28-A3 guidelines, all subjects underwent a standard physical and medical examination. The exclusion criteria were as follows. Girls with Turner syndrome, a suspected disorder of sexual development, PCOS, premature ovarian insufficiency, endocrine disease, inherited metabolic disease, liver malfunction, kidney malfunction, kidney disease, circulatory system diseases, acute or chronic infections, prescribed or non-prescribed medications and girls who had undergone surgery were excluded from the study. In total, 1702 healthy girls aged between 0 and 12 years (median age, 6.4 years) were included in the final analysis. This study was approved by the ethics committee of Wuhan Children’s Hospital. The guardians of all the recruited volunteers provided informed consent for the analysis of the data for publication. The data were supplied anonymously. Table 1 shows the baseline characteristics of the study population.
Descriptive information of biomarkers in a population-based cohort of healthy Chinese girls aged 0–12 years, at 1-year intervals.
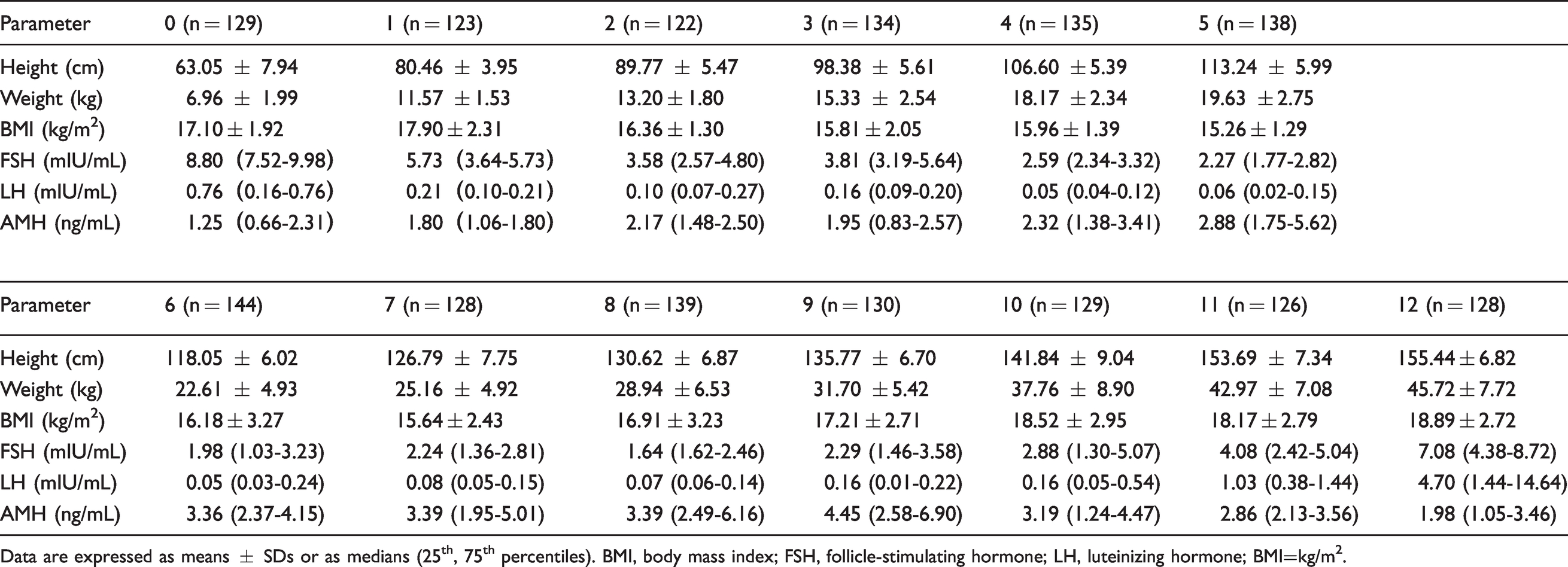
Data are expressed as means ± SDs or as medians (25th, 75th percentiles). BMI, body mass index; FSH, follicle-stimulating hormone; LH, luteinizing hormone; BMI=kg/m2.
