Abstract
Background
To establish cutoff values for salivary liquid chromatography tandem mass spectroscopy cortisol and cortisone in defining adequate adrenocortical function during a standard synacthen test.
Methods
We compared salivary liquid chromatography tandem mass spectroscopy cortisol and cortisone responses to those of serum cortisol measured on the Roche E170 immunoassay analyser and the Abbott Architect i2000 before and 30 min and 60 min following 0.25 mg of intravenous synacthen.
Results
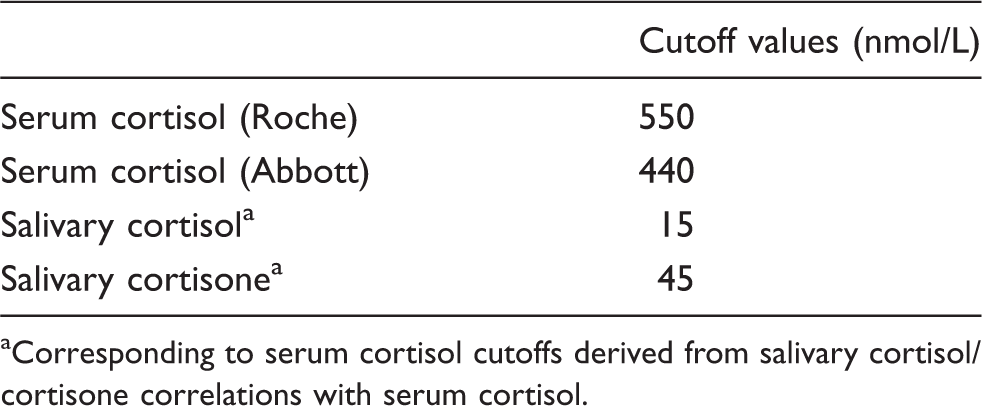
Correlations of salivary cortisol and cortisone were bimodal and linear, respectively. Based on these correlations, adequate salivary cortisol and cortisone responses to synacthen were extrapolated from a serum cortisol (Roche) cut-off of 550 nmol/L and defined as 15 nmol/L and 45 nmol/L, respectively. The Abbott method correlated well with the Roche but gave results that were about 20% lower than the Roche method.
Conclusions
Measurement of salivary cortisol and cortisone responses offers an alternative to those of serum cortisol during a synacthen test in the investigation of adrenal hypofunction.
Introduction
Cortisol circulates as free cortisol (<5%) but mostly bound to the carrier proteins cortisol binding globulin (CBG) and albumin. Serum cortisol, however, is a measurement of free and bound cortisol and in conditions of altered CBG concentrations, such as acute illness or oestrogen therapy, it may be misleading as it does not accurately reflect serum free cortisol. Cortisol in saliva, however, accurately reflects serum-free cortisol, as it is unbound and in equilibrium with circulating free cortisol.1–3
Cortisol in the salivary glands is deactivated by 11β-hydroxysteroid dehydrogenase type 2 (11β-HSD2) to cortisone. Salivary cortisol concentrations are, therefore, dependent upon circulating free cortisol and its metabolism by salivary 11β-HSD2 to cortisone. Circulating free and bound cortisone are stable in their basal state and following adrenal stimulation, and therefore their contribution to salivary cortisone is constant.1–3 Increases in salivary cortisone are, therefore, solely due to its conversion from salivary cortisol by 11β-HSD2. This indicates that salivary cortisol and cortisone may be better biomarkers of adrenal function than serum cortisol. The low salivary cortisone and cortisol concentrations, which are 10-fold and 30-fold lower than serum total cortisol concentrations respectively, however, precluded their accurate and reliable measurement until the advent of liquid chromatography tandem mass spectroscopy (LC-MS/MS).
We have encountered patients, referred for synacthen tests to exclude adrenal failure, who have extremely difficult venous access. Multiple attempts of often unsuccessful cannulation are extremely distressing for them. In these patients, assessment of adrenal function using salivary cortisol instead of serum cortisol would circumvent the problem. There is, however, paucity of data on salivary LC-MS/MS cortisol and cortisone responses to synacthen.
We, therefore, compared salivary cortisol and cortisone responses to those of serum cortisol during a standard 0.25 mg synacthen test. Appropriate cut-offs for salivary cortisol and cortisone defining adequate adrenocortical function during a synacthen test were extrapolated from widely accepted serum cortisol cutoff values.
Subjects and methods
Patients
In a six-month prospective evaluation study, outpatients attending the endocrine and metabolic dynamic function clinic for a short synacthen test (SST) for possible adrenal insufficiency were recruited. Inclusion criteria included those willing to participate who did not have periodontal disease and those who were not taking or had not recently received either oestrogens or glucocorticoids. All patients had simultaneous blood and saliva samples as detailed below.
