Abstract
Background
There are limited data on performance of biomarkers to predict pre-eclampsia (PE) in high-risk women. This study investigated the ability of FABP4, PAPP-A, PlGF, sFlt-1 and sEng to predict PE in a high-risk group.
Methods
Non-fasting samples were analysed at 11 + 0–13 + 6 (V1) and 19 + 0–21 + 6 weeks (V2) (n = 195). Logistic regression models were determined. Area under (AUC) the receiver operating characteristic (ROC) curve analysis was performed. The added value of biomarkers to clinical characteristics for PE prediction was quantified using integrated discrimination improvement (IDI) and net reclassification improvement (NRI) indices.
Results
Prevalence of PE was 12%. Lower concentrations of sFlt-1:PlGF (V1) and PlGF and PlGF:sEng (V2) were seen in women who developed PE. Controlling for baseline characteristics (V1), a doubling of sFlt-1 (pg/mL) (median 896.0, IQR 725.5–1097.0) and sFlt-1:PlGF (median 21.2, IQR 14.7–32.3) was associated with reduction in odds of PE (OR 0.20, 95% CI 0.06–0.65, P = 0.007 and OR 0.48, 95% CI 0.25–0.92, P = 0.04). Addition of sFlt-1 and sFlt-1:PlGF to baseline characteristics non-significantly improved AUC (0.74) (AUC 0.77, P = 0.40 and 0.76, P = 0.39). NRI and IDI analyses confirmed added clinical utility of sFlt-1 (NRI = 0.539, P = 0.01 and IDI = 0.052, P = 0.03). In V2, doubling of PlGF:sEng (median 71.9, IQR 47.0–102.8) was associated with reduction in the risk of PE (OR 0.56, 95% CI 0.35–0.98, P = 0.04). The addition of PlGF:sEng to baseline characteristics non-significantly improved AUC from 0.78 to 0.82 (P = 0.25) and improved reclassification of cases (NRI = 0.682, P = 0.002).
Conclusions
Screening tests incorporating first trimester sFlt-1 and second trimester PlGF:sEng have potential to aid PE prediction in high-risk pregnancies.
Keywords
Introduction
Pre-eclampsia (PE) is a leading cause of maternal and perinatal morbidity and mortality, affecting approximately 2% of all pregnant women. 1 Women with obesity, diabetes mellitus (DM), hypertensive disease, renal disease, a history of intrauterine growth restriction (IUGR), thrombophilia or autoimmune disease are at increased risk of developing PE, 2 with reported incidences of up to 20% in high-risk populations. 3
Over the last decade, growing evidence suggests that an imbalance between angiogenic factors, such as placental like growth factor (PlGF), anti-angiogenic factors, soluble Fms-like tyrosine kinase 1 (sFlt-1) and soluble endoglin (sEng), plays a key role in the pathogenesis of PE.4,5 In addition, altered concentrations of pregnancy-associated plasma protein-A (PAPP-A)6,7 and fatty acid-binding protein-4 (FABP4) 8 have been linked with the development of PE. To date, few studies have examined the role of maternal serum biomarkers for prediction of PE in high-risk pregnancies. In addition, while it is recognized that serum biomarker concentrations are influenced by maternal risk factors, 9 little is known about the impact of these high-risk obstetric co-morbidities on biomarker performance in models for prediction of PE.
An accurate screening test for PE incorporating serum biomarkers in a high-risk obstetric cohort has the potential to improve clinical management of these pregnancies. Furthermore, identification of pregnancies at greatest risk of PE would enable clinicians to identify and facilitate appropriate pathways of antenatal care, effect early prevention strategies in pregnancy and ensure timely management of complications.
Against this background, the PREDICT study (prediction of PE in high-risk women) aimed to investigate the ability of five maternal serum biomarkers: FABP4, PAPP-A, PlGF, sFlt-1 and sEng to predict PE in high-risk women.
Materials and methods
Subjects were high-risk women recruited at 11 + 0–13 + 6 weeks’ gestation from a tertiary referral centre between December 2014 and August 2016. PREDICT was a longitudinal observational cohort study designed to evaluate the clinical utility of whole volume-derived three-dimensional power Doppler placental vascularization indices (PVIs) and maternal serum biomarkers to predict PE in high-risk women in the first and second trimester of pregnancy. Eligible women aged ⩾18 years with singleton pregnancies were recruited to one of four high-risk groups: (1) diabetes (pre-existing type 1 and type 2 DM) (2) obesity (booking body mass index [BMI] >35 kg/m2) (3) hypertension (essential hypertension, an obstetric history of PE, IUGR or renal disease) (4) autoimmune (known thrombophilia or autoimmune disease). Written informed consent was obtained from all women. The Office for Research Ethics Committees NI (ORECNI) provided ethical approval (14/NI/1068).