Samples collection
Venous blood samples (5 mL) were collected in a separation gel vacutainer (BD Biosciences, New Jersey, USA) between 8 a.m. to 10 a.m. Blood samples were immediately placed on ice and transferred to the Eugenic Genetics Laboratory of Wuhan Children’s Hospital. The serum was separated by centrifugation from the blood specimen at 3500 r/min for 15 min. The centrifuged samples were stored at −80°C before analysis.
Body mass was measured by a trained research nurse to the nearest 0.1 kg by using calibrated electronic scales (Seca, Birmingham, UK), and height was measured by a trained research nurse to the nearest 0.1 cm using a stadiometer (Seca, Birmingham, UK). The BMI was calculated as the ratio of body weight to the square of body height (kg/m2).
Laboratory analyses
The concentrations of AMH, LH and FSH were assessed using a completely automated immunoassay on an Access 2 immunoassay analyser (Beckman Coulter, Villepinte, France) with corresponding reagents and calibrators at the Eugenic Genetics Laboratory of Wuhan Children’s Hospital. All these tests were performed according to standard operating procedures recommended by Clinical and Laboratory Standards Institute. To monitor the precision of the instrument, quality controls (QCs) were used before the analyses. The assay was evaluated for both intra- and inter-assay precision. To estimate intra-assay CV, three known QC materials with AMH concentrations of 1.0, 5.0 and 15.0 ng/mL were measured 10 times in a single assay. To evaluate inter-assay CV, three same concentrations AMH (AMH concentrations of 1.0, 5.0 and 15.0 ng/mL) were measured for 10 consecutive days. The mean, standard deviation (SD) and CV were calculated. Intra- and inter-assay coefficients of variation with serum AMH were 1.58−2.60% and 2.07−3.27%, respectively. Any sample that can be accurately measured within the lowest limit of quantitation is 0.02 ng/mL.
Statistical analyses
All data processing was performed using R for Windows, version 3.4.3 (R Foundation for Statistical Computing, China). Data distributions were evaluated using the Kolmogorov-Smirnov test. The data of normal distribution are expressed as the means ± SD, while non-normal distributions are expressed as medians with the 25th to 75th percentiles. Reference ranges were expressed between the 2.5th and 97.5th percentiles. Moreover, 95% confidence intervals (CIs) were computed using a non-parametrical percentile method. Spearman correlation coefficients were used to assess correlations between serum AMH concentrations and age, height, weight, BMI, LH/FSH ratio and the concentrations of FSH and LH. P < 0.05 was considered to be statistically significant.
Results
General clinical data of the subjects
The clinical and laboratory characteristics of the sampled girls are summarized in Table 1. The mean ± SD values of BMI of the study population were 16.6 ± 2.59 kg/m2. Medians with the 25th to 75th percentiles of the serum AMH, FSH and LH concentrations were 2.64 (1.60–3.9) ng/mL, 2.830 (1.94–4.452) mIU/mL and 0.09 (0.05–0.260) mIU/mL, respectively. The concentrations of serum FSH, LH and AMH were non-normally distributed in the study population. Height, weight and BMI are normally distributed. There were significant differences in height, weight and BMI among single-year-specific age groups using analysis of variance (P < 0.001). The Kruskal-Wallis test revealed significant differences in the concentrations of FSH, LH and AMH among single-year-specific ages (P < 0.001). General information and biomarkers of the sample are shown in Table 1.
Changes in the serum AMH, FSH and LH concentrations and the LH/FSH ratio in healthy girls
The changes in the serum AMH, the FSH, the LH concentrations and the LH/FSH ratio in the sampled girls are shown in Figure 1, along with median values and interquartile ranges. The concentrations of the serum AMH increased after birth, reached its peak at the age of 9 at 4.45 ng/mL, (IQR 2.58–6.90), and then began to decrease. At the age of 12, the median level of AMH is 1.98 ng/mL (IQR 1.05–3.46). In contrast, serum LH concentrations and the LH/FSH ratio in healthy girls were negligible before the age of 9, and from the age of 10 onwards, they gradually increased. Serum FSH concentrations started to decline from birth up to the age of 9, and from the age of 10 onwards, it increased.
