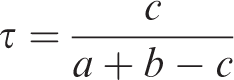Abstract
Background
Immunoassays are commonly used to test for drugs of abuse in patients in a variety of settings. The increasing prevalence of ‘designer’ drugs causes difficulties for the toxicology laboratory and may result in unexpected false positives and identification of unfamiliar compounds. Within the past decade, there have been a variety of ketamine and phencyclidine analogues identified, particularly as drugs of abuse.
Method
We present a case of intoxication with a novel ketamine analogue, deschloro-N-ethyl-ketamine, causing a false positive phencyclidine immunoassay. Additionally, we performed spiking studies and 2D molecular similarity calculations for deschloro-N-ethyl-ketamine, ketamine and three other analogues on the Siemens Viva-E EMIT-II phencyclidine assay to assess their cross-reactivity.
Results
Four of the tested compounds (deschloro-N-ethyl-ketamine, 3-methoxy-phencyclidine, 3-methoxy-eticyclidine and methoxetamine) cause false positive phencyclidine immunoassay results, while ketamine gives a negative result. The cross-reactivity data are in accord with the similarity calculations of these molecules, further validating the ability of 2D molecular similarity analysis to predict the molecular cross-reactivity in immunoassays.
Conclusions
The cross-reactivity data of phencyclidine and ketamine analogues presented in this study could help toxicology laboratories and clinicians in evaluating unexpected results, particularly when novel PCP and ketamine analogues are being considered.
Introduction/Background
Immunoassay drug screening tests are commonly used to evaluate patients for the presence of drugs of abuse in both inpatient and outpatient settings. As with all laboratory tests, these assays may be subject to a variety of factors which may cause false positive and false negative results. In some cases, this may result from other compounds with a similar chemical structure that can be recognized by the target antibody or antibodies of the assay. This issue may not only confuse clinicians, but also mislead them, resulting in negative effects on the overall clinical management. 1
Previous studies have described computational techniques to calculate chemical similarity as a method of predicting cross-reactivity of compounds to target molecules of immunoassays,2,3 adapting computational similarity algorithms often used in pharmaceutical research to interrogate databases of chemical structures.4,5 One such method is known as two-dimensional (2D) molecular similarity analysis and can use molecular descriptors such as MDL public keys to calculate similarity coefficients between two chemical structures.2,5,6 All of these described techniques compare various aspects of a drug’s molecular features to determine their similarity.7–9
Phencyclidine (1–(1-phenylcyclohexyl)piperidine), also known as PCP, is a drug of abuse with dissociative and hallucinogenic effects. A related compound, ketamine (2–(2-chlorophenyl)-2-(methylamino)cyclohexan-1-one), is most commonly used as an anaesthetic and has recently been investigated as an antidepressant,10,11 but it has also been known to be abused. Analogues of PCP and ketamine have been developed in both pharmacologic research and clandestine laboratories. Morris and Wallach provide a thorough review of analogues of both PCP and ketamine as well as other dissociative drugs. 12 One of the more commonly identified PCP analogues, which has been seen on several occasions at our institution since 2017, is 1-[1–(3-methoxyphenyl)cyclohexyl]-piperidine, also known as 3-methoxy-phencyclidine (hereafter referred to as 3-MeO-PCP). 12 A commonly described ketamine analogue frequently seen in the literature is 2–(3-methoxyphenyl)-2-(ethylamine)cyclohexan-1-one, also known as methoxetamine, 3-methoxy-2-oxo-PCE or MXE (hereafter referred to as MXE).13–17 Newer ketamine analogues, including 2-phenyl-2-(ethylamino)cyclohexan-1-one, also known as deschloro-N-ethyl-ketamine, eticyclidone or 2-oxo-PCE (hereafter referred to as 2-oxo-PCE), and 2–(3-methoxyphenyl)-2-(ethylamino)cyclohexane, also known as 3-methoxy-PCE, 3-methoxy-eticyclidine or methoxieticyclidine (hereafter referred to as 3-MeO-PCE), are incompletely characterized from a clinical and laboratory medicine perspective, including data on their metabolism, and are sparsely reported in the literature. 18
Here we report a case of 2-oxo-PCE intoxication with a suspected false positive PCP immunoassay and subsequent identification by GC-MS. Additionally, we perform urine spiking studies and chemical similarity calculations for ketamine and several analogues, including 2-oxo-PCE, to assess their structural similarity to PCP and cross-reactivity on the EMIT II PCP immunoassay.
