Abstract
The rising incidence of chronic liver disease continues to be an increasing health burden. The morbidity and mortality associated with chronic liver disease typically occur in patients with advanced fibrosis. Hence, early identification of those at-risk is of vital importance to ensure appropriate ongoing management. Currently, tools for appropriate risk stratification remain limited. Increasing awareness of the limitations of liver biopsy has driven research into alternative non-invasive methods of fibrosis assessment including serological markers assessing functional changes. One such biomarker, the Enhanced Liver Fibrosis test, was initially validated in a cohort of 1021 patients with mixed aetiology chronic liver disease and shown to perform well. Since this pathfinder study, it has been independently validated in cohorts of hepatitis C, non-alcoholic fatty liver disease, alcoholic liver disease, primary biliary cirrhosis and primary sclerosing cholangitis. In addition to performing well as a diagnostic tool, the Enhanced Liver Fibrosis test has been shown to outperform liver biopsy in prognostic studies and is the only non-invasive marker to do so. However, questions remain regarding the use of this test, particularly regarding the possible effect age and alcohol may have on test scores. This review examines the current literature published in relation to the Enhanced Liver Fibrosis test and its clinical utility and highlights areas requiring further study.
Keywords
Introduction
The rising incidence of chronic liver disease (CLD) continues to be an increasing health burden. 1 The morbidity and mortality associated with CLD typically occur in patients with advanced fibrosis and cirrhosis. Concerningly, these patients often remain asymptomatic until they develop decompensated disease. Hence, early identification of those at-risk is of vital importance to ensure appropriate ongoing management.
Currently, tools for appropriate risk stratification remain limited. Although liver biopsy remains the gold standard for identifying advanced fibrosis, it requires access to hospital services, is invasive, costly and not without risk. Increasing awareness of the limitations of liver biopsy has driven research into alternative non-invasive tests (NIT) of liver fibrosis severity, including either novel imaging techniques assessing liver stiffness or serological markers assessing functional changes. While transient elastography offers a user-friendly bedside test, it still requires access to a dedicated machine with a trained operator and may be inaccurate in patients with severe obesity, narrow intercostal spaces and mild steatosis.2–5 Conversely, serum biomarkers provide a standardized automated test with good reproducibility6–9 and do not require specialist equipment or personnel. The wider availability of serum biomarkers facilitates their utility in clinical practice, especially for screening of patients with compensated disease or in settings where liver biopsy is not pragmatic. Biomarkers can be direct (reflecting underlying fibrosis) or indirect markers (using markers of liver function). Table 1 illustrates a comparison between these biomarkers. Although indirect markers are inexpensive, as they rely upon routine blood tests and are not subject to patents, they are less accurate, less precise and not as reproducible.10,11
Comparison of serum biomarkers.
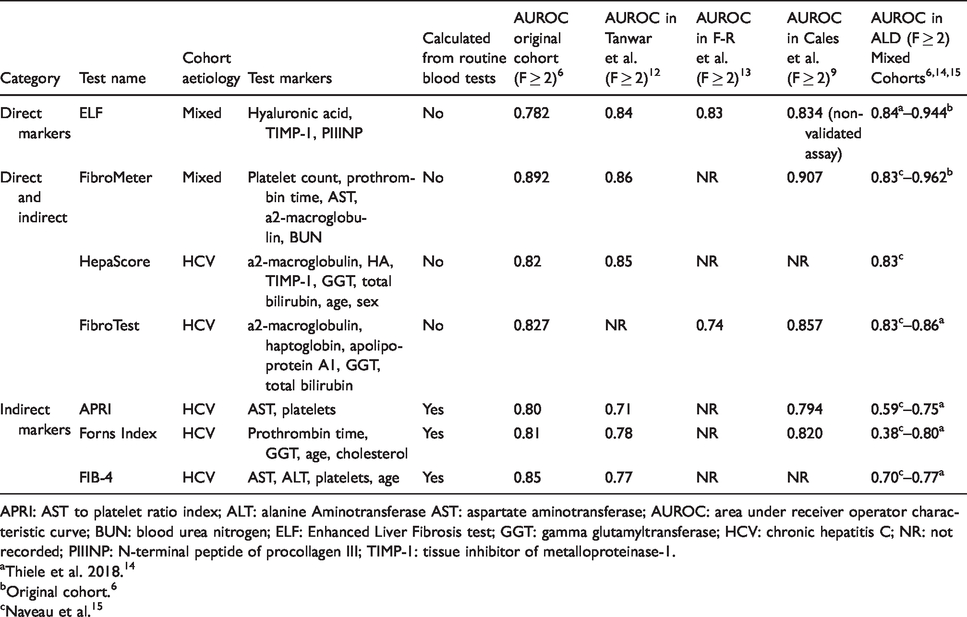
APRI: AST to platelet ratio index; ALT: alanine Aminotransferase AST: aspartate aminotransferase; AUROC: area under receiver operator characteristic curve; BUN: blood urea nitrogen; ELF: Enhanced Liver Fibrosis test; GGT: gamma glutamyltransferase; HCV: chronic hepatitis C; NR: not recorded; PIIINP: N-terminal peptide of procollagen III; TIMP-1: tissue inhibitor of metalloproteinase-1.
aThiele et al. 2018. 14
bOriginal cohort. 6
cNaveau et al. 15