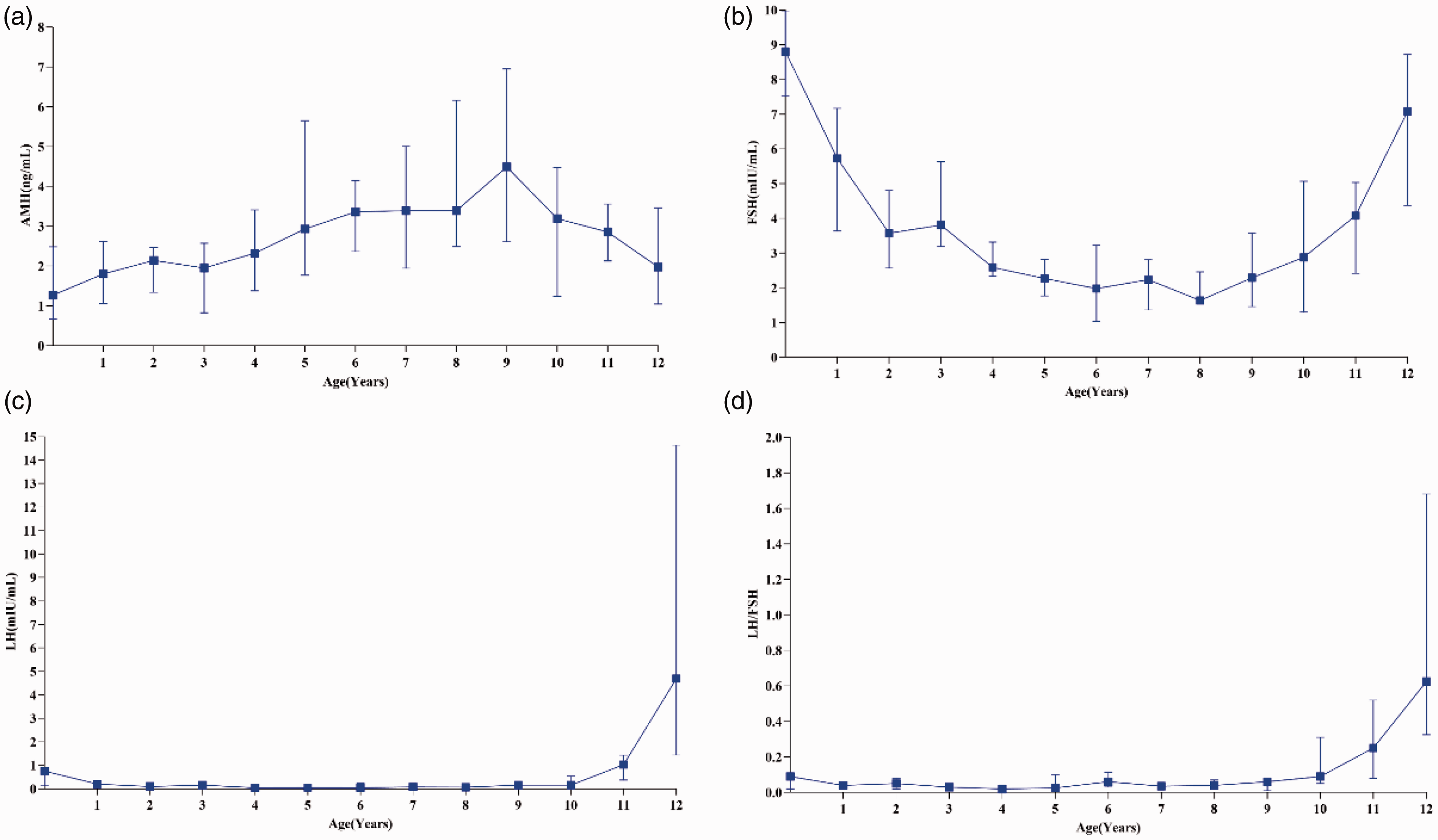
Age-specific serum AMH, FSH, LH and LH/FSH concentrations in healthy girls aged 0–12 years, at one-year intervals.