Case presentation
A young man in his 30s with history of several psychiatric disorders, including polysubstance abuse, presented to the hospital with altered mental status. He was last seen normal three days prior to admission. The patient endorsed using ketamine analogues ‘2-oxo-PCE’ and ‘3-MeO-PCE,’ which were purchased online, for approximately 5 days prior to admission. He was unable to quantify the amount or route of ingestion. Upon presentation, he was noted to be tachycardic with dry mucous membranes and multiple abrasions on his bilateral upper and lower extremities. On neurological examination he was found to have bilateral nystagmus, but was otherwise without focal deficits. He was directable throughout questioning, but he did have waxing and waning sensorium. Laboratory testing at admission revealed hypernatremia (150 mMol/L, reference range 136–146), acute kidney injury (creatinine 2.5 mg/dL, reference range 0.5–1.6; blood urea nitrogen (BUN) 32 mg/dL, reference range 8–26) and mild rhabdomyolysis (total creatine phosphokinase (CPK) 6000 IU/L, reference range 0–200). He was treated with aggressive intravenous hydration, and he did not require sedation for agitation throughout admission.
His urine comprehensive drug testing was positive for PCP on the immunoassay screen with identification of 2-oxo-PCE, ketamine, caffeine, naproxen, and salicylate metabolites by GC-MS. The 2-oxo-PCE peak was seen as a large peak in the total ion chromatogram (TIC), which is further visualized on the extracted ion chromatogram (EIC) (see Figure 1(a) for patient TIC, Figure 1(b) and (c) for EIC, and Figure 2 for reference and patient mass spectra of 2-oxo-PCE). No PCP or 3-MeO-PCE were identified by GC-MS. Approximately 36 h after presentation, the patient was back to his baseline mental status with improving laboratory values, and he was discharged home with plan for outpatient follow-up. The positive PCP immunoassay result was suspected to be a false positive, potentially due to 2-oxo-PCE. This hypothesis was tested by performed urine spiking studies as described below.
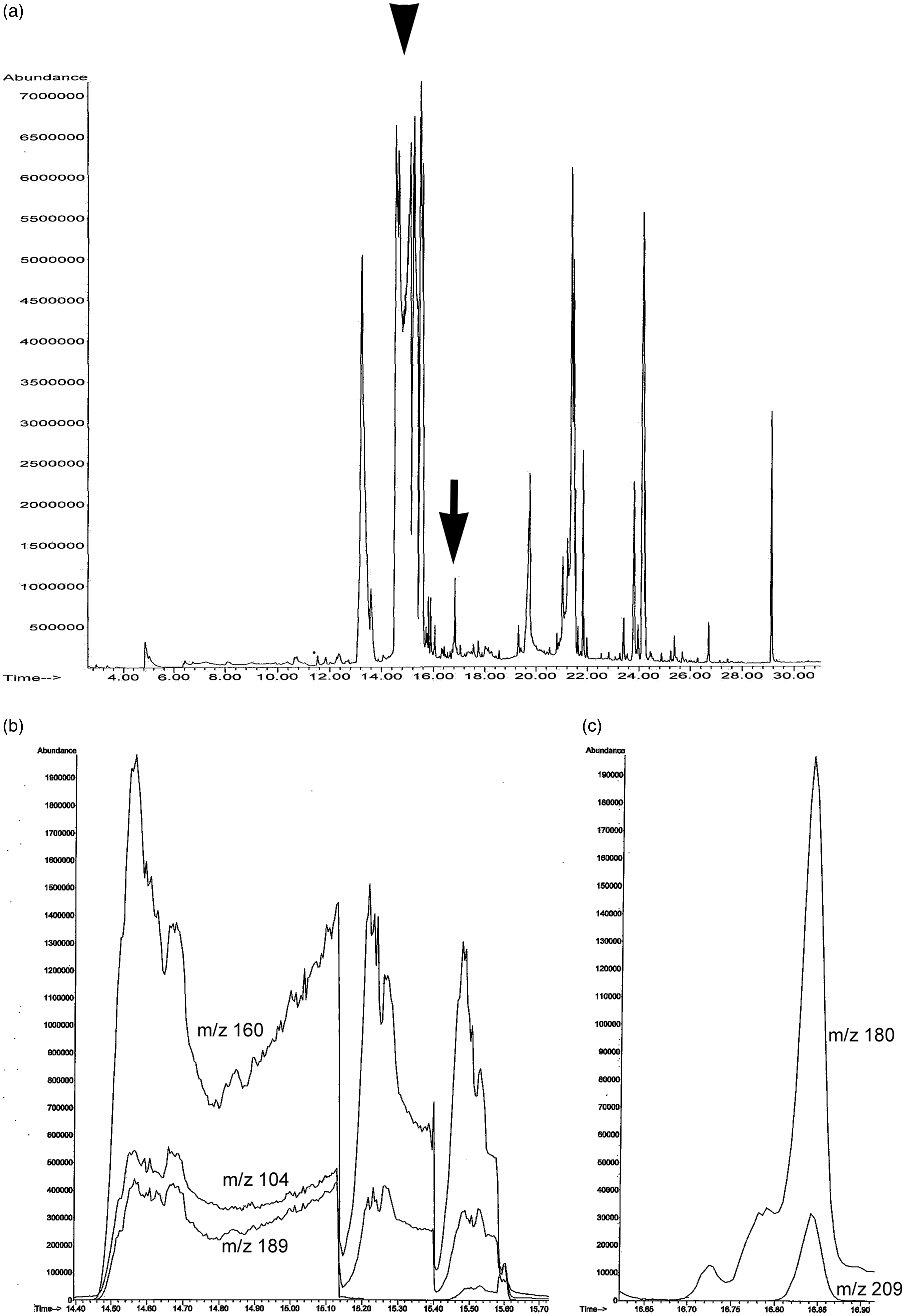
Total ion chromatogram (TIC) (a) and extracted ion chromatograms (EIC) (b and c) of the GC-MS for the patient. After liquid–liquid extraction with charcoal of the urine sample, the extract was dissolved in methanol and injected into the GC-MS (Agilent Technologies 5975 mass spectrometer, Santa Clara, CA) instrument operated in full scan mode using electron ionization. (a) The large TIC peaks (retention time between 14.5 min and 16.0 min) were all identified as 2-oxo-PCE (arrowhead) (mass spectrum is shown in Figure 2). An additional TIC peak (retention time =16.8 min) was identified as ketamine (arrow). Other detected drugs and compounds include ketamine metabolite, caffeine, naproxen, and salicylate metabolites (unlabelled). (b) EIC of m/z values 104, 160, and 189 in the retention time between 14.4 min and 15.7min for 2-oxo-PCE. (C) EIC of m/z values 180 and 209 in the retention time between 16.6 min and 16.9 min for ketamine.