The Enhanced Liver Fibrosis (ELF) test is one such direct serum biomarker. It is the result of a logarithmic algorithm combining quantitative serum measurements of three markers of hepatic extracellular matrix metabolism – hyaluronic acid (HA), tissue inhibitor of metalloproteinase-1 (TIMP-1) and N-terminal peptide of procollagen III (PIIINP). 6 HA, a polysaccharide synthesized by hyaluronic synthases on the plasma membranes of synovial lining cells and hepatic stellate cells,16–20 is almost entirely (90%) cleared by the synovial endothelial cells.17,21–24 HA has a short plasma half-life, is synthesized and excreted by the liver and concentrations have been shown to correlate with fibrosis stage.17,18,24–28 TIMP-1 is produced by activated hepatic stellate cells during liver injury and inhibit matrix metalloproteinases, resulting in fibrotic tissue accumulation.6,29,30 PIIINP is cleaved from type III procollagen during synthesis of type III collagen. 31 Thus, serum PIIINP concentrations reflect collagen synthesis and indicate fibrotic transformation of liver parenchyma. Therefore, greater concentrations of these three analytes (HA, TIMP-1 and PIIIMP) combine to generate higher ELF score indicative of more fibrosis.6,12,32,33
ELF performs best when two thresholds are used; an upper threshold to confirm advanced fibrosis (high specificity and positive predictive value [PPV]) and a lower threshold to exclude fibrosis (high sensitivity and negative predictive value [NPV]). 34 According to the manufacturer, ELF should be interpreted as follows: <7.7 – no to mild fibrosis; ≥7.7 to <9.8 – moderate fibrosis (F ≥ 2); ≥9.8 – significant fibrosis (F ≥ 3). 6 Recent evidence has suggested additional thresholds of <8.3 to exclude moderate fibrosis, ≥10.5 to indicate advanced fibrosis (F ≥ 3) and ≥11.3 for cirrhosis.14,35
In the primary care setting, use of serum biomarkers such as ELF enables testing for fibrosis severity in patients unable to access or unsuited to liver biopsy or transient elastography (TE). Patients at risk may be screened for fibrosis, possibly in combination with simple tests, to reduce the need for biopsy and facilitate more appropriate referrals to care, with consequent health economic benefits.36,37 A significant role for ELF has also been outlined in secondary and tertiary care, where it has been shown to not only accurately stratify fibrosis severity but also provide greater prognostic insight than liver biopsy. The prognostic utility of ELF has been shown to be graded, whereby a one unit increase in ELF is associated with a doubling in the risk of a liver-related event. 38 ELF is one of only two serum biomarker panels designed for all aetiologies. While originally validated in a mixed cohort of 1021 CLD patients, 6 it has since been validated in cohorts of hepatitis C virus (HCV),12,34 primary biliary cirrhosis (PBC), 39 primary sclerosing cholangitis (PSC), 40 alcoholic 41 and non-alcoholic fatty liver disease (NAFLD) 11 ; however, it has been shown to perform poorly in autoimmune hepatitis. 42
In comparison to alternative patented serum biomarkers, ELF does not have an upper threshold, meaning it can be used to evaluate fibrotic liver damage in cirrhotic patients to provide a quantitative measure that correlates with portal hypertension and prognosis.43–45 Further, its linear relationship to fibrosis severity and non-invasive nature renders it an excellent tool to monitor disease course and response to antifibrotic therapies.46,47
Current clinical uses of ELF
Viral hepatitis C
Globally, approximately 71 million people are infected with HCV. Untreated HCV may progress to cirrhosis and hepatocellular carcinoma, causing an estimated 399,000 deaths annually. 48 Recent developments in antiviral medications have facilitated clearance of the virus in many patients; however, the significant global prevalence necessitates ongoing progress be made in both treatment and monitoring of this disease. ELF has shown good efficacy in the stratification of fibrosis severity in both chronic HCV infection and HIV/HCV co-infection,6,12,34,49,50 is able to predict evolution of fibrosis in response to antiviral treatment51–53 and is cost-effective. 54 The European Association for the Study of the Liver and The Asian Pacific Association for the Study of the Liver guidelines on the use of NIT have recommended the use of ELF in viral hepatitis and other CLD. ELF may prove particularly valuable in assessing fibrosis severity in prison populations, where HCV prevalence is high, but liver biopsy is not routinely performed due to practical restrictions. 55
