Abstract
Aims
To evaluate the analytical performance of a fully automatic iFlash anti-Müllerian hormone immunoassay (YHLO Biotech) and establish age-specific reference range for anti-Müllerian hormone in Chinese women based on a multicentre population study.
Design and method
iFlash anti-Müllerian hormone assay was evaluated for sensitivity, imprecision, serial dilution for linearity, impact of sample type, storage and stability. Method comparison of iFlash anti-Müllerian hormone with Elecsys anti-Müllerian hormone was studied. Reference intervals in healthy females were established for iFlash anti-Müllerian hormone.
Results
The limit of blank and limit of detection were below 0.001 ng/mL and 0.02 ng/mL, respectively. Assay sensitivity defined as limit of quantitation was 0.08 ng/mL. The assay imprecision was similar at low and high concentration being 3.1% and 3.2%, respectively. The linearity was observed to be between 0.02 ng/mL and 27.22 ng/mL. The stability of AMH was most at −80°C and up to seven days at 4°C, −20°C. No significant difference was observed for anti-Müllerian hormone among different sample types. An excellent agreement of anti-Müllerian hormone concentration was found in 180 samples analysed by iFlash and Roche; the correlation coefficient was 0.975 and regression slope of 1.009. The AMH reference intervals for Chinese women aged between the ages of 20 and 49 years with five-year intervals were 1.20–10.21 ng/mL, 1.14–9.17 ng/mL, 0.55–8.18 ng/mL, 0.25–7.02 ng/mL, 0.07–4.59 ng/mL and 0.01–2.11 ng/mL, respectively (1 ng/mL = 7.14 pmol/L).
Conclusion
The fully automated iFlash anti-Müllerian hormone immunoassay demonstrates excellent analytical performance. Consequently, the availability of iFlash anti-Müllerian hormone assay will represent a robust, fast, sensitive and precise immunoassay for the determination of anti-Müllerian hormone concentration.
Introduction
Anti-Müllerian hormone (AMH) is a member of the transforming growth factor-β superfamily. AMH is a 140 kDa homodimer glycoprotein which is composed of two identical 70 kDa monomer subunits linked by disulphide bridges. The synthesis of AMH in women comes from follicular granulosa cells in the early stage of follicular development. AMH is the key hormone to regulate follicular maturation. It reflects the number of ovarian antral follicles and preantral follicles (ovarian reserves) and is also helpful in evaluating ovarian function. 1
Detection of AMH concentration with fully automated immunoassays has provided more reproducible results with much higher sensitivity than the traditional manual ELISA assays. Manual ELISA techniques are time consuming and labour intensive, and their results are influenced by the operator’s handling practices. Therefore, the need of the hour is an automated AMH assay which meets the requirements of reproducibility and accurate results. Three such automatic AMH immunoassays have been recently released in the market: (1) Elecsys AMH manufactured by Roche Diagnostics (Indiana, USA); (2) Access AMH manufactured by Beckman Coulter, Inc. (California, USA) and (3) iFlash AMH manufactured by YHLO Biotech (Shenzhen, China).2–5 We examined the analytical performance of the newly released iFlash AMH for its sensitivity, imprecision, linearity, method comparison, sample stability, and most importantly, the establishment of reference range in healthy adult Chinese women.
Materials and methods
Assay principle
The iFlash AMH assay is a one-step sandwich chemiluminescence immunoassay performed on the iFlash 3000 immunoassay analyzer (manufactured by YHLO Biotech, Shenzhen, China). It uses two mouse monoclonal antibodies directed at the pro- and mature-regions on the AMH molecule, respectively. The assay detects both the cleaved non-covalent complex form of AMH and the uncleaved proAMH. Fifty microlitres of a sample are added to the paramagnetic particles coated with the mouse monoclonal antibody (Clone No. 32) and the mouse monoclonal antibody (Clone No. 24) conjugated to acridinium ester. 6 After incubation and final wash, pre-trigger and trigger solutions are added to the reaction tubes to produce a light signal, which indicates the concentration of AMH in the sample. The AMH concentration is determined using a stored calibration curve. Total assay time is approximately 18 min.