Short synacthen test
SSTs were performed in the afternoon. An intravenous cannula was inserted into an antecubital fossa vein, and baseline blood and saliva samples were collected between 14.00 and 14.30. Tetracosactide (Synacthen, Alliance Pharmaceuticals Ltd, Chippenham, UK) (250 µg) was then administered intravenously and further blood and salivary samples collected 30 and 60 min later. Blood and saliva samples were collected into Sarstedt serum/z4 gel tube (Sarstedt Safety Monovette serum Z/4 2, Aktiengesellschaft & Co, Germany) and Salivette tubes (Plain cotton swab; Sarstedt, Numbrecht, Germany), respectively.
Blood was separated and serum cortisol was measured by electrochemiluminescence immunoassay (Elecsys Cortisol, Roche Diagnostics GmbH, Mannheim, Germany) on the Roche Modular Analytics E170 immunoassay analyser and by a chemiluminescence microparticle immunoassay on an Abbott Architect i2000 (Abbott Laboratories, USA). Salivary samples were centrifuged at 3000 rpm for 5 min. The cotton swab was then discarded, and the extracted saliva was frozen at −80C until analysis for cortisol and cortisone by LC-MS/MS. LC-MS/MS analysis was performed on a Shimadzu Prominence HPLC system coupled to an ABSciex 3200 mass spectrometer based on a previously described method.4,5 The only differences were the use of phenomenex C8 guard cartridges (AJO-4289) for the solid phase extraction steps and the platform used. The method was validated in-house and via sample exchange prior to the study.
The Elecsys cortisol assay has a detection limit of 2 nmol/L (pack insert; Elecsys Cortisol, Roche Diagnostics GmbH, Mannheim, Germany) with intra-assay and inter-assay coefficients of variation (CV) of 1.1% at 418 nmol/L and 1.7% at 410 nmol/L, respectively (in-house data). The Abbott Architect cortisol assay has a detection limit of 22 nmol/L (pack insert; Abbott Laboratories, USA) with intra-assay and inter-assay CVs of 4.3% and 3.2% at 103 nmol/L and 374 nmol/L, respectively. Cross-reactivity in the cortisol assay due to cortisone is 2.7% for the Abbott Architect (pack insert; Abbott Laboratories, USA) and 0.3% for the Elecysys (pack insert; Elecsys Cortisol, Roche Diagnostics GmbH, Mannheim, Germany). Both the LC-MS/MS cortisol and cortisone assays have a detection limit of 0.25 nmol/L. The intra-assay CV for cortisol and cortisone were 4.9% at 9.5 nmol/L and 4.9% at 9.5 nmol/L, respectively, and the interassay CV for cortisol and cortisone were 10.8% at 9.7 nmol/L and 6.0% at 10 nmol/L, respectively (in-house data).
A normal cortisol response to synacthen was defined as a post-stimulation peak Roche Elecsys cortisol value of ≥550 nmol/L at either 30 or 60 min. 6
Results
Thirty-six patients (16 female) with a mean age 51 y (range 15–88) were studied. Of these, 32 patients had normal (NR) and four patients had abnormal (AR) serum cortisol responses to synacthen as defined by the cutoff for the Roche method.
Median, lower limit and upper limit for normal responders and abnormal responders for each method and analyte.
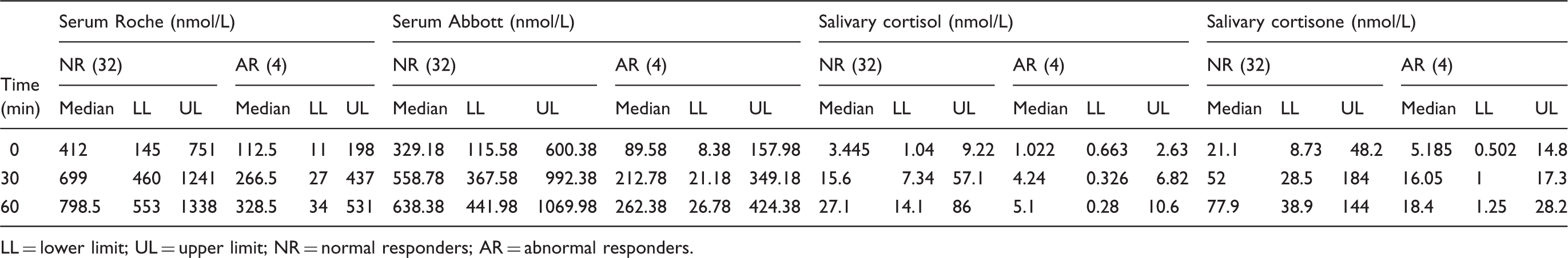
LL = lower limit; UL = upper limit; NR = normal responders; AR = abnormal responders.
