Abstract
Background
Pregnancy-associated plasma protein A (PAPPA) is an emerging biomarker used in various medical fields but has yet to be evaluated in critical care medicine. This study evaluates the value of PAPPA as a biomarker in predicting myocardial dysfunction and 28-day mortality in patients with severe sepsis.
Methods
Serum concentrations of PAPPA and traditional cardiac biomarkers including cardiac troponin I (cTnI) and B-type natriuretic peptide (BNP) were measured on admission in 118 severely septic patients. The value of PAPPA for the diagnosis of sepsis-related myocardial dysfunction (SRMD) and for the prediction of 28-day mortality was subsequently evaluated using statistical methods.
Results
PAPPA was elevated (≥4.5 ng/mL) in 76 (64.4%) patients; patients with elevated PAPPA were more likely to have SRMD (76.3% vs. 38.1%, P < 0.001). By comparison of the area under the curve (AUC) in receiver operating characteristics analysis, PAPPA had comparable value (AUC 0.661, P = 0.003) to conventional biomarkers (BNP: AUC 0.699, P < 0.001; cTnI: AUC 0.647, P = 0.007) in the diagnosis of SRMD and offered superior value (AUC 0.771, P < 0.001) over them (all AUC<0.7, P > 0.05) in the prediction of 28-day death. Increased PAPPA (≥5.4 ng/mL) was associated with lower 28-day survival (χ2 = 19.78, P < 0.001) and independently predicted 28-day mortality in septic patients.
Conclusions
Serum PAPPA concentration frequently increases in patients with severe sepsis and appears to be associated with SRMD. PAPPA can be used as a novel biomarker for the diagnosis of SRMD and the prediction of outcomes in critically ill patients.
Introduction
Sepsis-related myocardial dysfunction (SRMD) characterized by transient depression in left ventricular function in septic patients is an emerging challenge in clinical practice. 1 It is reported that up to 50% of septic patients may have SRMD that contributes to an elevated mortality rate compared to those without cardiovascular impairment. 2 However, early recognition of SRMD is not easy for clinicians partly due to the non-specificity of clinical signs and symptoms. Cardiac biomarkers such as cardiac troponins (cTns) and B-type natriuretic peptides (BNP) are commonly used tools for the early recognition of SRMD.3,4 However, these biomarkers may be affected by several conditions including severity of the disease, coexisting organ dysfunction, multi-organ involvement or altered synthesis/clearance; thus, interpretation of them may be confounded and,5,6 in this regard, there is still a need for reliable markers.
Recent research suggests that pregnancy-associated plasma protein A (PAPPA) might have the potential as a candidate novel biomarker for SRMD. PAPPA belongs to the metzincin metalloproteinase superfamily and is expressed in a wide variety of reproductive and non-reproductive organs and cells.7,8 PAPPA relates to regulation of local insulin-like growth factor (IGF) action and pathophysiologic modulation in patients with renal impairment, asthma and acute coronary syndromes.9–11 Importantly, PAPPA elevation has been suggested to reflect instability of coronary and carotid plaque and has been proved to be promising in diagnosis of ischemic myocardial damages and prediction of cardiovascular events and death in patients with coronary artery diseases.12–15 Elevated PAPPA represents an enhanced local inflammatory state involving remodelling of subendothelial extracellular matrix (ECM) that contributes to atherosclerotic plaque fragility and rupture13,14,16 and is closely related to chronic inflammation and oxidative stress in uraemic and asymptomatic hyperlipidemic patients. 9 Therefore, PAPPA is not only a biomarker of myocardial damage with prognostic value but also can reflect inflammatory burden thus fulfills the criteria of ideal biomarker for sepsis.
In the present study, we aim to investigate the relationship between concentration of PAPPA and prevalence of SRMD and to evaluate the value of PAPPA for prediction of adverse outcomes in septic patients.
Materials and methods
Study population
One hundred and eighteen patients (males: 64, females: 54), aged 36–94 years, hospitalized in a 27-bed intensive care unit (ICU) of Zhejiang Hospital from September 2008 through October 2010, were enrolled when they met the criteria of severe sepsis or septic shock defined by the 2001 International Sepsis Definitions Conference. 17 Pregnant women; patients with pre-existing cardiovascular impairments including left ventricular dysfunction, dilated cardiomyopathy, valvular disease and prior coronary artery diseases determined by angiography; and patients with non-infectious shock, acute and end stage hepatic and renal disease and life expectancy less than 24 h were excluded from the study.