The original validating ELF cohort included a substantial HCV population (49%), whereby subgroup analysis found an area under receiver operator characteristic curve (AUROC) of 0.773 (95% CI: 0.697–0.848) for the diagnosis of significant fibrosis. 6 Perhaps of greater clinical utility, a NPV of 94.9% and PPV of 90.0% were found. Recruitment of this cohort was completed in tertiary centres; therefore, generalization of PPV and NPV to primary care should be undertaken with caution. Further investigation of ELF in HCV in populations where the spectrum of disease reflects that seen in primary care is required. The heterogenous aetiology of this cohort has also been criticized, 56 and subsequently, further validation in HCV exclusive cohorts has been undertaken. One independent validation study recruited patients in three different cohorts. 34 They identified similar AUROCs to the original ELF cohort and determined that if sensitivity and specificity of 85% are accepted, biopsy could be avoided in 81% of patients. As the study did not use a centralized pathologist to review all histology, inter-observer variation may compromise the validity of these findings. Further investigation into the avoidance of biopsy found that 63% of patients in the Martinez et al. cohort and 74.7% in the Petersen et al. cohort could avoid biopsy by using ELF as a surrogate fibrosis marker.53,57 Each of these studies utilized different acceptable error rates when calculating the proportion of biopsies which might have been avoided in their respective cohorts, with sensitivities and specificities ranging from 76% to 90%. This highlights the variability in acceptable error rates between clinicians, which impacts the number of biopsies potentially avoided. The largest independent study to date assessed 512 patients with HCV and confirmed ELF’s utility in predicting fibrosis or cirrhosis in HCV. 50 Their findings were similar to those of the original validating cohort, whereby significant and severe fibrosis and cirrhosis could be detected with AUROCs of 0.78 (95% CI: 0.74–0.82), 0.82 (0.78–0.86) and 0.85 (0.81–0.90), respectively. These results were not significantly different to FibroTest or Hepascore when statistical comparisons were completed. However, the greater stability of the ELF analytes means that the ELF test is more widely applicable to clinical settings, in which there may be delays in sample processing or analysis.58–60 Although it is difficult to compare AUROCs from different studies given the potential influence of spectrum bias, generally, the utility of ELF in fibrosis assessment has been shown to be relatively consistent. Significant fibrosis can be identified with AUROCs ranging from 0.74 to 0.87, advanced fibrosis 0.82 to 0.89 and cirrhosis 0.82 to 0.90.6,34,50,51,53,57,61
NAFLD
NAFLD is estimated to affect approximately 25% of the adult population worldwide 62 ; it is the commonest cause of CLD seen in primary care and is now the second leading cause of liver disease, 63 primary liver cancer and decompensation among adults awaiting liver transplantation 64 in the USA.65,66 Therefore, identifying either those patients who require ongoing management or those who are suitable for either existing or emerging remedies remains essential. Although NAFLD screening in the community is not currently recommended, European clinical practice guidelines recommend the use of serum biomarkers (NAFLD fibrosis score [NFS], fibrosis 4 [FIB-4] or ELF test) as first-line risk stratification to help identify at-risk patients with NAFLD and advanced fibrosis due to its prognostic implications. In contrast to the European Associations, the United Kingdom National Institute for Health and Care Excellence (NICE) guidelines on the assessment and management of NAFLD recommend using the ELF test as the first-line test for advanced fibrosis in all patients who have been diagnosed with NAFLD. 