Subjects
A total of 1089 healthy female subjects with age ranging from 20 to 60 years were enrolled in this study between January 2018 and December 2018 from: (1) Obstetrics & Gynecology Hospital of Fudan University (349 attendees), Shanghai, China; (2) The Third Xiangya Hospital of Central South University (464 attendees), Hunan, China; (3) SUN YAT-SEN Memorial Hospital (276 attendees), SUN YAT-SEN University, Guangzhou, China. Exclusion criteria were: (1) being treated with steroid hormones; (2) pregnancy; (3) diagnosed with endometriosis; (4) previous or current history of endocrine or metabolic disorders. This study was approved by the Research Ethics Committee of the participating hospitals. Written informed consent was obtained from all the subjects participating in this study.
Samples
Five millilitres of peripheral blood were collected from subjects and allowed to clot for 30 min before centrifuging at 1000
The sample type comparison experiment was conducted on 11 paired patient samples. Serum and plasma were collected with different types of sample tubes such as coagulant-free vacuum tube, heparin lithium anticoagulant tube, sodium citrate anticoagulant tube and EDTA anticoagulant tube for each subject. Serum and plasma were collected after centrifugation at 1000
Sensitivity
The limit of blank (LoB), limit of detection (LoD) and limit of quantitation (LoQ) of iFlash AMH assay were determined according to the Clinical and Laboratory Standards Institute (CLSI) guideline EP17-A2. 7 LoQ represents the lowest AMH concentration that can be reproducibly measured with an inter-assay CV of ≤20%.
Imprecision
Repeatability, intermediate imprecision and total imprecision (within-laboratory precision) were determined according to CLSI EP05‑A3, with two runs per day in duplicate each for 20 days (
Linearity
Linearity of the reportable range was evaluated according to the CLSI EP06-A. 9 Nine dilutions were prepared by mixing the high concentration sample exceeding the upper measuring range with the low concentration sample below the measuring range. Duplicates of each dilution step were subsequently measured on an iFlash 3000 analyzer. The observed values were plotted against the expected values using multiorder linear regression.
Sample stability
Serum samples covering AMH concentrations of 0.68–18.48 ng/mL from 11 donors were collected and aliquoted. These aliquots were stored at room temperature (20–25°C), 4°C, −20°C and −80°C prior to being analysed with iFlash AMH in duplicate at different time points from day 0 (immediately) to day 7. Average per cent difference from the baseline (day 0) was then calculated for each time point.
Method comparison
Method comparison was performed on the iFlash AMH assay and the Elecsys AMH assay on 180 serum samples (assayed within 2 h since collection) using YHLO Biotech iFlash3000 and Roche Cobas e601 instruments. Results were analysed using Passing-Bablok regression and Bland-Altman bias plot. The assays were performed according to the instruction of the each manufacturer.
Reference interval
Samples from 1089 healthy female subjects with age ranging between 20 years and 60 years with regular menstrual cycles (21–35 days) were collected (Obstetrics & Gynecology Hospital of Fudan University, Shanghai, China; The Third Xiangya Hospital of Central South University, Hunan, China; SUN YAT-SEN Memorial Hospital, SUN YAT-SEN University, Guangzhou, China). Subjects with a BMI exceeding 30 and/or receiving hormone replacement therapy or using combined hormonal contraceptives were excluded from the study. Furthermore, subjects having medication with steroid hormones, pregnancy, diagnosed endometriosis, known previous or current endocrine or metabolic disorders were also excluded from the study.
Statistical analysis
For data with normal distribution and homogeneity of variance, an independent-sample
Results
Sensitivity
The LoB and LoD were 0.001 ng/mL and 0.02 ng/mL, respectively, for the iFlash AMH assay. The LoQ was 0.08 ng/mL for the iFlash AMH assay.
Imprecision
Repeatability, intermediate precision and total precision of iFlash AMH assay for low concentration are 1.3%, 2.2% and 3.2%, respectively. Repeatability, intermediate precision and total precision of iFlash AMH assay for high concentration are 1.6%, 2.1% and 3.1%, respectively. Results of imprecision study are summarized in Table 1.
Comparison of repeatability, between-run precision and intermediate imprecision of AMH assay.