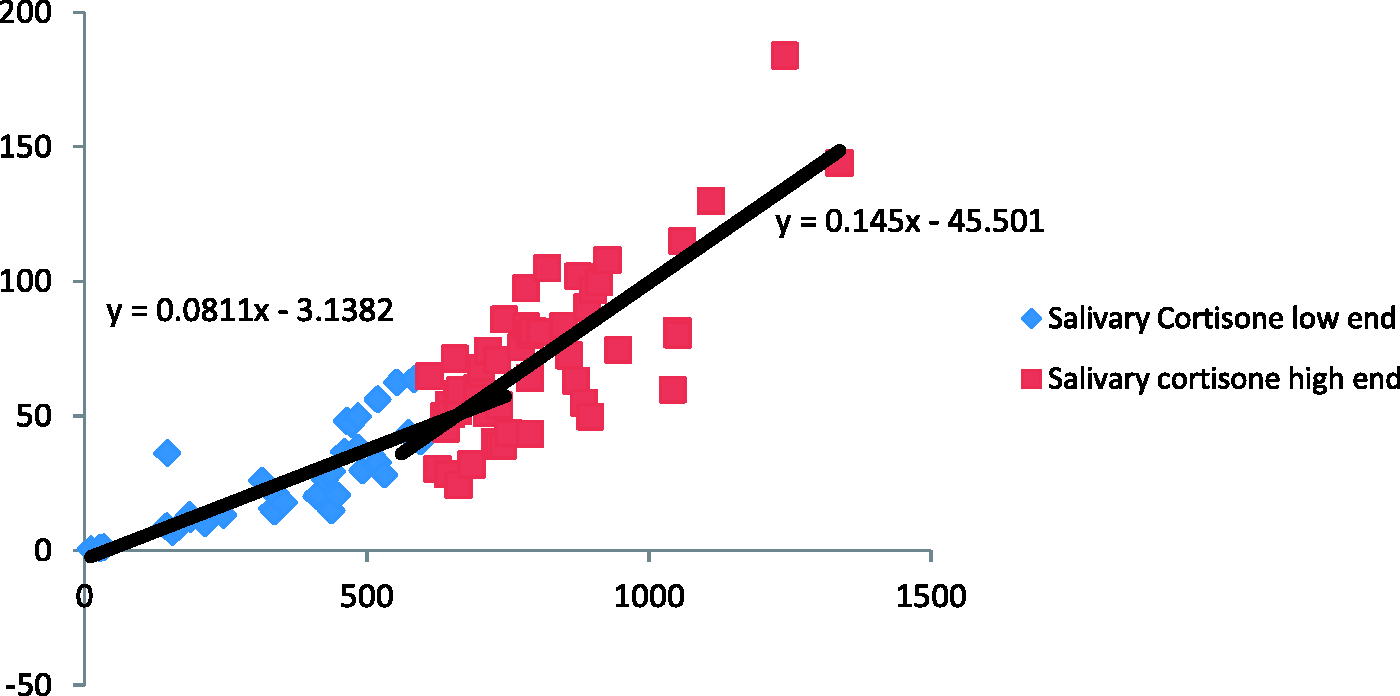
Correlation of salivary cortisol with the Roche serum cortisol was bimodal, intersecting at the CBG saturation level of a serum cortisol of 600 nmol/L (y = 0.0214x − 2.0105; Salivary cortisol vs. serum cortisol (nmol/L). Salivary cortisone vs. serum cortisol (nmol/L). Cut-off values for defining adequate adrenal function during a 0.25 mg synacthen test. Corresponding to serum cortisol cutoffs derived from salivary cortisol/cortisone correlations with serum cortisol.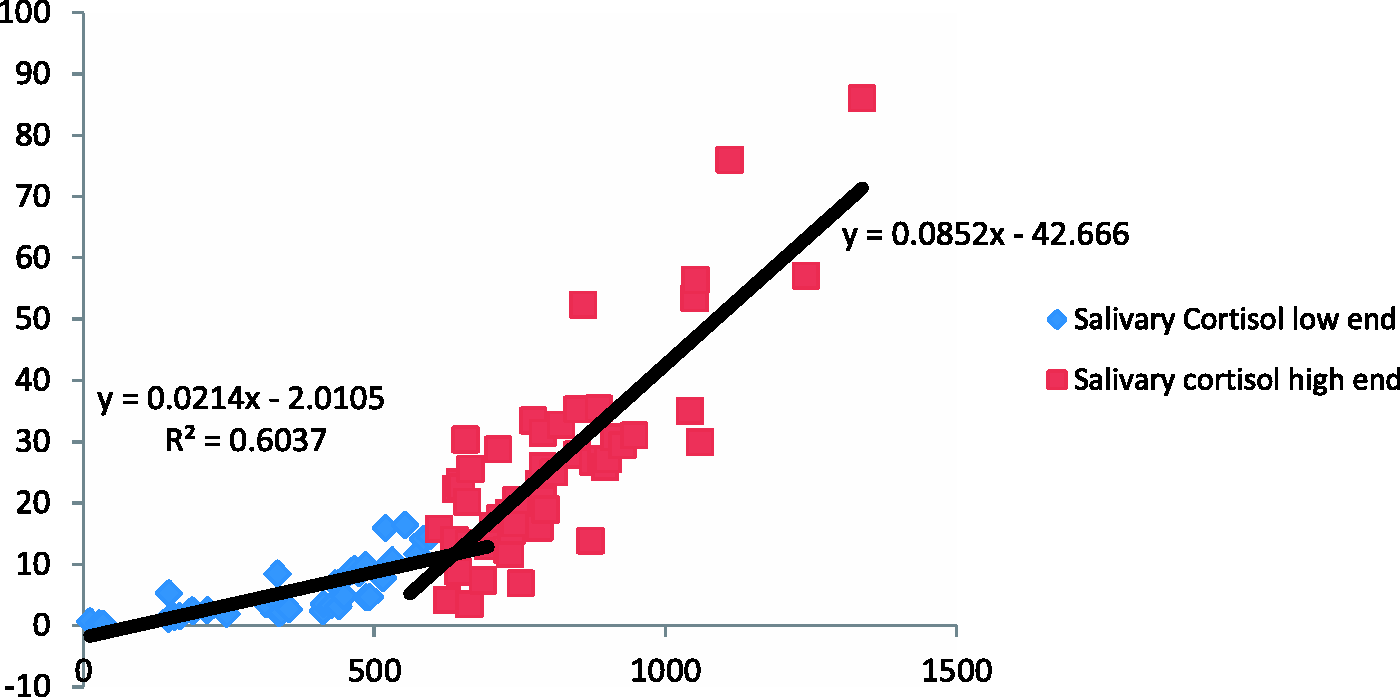
Discussion
We used an LC-MS/MS method for the analysis of salivary cortisol and cortisone based upon a previously described robust method4,5 to facilitate the investigation of adrenal disorders. The advantages of salivary cortisol analysis over blood cortisol analysis in the investigation of Cushing’s syndrome are well recognised. These include more accurate reflection of biologically active free plasma cortisol, non-invasiveness, stress-free for the patient, no risk of needle stick injury and ease of collection allowing potential home testing and posting of samples. Less well-recognised are the potential benefits of salivary analysis over blood analysis in investigating adrenal hypofunction including those patients with either difficult venous access and/or altered CBG concentrations.
Salivary cortisol had a bimodal correlation with serum cortisol, which is consistent with previous reports.1,3 The rapid increase in salivary cortisol at serum cortisol concentrations greater than 600 nmol/L suggests that the circulating CBG is saturated with cortisol at a serum cortisol concentration of 600 nmol/L. 3 Compared to salivary cortisol, salivary cortisone concentrations were threefold higher and had a closer linear correlation with serum cortisol. Salivary cortisol concentrations are dependent not only on circulating free cortisol but also on