Abstract
Background
Serum triglyceride concentrations are measured as total glycerides content in many Western countries. In Japan, glycerol-blanked triglycerides (TG-GB) are measured to identify postprandial hypertriglyceridaemia and to minimize the influence of glycerol formulation on serum triglyceride values. However, TG-GB measurements have not been standardized. Therefore, we developed an efficient quantification system for total glycerides and free glycerol that allows the calculation of TG-GB concentrations.
Methods
We measured total glycerides and free glycerol in human serum by using gas chromatography-isotope dilution mass spectrometry and compared its performance to the reference method of the US Centers for Disease Control and Prevention (CDC).
Results
Our practical method of total glycerides and free glycerol quantification achieved excellent precision for both within-run and among-run coefficients of variation (<1.5% and <2.7%, respectively), with an average recovery of 99.8% for free glycerol. However, we noted an average %bias of –0.26% for total glycerides and –3.15% for free glycerol between our TG-GB method and the CDC reference method.
Conclusions
This practical method of total glycerides and free glycerol quantification enables traceability assessment of TG-GB measurements. Differences between the output values of TG-GB and the CDC reference method might result from the differences in free glycerol values.
Keywords
Introduction
High serum triglycerides (TG) concentrations have been associated with the risk of ischaemic cardiovascular diseases (ICVDs), independent of other cardiovascular risk factors, including total cholesterol, LDL-C, and HDL-C.1–10 TG is one of the factors included in the Friedewald equation, 11 and it approximates to the concentration of LDL-C, a monitoring target for therapy. The concentrations of small-dense LDL, the most atherogenic LDL subfraction, is highly correlated with TG concentrations;12,13 therefore, the accuracy of TG measurements is essential for monitoring the risk factor profiles for ICVD. In many Western countries and most regions of China, TG concentrations are expressed as total glycerides (TLG), which include free glycerol, mono-glyceride, di-glycerides and triglycerides. In contrast, in Japan, these are measured using the glycerol elimination method and are reported as glycerol-blanked triglycerides (TG-GB). 14
Postprandial hypertriglyceridaemia is known to be positively associated with the risk of ICVD;1–4 especially, patients with high body mass index (BMI) have higher serum TLG concentrations during fasting than the postprandial concentrations because of the high free glycerol concentration during fasting. It has recently been reported that, as a consequence of this condition, the postprandial hypertriglyceridaemia of such patients may be missed if their TG concentrations are not measured using the TG-GB measurement reagent. 15 Therefore, calculation of TG-GB requires the standardization of an efficient and effective measurement system for TLG and free glycerol.
The traceability of TG measurements can be verified by comparing the methods provided by manufacturers or clinical laboratories with the chromotropic acid reference measurement procedure (CA-RMP) recommended by the US Centers for Disease Control and Prevention/Cholesterol Reference Method Laboratory Network (CDC/CRMLN) certification programme.16–18 In our Osaka CRMLN laboratory, a fluorometric method of TG estimation was used between April 1975 and August 1985 through the World Health Organization (WHO)-CDC Cooperative Cholesterol-Triglyceride Standardization Program, 19 and an enzymatic method for TG was used between September 1985 and April 2011 through the CDC – National Heart, Lung, and Blood Institute Lipid Standardization Program. 20 As the CA-RMP requires highly toxic reagents and is not specific for the measurement of TG, 21 the CDC in 2012 introduced an RMP for TLG by using gas chromatography-isotope dilution mass spectrometry (GC-IDMS) as a substitute for CA-RMP. Edwards et al. have reported that the CDC GC-IDMS method can accurately measure TLG in a short time (within 4 h) and demonstrated that the method shows sufficient agreement with the National Institute of Standards and Technology (NIST) primary RMP for TLG measurements. 22 Whereas the CDC GC-IDMS method measures only TLG, the Osaka GC-IDMS has continuously used the reference method to measure not only TLG but also free glycerol to calculate the TG-GB.