Reference intervals for healthy girls
The Kolmogorov-Smirnov test revealed that the serum AMH concentrations were non-normally distributed in healthy girls (P < 0.05). The age-specific AMH reference intervals for healthy girls determined by using the Beckman Coulter Access 2 immunoassay systems were presented as 2.5th and 97.5th percentiles, and the 95% CIs were computed. The results are shown in Table 2.
Serum AMH levels in healthy girls of different age groups (ng/mL).
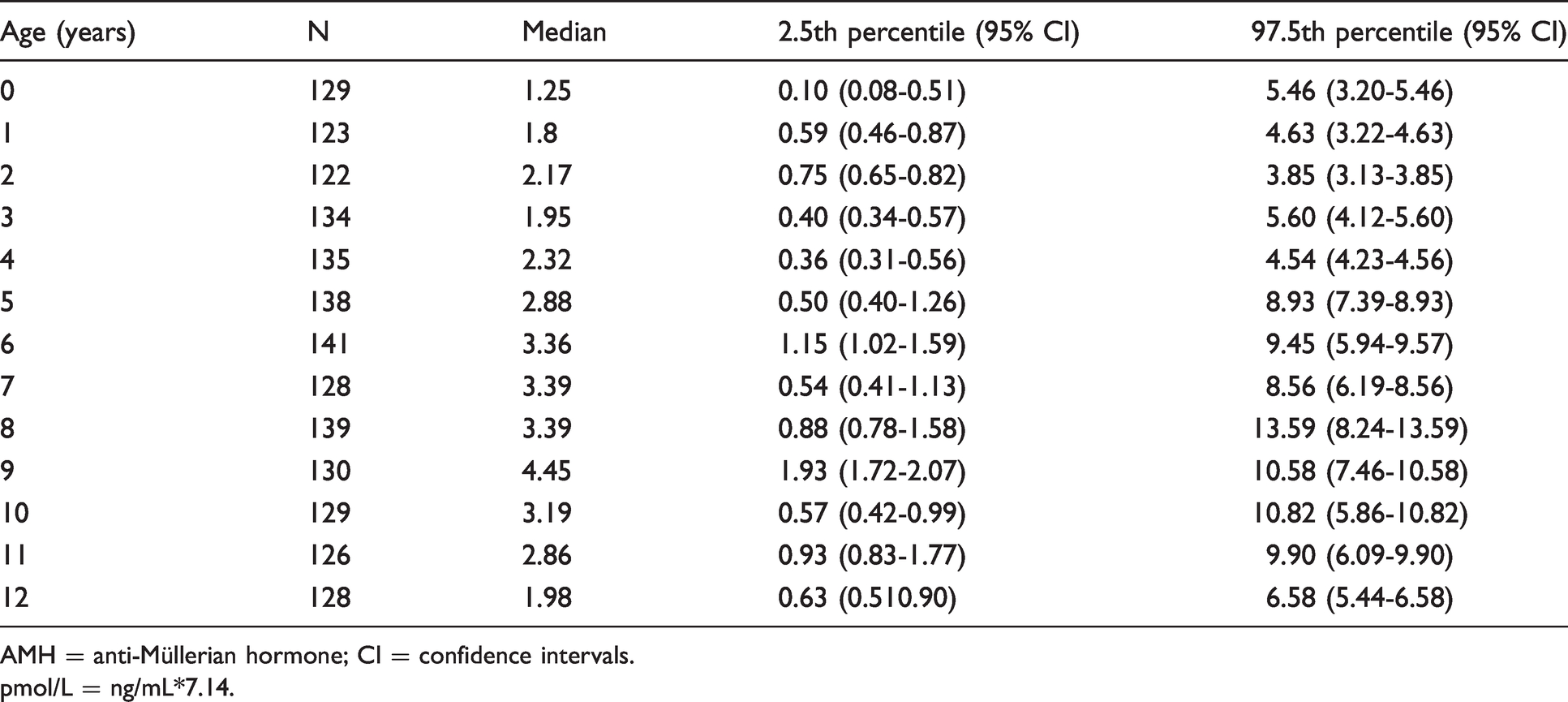
AMH = anti-Müllerian hormone; CI = confidence intervals.
pmol/L = ng/mL*7.14.