The primary outcome was PE, defined as hypertension after 20 weeks’ gestation and the co-existence of one or more of the following new-onset conditions: (1) proteinuria (2) maternal organ dysfunction or (3) uteroplacental dysfunction (i.e.) fetal growth restriction, in accordance with the International Society for the Study of Hypertension in Pregnancy (ISSHP) guidelines. 10 The diagnosis of PE was independently confirmed by three clinicians.
Non-fasting peripheral venous blood samples were collected from participants at 11 + 0–13 + 6 (visit 1) and 19 + 0–21 + 6 weeks’ gestation (visit 2). Samples were centrifuged (3000 r/min for 15 min) and stored at −80°C. Serum concentrations of five biomarkers (FABP4, PAPP-A, PlGF, sFlt-1, sEng) were measured at both time points. Samples were batch-analysed centrally in the Nutrition and Metabolism Laboratories, Centre for Public Health, Queen’s University Belfast. Analysis was performed according to the manufacturer’s recommendations. In order to minimize variation, all kits were from the same batch, and analysis of both participant samples was carried out on the same plate blinded to PE outcome.
Serum FABP4 and PAPP-A concentrations were measured using a commercially available ELISA kit (Biovendor, Modrice, Czech Republic) on a Triturus® automated immunoassay analyser (Grifols, Barcelona, Spain). For FABP4, inter-assay and intra-assay coefficient of variation (CVs) were 4.3% and 2.0%, respectively. Serum PAPP-A inter-assay and intra-assay CVs were 13.9% and 6.1%, respectively. Serum PlGF, sFlt-1 and sEng concentrations were also measured using a commercially available ELISA kit (R&D systems, Abingdon, United Kingdom) on a Triturus® automated immunoassay analyser (Grifols, Barcelona, Spain). Inter-assay and intra-assay CVs for PlGF were 11.5% and 1.9%, respectively. Serum sEng inter-assay and intra-assay CVs were 5.9% and 0.9%, and serum sFlt-1 inter-assay and intra-assay CVs were 7.9% and 1.8%, respectively.
Statistical analysis
All biomarker results were positively skewed and were therefore logarithmically transformed prior to further analysis. Values are reported as geometric mean and interquartile range (IQR). Comparison of biomarkers across groups was performed using one-way analysis of variance (ANOVA). Post hoc analysis using Duncan’s multiple range test was undertaken to determine which groups differed significantly.
Logistic regression analysis was performed to assess the association of the biomarkers with development of PE and a number of confounding variables were controlled for, including: study group, age, BMI, smoking status, aspirin use, parity, material deprivation of area of residence and mean arterial pressure (MAP). For this analysis, biomarkers were logarithmically transformed to base 2, so that odds ratios have the interpretation of multiplicative effects on the odds of PE associated with a doubling of the biomarker. Where appropriate, the area under (AUC) the receiver operating characteristic (ROC) curve was used to assess the ability of each serum biomarker and selected ratios to predict PE in high-risk women. Logistic regression analysis provided predicted probabilities of PE for a base model containing baseline maternal characteristics and for models obtained by the addition of each biomarker to the base model. These predicted probabilities were used to derive ROC curves, and the increase in the area under these curves was assessed for significance. 11
To quantify the additional value of maternal serum biomarkers to baseline clinical risk factors for prediction of PE, integrated discrimination improvement (IDI) and net reclassification improvement (NRI) indices were calculated. 12 These metrics incorporated predicted probabilities derived from logistic regression models calculating the probability of development of PE for each woman. 13 The NRI was calculated on a continuous, uncategorized basis (with no predefined PE risk categories). 12 Defining PE as the event, Pickering and Endre describe the NRI as the sum of NRI(events) and NRI(non-events). 13 It is interpreted as the proportion of women reclassified to a more appropriate risk category on addition of the biomarker of interest to the logistic regression model. In women who developed PE, if the addition of the biomarker results in more individuals being reclassified to a higher risk, then the NRI(events) is positive. For women who did not develop PE, if more women are assigned as lower risk, then the NRI(non-events) is positive. The IDI was defined as the average increase in predicted risk in women without PE added to the average decrease in predicted risk of PE in women without PE. 13 Statistical analysis was performed using SPSS version 21 (IBM Corp., Armonk, NY), Stata release 14 (StataCorp, College Station, TX) and the Hmisc package in R version 3.1.3 (R Core Team, Vienna, Austria).
Results
A total of 195 women were recruited to the PREDICT study. Analysis of biomarkers by primary outcome (PE) was performed at two time points during pregnancy; 11 + 0–13 + 6 weeks’ and 19 + 0–21 + 6 weeks’ gestation. The overall rate of PE was 12% (n = 23). There is variation in the number of results reported for each biomarker: (1) results were excluded if out of working range (OWR) even following repeat analysis at an appropriate dilution; (2) results were also excluded if there was analytic interference (e.g.) due to the presence of autoantibodies. Baseline maternal characteristics in women who developed PE and those who did not are presented in Table 1. No differences in age, BMI, ethnicity, gestational age at booking, full-time education, parity, smoking status, alcohol intake, social deprivation, aspirin use or study grouping were observed between the two groups. Table 2 summarizes geometric mean (IQR) results for all participants. At visit 1, mean sFlt-1:PlGF ratio was significantly lower in women who developed PE compared with those who did not. In the second trimester, mean PlGF and mean PlGF:sEng ratio were significantly lower in women who developed PE.