its metabolism by salivary 11β-HSD2 to cortisone, whereas changes in salivary cortisone concentrations are solely due to its conversion from salivary cortisol since adrenal stimulation does not affect circulating cortisone concentrations.2,3 These data suggest that salivary cortisone may be the preferred analyte in the assessment for adrenal function.
Abbott serum cortisol results were 20% lower than Roche serum cortisol results, which is well-recognised 7 and evident from EQA schemes. This has, therefore, led to the concept of assay specific cutoffs for defining normal serum cortisol responses to synacthen.6,7 Variability in immunoassay and non-immunoassay performance indicates that LC-MS/MS salivary cortisol cutoffs defined in this study may not be transferable to salivary cortisol immunoassays.
There are insufficient data to define reference intervals for salivary cortisol and cortisone. Cutoff values of 15 nmol/L for salivary cortisol and 45 nmol/L for salivary cortisone were, therefore, extrapolated from established serum cutoff cortisol value of 550 nmol/L and would have correctly classified all patients into those with normal and those abnormal responses to synacthen. Cortisol responses to intramuscular synacthen are similar to those following intravenous synacthen. 8 We, therefore, plan to offer synacthen tests with salivary cortisol and cortisone measurement for suspected adrenal hypofunction routinely in patients with limited venous access and those with suspected altered CBG concentrations.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
Ethical approval
Not required. This was a service evaluation.
Guarantor
RG.
Contributorship
HA, YK and HB were responsible for patient recruitment and sample collection. MC set up and analysed all specimens. HB, RG and CF conceived the idea. MC and RG wrote the initial manuscript. All authors reviewed the data and edited the paper.