However, the previously reported methods for free glycerol measurement by using GC-IDMS were not practical. For example, the procedure reported by Bernert et al. was a simple procedure to detect glycerol as a trimethylsilyl derivative, but the reactions were highly sensitive to moisture. 23 In addition, the method reported by Chen et al. was a time-consuming and complicated procedure. 24
We required a practical method to measure both TLG and free glycerol in parallel by using our existing instrumentation because our purpose is to assess the clinical laboratory method for TG-GB measurement mainly used in Japan. Therefore, we attempted to modify the Edwards method to render our measurement procedure practical for both TLG and free glycerol by using GC-IDMS with an acceptable time duration for measurement. This study aimed to determine the performance of the practical Osaka GC-IDMS measurement method for TG-GB.
Materials and methods
Materials
Standard reference materials (SRMs) for pure tripalmitin (SRM 1595) were obtained from NIST (Gaithersburg, MD, USA). Isotope-labelled glycerol, 1,3-13C2-glycerol (100 mg; purity, 99%), used as the internal standard (IS), was obtained from Cambridge Isotope Laboratories, Inc. (Andover, MA, USA). The detergent Emulgen108 was procured from Kao Chemicals (Tokyo, Japan). All standardization pools for TG were prepared according to the Clinical Laboratory Standards Institute document C37-A, 25 which implies that no preservatives or additives were added. They were shipped frozen from the CDC and stored at –70°C before analysis. Other serum pools were obtained from many healthy volunteers and were prepared and stored in microtubes at –70°C until analysis. Other chemicals, including HPLC-grade ethanol, ethyl acetate and pyridine, were from FUJIFILM Wako Pure Chemical Corporation (Osaka, Japan).
Instrumentation
We used a DIGIFLEX TP (Titertek, Pforzheim, Germany) automatic pipettor with a dual microprocessor-controlled direct drive stepper motor for aspirating and dispensing solutions, and a Turbovap LV evaporator (Biotage AB, Kungsgatan, Uppsala, Sweden) for drying samples. To weigh samples precisely, we used a Mettler Toledo analytical balance. The automatic pipettors and analytical balance were gravimetrically calibrated using Japan Calibration Service System (JCSS)-certified weights.
Until April 2016, TG assays were conducted using an Agilent 6890 gas chromatograph incorporating a G2614A autosampler (Agilent Technologies, Santa Clara, CA, USA) coupled to a magnetic sector-type mass spectrometer (JMS-GC mate II, JEOL Ltd, Tokyo, Japan). Subsequently, we used an Agilent 7890A gas chromatograph incorporating a 7693 autosampler (Agilent Technologies) coupled to quadrupole mass spectrometer JMS-Q1050GC (JEOL Ltd). A DB17-ms (50% phenyl)-methylpolysiloxane capillary column (Agilent Technologies) was installed between the gas chromatograph and mass spectrometer.
Preparation of standard solutions
Tripalmitin SRM1595 was weighed into a measuring flask, dissolved in toluene and diluted to prepare the working calibrator solutions. For example, the concentrations of calibrator solutions for free glycerol measurement were typically 2.5, 5.0, 10.8 and 19.6 mg tripalmitin/dL, and these concentrations are comparable to 2.8, 5.5, 11.8 and 21.5 mg triolein/dL. An aqueous solution of 1,3-13C2-glycerol (approx. 2 mg/dL) as the IS for free glycerol measurement was prepared in deionized water containing 0.05% w/v sodium azide at room temperature (18–26°C). Aliquots (10 mL) of this internal standard solution were dispensed into clean capped tubes. These stock solutions were stored in a refrigerator at temperatures below 5°C until analysis.
Sample preparation and GC-MS measurements
Our measurement procedure for TLG and free glycerol by using GC-IDMS was based on a modified version of the Edwards method.