Analysis of maternal baseline characteristics by pre-eclampsia outcome.
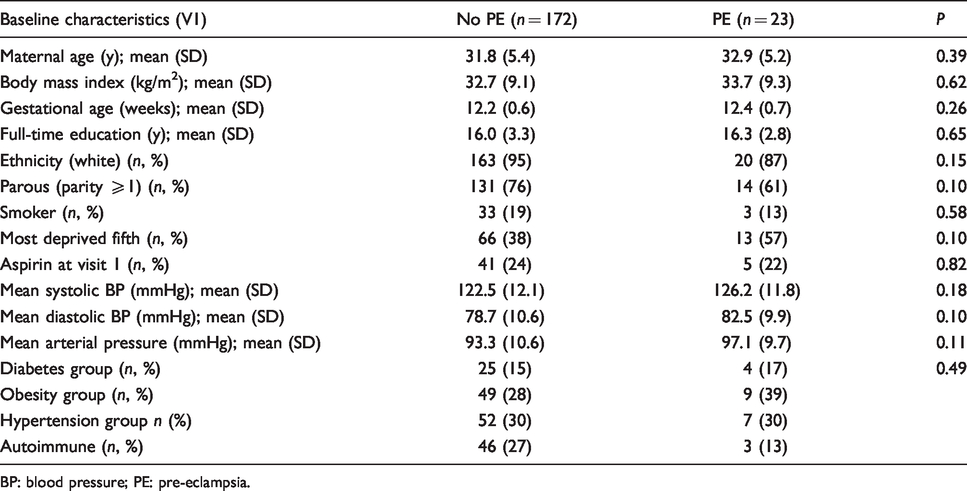
BP: blood pressure; PE: pre-eclampsia.
Serum biomarker concentrations across groups by pre-eclampsia outcome at 11 + 0–13 + 6 and 19 + 0–21 + 6 weeks’ gestation.
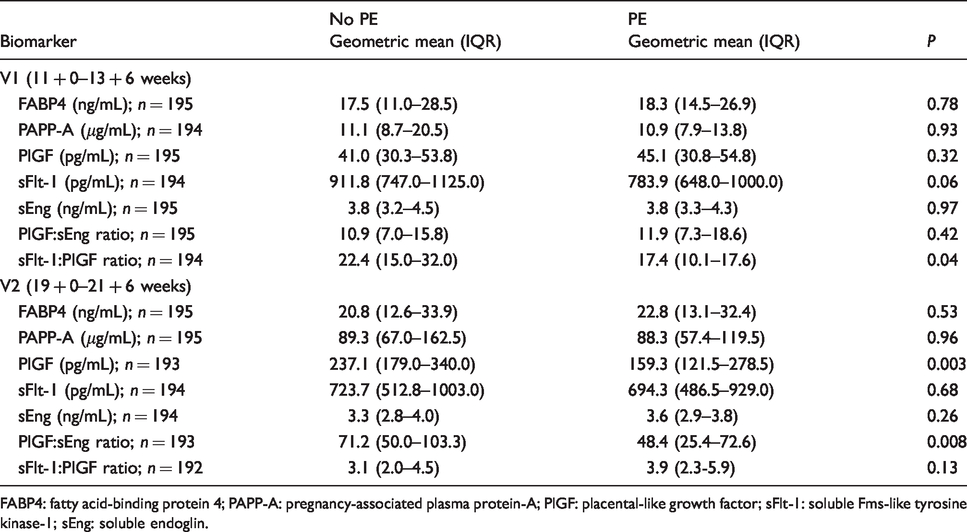
FABP4: fatty acid-binding protein 4; PAPP-A: pregnancy-associated plasma protein-A; PlGF: placental-like growth factor; sFlt-1: soluble Fms-like tyrosine kinase-1; sEng: soluble endoglin.
The relationship between serum biomarkers and prediction of PE was further examined using logistic regression models. The unadjusted and adjusted odds of developing PE controlled for baseline maternal characteristics (study group, age, BMI, smoking status, aspirin use, parity, material deprivation and MAP) are summarized in Table 3. After adjustment, significant associations remained between sFlt-1 and the ratio of sFlt-1:PlGF in the first trimester. A doubling in mean sFlt-1 was associated with an 80% reduction in the odds of developing PE (OR 0.20, 95% CI 0.06–0.65, P = 0.007), while a doubling in sFlt-1:PlGF ratio was associated with a 52% reduction in the odds of developing PE (OR 0.48, 95% CI 0.25–0.92, P = 0.03). In the second trimester, only the PlGF:sEng ratio was a useful predictor of PE after controlling for baseline maternal characteristics (OR 0.56, 95% CI 0.35–0.98, P = 0.04 per doubling).