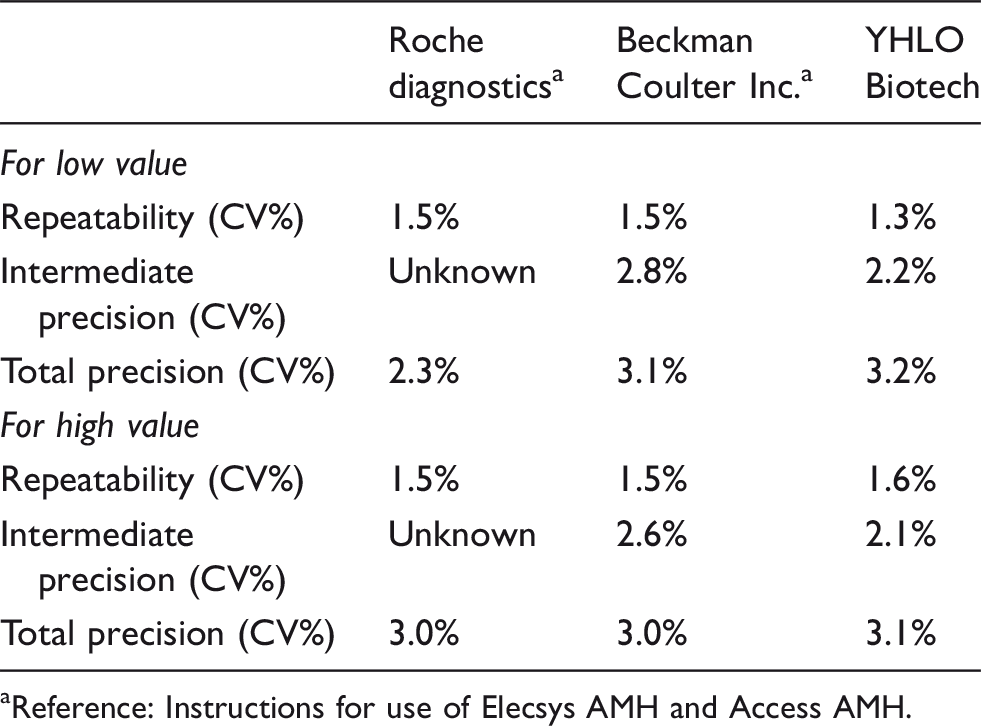
aReference: Instructions for use of Elecsys AMH and Access AMH.
Linearity
The results of multiple regression showed that the concentration was linear in the range of 0.02 to 27.22 ng/mL (Figure 1).
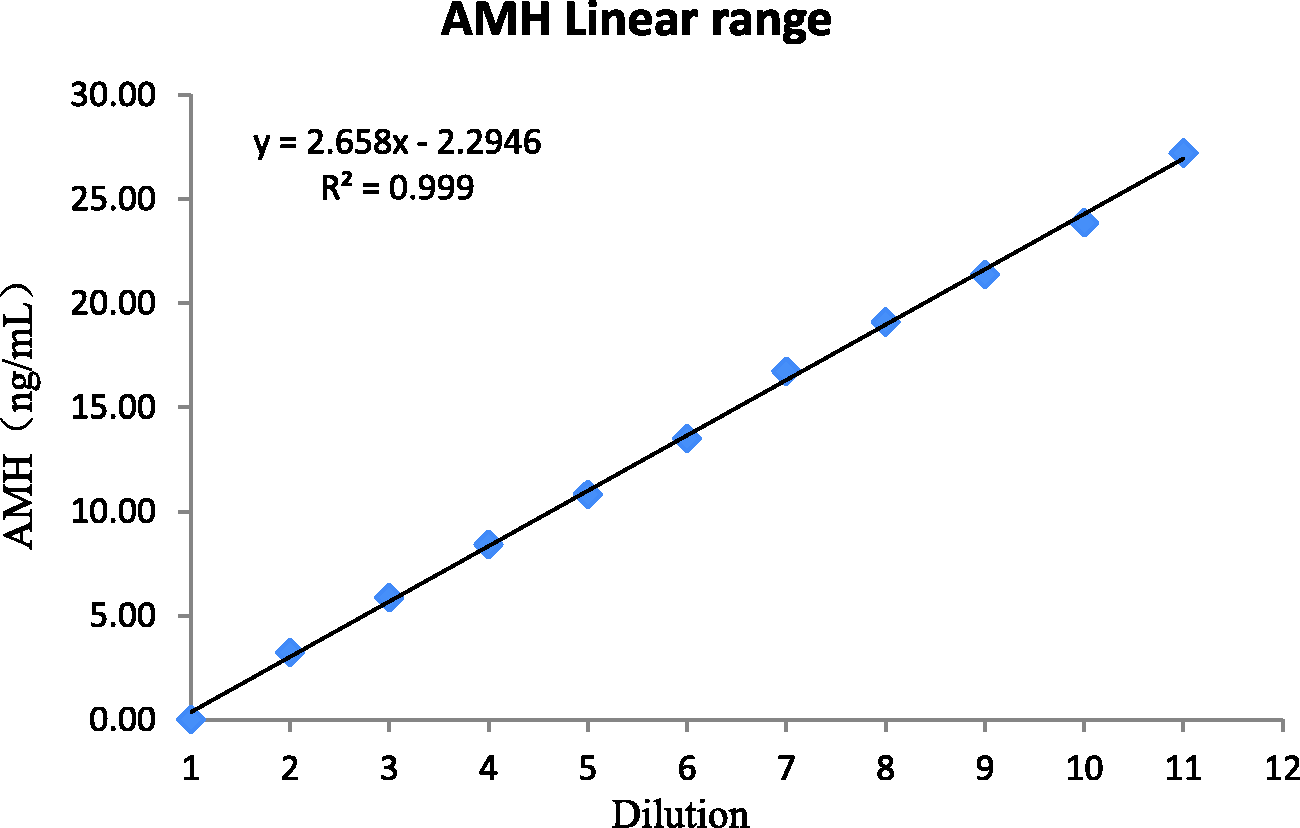
Linear range.