The baseline clinical characteristics including demographic data, underlying diseases and parameters of serum biochemical panel and arterial blood gas analysis of all patients were recorded on ICU admission. All patients underwent continuous electrocardiographic monitoring, evidence-based 6-h resuscitation and 24-h management bundles according to the Surviving Sepsis Campaign guidelines for the management of severe sepsis and septic shock. 18 The efficacy of therapy was based on 28-day survival from study enrolment. Twelve-lead electrocardiograms were taken within first 2 h, then every 6 h in the next 24 h, then every 12 h in the following days. Cardiac markers (cTnI, BNP and PAPPA) and an inflammatory marker (procalcitonin, PCT) 19 were measured on ICU admission. Severity of disease was quantified by the Acute Physiological and Chronic Health Evolution II (APACHE II) 20 and the Sequential Organ Failure Assessment (SOFA) score systems in all subjects at enrolment. 21 Both the need for and the timing of inotrope support defined as a continuous infusion of milrinone, dobutamine, dopamine (>5 µg kg−1 min−1), epinephrine, norepinepherine or vasopressin based on the professional guidelines 18 were recorded. All clinical events and outcomes were followed throughout the hospitalization. The follow-up was limited to the period of hospitalization.
This study was carried out according to the principles of the Declaration of Helsinki and approved by the Ethics Committee of Zhejiang Hospital. Written informed consent was obtained from all patients or their relatives, or legal guardians before enrolment.
Blood sampling and biochemical assays
The blood samples from the internal carotid vein were drawn for routine complete blood count and biochemical panel, cardiac biomarkers and PCT detection. Serum samples for detection of cTnI, PAPPA and PCT, and plasma samples for BNP were collected and stored prior to assay. Quantitative biochemical assays were performed as recently described by our group. 22 Briefly, complete blood count was tested using the Coulter LH 700 hematology system (Beckman Coulter Inc., USA); plasma BNP concentration by immunoassay with Triage BNP analyzer and matching kit (Biosite Inc., CA, USA); PCT by immunoluminometric assay (LUMI test PCT; BRAHMS GmbH, Berlin, Germany); cTnI by chemiluminescent immunoassay in a scintillation Beckman Access 2 Counter with matched reagent (AccuTnI Troponin I, Beckman Coulter Inc., USA). In addition, 0.5 mL of arterial blood was also obtained for instant blood gas analysis plus lactate detection using an automated Easystat Analyzer with matched reagent (Medica Co., USA). Cutoff values considered indicative of cardiac injury when equalled or exceeded were 100 pg/mL for BNP and 0.1 ng/mL for cTnI. Cutoff values for positivity were 2 ng/mL for PCT and 2 mmol/L for lactate. All measurements were done in triplicate; the mean value of the three time measurements was used for subsequent statistical analysis.
Serum PAPPA (non-complexed) concentration was detected by a sandwich enzyme-linked immunosorbent assay (ELISA) using commercially available kits (Kangchen Bioscience, Shanghai, China). The detection limit of PAPPA with sandwich-ELISA is 0.5–200 ng/mL; the total imprecision of the detection is 5.1–7.7%. A value ≥4.5 ng/mL represents abnormality according to the manufacturer. In order to minimize the possible stimulating effect of heparin products on PAPPA, the heparin-based pipeline maintenance was suspended for at least 1 h prior to blood sampling.
Transthoracic echocardiographic detection
For the recognition of SRMD, a transthoracic echocardiographic evaluation (GE vivid 7, USA) was performed on every subject within 24 h of enrolment and the left ventricular ejection fraction (LVEF) was visually assessed. Patients with LVEF less than 50% were labelled as cardiac dysfunction. 23
Statistical analysis
Categorical variables were analysed using the chi-square test. Continuous variables were compared using Student’s t-test or Mann-Whitney’s U test between groups, depending on distribution. The receiver-operating characteristic (ROC) curve was used to compare the powers of biomarkers as predictors of SRMD and 28-day mortality; the area under the curve (AUC) and the best cutoff value for each biomarker were calculated. Hazard ratios (HRs) and 95% confidence interval (CI) of potential predictors of 28-day death were identified by Cox proportional hazards models; significant variables in univariate model were then tested in a multivariate model performed as stepwise regressions with backward elimination to identify independent predictors. Cumulative survival was estimated by Kaplan–Meier method with the log-rank test. All statistical analyses were performed using SPSS for Windows 17.0 (SPSS Inc, Chicago, IL). A two-tailed P < 0.05 was considered significant.
Results
Baseline characteristics
Baseline characteristics of study population.