Summary of logistic regression models for unadjusted and adjusted analysis of serum biomarkers at visit 1 (11 + 0–13 + 6 weeks) and visit 2 (19 + 0–21 + 6 weeks).
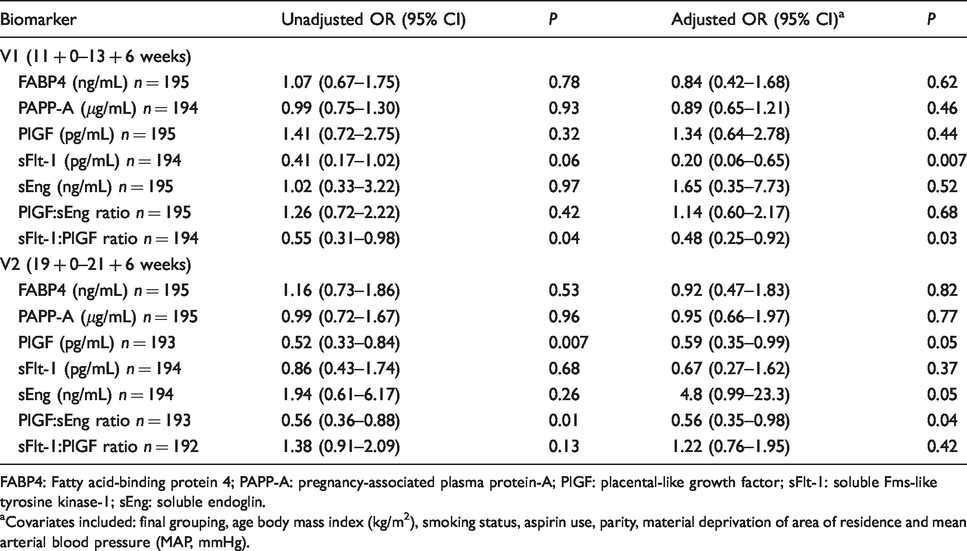
FABP4: Fatty acid-binding protein 4; PAPP-A: pregnancy-associated plasma protein-A; PlGF: placental-like growth factor; sFlt-1: soluble Fms-like tyrosine kinase-1; sEng: soluble endoglin.
aCovariates included: final grouping, age body mass index (kg/m2), smoking status, aspirin use, parity, material deprivation of area of residence and mean arterial blood pressure (MAP, mmHg).
Additional ROC analysis of the biomarkers which remained significant predictors of PE in adjusted logistic regression models was performed. This enabled the assessment of serum biomarkers to predict PE when added to baseline maternal characteristics (Table 4). In the first trimester, the AUC for baseline maternal characteristics (0.74) improved modestly with the addition of sFlt-1 and sFlt-1:PlGF (AUC 0.77 and 0.76, respectively); however, comparison of ROC curves was not significant (P = 0.40, P = 0.39, respectively). At 19 + 0–21 + 6 weeks, an improvement in AUC (0.78) for a model incorporating only baseline maternal characteristics was demonstrated with the addition of the PlGF:sEng ratio (AUC 0.82); however, comparison of ROC curves was not statistically significant (P = 0.25).
Performance of selected biomarker(s) for prediction of pre-eclampsia.
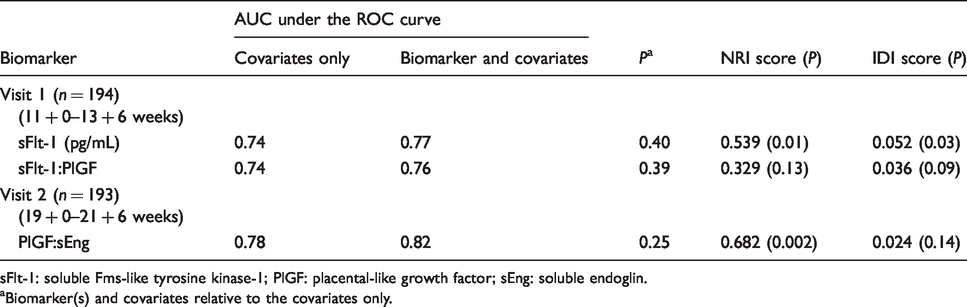
sFlt-1: soluble Fms-like tyrosine kinase-1; PlGF: placental-like growth factor; sEng: soluble endoglin.
aBiomarker(s) and covariates relative to the covariates only.
