Abstract
Background
The 0.25 mg short synacthen test is used to assess recovery from hypothalamic–pituitary–adrenal suppression due to chronic glucocorticoid administration. We assessed the potential role of salivary cortisol and cortisone in predicting hypothalamic–pituitary–adrenal function using the short synacthen test as the gold standard test.
Method
Between 09:00 and 10:30, salivary and blood samples were collected just prior to a short synacthen test to assess hypothalamic–pituitary–adrenal axis recovery in patients previously treated with oral glucocorticoids. The cut-off for a normal short synacthen test was a 30-min cortisol ≥450 nmol/L.
Results
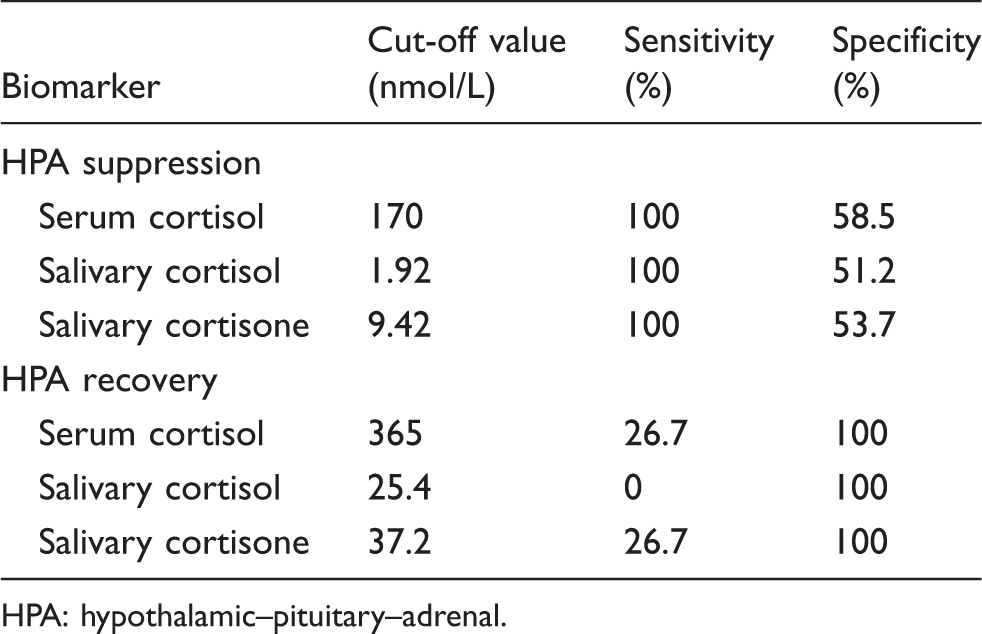
Fifty-six short synacthen tests were performed on 47 patients. Of these, 15 were normal. The area under receiver operating characteristic curves for serum cortisol, salivary cortisone and salivary cortisol were 0.772, 0.785 and 0.770, respectively. From the receiver operating characteristic analysis, the cut-offs for baseline serum cortisol (≥365 nmol/L) and salivary cortisone (≥37.2 nmol) predicted hypothalamic–pituitary–adrenal axis recovery with 100% specificity in 26.7% of pass short synacthen tests, whereas salivary cortisol predicted none. Baseline serum cortisol (≤170 nmol/L), salivary cortisone (≤9.42 nmol/L) and salivary cortisol (≤1.92 nmol/L) predicted hypothalamic–pituitary–adrenal suppression with 100% sensitivity in 58.5%, 53.7% and 51.2% of failed short synacthen tests, respectively. Using these cut-offs, baseline serum cortisol, salivary cortisone and salivary cortisol could reduce the need for short synacthen tests by 50%, 46% and 37%, respectively.
Conclusion
Although marginally inferior to early morning serum cortisol, early morning salivary cortisone may be used as a first-line test for assessing hypothalamic–pituitary–adrenal function. We plan to incorporate salivary cortisone into a home-based patient pathway to identify patients with hypothalamic–pituitary–adrenal recovery, continuing hypothalamic–pituitary–adrenal suppression and those who require a short synacthen test.