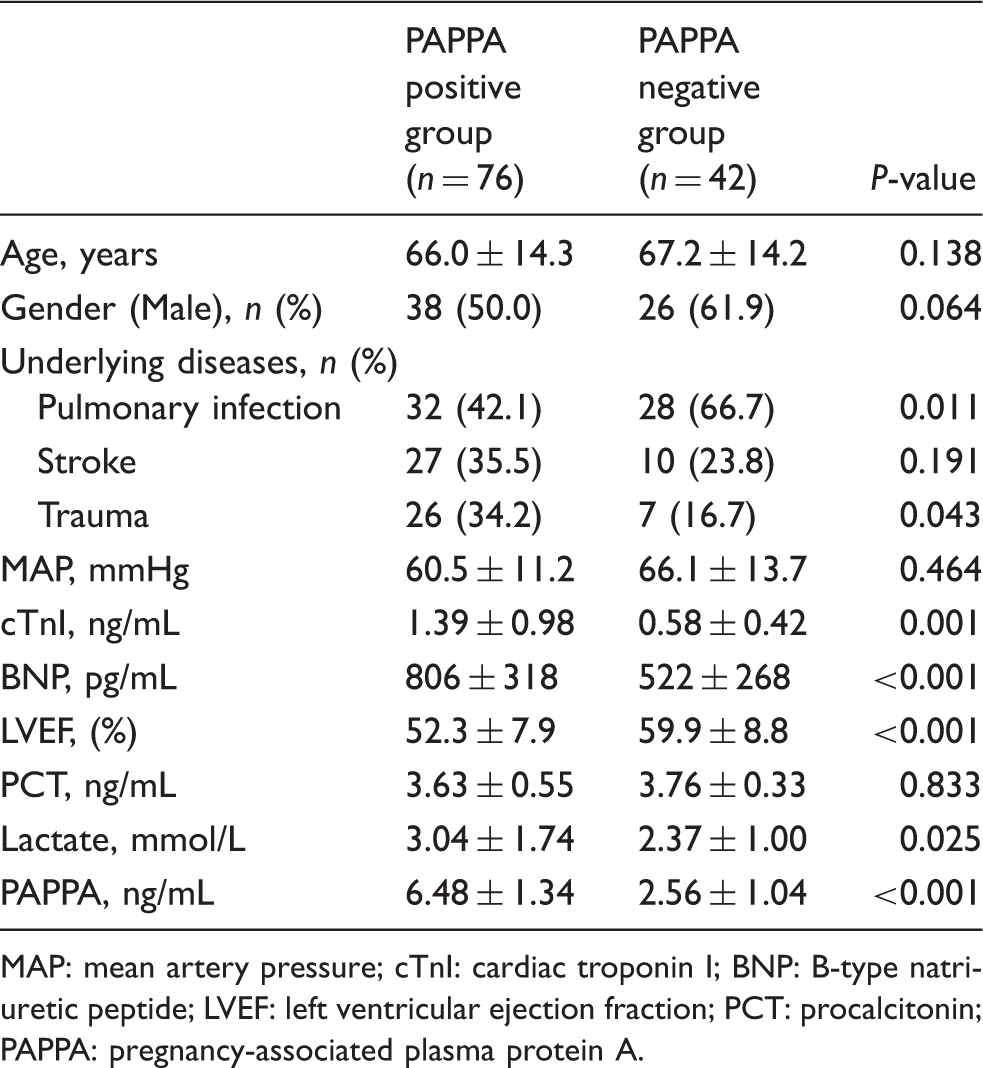
MAP: mean artery pressure; cTnI: cardiac troponin I; BNP: B-type natriuretic peptide; LVEF: left ventricular ejection fraction; PCT: procalcitonin; PAPPA: pregnancy-associated plasma protein A.
Severity of illness and clinical outcomes
Compared to PAPPA-negative group, the PAPPA-positive patients were more severe in illness presenting with higher APACHE II (20.7 ± 2.9 vs. 18.5 ± 2.0, P < 0.001) and higher SOFA score (11.5 ± 2.6 vs. 10.3 ± 2.2, P = 0.013); they were more likely to have cardiac dysfunction (76.3% vs. 38.1%, P < 0.001) and needed more mechanical ventilation (65.8% vs. 38.1%, P = 0.004); more PAPPA-positive patients died within 28 days (42.1% vs. 14.3%, P = 0.002) during hospitalization.
Correlations between PAPPA, BNP and cTnI
By bivariate correlation analysis, there were weak but statistically significant correlations between PAPPA and BNP (Pearson r = 0.393, P < 0.001), and cTnI (Pearson r = 0.349, P < 0.001).