To determine the ability of these selected biomarkers to improve prediction of PE in addition to maternal baseline characteristics, NRI and IDI metrics were calculated (Table 4). When applied to a large group of patients, the NRI and IDI statistics indicate improved performance of the predictive model with the addition of biomarkers. In the first trimester, the NRI statistic demonstrated that the addition of sFlt-1 to baseline maternal characteristics significantly increased correct classification of cases of PE (NRI = 0.539, P = 0.01) and improved discrimination between cases and non-cases (IDI = 0.052, P = 0.03). These metrics did not demonstrate a statistically significant clinical benefit in screening with sFlt-1:PlGF for prediction of PE (NRI = 0.329, P = 0.13 and IDI = 0.036, P = 0.09). In the second trimester, only the NRI statistic demonstrated that the addition of PlGF:sEng to baseline maternal characteristics significantly improved classification of cases (NRI = 0.682, P = 0.002).
An earlier publication from our group reports the ability of PVIs to predict pre-eclampsia in high-risk women. 14 Additional analysis was undertaken to derive combined screening models for the prediction of PE in the first and second trimester of pregnancy. As previously described, logistic models were generated using PE as the primary outcome. Values of PVIs were standardized to a mean of zero and a standard deviation (SD) of one. The resulting standardized odds ratios obtained by logistic regression indicate the multiplicative effect on the odds of PE associated with a 1 SD change in the PVI. This approach facilitates comparison of the odds ratios for each PVI. In addition, as biomarker concentrations were positively skewed, logarithmic transformation was followed by standardization to provide standardized odds ratios in the logistic regression analysis. A final first trimester screening model combining vascularization flow index (VFI), sFlt-1 and baseline maternal characteristics demonstrated that a 1 standard deviation (SD) increase in VFI was associated with a 67% reduction in the odds of developing PE (OR 0.33, 95% CI 0.14–0.78, P = 0.01), while a 1 SD change in the logarithmically-transformed concentration of sFlt-1 (pg/mL) reduced the odds of developing PE by 54% (OR 0.46, 95% CI 0.24–0.87, P = 0.02). Addition of sFlt-1 and VFI to baseline characteristics increased AUC non-significantly from 0.74 to 0.83, P = 0.05. However, NRI and IDI analyses confirmed added clinical utility of VFI and sFlt-1 (NRI 0.709, P = 0.0006, IDI 0.103, P = 0.003). The final combined screening model for PE in the second trimester incorporated measurement of FI in addition to baseline maternal characteristics. No improvement in prediction of PE was noted with the addition of PlGF:sEng.
The differences in biomarker concentrations across study groups are shown in Table 5. Additional discussion of the analysis (Duncan’s test) is provided in a supplemental appendix (SA 1). Serum concentrations of FABP4 were highest in the obese group compared with the other groups at both study visits. Significant differences in first trimester concentrations of sFlt-1 and sEng were also noted between the obesity and autoimmune group. No significant differences in first trimester concentrations of PAPP-A and PlGF were noted between groups. Lowest mean concentrations of PlGF:sEng and highest concentrations of sFlt-1:PlGF were observed in the autoimmune group at visit 1, which differed from all other groups. In the second trimester, geometric mean (IQR) concentration of PAPP-A was lowest in the obese group (59.8 [42.7–101.2] μg/mL), and this was significantly different from all other groups. Differences in mean concentrations of PlGF were also noted between the obesity and hypertension groups (184.1 [130.0–276.5] vs. 260.7 [194–369.0] pg/mL) in comparison to all other groups. Mean concentrations of sFlt-1 and sEng were significantly higher in the autoimmune group, and these results were significantly different from other groups. Lowest mean concentrations of PlGF:sEng and highest concentrations of sFlt-1:PlGF were observed in the autoimmune group at visit 2; significant differences were noted between the hypertension and autoimmune subgroups. Given the significantly higher concentrations of sFlt-1 and sEng in the autoimmune group, the logistic regression analysis was repeated without the autoimmune group to investigate the impact of this group on the performance of the final model. In the first trimester model, including sFlt-1, no difference was observed (unadjusted OR 0.35, 95% CI 0.13–0.97, P = 0.04, adjusted OR 0.16 [0.04–0.58], P = 0.005). However, the second trimester model, including PlGF:sEng, became non-significant (unadjusted OR 0.77, 95% CI 0.49–1.2, P = 0.24, adjusted OR 0.84 [0.52–1.37], P = 0.48) when the autoimmune group was excluded.
Comparison of serum biomarker concentrations across groups at visit 1 and visit 2 using ANOVA (one-way analysis of variance).