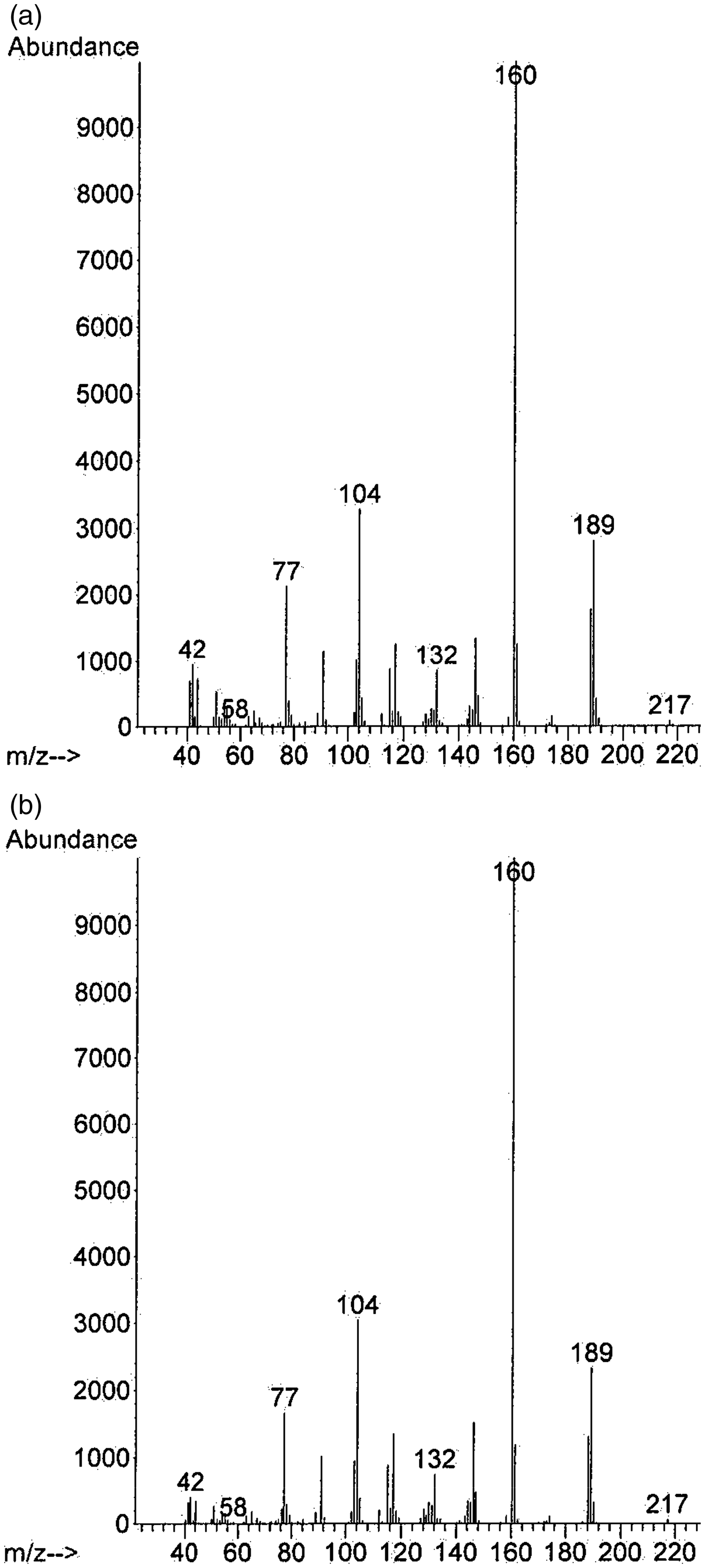
The mass spectrum of the unknown peak at 15.5 min in the TIC in Figure 1(a) and that of 2-oxo-PCE in the reference spectral library (b). The unknown compound in the unknown peak in the TIC was identified as 2-oxo-PCE through library match of the mass spectrum by using Cayman Spectral Library (Ann Arbor, MI).
Materials and methods
Patient urine testing
Urine drug testing was performed at the University of Pittsburgh Medical Center Toxicology Laboratory by the immunoassay testing panel on the Siemens VIVA-E platform (Siemens Healthcare Diagnostics, Inc., Tarrytown, NY, USA) using EMIT-II Plus kits for amphetamine, barbiturates, benzodiazepines, buprenorphine, cannabinoids, cocaine/cocaine metabolites, methadone, opiates and PCP and by gas chromatography-mass spectrometry (GC-MS) on the Agilent (Agilent, Santa Clara, CA, USA) system with an HP5-MS capillary column. The positive cut-off for the PCP immunoassay is 25 ng/mL and the lower detection limit of PCP by GC-MS is 2.5 ng/mL.