Diagnostic value of PAPPA for SRMD
By ROC curve analysis, PAPPA appeared to have comparable power to traditional biomarkers (BNP and cTnI) in the diagnosis of SRMD. The AUC for PAPPA (0.661, P = 0.003) was similar to AUCs for BNP (0.699, P < 0.001) and cTnI (0.647, P = 0.007); at the best cutoff values for PAPPA (4.5 ng/mL), BNP (534 pg/mL) and cTnI (1.125 ng/mL), the sensitivity in diagnosis of SRMD was 0.81, 0.86 and 0.56, and the specificity was 0.57, 0.61, and 0.83, respectively. Detailed information was showed in Figure 1.
ROC curve analysis on the diagnostic value of PAPPA for SRMD compared with traditional cardiac biomarkers. PAPPA had comparable power (AUC 0.661, 95% CI 0.548–0.774, P = 0.003) to BNP and cTnI in the recognition of SRMD. The optimal cutoff value of PAPPA for diagnosis of SRMD was 4.5 ng/mL, with a sensitivity of 0.81 and specificity of 0.57.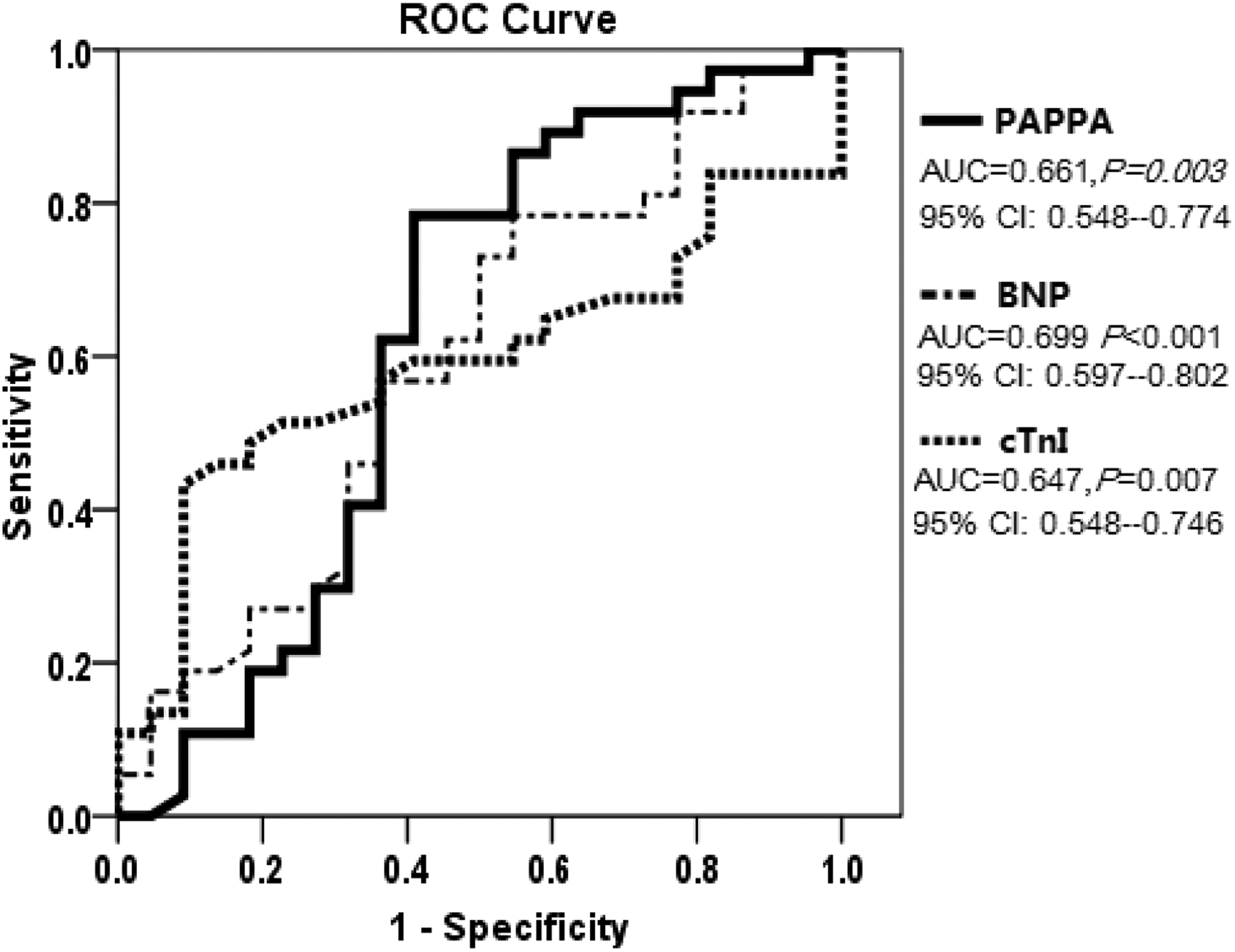
Prognostic value of PAPPA for 28-day mortality
ROC curve analysis shown in Figure 2 revealed that PAPPA offered superior power over conventional biomarkers in the prediction of 28-day mortality. AUC for PAPPA (0.771, P < 0.001) was higher than AUCs for BNP (0.568, P = 0.231) and cTnI (0.605, P = 0.065). The best cutoff value for PAPPA in the prediction of 28-day mortality was 5.4 ng/mL with a sensitivity of 0.79 and specificity of 0.65.