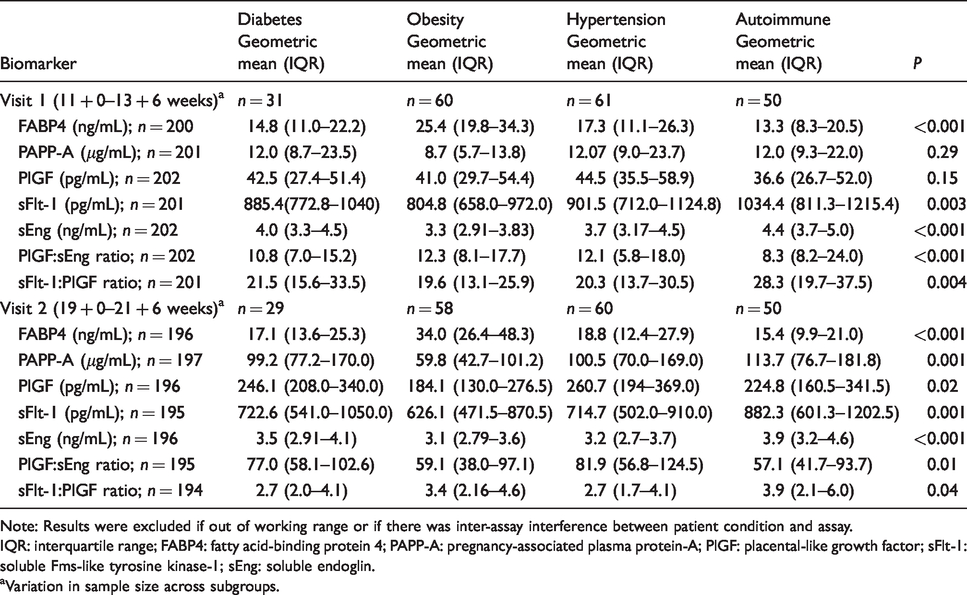
Note: Results were excluded if out of working range or if there was inter-assay interference between patient condition and assay.
IQR: interquartile range; FABP4: fatty acid-binding protein 4; PAPP-A: pregnancy-associated plasma protein-A; PlGF: placental-like growth factor; sFlt-1: soluble Fms-like tyrosine kinase-1; sEng: soluble endoglin.
aVariation in sample size across subgroups.
Supplemental Tables (S1 to S4) summarize biomarker concentrations by PE outcome for each high-risk group. A number of significant findings were apparent from this subgroup analysis. Lower concentrations of second trimester PlGF were detected in women with DM who subsequently developed PE. In addition, mean concentrations of sFlt-1 were significantly lower at visit 1 in obese women who developed PE. In the autoimmune group, concentrations of PlGF:sEng were significantly lower in women who developed PE at both time points.
Discussion
PE is a highly significant and clinically relevant global health problem, and development of an effective screening test for at-risk women remains elusive. PREDICT addresses prediction of PE in a high-risk obstetric cohort using maternal serum biomarkers.
Significantly lower first trimester concentrations of sFlt-1:PlGF were seen in women who subsequently developed PE. However, first trimester logistic regression models controlling for baseline maternal characteristics demonstrated an association between sFlt-1, sFlt-1:PlGF and development of PE. Additional NRI and IDI analysis confirmed the added clinical utility of first trimester sFlt-1 for prediction of PE. These analyses support a potential role for sFlt-1 as a first trimester screening biomarker for PE in high-risk populations.
Concentrations of sFlt-1 remain fairly constant in the maternal circulation until 33–36 weeks’ gestation and are known to increase approximately five weeks before the onset of PE.15–17 This expected rise was not apparent within our study at either time point; however, this finding may be attributed to the earlier gestation at which sFlt-1 was sampled. Additional sampling of sFlt-1 at a later gestation could provide additional information. Measurement of sFlt-1 for prediction of PE remains controversial; a number of studies have reported no difference in first trimester serum concentrations of sFlt-1 in women who develop PE.18–20 Specifically, little is known about the role of first trimester sFlt-1 as a predictive maker for PE in high-risk populations. In a prospective observational study of a high-risk population of women (n = 226), sFlt-1 did not improve prediction of PE in multivariate models combining sFlt-1, PlGF and uterine artery Doppler measurements. 21 By contrast, a recent prospective nested case–control study of low-risk participants (n = 740) demonstrated significantly higher concentrations of first trimester sFlt-1 in women who developed PE (n = 44). 22 However, first trimester sFlt-1 was not reported as a significant predictor variable for PE in logistic regression models. 22 Further research investigating the ability of first trimester sFlt-1 to predict PE within a high-risk cohort is warranted to fully elucidate findings from the PREDICT study.