Sample stability
Table 2 and Figure 2 show the average per cent difference from the baseline (day 0) for each time point after the samples were stored at different conditions from 1 to 7 days. The detection of measured AMH concentration was stable up to seven days at 4°C, −20°C and −80°C, and three days at room temperature.
Per cent difference of measured AMH concentrations from day 0.
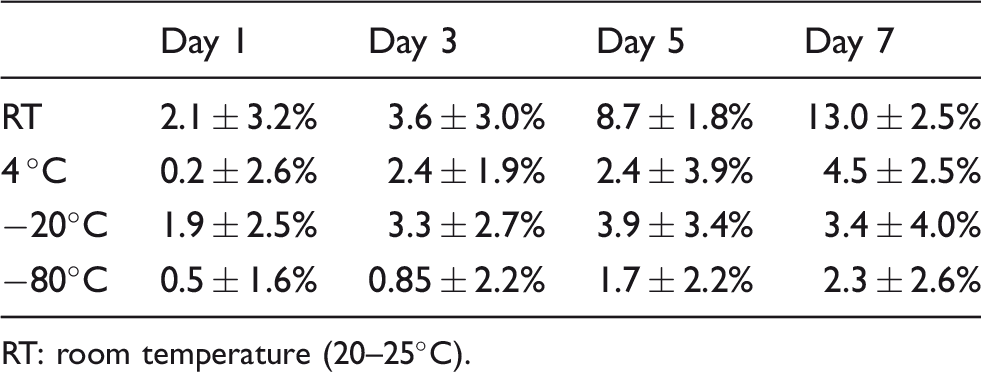
RT: room temperature (20–25°C).

Stability of AMH in samples under different storage conditions.
Comparison of sample types
Comparison between different specimen types showed a small variation. The correlation coefficient was 0.99 with a slope of 1 at 95% level of confidence. There was a linear correlation between the detection results of serum and plasma samples, and there was no significant deviation in the linearity. Comparison of serum vs. plasma (in different anticoagulants) is shown in Table 3.
Comparison between different specimen types.
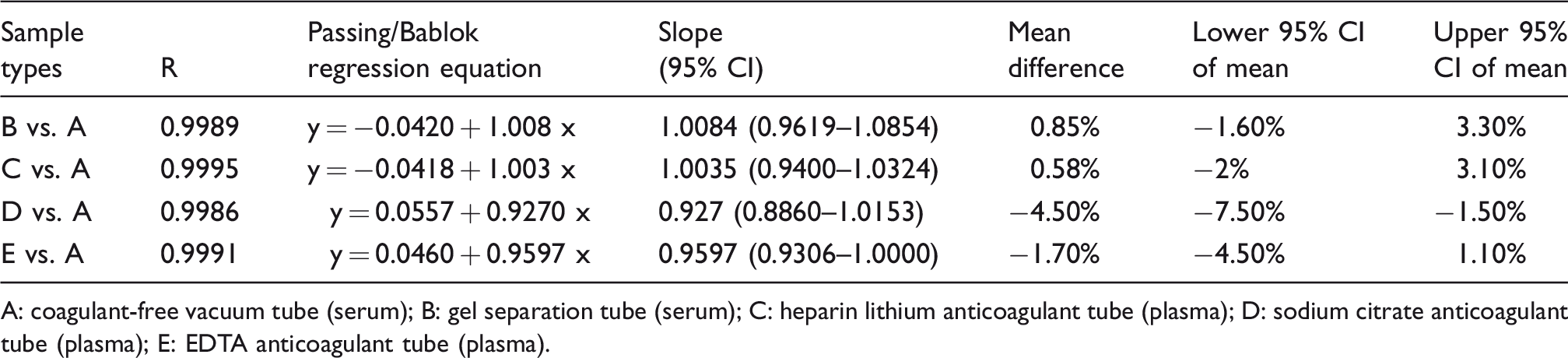
A: coagulant-free vacuum tube (serum); B: gel separation tube (serum); C: heparin lithium anticoagulant tube (plasma); D: sodium citrate anticoagulant tube (plasma); E: EDTA anticoagulant tube (plasma).