ROC curve analysis on the prognostic value of PAPPA for 28-day mortality compared to traditional cardiac biomarkers. PAPPA offered superior power (AUC 0.771, 95% CI 0.681–0.881) over BNP and cTnI in the prediction of 28-day mortality. The optimal cutoff value of PAPPA for prediction of 28-day mortality was 5.4 ng/mL, with a sensitivity of 0.79 and specificity of 0.65.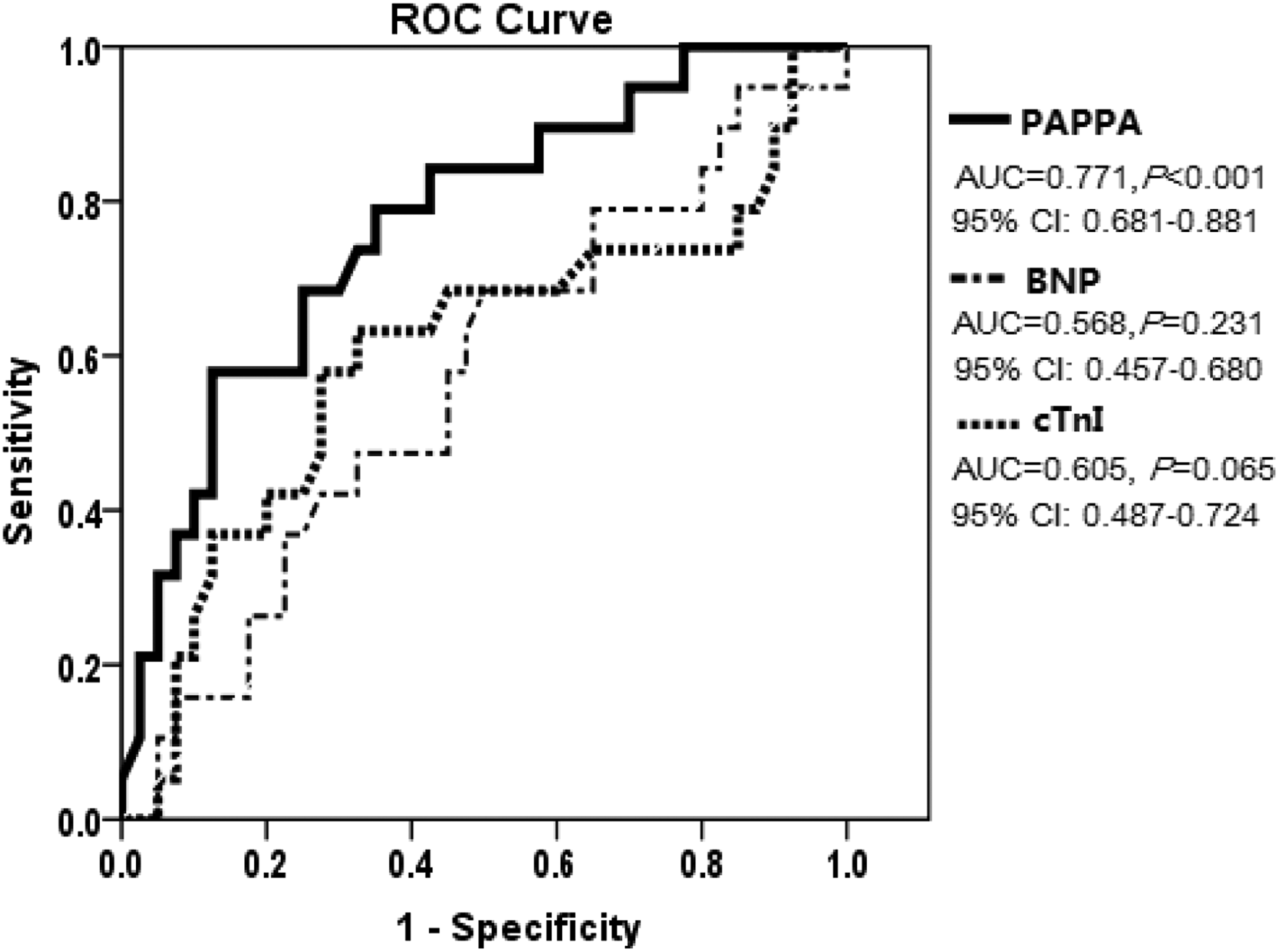
Kaplan–Meier analysis revealed that patients with serum PAPPA concentrations at or over 5.4 ng/mL had decreased 28-day survival compared to those with PAPPA under 5.4 ng/mL (48.3% vs. 86.7%, Log-rank χ
2
= 19.78, P < 0.001), as showed in Figure 3.