To date, few studies have evaluated the effectiveness of the sFlt-1:PlGF ratio as an early predictive marker of PE in the first trimester. A retrospective case–control study measuring first trimester sFlt-1:PlGF in women who developed late-onset PE detected no significant difference between groups. 23 In addition, previous studies evaluating sole measurement of sFlt-1 and PlGF have yielded contradictory results.24–26 This is reflected in a recent systematic review investigating the relationship between sFlt-1:PlGF and the development of PE. The review included 15 studies (n = 11,779) with a gestational age range between 6 and 24 weeks. 27 The inclusion criteria did not discriminate between high- and low-risk participants. Overall, serum sFlt-1:PlGF was elevated in women who later developed PE; however, this finding was inconsistent. In both high- and low-risk populations, increased ratios were observed when the test was performed later in pregnancy. This finding is consistent with the expected trend of sFlt-1:PlGF, which typically decreases during the first trimester and increases in the second trimester. 28 Current guidance from the National Institute for Health and Care Excellence (NICE) supports the use of PlGF and sFlt-1:PlGF immunoassay devices alongside standard clinical assessment as diagnostic tests to help rule-out PE in women presenting with suspected PE between 20 + 0–34 + 6 weeks’ gestation. 29
A potential explanation for the low first trimester concentrations of sFlt-1:PlGF observed in women who developed PE, could be that the expected reduction in first trimester serum concentration of PlGF was not evident;15,30–32 in addition, sFlt-1 concentrations were non-significantly lower in women with PE at the time of visit 1. Powers et al. previously described differing patterns of PlGF concentrations in women with PE; in particular that low first trimester PlGF concentrations are more likely to be associated with early-onset, severe disease; therefore, only identifying a subset of women with PE. 26 Given our sample size, it was not practical to subdivide PE outcome according to severity; therefore, we cannot comment on whether the initial observations noted at visit 1 resulted in similar clinical outcomes to those previously reported. 26
In the second trimester, significantly lower concentrations of PlGF and PlGF:sEng were noted in women who later developed PE.15,26,33–37 Following adjustment for confounding factors in logistic models, the PlGF:sEng ratio remained a significant predictor of PE. Additional analysis demonstrated a significant improvement in classification of cases and non-cases of PE using the NRI statistic. Our results are supported by findings from Chaiworapongsa et al. who conducted a retrospective cohort study investigating the role of PlGF:sEng as a prognostic test for women suspected of having PE between 20 and 36 weeks’ gestation (n = 87). 38 The ratio had a 76% sensitivity and 91% specificity for prediction of severe PE. In women with PE presenting prior to 34 weeks’ gestation, the ability of PlGF:sEng to predict delivery within 14 days was also demonstrated. These results indicate a promising role for PlGF:sEng both as a screening and diagnostic biomarker for PE, and in supporting clinical decision-making for at-risk women.
Comparison of maternal serum biomarkers across groups revealed a number of striking differences. At both study visits, significantly higher concentrations of FABP4 were noted in the obesity group. FABP4 is known to be increased in overweight and obese subjects 39 ; however, little research exists to clearly ascertain the role of FABP4 for prediction of PE in high-risk women. Although not statistically significant, mean serum concentrations of FABP4 were higher in women with PE in both the first and second trimesters reflecting previous work from Scifres et al. in a low-risk population. 8 Wotherspoon et al. previously reported elevated concentrations of FABP4 in women with type 1 DM who developed PE 40 ; however, subgroup analysis did not replicate this trend in the PREDICT study. More recently, elevated second trimester FABP4 has also been shown to independently predict gestational hypertension and PE in women with gestational diabetes mellitus (OR 1.15, 95% CI 1.00–1.27, P = 0.045). 41 Given the heterogeneity of participants in the PREDICT study and potential for differing pathways for development of PE, it may not have been possible to demonstrate the differences between groups. Additionally, the PREDICT study was not specifically powered to detect differences in FABP4 concentrations in relation to PE across groups.
A number of additional findings were demonstrated within the obese cohort recruited to the PREDICT study. Mean concentrations of sFlt-1 were lowest at both sampling points, and first trimester concentrations were significantly lower in obese women who developed PE. This finding supports an association between maternal weight gain and reduction in sFlt-1 concentrations previously reported in the literature.42,43 In the second trimester, lowest PlGF concentrations were seen in the obesity group. Maynard et al. reported a similar finding in an observational cohort study measuring PlGF concentrations at 23–27, 28–31 and 23–35 weeks’ gestation; mean concentrations of PlGF were significantly lower in obese women (P = 0.04, P = 0.008, P = 0.003, respectively). 9 PlGF decreases with maternal weight gain and in the presence of type 1 or type 2 DM requiring treatment with insulin.44,45 Subgroup analysis also revealed significantly lower second trimester concentrations of PlGF in women with DM who developed PE. Corroborating evidence from the available literature, sEng concentrations were lowest in the obesity group at both time points.46,47 This analysis highlights the variation in performance of maternal serum biomarkers, particularly in the obese population. Data from the FINNPEC cohort (Finnish Genetics of Pre-eclampsia Consortium) demonstrate that in women with PE, increased maternal BMI is associated with lower concentrations of sFlt-1 and sEng throughout pregnancy (P = 0.004, P = 0.008, respectively). 48 Given the increasing prevalence of maternal obesity, these women are an important group requiring further study. There is a need for development of custom reference ranges in the obese population to enable accurate interpretation and performance of maternal biomarkers within screening models for PE. Future research must focus on better understanding of the mechanisms underlying PE and their impact on biomarker concentrations within different high-risk subgroups to enable refinement of existing screening methods.