The correlation between AMH concentration and age, the BMI, the FSH, the LH concentrations and the LH/FSH ratio
The Spearmen correlation analysis showed AMH concentrations were moderately positively associated with age (r = 0.33, P < 0.001). In addition, Table 3 shows that FSH (r = –0.29, P < 0.001) concentrations were weakly negatively correlated with the serum AMH concentration. However, the BMI (r = –0.03, P =0.53), LH (r = –0.02, P =0.73) or the LH/FSH ratio (r = –0.01, P = 0.88) were not related to AMH concentration.
Correlations between AMH and age, height, weight, BMI, FSH, LH, and LH/FSH.
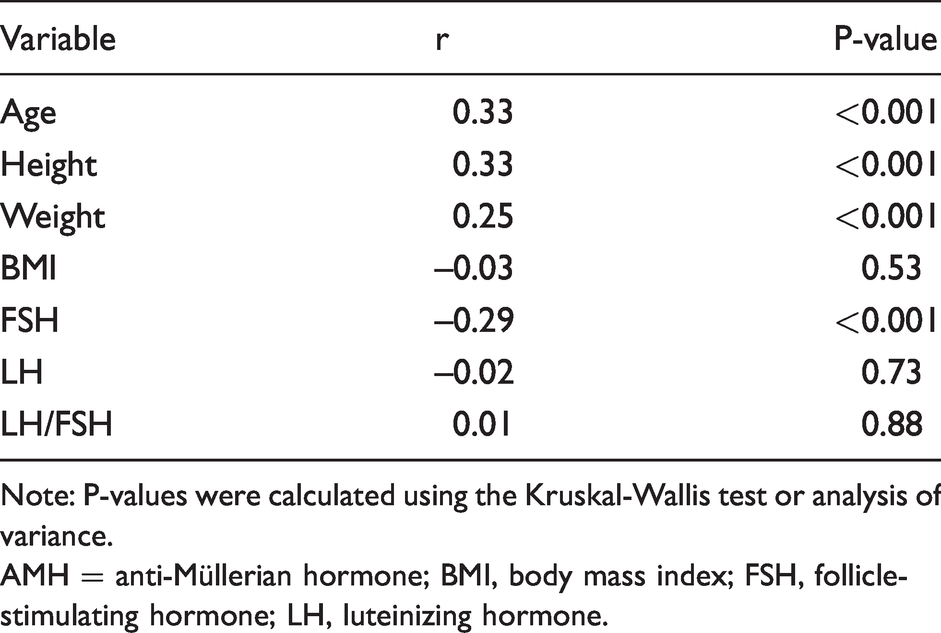
Note: P-values were calculated using the Kruskal-Wallis test or analysis of variance.AMH = anti-Müllerian hormone; BMI, body mass index; FSH, follicle-stimulating hormone; LH, luteinizing hormone.
Discussion
This is the largest study to date that established single-year-specific reference intervals for AMH in Chinese girls. Despite considerable attention to AMH reference intervals of adult females (e.g. Chinese women-specific reference intervals have been established), which are now widely applied, 20 the unavailability of unchallenged reference intervals of girls is a limitation. Jopling et al. reported female participants’ AMH reference ranges that were proposed based on data from only 235 female participants aged from one day to 18 years. The number of female participants in each of the seven age groups and at each stage was about 40, which does not meet the requirements of the minimum number, which is 120. 21 We established age-specific reference ranges for AMH based on large populations of healthy girls aged 0 to 12 years. Our reference intervals can be used to differentiate between abnormal and normal levels of AMH and can thus assist in clinical diagnoses and in the treatment of premature ovarian insufficiency in girls. In addition, Beckman Coulter developed a completely automated chemiluminescence immunoassay for detecting AMH, which is superior to the traditional ELISA-based method in terms of speed and specificity. 22 We conducted the current study to establish age-specific reference intervals for AMH concentrations using the Beckman Access 2 immunoassay system analyser. The technology platform created by Beckman Coulter effectively supports the extensive application of AMH in clinics.