Serum PAPPA at 5.4 ng/mL concentration and 28-day survival in septic patients. Survival curves were created by Kaplan–Meier method and analysed by a log-rank test. (5.4 ng/mL is the best cutoff value of PAPPA for prediction of 28-day death according to ROC curve analysis.)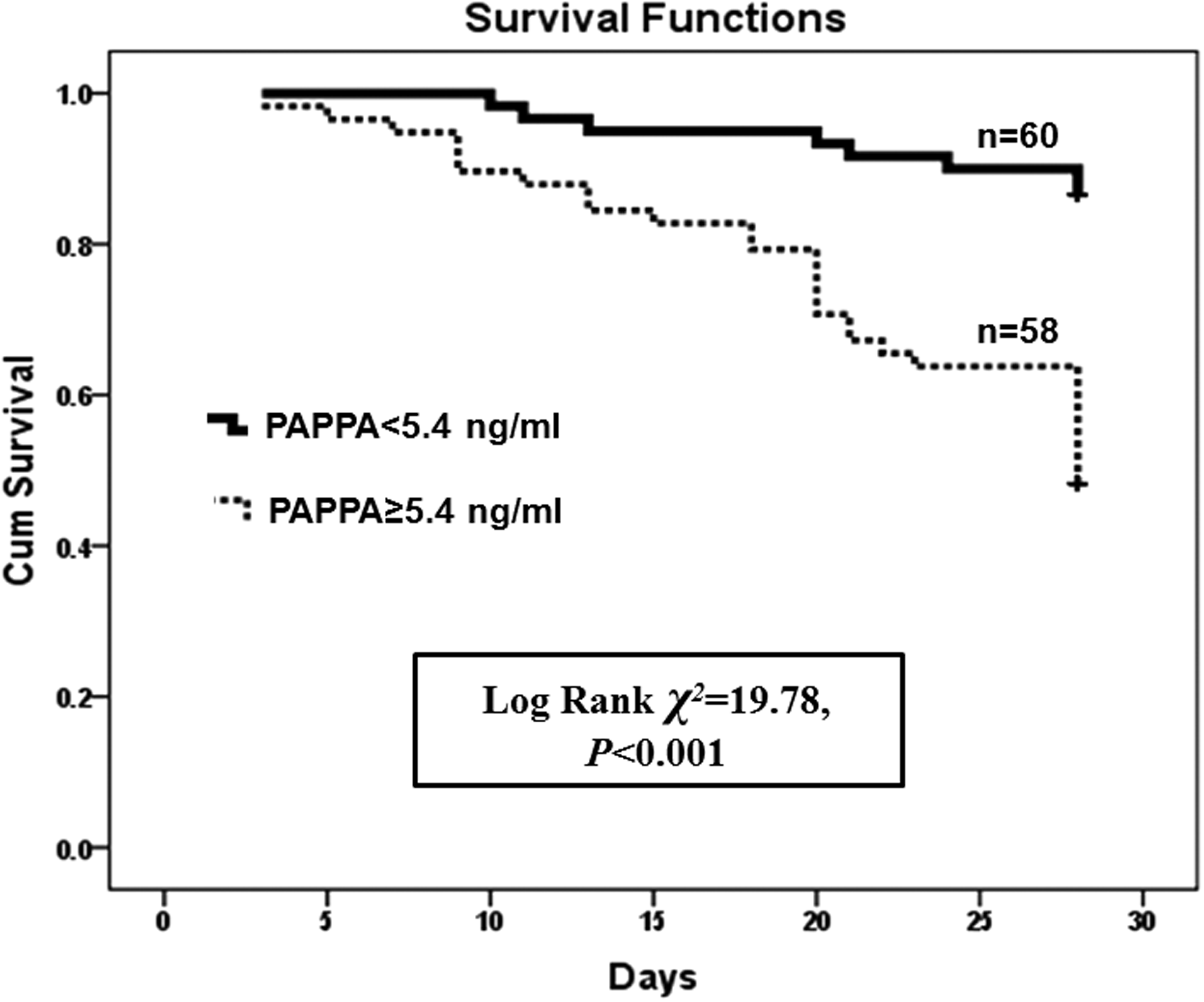
Univariate and multivariate Cox regression analysis of 28-day mortality.