In the autoimmune group, sFlt-1 and sEng concentrations were significantly higher at both study visits. In addition, biomarker ratios in the autoimmune group at both study visits were significantly different from the other groups. PlGF:sEng was lowest and sFlt-1:PlGF was highest at both visits. The relationship between autoimmune disease and performance of maternal serum biomarkers is poorly defined. Data from the PROMISSE study (Predictors of Pregnancy Outcome: Biomarkers in APLS and SLE) showed that in cases of SLE (systemic lupus erythematous) and APLS (antiphospholipid syndrome), concentrations of sFlt-1, PlGF and sEng at 12–15 weeks’ and 16–19 weeks’ gestation were markedly altered in those who went on to develop adverse pregnancy outcomes (APO). 49 In women who developed PE <34 weeks’ gestation, concentrations of PlGF were reduced and concentrations of sFlt-1 and sEng were increased in comparison to control subjects. 49 Leanos-Miranda et al. reported similar findings among women with SLE who developed PE. The group noted lower PlGF concentrations and higher sFlt-1 and sEng concentrations and a higher sFlt-1:PlGF ratio than normotensive pregnancies. These findings were significant from 12 weeks’ gestation in women who developed both early-onset (<34 weeks, P ⩽ 0.003) or late-onset PE (⩾34 weeks, P ⩽ 0.02). 50 Rodríguez-Almaraz et al. also demonstrated significantly higher sFlt-1:PlGF ratio at 24–29 weeks’ gestation in women with APLS or SLE who had adverse obstetric outcomes (PE or IUGR). 51 A study from Tsiakkas et al. investigating the effect of maternal characteristics on sFlt-1 concentrations in each trimester 43 included a small number of patients with SLE and APLS in the analysis; however, no significant impact of these conditions on sFlt-1 concentrations was reported. 43
The PREDICT study has a number of strengths including compliance with clearly defined inclusion and exclusion criteria, ensuring that pregnancies were carefully characterized prior to enrolment. There was a low attrition rate and sample size was achieved. The primary outcome (PE) was well defined and independently verified by three clinicians. 10 A detailed and comprehensive statistical analysis strategy enabled construction of screening models involving potential predictor variables for PE. There are a number of limitations; in particular, the lower than anticipated rate of PE (12%) which reduced the power of the study. Sample size was calculated on the assumption of a 20% incidence of PE. 3 The reduced incidence of PE may have been due to the influence of prevention strategies, such as aspirin use or availability of specialist care within a tertiary setting. Overall, the study was not powered to allow valid analysis of individual subgroups and so we have avoided subgroup comparisons. In addition, the numbers within each of the subgroups were relatively small, as some women, such as those with diabetes, were difficult to recruit. Serum samples were also batch analysed within a research environment following completion of recruitment. Further research is required to validate the performance of sFlt-1 and PlGF:sEng in a clinical setting.
In summary, the PREDICT study demonstrates a definite role for measurement of maternal serum biomarkers in screening for PE within a high-risk cohort. Chiefly, the PREDICT study highlights the potential of first trimester sFlt-1 and second trimester PlGF:sEng as predictive biomarkers for PE in high-risk women.
Supplemental Material
ACB894022 Supplemental Material1 - Supplemental material for The role of biomarkers in predicting pre-eclampsia in high-risk women
Supplemental material, ACB894022 Supplemental Material1 for The role of biomarkers in predicting pre-eclampsia in high-risk women by Kelly-Ann Eastwood, Alyson J Hunter, Christopher C Patterson, David R Mc Cance, Ian S Young and Valerie A Holmes in Annals of Clinical Biochemistry
Supplemental Material
ACB894022 Supplemental Material2 - Supplemental material for The role of biomarkers in predicting pre-eclampsia in high-risk women
Supplemental material, ACB894022 Supplemental Material2 for The role of biomarkers in predicting pre-eclampsia in high-risk women by Kelly-Ann Eastwood, Alyson J Hunter, Christopher C Patterson, David R Mc Cance, Ian S Young and Valerie A Holmes in Annals of Clinical Biochemistry
Footnotes
Acknowledgements
We wish to thank all the women who took part in the PREDICT study for their involvement. The authors also wish to thank Dr Sarah Gilchrist and Ms Christine Belton, Nutrition and Metabolism Laboratory, Queen’s University Belfast, for their assistance with the biomarker analysis.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by a Health and Social Care Research and Development Award from the Public Health Agency, Northern Ireland (EAT/4904/13)
Ethical approval
Ethical approval was obtained from Office for Research Ethics Committees Northern Ireland. ORECNI: 14/NI/1068.
Guarantor
KAE.
Contributorship
All authors accept responsibility for the content of this article. KAE wrote the article, collected the data and reviewed and edited the article. AJH, VAH, DRM and ISY contributed to the discussion, reviewed and edited the article. CCP oversaw statistical design and conduct of the research and reviewed and edited the article.
Supplemental material
Supplemental material is available for this article online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