67 The ELF test was validated for diagnosing moderate and severe fibrosis in an independent cohort of patients with NAFLD with an AUROC of 0.90 (95%CI 0.84–0.96) and 0.93 (95%CI 0.88–0.98), respectively. 11
To date, the utility of ELF as a screening tool for NAFLD has been tested in two international prospective studies. The UK-based study used a stepwise biomarker protocol with ELF as a secondary direct biomarker to triage primary care referrals requiring secondary assessment. Although the addition of the secondary ‘direct biomarker’ ELF test was only required in 26.7% of cases, its use enabled the primary care practice to allocate 59.9% of the remaining cases of NAFLD for referral to secondary care, leaving 40.1% to be managed in primary care. The use of the biomarker pathway resulted in 20% cost-saving compared with standard care. 36 The Australian-based study used ELF in combination with TE and demonstrated that together they have a NPV of 91.7% for excluding advanced fibrosis and a PPV of 95.8% for identifying advanced fibrosis. 68 Interestingly, the subanalysis highlighted that older age was significantly associated with higher odds of having an ELF > 9.8 independent of fibrosis status (P < 0.001). 68 Although both studies have highlighted the potential value of the use of ELF in a pragmatic way, due to comorbidities in the cohorts investigated, histological confirmation of fibrosis status was only available in a subset of patients.
Alcoholic liver disease
The spectrum of alcoholic liver disease (ALD) ranges from simple steatosis in almost all heavy drinkers through to steatohepatitis and ultimately fibrosis and cirrhosis, which develops in 20% of chronic alcoholics.33,69 While the performance of serum biomarkers in ALD is comparable to other aetiologies in that they better diagnose more severe stages of fibrosis, supporting literature is relatively sparse compared with viral hepatitis and NAFLD populations. The original ELF cohort included 64 patients with ALD and found an AUROC of 0.944 (95% CI: 0.836–1.000) for diagnosis of significant fibrosis (F ≥ 2). 6 Although a small cohort, the perfect specificity and sensitivity (100% when using two different thresholds; no CIs published) indicated good marker performance. More recent validation of ELF in a population of 289 ALD patients completed in 2018 14 found that the utility of ELF in detecting significant and advanced fibrosis and cirrhosis was excellent, with AUROCs of 0.84 (95% CI: 0.80–0.89), 0.92 (0.89–0.96) and 0.94 (0.91–0.97), respectively. Sensitivity (89%) and specificity (91%) were only published for advanced fibrosis. A comparison of ELF with FibroTest, TE, AST to Platelet Ratio Index (APRI), FIB-4 and Forns Index found no significant difference between FibroTest and ELF; however, they both outperformed indirect markers (P < 0.001–0.008). This corroborates findings, whereby indirect markers have been shown to perform poorly in ALD populations relative to other aetiological cohorts.14,15,71–76 TE performed significantly better than ELF (P < 0.002) in the per protocol analysis, although this difference was not observed in the intention to treat analysis due to the 5–15% failure rate of TE. 5