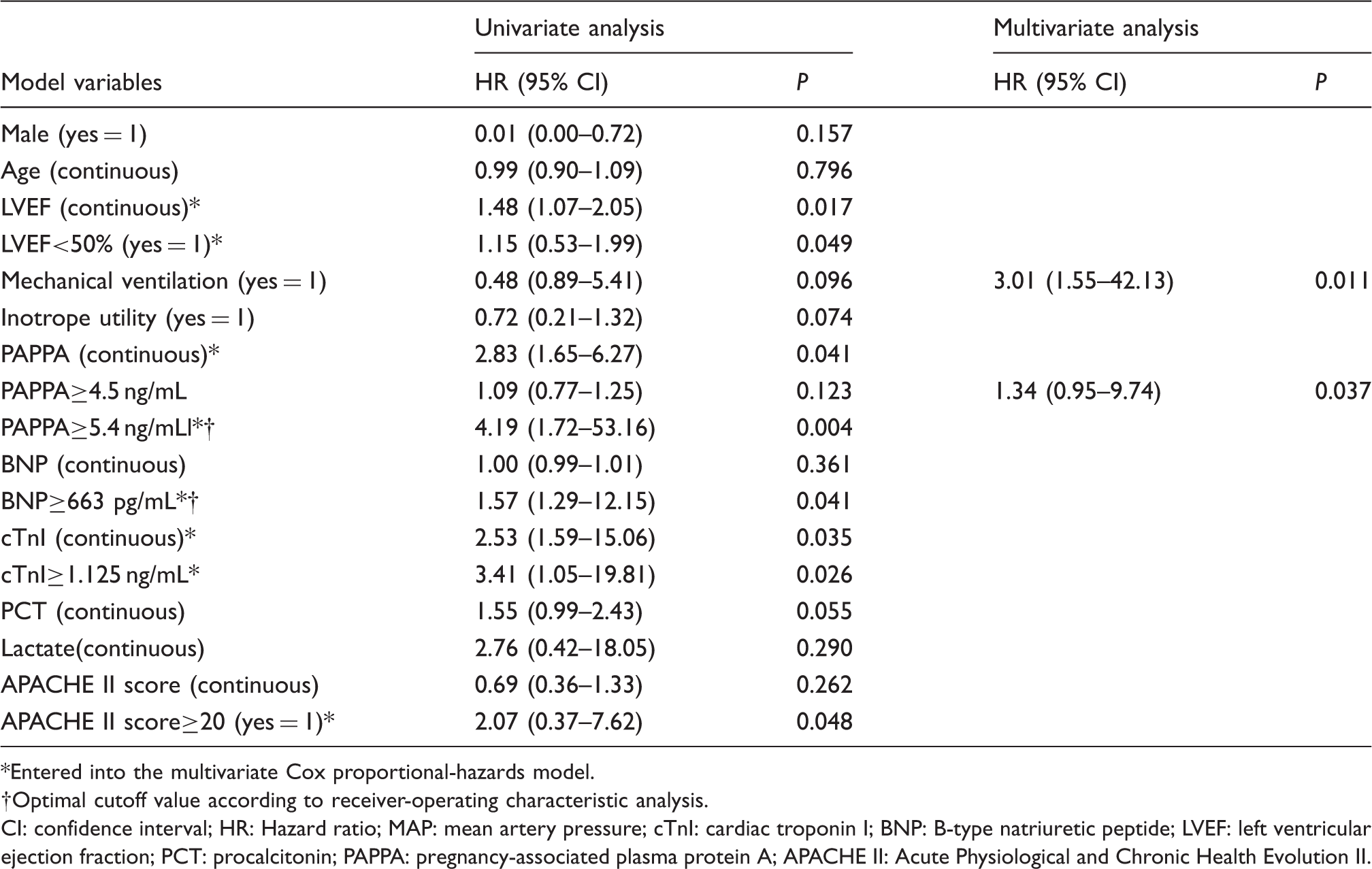
Entered into the multivariate Cox proportional-hazards model.
Optimal cutoff value according to receiver-operating characteristic analysis.
CI: confidence interval; HR: Hazard ratio; MAP: mean artery pressure; cTnI: cardiac troponin I; BNP: B-type natriuretic peptide; LVEF: left ventricular ejection fraction; PCT: procalcitonin; PAPPA: pregnancy-associated plasma protein A; APACHE II: Acute Physiological and Chronic Health Evolution II.