Background
Hypothalamic–pituitary–adrenal (HPA) axis suppression may occur as a result of prolonged use of glucocorticoids at supraphysiological doses and abrupt withdrawal of glucocorticoid treatment is the commonest cause of adrenal insufficiency. 1 HPA axis suppression may persist for an unpredictable and prolonged period of time. The 0.25 mg synacthen test (SST) is commonly used to evaluate HPA axis recovery. A normal cortisol response to synacthen indicates adequate function of the HPA axis and glucocorticoids may be safely withdrawn. If the HPA axis has not recovered, glucocorticoid replacement is continued and the HPA axis is reassessed periodically with a SST. 2
The SST is an invasive test requiring cannulation or repeated venepunctures and requires the patient to attend hospital. This is particularly relevant in patients with HPA suppression who may require repeated SSTs to confirm HPA recovery before withdrawal of glucocorticoids. 2 In these patients, early morning cortisol has been used to predict HPA recovery, HPA suppression and the need for a SST in those with indeterminate cortisol concentrations. 3
Similarly use of salivary cortisol or salivary cortisone or both to predict HPA functionality and the need for SST offers an attractive alternative assessment strategy. There are, however, limited data on the use of salivary biomarkers in the evaluation of HPA axis suppression and recovery. We, therefore, investigated the potential value of salivary cortisol and cortisone in the assessment of HPA axis recovery using the standard SST as the gold standard test.
Patients and methods
In our unit, withdrawal from long-term therapeutic doses of steroids involves tapering the steroid dose to 5 mg prednisolone or equivalent and then switching to hydrocortisone 10 mg in the morning, 5 mg at the midday and 5 mg in the early evening. Assessment of HPA axis with a SST is arranged once the patient has been on this replacement hydrocortisone regimen for at least four weeks.
Between March 2016 and January 2019, consecutive patients attending the endocrine dynamic function clinic for a SST to evaluate HPA recovery were invited to participate in a trust approved and registered quality improvement project assessing utility of salivary markers in predicting HPA function. All patients had previously been on prolonged supraphysiological therapeutic doses of oral glucocorticoids. Those with periodontal disease were excluded. Patients had blood and saliva samples collected as detailed below.
SSTs were performed between 09:00 and 10:30 after oral and inhaled glucocorticoid withdrawal for at least 24 h. Baseline salivary sample was collected immediately before the baseline serum sample and then 0.25 mg of tetracosactide acetate (synacthen) was injected either intravenously or intramuscularly and a further blood sample collected 30 min later. A SST was labelled as ‘pass’ (adequate response) if the 30-min serum cortisol was ≥450 nmol/L.
Saliva was collected in Salivette® tubes (plain cotton swab; Sarstedt, Germany) according to manufacturer’s instructions and blood in S-Monovette® 4.7 mL Z-gel tubes (Sarstedt, Aktiengesellschaft & Co, Germany). Blood was separated and serum cortisol measured by a chemiluminescence microparticle immunoassay on an Abbott Architect i2000 (Abbott Laboratories, USA). Salivary samples were centrifuged at 1500 g for 5 min, the cotton was discarded and the extracted saliva frozen at −80°C until analysed for cortisol and cortisone using liquid chromatography with tandem mass-spectrometry (LC-MS/MS) on a Shimadzu Prominence HPLC system coupled to an AB Sciex 3200 mass spectrometer based on previously described method. 4 , 5 The method summary and performance characteristics are included in the Supplemental material. The method was validated in-house and via sample exchange. The Abbott Architect cortisol assay has a quantitation limit of 22 nmol/L with an intra-assay and inter-assay coefficients of variation (CV) of 2.5% at 118 nmol/L and 3.5% at 110 nmol/L, respectively. The LC-MS/MS salivary cortisol and salivary cortisone assays both had a quantitation limit of 0.83 nmol/L. The intra-assay CVs were 4.9% at 9.5 nmol/L for both salivary cortisol and salivary cortisone and the inter-assay CVs were 10.8% at 9.7 nmol/L and 6.0% at 10 nmol/L, respectively.