Concerns regarding the effect of alcohol on the constituents of ELF have been expressed; however, a comparison of histologically matched cases and controls to properly investigate this is yet to be completed. Studies which have attributed differences in analyte concentrations to alcohol have not matched patients according to fibrosis severity, thus making the results difficult to interpret accurately, as the differences in ELF scores may be due to early fibrotic changes in alcoholics rather than alcohol consumption. 77 A small study (n = 10) assessing the impact of acute intoxication on the markers used in the ELF algorithm found no significant differences in analyte concentrations measured in blood taken while patients were intoxicated compared with bloods taken following two weeks of sobriety (P > 0.01). 78
PBC
ELF is particularly useful in PBC, as in typical presentations, liver biopsy is not required to confirm a diagnosis, 79 and a single biopsy may well not be representative of the whole liver. A non-invasive assessment of fibrosis severity with ELF in these situations aids clinicians in making the most appropriate treatment decisions and predicts clinical outcomes. While the original ELF cohort included 53 PBC/PSC patients, there is no published subgroup analysis. 6 A prospective cohort study of 161 PBC patients followed up for 7.3 years demonstrated that ELF scores correlate well with histology, albeit with significant overlap between ELF scores in the intermediate stages of fibrosis (AUROC = 0.75 for significant fibrosis and 0.76 for cirrhosis). 39 This good performance has been corroborated by subgroup analysis of 28 PBC patients in a comparison of ELF, biopsy, FibroTest and TE. 13 ELF may also be used to monitor fibrosis progression, as it has been shown to increase on average by 0.032 per year in those with fibrosis and has also performed as well as histology as a prognostic marker, whereby each unit increase in ELF was associated with a three-fold increase in future complications. 39
PSC
Patients with PSC have an unpredictable disease course with a median transplant-free survival of 12–21 years.80,81 Studies to date investigating potential predictors of poor outcome in PSC patients have either relied upon biochemical variables81–87 or clinical evidence of decompensated disease.81,88,89 Currently, there remain no reliable prognostic tools for use in patients with PSC. 90 However, recent literature has demonstrated promising results utilizing the ELF test. A Norwegian study using ELF to stratify PSC patients demonstrated that higher ELF scores at baseline were associated with shorter survival. The ELF test reliably distinguished between mild and severe disease with an AUROC of 0.81 (95% CI: 0.73–0.87). They also identified that ELF score was associated with transplant-free survival independent of the Mayo Risk Score (hazard ratio: 1.9, 95% CI: 1.4–2.5, and 1.5, 95% CI: 1.1–2.1, respectively). 40 The multivariate Cox regression analysis identified that both ELF score and Mayo Risk Score were independently associated with transplant-free survival. Interestingly, among the other variables in the regression model, only PSC duration and age at diagnosis were also independently associated with survival time.
A subsequent international multicentre retrospective PSC population study estimated rates of transplant-free survival. They identified that concentrations of ELF analytes were higher in patients with the combined outcome of liver transplantation or death (median 10.9 [interquartile range (IQR): 9.8–12.1]; n = 24 deaths, n = 79 liver transplantations, P < 0.001). They also identified that ELF independently predicted clinical outcomes with an AUROC of 0.79. 91 Unfortunately, in this study, they were unable to calculate the Mayo Risk Score, the most commonly used prognostic tool in PSC due to unreliable clinical data. Therefore, the authors could not validate whether ELF added incremental value to the Mayo Risk Score as identified by the Norwegian study. 40 As liver biopsies are not indicated in the management of PSC other than to diagnose concomitant autoimmune hepatitis, neither study had histological confirmation of fibrosis status and instead used endpoints of either a composite of all-cause mortality and liver transplantation, or transplant-free survival.