Method comparison
iFlash AMH and Roche Elecsys AMH displayed a strong correlation coefficient. The Passing-Bablok regression analysis resulted in a correlation coefficient of 0.975 with the slope values ranging from 0.974 to 1.046 (Figure 3(a)). iFlash 3000 and Roche Elecsys AMH assay were also evaluated by Bland-Altman with mean values of the two methods (x axis) plotted against the difference between the two methods (y axis).
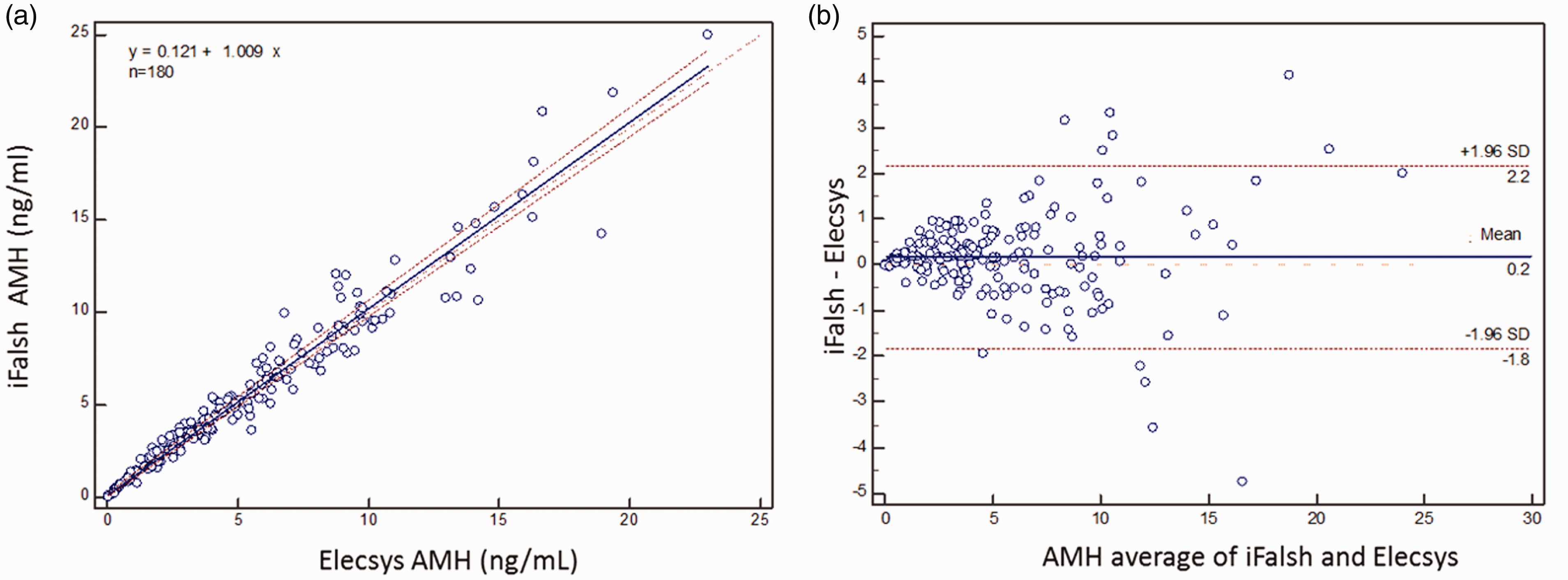
Passing-Bablok regression analysis and Bland-Altman bias evaluation of the association between AMH concentrations obtained from matched serum. (a) Solid lines represent the true regression line; dotted line represents the 95% confidence interval. (b) The upper and lower dashed lines indicate a 95% confidence interval (assuming a bias of 0). The middle-dotted line indicates the difference of mean value of 0 (Y=0). The solid line shows the mean difference.