Statistical analysis was performed using IBM SPSS Statistics for Windows, version 25 (IBM Corp., Armonk, N.Y., USA). As the data were non-parametric, Spearman rank correlation was used to measure the degree of association between variables.
Results
We studied 56 SSTs performed on 47 patients (31 females; median age 60 years, IQR 48.3–69.5). Females did not have higher serum cortisol than males. Seven patients underwent two SSTs each and one patient three SSTs. Of the 56 SSTs, 15 SSTs had normal serum cortisol responses to synacthen. There were no significant sex differences in baseline serum cortisol in those with and without HPA suppression.
Receiver operating characteristic (ROC) curves for baseline serum cortisol, salivary cortisol and salivary cortisone with 30-min post-synacthen cortisol as the reference test are shown in Figure 1. The areas under the curve (AUC) for serum cortisol, salivary cortisol and salivary cortisone were 0.772, 0.770 and 0.785, respectively. From the ROC analysis, the optimal cut-offs for the three biomarkers for HPA recovery with 100% specificity and HPA suppression with 100% sensitivity are shown in Table 1 and Figure 2.
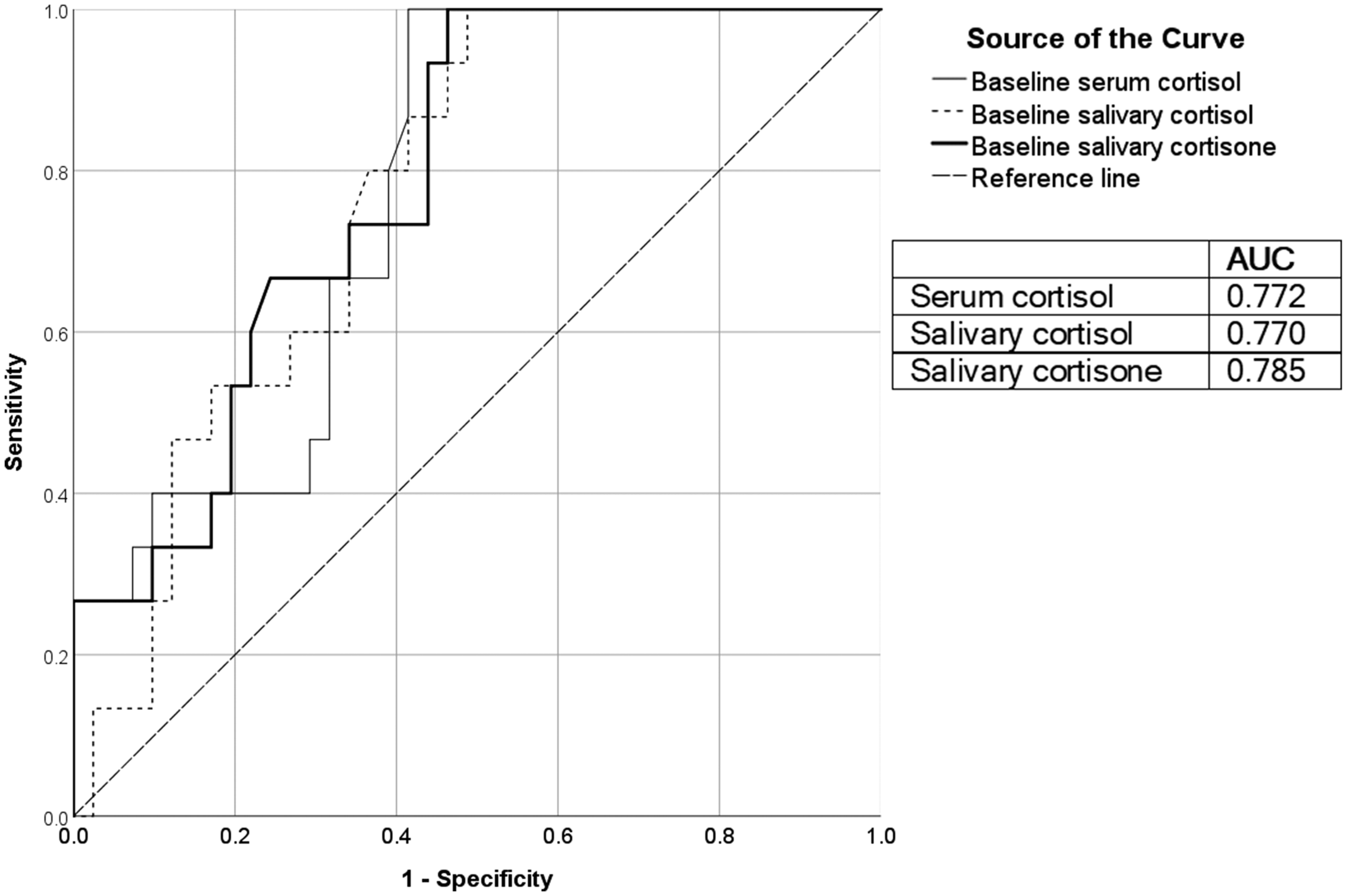