22
A flow chart of the sample preparation procedure for the GC-IDMS measurement of TLG or free glycerol is shown in Figure 1. The Osaka practical method and CDC method differ in the type of IS, sample treatment after acylation and target ions in the selected ion monitoring (SIM) mode of MS analysis. TG assays were conducted using the GC-IDMS method. We performed mass ion detection in SIM mode with the electron voltage set at 70 eV. The selected ions used for quantification were

Flow chart of the procedures of sample preparation by using the CDC method for measuring total glycerides and the Osaka practical method for measuring total glycerides or free glycerol by using gas chromatography-isotope dilution mass spectrometry. The IS of the Osaka practical method is 1,3-13C2-glycerol, whereas that of the CDC method is 13C3-glycerol.
Statistical analysis
Except for Bland–Altman analysis, all statistical analyses were performed using ‘EZR’ (Easy R) statistical software, which is a modified version of R commander designed to add mathematical function frequently used in biostatistics. 26 The statistical data of the Bland–Altman analysis containing the limits of agreement and confidence intervals were obtained using Microsoft Excel (Microsoft Corporation).
Results
The GC-MS chromatograms of the total ions monitored for unlabelled or labelled tri-acetyl glycerol (triacetin), which were eluted at the same time (5.15 min), are shown in Figure 2(a). The mass spectra obtained from the peaks of the unlabelled or labelled triacetin samples are shown in Figure 2(b). We selected the typical fragment ion peaks
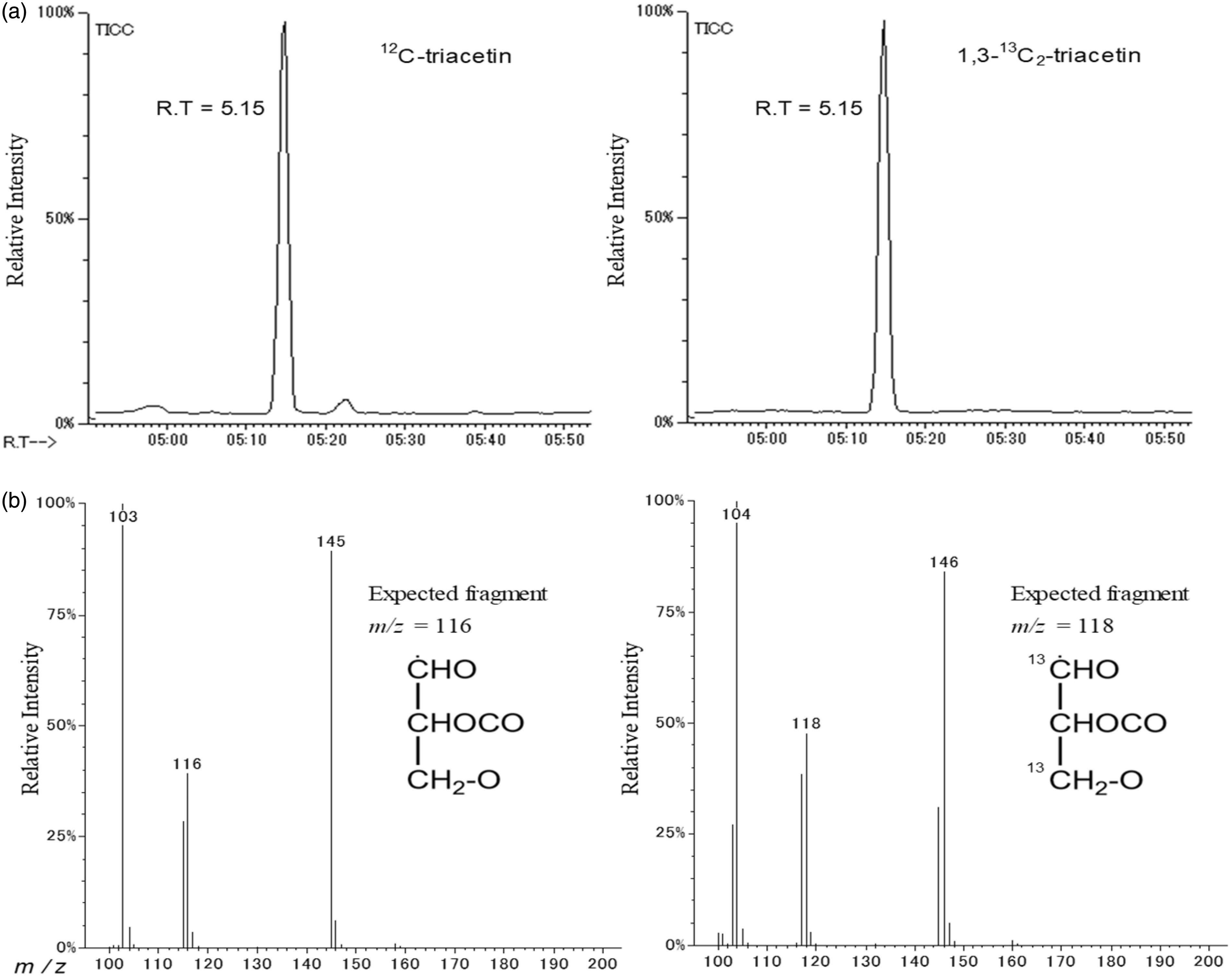
A total ion chromatogram of triacetin derived from unlabelled and labelled glycerol obtained by using gas chromatography-isotope dilution mass spectrometry (a) Mass spectrum of the peak eluted at a retention time of 5.15min. (b) The fragment peaks of m/z=116 and m/z=118 were derived from unlabelled and labelled triacetin, respectively.