Regarding the GC-MS-based drug testing, the urine sample was processed with liquid–liquid extraction. In brief, the urine specimen was split and each portion underwent liquid–liquid extraction with methylene chloride spiked with barbital as an internal standard and activated charcoal, one at pH 11 and the other at pH 6 or less. The sample extracts were then combined and dried under a nitrogen gas stream, and the evaporated sample was reconstituted with methanol before injection into the GC-MS instrument operated in full scan mode using electron ionization. Blank samples are run in between all patient samples at our institution to prevent carry-over of the previous sample. The molecules in the extracted sample were screened in an untargeted manner and identified by their retention time in the TIC and mass spectra through library match with pre-established reference mass spectra in the Pfleger-Maurer-Weber Drugs and Pesticides Library for Toxicology (PMW_3.1 Tox, Agilent Technologies, Santa Clara, CA, USA), Mass Spectra of Designer Drugs 2012 (Wiley, Hoboken, NJ, USA), and Cayman Spectral Library (Cayman Chemical, Ann Arbor, MI, USA). The retention times were calculated using retention indices of analytes and barbital (internal standard). Quality control samples of known compounds are run daily for both the immunoassay and GC-MS methods. Quantification of compounds identified by GC-MS is not routinely performed, and patient samples are stored for approximately 1 month before being discarded.
Spiking studies
One milligram of ketamine, 2-oxo-PCE, MXE and 3-MeO-PCE (Cayman Chemical, Ann Arbor, MI, USA) were each reconstituted in 100 µL of methanol. Five milligrams of 3-MeO-PCP (Cayman Chemical, Ann Arbor, MI, USA) was reconstituted in 500 µL of methanol. Next, 10 µL of each solution was diluted with 90 µL of synthetic urine (UTAK Laboratories, Valencia, CA) for a concentration of 1000 µg/mL. Three serial dilutions were performed for each drug as in the previous step, resulting in concentrations of 100 µg/mL, 10 µg/mL and 1 µg/mL. Additional dilutions were subsequently performed for 2-oxo-PCE (800 µg/mL, 600 µg/mL, 400 µg/mL, 200 µg/mL), 3-MeO-PCE (80 µg/mL, 60 µg/mL, 40 µg/mL, 20 µg/mL), MXE (800 µg/mL, 600 µg/mL, 400 µg/mL, 200 µg/mL), and 3-MeO-PCP (0.1 µg/mL, 0.05 µg/mL, 0.025 µg/mL, 0.01 µg/mL) to further assess the respective positive cut-offs. A 50–75 µL aliquot of each sample was singly analysed on the Siemens VIVA-E platform using the EMIT-II PCP immunoassay.
Chemical similarity calculations