Receiver operating characteristic curves for baseline serum cortisol, salivary cortisol and salivary cortisone with 30-min post-synacthen cortisol as the reference test. AUC is area under the curve.
Receiver operating characteristic curve derived values for HPA suppression and HPA recovery with 100% sensitivity and 100% specificity respectively.
HPA: hypothalamic–pituitary–adrenal.
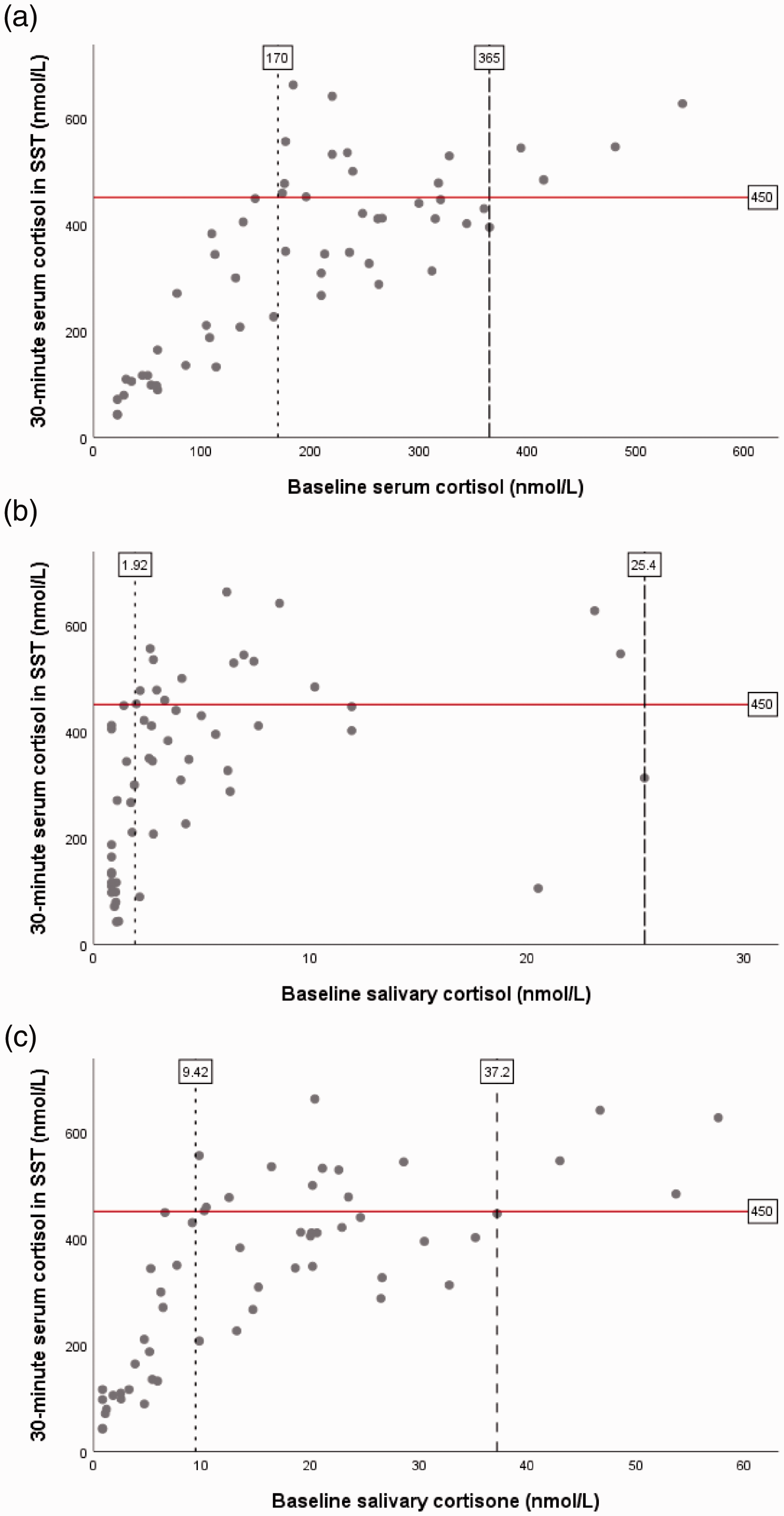
(a) Baseline serum cortisol, (b) baseline salivary cortisol and (c) baseline salivary cortisone plotted against 30-min serum cortisol. ROC analysis derived cut-offs for 100 % sensitivity and 100 % specificity are denoted by dotted and dashed vertical lines respectively.
Baseline serum cortisol was superior to salivary cortisone and both were superior to salivary cortisol in predicting HPA suppression. Baseline serum cortisol was similar to salivary cortisone and both were superior to salivary cortisol in predicting HPA recovery but all had low sensitivity.
Baseline serum cortisol correlated with salivary cortisone (ρ 0.92; P < 0.001), salivary cortisol (ρ 0.73; P < 0.001) and 30-min cortisol (ρ 0.77; P < 0.001). Thirty-minute serum cortisol correlated with salivary cortisone (ρ 0.76; P < 0.001) and salivary cortisol (ρ 0.59; P < 0.001). Salivary cortisone correlated with salivary cortisol (ρ 0.77; P < 0.001).
Discussion
We investigated baseline serum cortisol, salivary cortisol and salivary cortisone in evaluating HPA recovery in those with possible HPA suppression. The areas under the ROC curve indicated that salivary cortisone was the best of the three biomarkers studied for assessing HPA function but they all lacked sensitivity and specificity to be of clinical value as stand-alone tests for HPA axis assessment. We, therefore, investigated their potential value as first-line tests to identify patients with HPA recovery, continuing HPA suppression and those who require a SST.
Baseline serum cortisol (≥365 nmol/L) and salivary cortisone (≥37.2 nmol/L) were both able to predict a normal HPA axis with 100% specificity in 4 (26.7%) of the 15 patients with normal SSTs, whereas salivary cortisol had no value in predicting a pass SST outcome. Baseline serum cortisol (≤170 nmol/L), salivary cortisone (≤9.42 nmol/L) and salivary cortisone (≤1.92 nmol/L) predicted HPA suppression with 100% sensitivity in 24 (58.5%), 22 (53.7%) and 21 (51.2%) respectively of 41 patients with failed SSTs. These data indicate that compared with salivary biomarkers, serum baseline cortisol was the superior test in predicting SST outcome and that salivary cortisone was superior to salivary cortisol.