Precision of the practical measurement procedure for free glycerol.
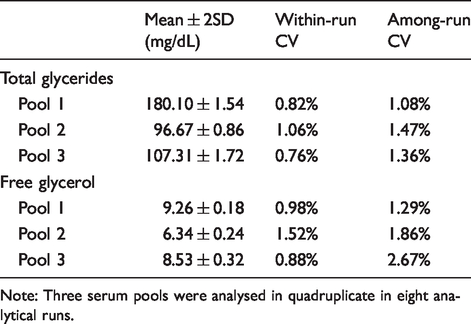
Note: Three serum pools were analysed in quadruplicate in eight analytical runs.
The results of the recovery test for free glycerol measurement are shown in Table 2. The concentrations of the original three pooled serum samples and the two additive glycerol specimens were measured in quadruplicate in advance. The average concentrations of the two additive specimens were 3.86 mg/dL and 5.62 mg/dL, and the recoveries ranged from 99.1 to 100.5%, with an average recovery of 99.8%.
Recovery test for the measurement of free glycerol in human serum by using GC-IDMS.
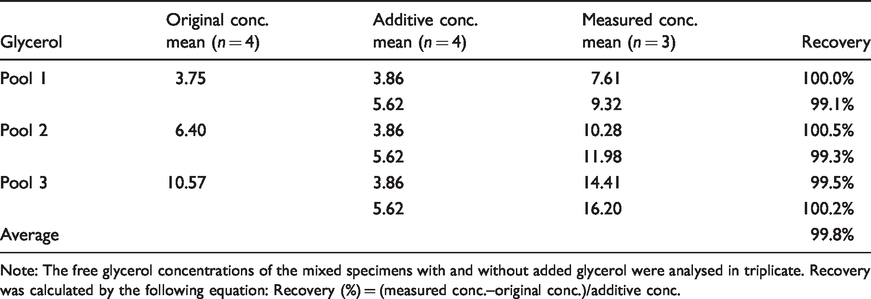
Note: The free glycerol concentrations of the mixed specimens with and without added glycerol were analysed in triplicate. Recovery was calculated by the following equation: Recovery (%) = (measured conc.–original conc.)/additive conc.
Between April 2011 and January 2017, 94 CDC/CRMLN survey samples were analysed from 43 survey runs, in which each survey run consisted of four or five different pools. All survey pools from the CDC were blinded to the CRMLN laboratories. In the report from the CDC, the reference value of TG-GB evaluated using the chromotropic acid method as the previous CDC reference method (CA-RMP) was listed for 56 survey samples. To assess the difference between the Osaka practical method and the CDC method, we performed Bland–Altman analysis. The data obtained are summarized in Table 3. For the CDC reference value, the concentrations of the pools of TLG ranged from 72.34 to 300.94 mg/dL. The average of the CDC reference value and that obtained using the Osaka GC-IDMS method were 166.03 mg/dL and 165.59 mg/dL, respectively (
Summarization of the results of the CDC/CRMLN survey.