Adult female reference interval
This multicentre study covered the population from three different provinces in China, and no regional difference in AMH concentrations was observed among each study centre. The median AMH values for healthy females decreased with increase in age from 3.98 ng/mL to 0.15 ng/mL for 20–24 years old to 45–49 years old, respectively. After 49 years of age, the median AMH value for females was 0.05 ng/mL. The reference interval results are displayed in Table 4. Analysis with quadratic power regression model also indicated the decline of AMH serum concentrations with age, as shown in Figure 4.
AMH reference range for Chinese adult women.
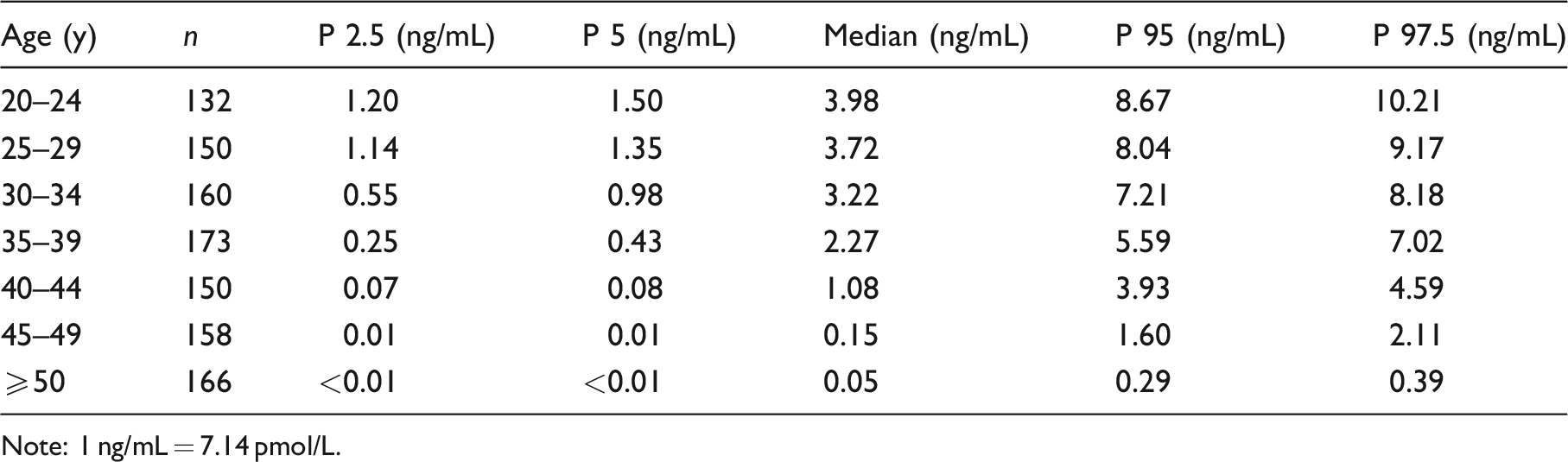
Note: 1 ng/mL = 7.14 pmol/L.