Compared to salivary cortisol, the closer correlation of salivary cortisone to baseline and 30-min serum cortisol explains its better performance and supports the notion that salivary cortisone better reflects serum cortisol and is preferable to salivary cortisol in assessing HPA function. 6 , 7 Cortisol in the salivary glands is converted by 11β-hydroxysteroid dehydrogenase type 2 (11β-HSD2) to cortisone. Salivary cortisol concentrations are, therefore, dependent upon circulating free cortisol and its metabolism by salivary 11β-HSD2 to cortisone. Plasma free and bound cortisone are stable in their basal state and following adrenal stimulation and therefore their contribution to salivary cortisone is constant. 8 Changes in salivary cortisone are, therefore, solely due to the conversion of salivary cortisol by 11β-HSD2. This may, therefore, clarify why salivary cortisone may offer a superior assessment of adrenal function compared with salivary cortisol. 8 , 9
There are various serum cortisol cut-offs for defining HPA suppression and HPA recovery in the literature. 3 , 10 , 11 These may be related to differences in assays and patient cohorts. The concept of assay and sex specific cut-offs for defining normal serum cortisol responses to synacthen is well-established. 12 , 13 The reported variability in immunoassay and non-immunoassay performance indicates that not only serum cortisol immunoassay but also LC-MS/MS salivary cortisol cut-offs defined in this study may, therefore, not be transferable to other serum cortisol and salivary assays. The cut-offs derived in this study may not be applicable to the evaluation of HPA function in cohorts without HPA suppression. Premenopausal females, except those on oral oestrogen, 14 have lower serum total cortisol concentrations than males 15 , 16 which, in part, could be due to analytical aspects. 17 This difference in serum cortisol, however, does not exist beyond age 50 years. 15 This may explain the absence of sex difference in baseline serum cortisol in our study cohort as most females were perimenopausal or postmenopausal. Our cut-offs may, therefore, not be applicable to premenopausal females and females on oral oestrogen.
In this study, most patients had HPA suppression and will require repeat SSTs until HPA recovery is documented to allow safe withdrawal of steroids. It would, therefore, be cost-effective and of patient benefit to safely reduce the number of SSTs. Using the derived cut-offs for serum cortisol, salivary cortisone and cortisol would reduce the need for SSTs by 50%, 46% and 37%, respectively. Although early morning salivary cortisone is marginally inferior to serum cortisol in predicting SST outcome, it offers other advantages. These include non-invasiveness, stress free for the patient, absence of needle stick injury risk and, importantly, ease of collection allowing potential home testing and posting of samples with avoidance of hospital visits.
We currently batch analyse salivary cortisol and cortisone and this laboratory practice would be unsuitable for assessment of adrenal insufficiency in other patient cohorts, since a prolonged turnaround time in salivary results may present clinical risk in delaying a diagnosis of adrenal failure. There, however, would be no clinical risk when assessing recovery from HPA suppression as patients would be covered with replacement glucocorticoids while awaiting results.
In summary, our data indicate that salivary cortisol has a limited role in the assessment of recovery from HPA suppression. Although marginally inferior to early morning serum cortisol, we suggest that early morning home collected salivary cortisone is a valuable first line test to identify patients with HPA recovery and continuing HPA suppression and thereby significantly reducing the need for SSTs to those with indeterminate values. We plan to incorporate morning salivary cortisone into our patient pathway for glucocorticoid withdrawal (Figure 3). Ideally, its clinical effectiveness should be assessed with a randomized study comparing the current pathway to the salivary cortisone pathway to demonstrate at least non-inferiority in patient outcomes.