Our cross-sectional study showed that the concentrations of the serum AMH continued increasing from birth, reached its peak at the age of 9 at which the concentrations of AMH increased by 24% from the age of seven. Our conclusion is consistent with a longitudinal study, showing a 20% increase in AMH concentrations between the ages of 7 and 9 as well as a cross-sectional study showing the doubling in AMH concentrations between the ages of 4 and 8 in girls.23,24 Lashen et al. speculated that the secretion of AMH increases steadily as girls approach the onset of puberty. 25 The increase in AMH concentrations during prepubertal childhood, therefore, supports the intriguing concept that increasing ovarian primordial follicle recruitment and follicle activity precede the onset of central puberty. 26 The concentrations of AMH gradually decreased from the age of 9 until the age of 12. Broekmans et al. argued that the reason for the reduction in circulating AMH concentrations is that the onset of puberty is characterized by the progression of small antral follicles to antral follicles in the ovary, which do not produce AMH. 27 However, we have reported that in boys, the concentration of AMH increased slightly after birth and decreased consistently from the age of 2 until the age of 12, and then remains low. 28 Evidently, the secretion of AMH in boys is significantly different from that in girls.
We found that AMH concentrations were moderately positively correlated with age (r = 0.33, P < 0.001) in healthy girls, which is similar to findings reported in other studies. 29 In addition, FSH (r = –0.29, P < 0.001) concentrations were weakly negatively correlated with the serum AMH concentration. Fleming et al. argued that FSH might indirectly affect circulating AMH concentrations by inducing follicle growth, which reduces the number of AMH-producing follicles. 30 Most previous studies on the correlation between AMH concentration and the BMI focused on infertile women. Tehrani et al. have reported that AMH concentrations correlated negatively with BMI in females aged 18–50 years, whereas other studies have reported that AMH concentrations were independent of BMI.31,32 Our study showed that the BMI (r = –0.03, P =0.53) was not related to the AMH concentration in girls aged 0–12 years.
This study had several limitations. First, we did not study whether the single-year-specific reference intervals of AMH in Chinese girls that were established based on the Beckman Access 2 automated chemiluminescence immunoassay can be applied to the Roche Elecsys/Cobas electrochemiluminescence immunoassay system analyser or to the traditional ELISA method. Second, in this study, we only focused on girls aged 0–12 years. We have plans to carry out further studies on the concentrations of AMH in girls aged 13–18 years and to establish the paediatric female reference range classified on the basis of Tanner stages.
Conclusions
This is the first study to establish single-year-specific reference intervals for AMH concentrations in Chinese girls aged 0 to 12 years using the Beckman chemiluminescent platform system. The concentrations of the serum AMH increased after birth up to the age of 9 and then began to decrease until the age of 12. Furthermore, the serum AMH concentrations showing positive correlation with age were negatively correlated with the FSH concentrations. However, BMI, LH and the LH/FSH ratio are not related to AMH concentration. Our data are useful to understand the changes in AMH secretion during the normal growth of a girl and may provide a basis for the clinical use of AMH.
Footnotes
Acknowledgements
We express our gratitude to the Beckman Coulter and Sinopharm Hubei Medical Device Co., Ltd for their assistance and to all persons who participated in this population-based study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Eugenic Genetics Laboratory, Wuhan Children’s Hospital, Tongji Medical College, Huazhong University of Science and Technology.
Ethical approval
This study was approved by the Ethics Committee of Wuhan Children’s Hospital: Number: 2020R094.
Guarantor
JW.
Contributorship
JW analysed the data, drafted the article. TY revised the article. XZ, QC, SG and LJ have collected samples for the reference interval study. HY and XH contributed to study design. All authors reviewed and edited the article and approved the final version of the article.