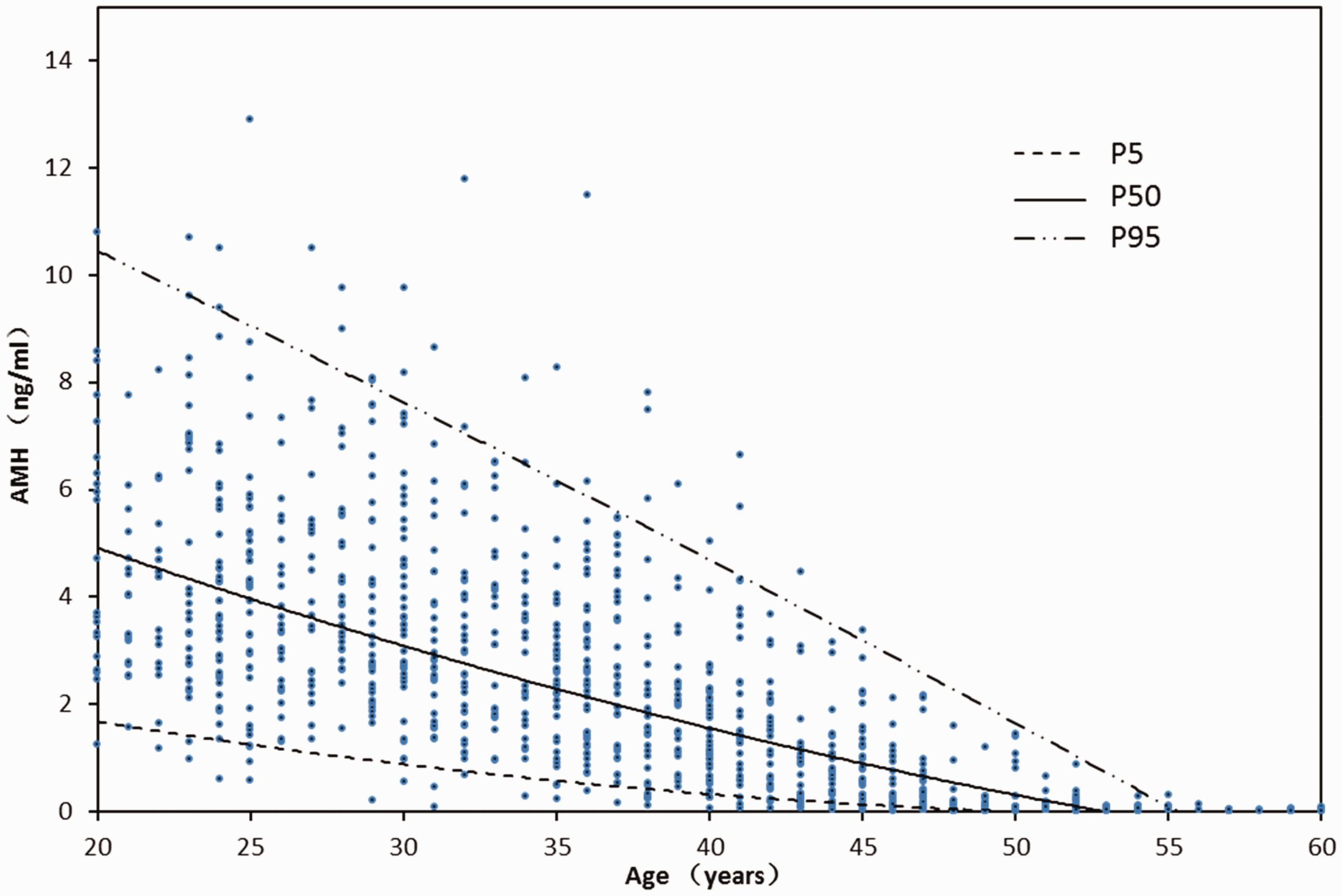
Serum AMH concentrations of different age groups.
Discussion
The analytical performance characteristic study was performed on a novel, automated AMH assay for the iFlash immunoassay. In order to ensure the quality of the inspection, any new laboratory equipment and testing methods need to be verified for their analytical performance for meeting specifications provided by the manufacturer and the clinical requirements before it is formally used in the clinical examination work. These will guarantee the test quality and its analysis for the mutual recognition of the test results. Under routine conditions, the fully automated iFlash AMH assay has demonstrated good precision with CV values ranged between 1.3 and 3.2%, similar to the two existing automated AMH assays in the market, Roche Elecsys AMH and Beckman Access AMH. Furthermore, iFlash automated assay has a wider linearity range from 0.02 to 27.22 ng/mL and significantly higher sensitivity with LoQ of 0.08 ng/mL, making it superior to the manual AMH assays. The LoQ of iFlash AMH is the same or close to that of Access AMH (0.08 ng/mL) and Elecsys AMH (0.03 ng/mL). Samples for AMH in serum and three types of plasma are stable up to seven days at 4°C, −20°C and −80°C, and three days at room temperature, facilitating the storage and transportation.