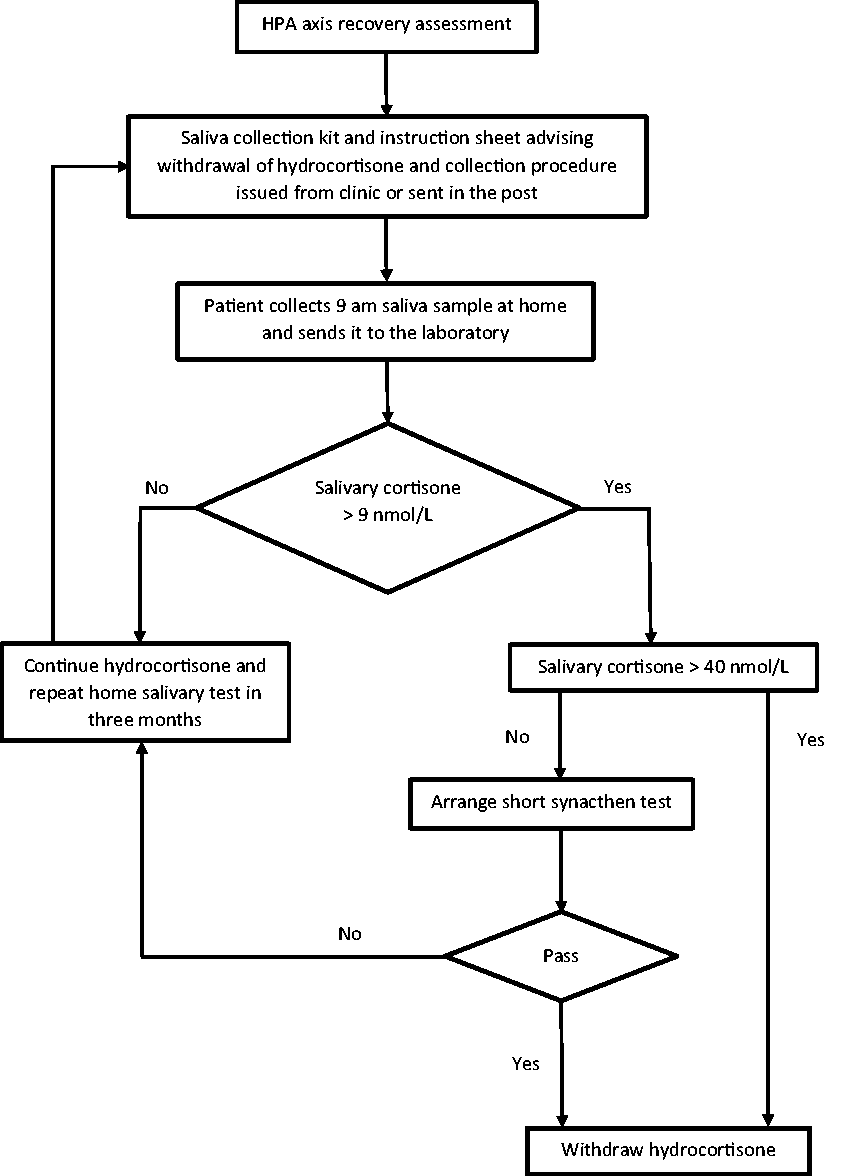
Proposed sequential testing pathway for assessment of HPA axis recovery. HPA: hypothalamic–pituitary–adrenal.
Supplemental Material
sj-pdf-1-acb-10.1177_0004563220961745 - Supplemental material for Hypothalamic–pituitary–adrenal axis suppression – The value of salivary cortisol and cortisone in assessing hypothalamic–pituitary–adrenal recovery
Supplemental material, sj-pdf-1-acb-10.1177_0004563220961745 for Hypothalamic–pituitary–adrenal axis suppression – The value of salivary cortisol and cortisone in assessing hypothalamic–pituitary–adrenal recovery by Tejas Kalaria, Mayuri Agarwal, Sukhbir Kaur, Lauren Hughes, Hayley Sharrod-Cole, Rahul Chaudhari, Carolina Gherman-Ciolac, Roopa Chopra, Victor Okeke, Clare Ford, Harit Buch and Rousseau Gama in Annals of Clinical Biochemistry
Footnotes
Acknowledgements
None.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Registered quality improvement project (QIP) ID 5334. Permission to publish data has been granted by the trust Caldicott Guardian.
Guarantor
RG.
Contributorship
HB and RG designed the QIP. TK contributed to sample collection, compiled and analysed the data and wrote the initial manuscript. MA collected samples and helped with data compilation. RC, CGC, RC and VO performed SSTs, collected samples and helped with data collection. SK, LH and SHC analysed salivary samples. All authors critically reviewed the article and approved the final version of the article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