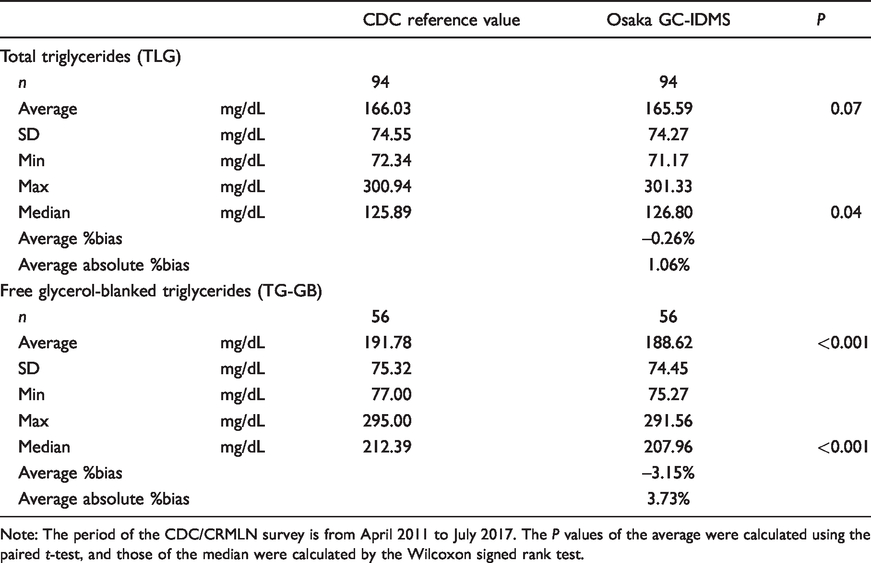
Note: The period of the CDC/CRMLN survey is from April 2011 to July 2017. The
The correlation between the CDC reference method (x) and the Osaka practical method for TLG (y) was y = 0.996x + 0.27 (
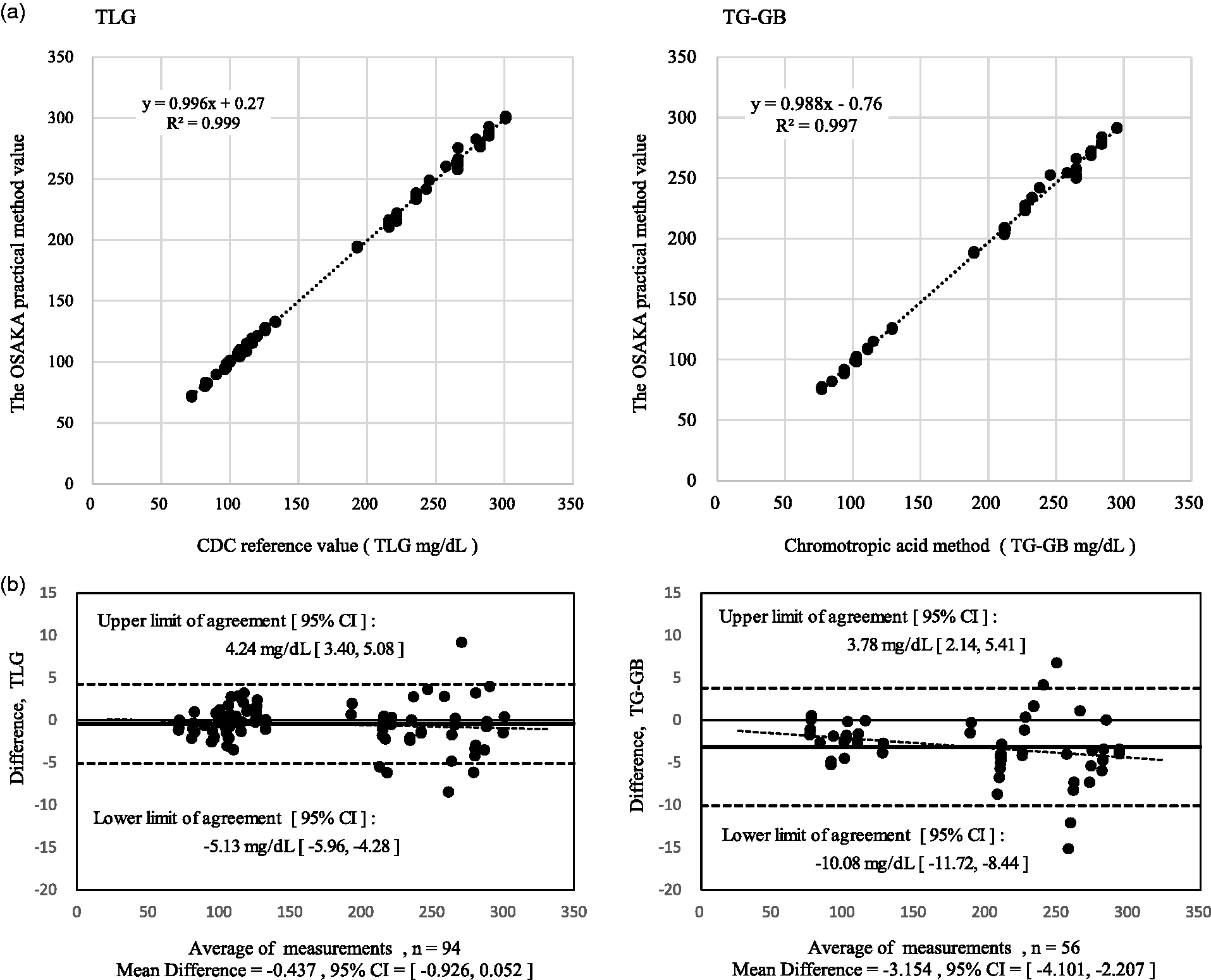
(a) The correlation between the CDC method and the Osaka practical method was y = 0.996x + 0.27 (
Discussion
In this study, we used a modified version of the CDC method for the measurement of TLG and free glycerol by using existing equipment with minor changes in the GC-MS system, the IS and sample preparation procedures. More specifically, we employed the procedures described below to clean up the ethyl acetate extract because residual pyridine might not be completely removed by sodium bicarbonate. The residual pyridine was oxidized using hydrochloric acid before the ethyl acetate extraction process, because the pyridinium hydrochloride salt is relatively insoluble in ethyl acetate. Next, the residual by-product, acetic acid, in the samples was neutralized with sodium bicarbonate. In our free glycerol measurement system without the hydrolysis process, no fatty acid was liberated from any acyl-glycerol, and no transesterification was obtained under the alcohol-free conditions of the derivatization process. 27 Although the base-catalysed transesterification requires alcohol, alcohol was not present in all tubes owing to the drying process performed before acylation in our sample preparation method. Thus, the free glycerol measurement values obtained using this method had extremely high specificity, as the samples did not include any acyl-glycerol, such as monoglyceride, diglycerides and triglycerides. The within-run and among-run precision and accuracy of our practical method for free glycerol were found to be satisfactory.
Triglycerides play a central role in dietary lipid metabolism, including in digestion, absorption, transport by chylomicron or VLDL, degradation, recomposition and lipid storage in the liver. However, the clinical significance of free glycerol, which is a degradation product of triglycerides, is not clear. In Western populations, free glycerol occurs in only trace amounts compared with TLG, and thus TLG, including free glycerol, is adopted for TG measurement in clinical settings. In contrast, to avoid the potential effect of glycerol formulation on TG measurements, in one instance, glycerol formulation was used normally, considering that there are more stroke patients in Japan than in Western countries. 28 In addition, to obtain more accurate TG values, the TG-GB measurement reagent has become widely used in Japan. Under these circumstances, the TG measurement reagent has been used in Japanese clinical tests, whereas the reagent for TG-GB is replaced by that for TLG in many Western countries. Recently, the postprandial hypertriglyceridaemia of the patients with high BMI has been reported to be likely missed if TG concentrations are not measured using the TG-GB measurement reagent. 15 Accordingly, the glycerol elimination method for TG measurement is beneficial and required for such patients and should be standardized through the development of a sound measurement system for both TLG and free glycerol.