Chemical similarity calculations for ketamine, 2-oxo-PCE, MXE, 3-hydroxy-PCE, 2-MeO-PCE and 3-MeO-PCP in reference to PCP were performed using 2D molecular similarity analysis. Molecular similarity compares how similar two molecules are with respect to a variety of chemical features.7,8 In summary, this method evaluates multiple structural features using particular descriptors which describe the molecule (e.g., Number/types of atoms, etc.) to produce a binary vector. For each molecule these vectors are compared and the similarity determined using the Tanimoto coefficient as:
In this equation, ‘a’ is the number of features present in the reference molecule, ‘b’ is the number of features present in the comparative molecule, and ‘c’ is the number of features shared by the molecules.9,19 This approach has been previously used by the authors.2,20 Advantages of this method include relative ease to perform given the available software and a documented correlation with cross-reactivity for toxicology immunoassays.2,5,6 Potential disadvantages include possible exclusion of other differentiating molecular features or properties, 7 an inability to account for the complex 3D antibody–antigen interaction, and an inability to exactly predict the cross-reactivity concentration. 2
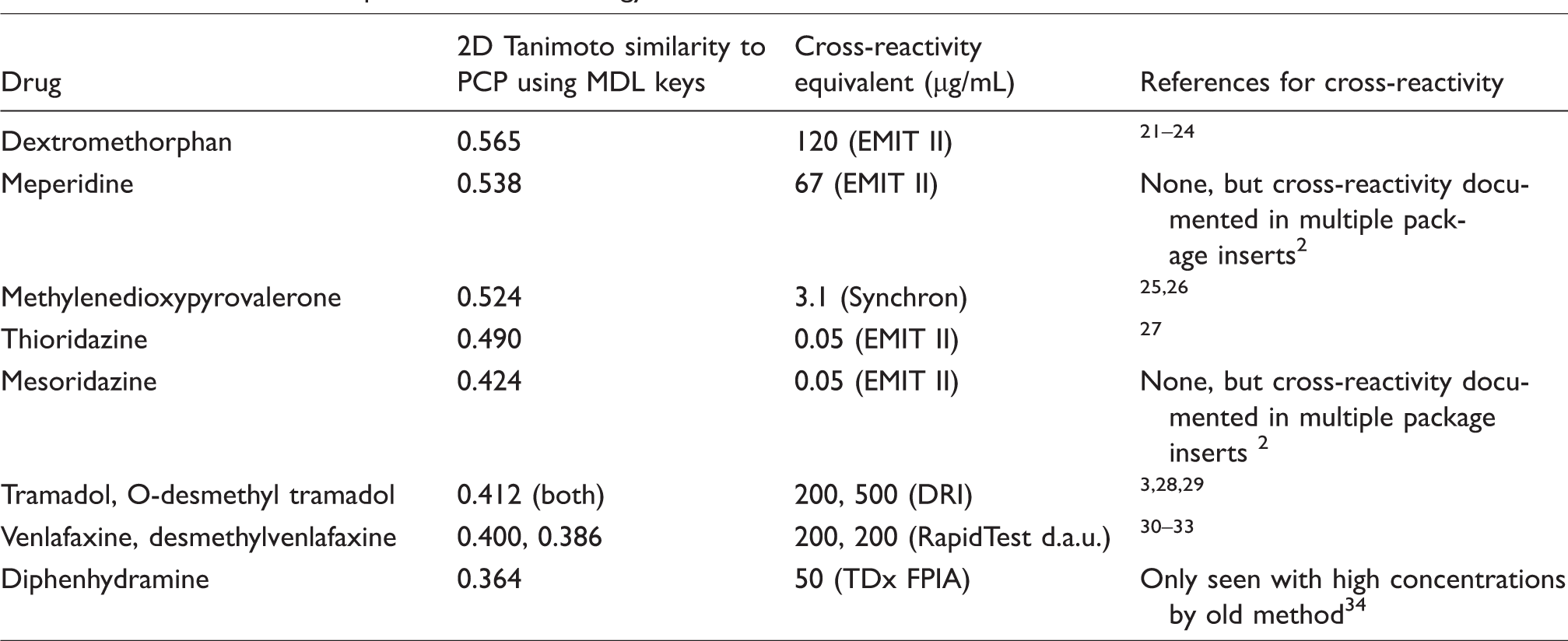
2D similarity searching analysis was performed using the ‘find similar molecules’ protocol in Discovery Studio version 3.5 (Biovia Inc., San Diego, CA, USA). MDL public keys for the drugs of interest were used as the input query and the Tanimoto similarity coefficients as the output. A similarity value of 1 implies maximum similarity (e.g., a molecule compared with itself or a very closely related compound), whereas 0 indicates maximal dissimilarity. 2D similarity for 2-oxo-PCE and related compounds were calculated. As a comparison, other compounds known to be cross-reactive with PCP immunoassays (according to manufacturer package inserts and/or reported in the literature) were included, and the data are summarized in Table 1.
2D similarity data for select drugs with reported cross-reactivity on PCP immunoassays. See the Materials and methods section for details on the computational methodology.
Results
Spiking studies
A 2-oxo-PCE concentration of 200 µg/mL and above produced cross-reactivity stronger than 25 ng/mL PCP (cut-off concentration of the PCP immunoassay), but that of 100 µg/mL failed to produce cross-reactivity equivalent to 25 ng/mL PCP (see Figure 3(a)). Therefore, 2-oxo-PCE causes a false positive PCP immunoassay result at a concentration of approximately 200 µg/mL and above. 3-MeO-PCE concentrations of 20 µg/mL and above produced cross-reactivity stronger than 25 ng/mL PCP, but that of 10 µg/mL failed to produce cross-reactivity equivalent to 25 ng/mL PCP (see Figure 3(b)). Thus, 3-MeO-PCE resulted in positive PCP immunoassay tests at a concentration of approximately 20 µg/mL and above.