Discussion
SRMD occurs in almost 50% septic patients and can increase the risk of death in sepsis as much as two times.2,24 Therefore, early recognition of SRMD and aggressive supportive therapy are mandatory. 25 However, the ambiguity of clinical findings and unclear risk stratification in sepsis has been major problems in sepsis intervention trials. 26 Within this context, there is a need for biomarkers to tackle the challenges of sepsis monitoring and treatment.
BNP and cTnI are commonly used biomarkers in ICUs. Both of them have been identified as predictors of SRMD and death in septic patients;1,3 however, BNP alone has been proven not to be practical in the differentiation of septic patients with or without SRMD,3,27 and the specific use of BNP in diagnosing SRMD is not recommended because of a number of confounding factors of BNP as previously reported.5,6 The cardiac-specific cTnI has been suggested to be sensitive in the detection of minor myocardial injury in systemic inflammation, sepsis or septic shock; but the low specificity limits the use of cTnI as a diagnostic tool for SRMD.4,27,28
The present study indicated that PAPPA as a novel biomarker was encouraging for both the diagnosis of SRMD and the prediction of adverse outcomes in septic patients. PAPPA appeared comparable to conventional biomarkers such as BNP and cTnI in reflecting SRMD. The reason responsible for this diagnostic power is unclear but is suggested by previous studies. Serum PAPPA concentrations have recently been reported to be associated with the severity of non-septic heart failure 29 and increased concentrations of PAPPA in patients with cardiac dysfunction might reflect the degree of heart damage against inflammation, reactive oxygen species and mechanical stress.29–31 The reason for the superiority of PAPPA over classic biomarkers in the prediction of ICU death is also to be elucidated. The most plausible explanation might be that the elevation of PAPPA in septic patients represents not only continuous tissue damages including cardiomyocytes injury due to its metalloproteolytic activity but also indicates systemic inflammatory burden. PAPPA can serve as targets of inflammatory cytokines such as TNF-α and IL-1β, IL-6 abundant during sepsis through nuclear factor κB (NF-κB) pathway, these cytokines can in turn stimulate the overexpression of PAPPA by several cell types.7,13,14
It should be noted that the accuracy of four studied cardiac biomarkers for the diagnosis of SRMD as quantified by the AUC was relatively low (AUC<0.7), and this unsatisfactory result might attribute to multiple confounding factors existed in the coexisting diseases of septic patients and sepsis-related clinical monitoring and intervention. Diseases of the central nervous system, the electrolyte concentration, early fluid resuscitation and catecholamine therapy, and cytokine up-regulation by early antibiotic treatment may all affect BNP and cTnI release.5,6 As to PAPPA, mechanisms other than systemic inflammation and sepsis-related vascular damages, the intensive insulin therapy used in certain septic patients, 32 heparin administration in pipeline flushing and renal replacement, if necessary, 33 the senility of the study population may also affect the kinetics of PAPPA as taking the reduced ability and efficiency of PAPPA clearance into account. All the aforementioned confounder might led to false-positive and/or false-negative results in the measurement of PAPPA, BNP and cTnI in critically ill patients thus reduce the diagnostic accuracy.
Potential limitations of the present study merit consideration. The small sample size and single-centre setting of our study may result in reduced applicability to patients with sepsis. The definition of SRMD by either LVEF decrease or inotrope support requirement or both might overestimate the prevalence of SRMD (62.7% for all subjects) compared to previous investigation, 26 although it is practical and comprehensive in the ICU. The limited sensitivity of the PAPPA ELISA and the time taken to obtain the result will limit its utility clinically. The kinetics of PAPPA alteration and the impact of PAPPA on the long-term outcome in septic patients need further investigation.
In conclusion, serum PAPP-A frequently increases in patients with severe sepsis, and this elevation is associated with an increased prevalence of SRMD, and independently predicts adverse outcomes. These data indicate that serum PAPP-A can serve as a useful biomarker for the evaluation of septic patients.
Footnotes
Acknowledgements
We would like to thank Dr. Xiaoqiong Zou for her assistance in laboratory analysis.
Declaration of conflicting interests
None.
Funding
This research was funded by the Joint Foundation of Zhejiang Bureau of Health and Ministry of Health of China (No. WKJ2005-2-035).
Ethical approval
This study approved by the ethics committee of Zhejiang Hospital (REC number: ZJYY208029C).
Guarantor
ZZ.
Contributorship
ZZ designed research, analysed data and wrote the first draft of manuscript; YY performed statistics; JC and LW performed clinical monitoring and laboratory detection; JY performed echocardiography; HD collected data. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