Method comparison of a newly launched assay with an already marketed system is important means to evaluate the performance for meeting clinical laboratory requirements. In this paper, the correlation and bias of YHLO iFlash AMH and Roche Elecsys AMH were studied and verified by univariate linear regression analysis and Bland-Altman method. The results show that the bias between two kinds of methods is only 0.2 ng/mL, and the 95% confidence interval of the bias includes 0 ng/mL (Figure 3(b)), indicating that there is no significant bias between YHLO iFlash AMH and Roche Elecsys AMH. The linear regression equation was y = 0.121 + 1.009x, and the correlation coefficient was 0.975, indicating that the two methods were in excellent agreement. In the absence of international reference material for AMH (under the development by WHO), the iFlash AMH has been standardized against the existing Elecsys AMH, and hence the excellent correlation coefficient of 0.975 and minimum bias (0.2 ng/mL) using univariate linear regression analysis and Bland-Altman method and consistent results with the previous report. 4 The excellent correlation coefficient of iFlash AMH with Elecsys AMH may be attributed to the antibodies used in each assay which bind to the same regions of AMH molecule, pro- and mature-regions. A part of AMH is cleaved at a specific site between the pro-region and mature-region during cytoplasmic transit, and then associated in a non-covalent complex, the biologically active form of AMH in circulation. 10 Although the antibody pairs used in iFlash AMH and Elecsys AMH are not identical, their binding regions are the same, i.e. one antibody directs against pro-region and the other directs against the mature region. The antibodies used in Elecsys AMH were originally developed by Diagnostic Systems Laboratories (DSL, Webster, TX),2,11 and iFlash AMH used a new version of AMH antibodies developed by Anshlabs (Webster, TX, the same team from DSL),12–14 avoiding the issue of complement interference of the DSL AMH antibodies when used on Beckman Gen II AMH assay. 15 Harmonization of AMH measurement with different systems is important to compare and interpret the test result among different laboratories.
Reliable reference intervals for the measurement of serum AMH concentration are critical for the clinical application of AMH in the evaluation of ovarian reserve and IVF treatments. As the antibodies and antigens used in immunoassays among different manufacturers are basically not identical, standardization of immunoassays among different system is a big challenge even with international reference materials. One of the major solutions is the establishment of the reference intervals for the individual system. iFlash AMH developed by YHLO Biotech is one of the three fully automated AMH immunoassays in the world by now. To our knowledge, our study is the first to establish age-specific reference intervals for serum AMH concentrations using iFlash AMH in Chinese women based on a multicentre population. It has been reported that only age had a deterministic effect on the AMH concentrations being highest between the ages of 18 and 24 years.16–18 Our result indicated that female AMH concentrations peak at the age of 20–24 years and gradually decrease to almost undetectable level at menopause. Differences in the AMH detection methods used in dissimilar ethnic groups studied may however influence the exact time of AMH peak concentrations. 19 Jopling et al. established the AMH reference intervals for females aged 0–18 years and reported the median AMH concentration of 21.14 at the age of 15–18 years. 5
A recent publication by Yates et al. indicated that AMH concentrations measured with Elecsys AMH were significantly different from that measured with Access AMH assay (Beckman Coulter). 20 Although the median AMH concentrations decrease gradually with age, the range of AMH concentrations at each age group showed significant overlap between age groups throughout 20–50 years old and indicates a significant individual variation of AMH concentrations. This highlights the clinical utility of measurement of serum AMH concentrations which reflect and correlate the antral follicle count and ovarian reserve. 21 Serum AMH concentrations can help the female individuals to estimate the time when a pregnancy might be postponed. It can also be used for more accurate prediction of menopause, helping in perimenopausal management.
In conclusion, the results of our evaluation of the novel iFlash AMH indicate that the analytical performance of this fully automated immunoassay is excellent. There was a good correlation between the measured values of iFlash AMH and Elecsys AMH. The AMH reference intervals of this multicentre study provide clinicians with age-dependent reference intervals in adult women.
Footnotes
Acknowledgements
All the immunoassay kits used in this study were kindly supplied by the manufacturers, namely, YHLO Biotech (Shenzhen, China).
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The reagents used in this study were provided by YHLO, but the company played no role in the study design; in the collection, analysis, and interpretation of data; in the writing of the report; or in the decision to submit the report for publication.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This study was approved by the Ethics Committee of the Obstetrics and Gynecology Hospital of Fudan University, Number: kyy2018-15.
Guarantor
C-yY.
Contributorship
C-yY analysed the data, drafted the article and contributed to study design. YW, C-hD contributed to study design. JW, DZ, X-hL and Q-LZ contributed to data gathering. C-MY revised the article. All authors reviewed and edited the article and approved the final version of the article.