In our Osaka CRMLN laboratory, we have been using the GC-IDMS method for TLG since April 2011 through the TG standardization programme. 19 Between April 2011 and January 2017, 94 CDC/CRMLN survey samples were analysed using 43 survey runs. Although the CDC/CRMLN survey is now conducted only for TLG measurement, the reference value of TG-GB estimated using the CA-RMP was listed for 56 survey samples in the CDC report until January 2016. By using Bland–Altman analysis, we attempted to assess the difference between our practical method and the CDC reference method for both TLG and TG-GB. As for TLG, the average %bias was –0.26%, and the area of the limits of agreement was narrow, indicating that the difference between the two methods was acceptable, and that the Osaka practical method can be used as an alternative to the CDC reference method. Conversely, as for TG-GB, the average %bias was –3.15%, and the area of the limits of agreement was wider; hence, we failed to obtain conclusive proof for the equivalence of the two methods for TG-GB analysis. This difference could be attributed to the fact that the CA-RMP for TG-GB analysis involves the elimination of free glycerol by using silicic acid for non-specific adsorption, 18 which is not included in our method. As our method for free glycerol measurement has sufficient specificity, owing to the use of the GC-IDMS measurement system, we attributed the difference in free glycerol concentrations obtained using the two TG-GB methods to the different specificities of the free glycerol adsorption process in the CA-RMP.
Conclusion
We developed a practical reference method based on GC-IDMS analysis for the measurement of both TLG and free glycerol, which enabled us to calculate the TG-GB in human serum. This procedure involved a one-step derivatization process in measuring both TLG and free glycerol and in calculating TG-GB, with excellent precision and accuracy comparable to those of conventional methods. Therefore, the traceability of the glycerol elimination method for TG-GB measurement in manufacturer or clinical laboratories can be verified by comparing this practical measurement procedure for both TLG and free glycerol.
Supplemental Material
sj-pdf-1-acb-10.1177_0004563220921884 - Supplemental material for A method for measuring glycerol-blanked triglyceride concentrations by using gas chromatography-isotope dilution mass spectrometry
Supplemental material, sj-pdf-1-acb-10.1177_0004563220921884 for A method for measuring glycerol-blanked triglyceride concentrations by using gas chromatography-isotope dilution mass spectrometry by Isao Koyama, Hironori Imano, Masakazu Nakamura, Akihiko Kitamura, Masahiko Kiyama, Yoshihiro Miyamoto and Hiroyasu Iso in Annals of Clinical Biochemistry
Supplemental Material
sj-pdf-2-acb-10.1177_0004563220921884 - Supplemental material for A method for measuring glycerol-blanked triglyceride concentrations by using gas chromatography-isotope dilution mass spectrometry
Supplemental material, sj-pdf-2-acb-10.1177_0004563220921884 for A method for measuring glycerol-blanked triglyceride concentrations by using gas chromatography-isotope dilution mass spectrometry by Isao Koyama, Hironori Imano, Masakazu Nakamura, Akihiko Kitamura, Masahiko Kiyama, Yoshihiro Miyamoto and Hiroyasu Iso in Annals of Clinical Biochemistry
Footnotes
Acknowledgements
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Guarantor
IK.
Contributorship
MN and HI researched literature and conceived the study. HI analysed data and contributed the scientific discussion. AK, MK and YM contributed to the scientific discussion. IK was involved in protocol development, performed experiments, analysed the data and wrote the first draft of the article. All authors reviewed and edited the article and approved the final version of the article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