Cost-effectiveness
Cost-effectiveness analyses have demonstrated the utility of ELF in populations of HCV, ALD and NAFLD.36,37 Theoretical cohorts of ALD and HCV have compared the use of annual ELF, annual sequential TE/ELF and single biopsy. ELF test alone was the most effective strategy in both HCV and ALD; however, it was also the most expensive (€11,484 per quality adjusted life year [QALY] in HCV and €189 per QALY in ALD). The significantly greater cost associated with HCV is due to the cost of antiviral therapies. At a willingness-to-pay threshold of €30,000 per QALY, sequential ELF/TE was found to be a cost-effective strategy in 90.1% of HCV cases and >99.9% of ALD cases.
A previous cost-effectiveness investigation into the use of NIT in HCV found that treatment with antiviral agents without liver testing was the most cost-effective strategy to reduce liver fibrosis occurrence and progression; however, this study was completed during the age of interferon-only therapy. Since the development of direct acting antivirals, which are substantially more costly, NITs are able to identify those in early stages of fibrosis requiring access to treatment in countries where not everyone with a positive test can receive treatment.
As previously discussed, the sequential use of ELF and an indirect fibrosis marker in NAFLD in a UK primary care setting is associated with a five-fold increase in the identification of advanced fibrosis and cirrhosis and a 20% cost-saving compared with standard care. 36 There are yet to be cost-effectiveness analyses for ELF performed in other aetiologies of CLD. All studies of cost-effectiveness are dependent on determining the cost of the ELF test. As with most diagnostic tests, the cost has fallen since first introduction and the cost of the test is likely to be inversely proportional to the number of tests performed up to a certain value. Few of the cost-effectiveness studies performed to date have incorporated the current NHS price of ELF of £42.
Areas requiring further study
Although the promising utility of ELF has been highlighted in a variety of pathologies of CLD, there are still areas that require further clarification. Age has been highlighted as an independent factor influencing the ELF score in some studies in HCV, 92 NAFLD 68 and PSC. 40 This may, in part, be explained by the higher incidence of cardiovascular and connective tissue disease seen in elderly populations, which are known to impact ELF. The original ELF algorithm did take age in account; however, subsequent studies designed to specifically address the necessity for incorporating age in the ELF algorithm found that age could be omitted with no loss in diagnostic performance. 93 One potential reason for this may have been the relatively young age of the cohorts that were used, compared with the higher age seen in the subsequent studies. 68 Clearly, further analysis is required as to clarify the impact of age on the ELF score. Assessment of ELF in ALD has shown promising results; however, there is a need for studies assessing ELF’s performance in the setting of severe alcoholic hepatitis, decompensated liver disease and HCV co-infection. The impact of alcohol on ELF scores must be more thoroughly investigated, as it is unclear if this will affect ELF performance as a diagnostic or prognostic test and if so, for better or worse. Further studies assessing its longitudinal utility, either alone or in combination with other risk scores or non-invasive tests, are required to help us understand the full potential of ELF in clinical practice. To this point, no statistically significant differences have been shown between ELF and other patented panels used to assess fibrosis stage (including Hepascore, FibroMeter, FibroSure, FibroTest); however, generally, ELF has been shown to consistently outperform simple panels (including FIB-4, Forns Index and APRI).
Conclusion
The ELF test is a combination of serological biomarkers that has been developed and validated in multiple underlying aetiologies of CLD. Its utility both in risk stratification and prognostication has been subsequently determined and validated by multiple international studies in several different populations and settings.11,38,53,94,95 The analytical stability and excellent reproducibility of the test differentiate it from other non-invasive tests for liver fibrosis. Its value appears particularly promising, where access to imaging modalities is limited, such as in prisons or rural areas; however, further studies to clarify its uses with regard to longitudinal data and the concerns related to older age, and concomitant heavy alcohol use are required.
Footnotes
Acknowledgements
This article was prepared at the invitation of the Clinical Sciences Reviews Committee of the Association for Clinical Biochemistry and Laboratory Medicine.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: WR is an inventor of the ELF test and has received support for research and speakers fees from Siemens Healthineers.
AS has received support for research and speaker fees from Siemens Healthineers.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Guarantor
WR.
Contributorship
All authors contributed equally to the review.