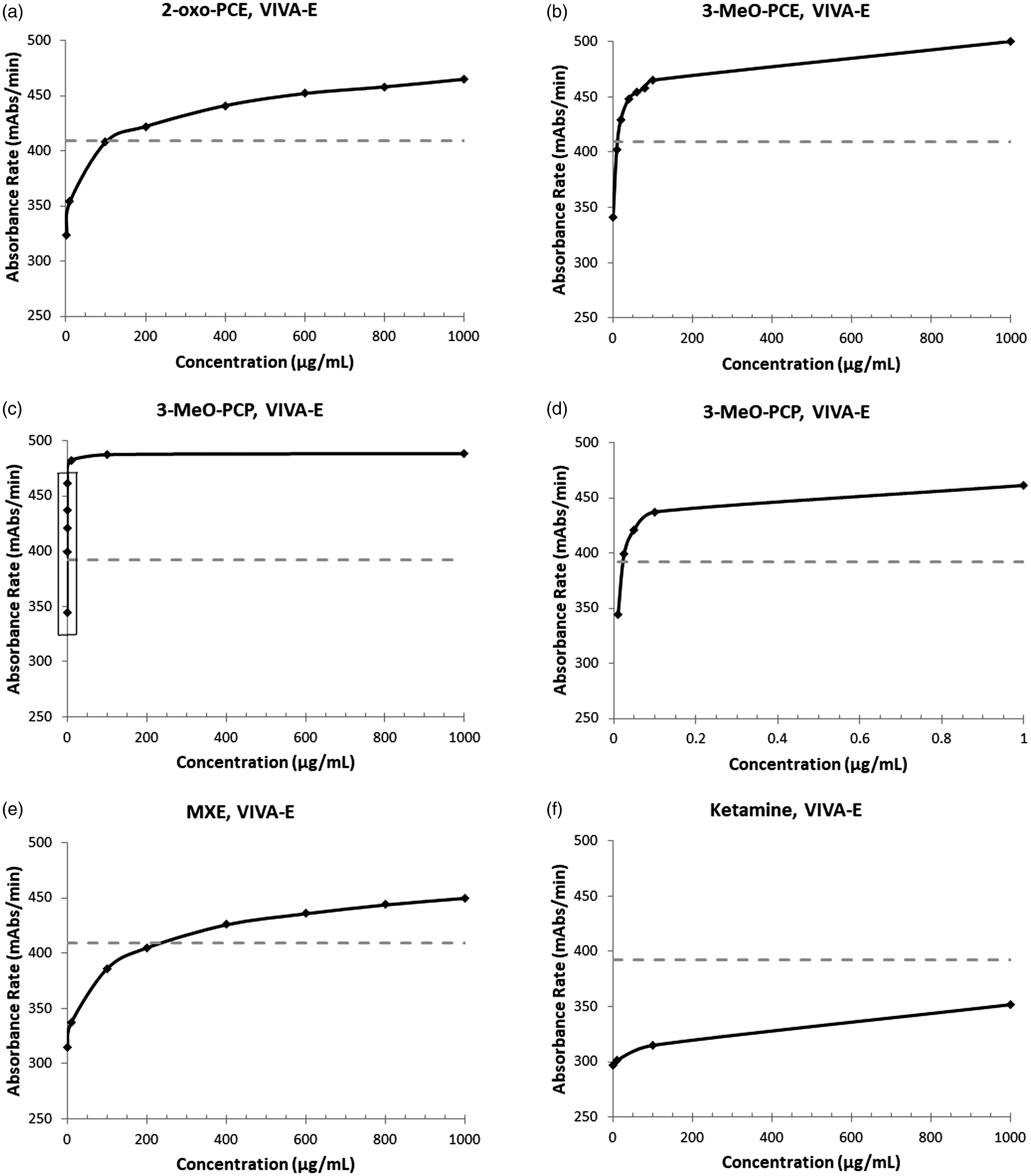
The dose–response curves of ketamine and PCP analogues in the EMIT-II PCP immunoassay. Blank urine samples spiked with 2-oxo-PCE (a), 3-MeO-PCE (b), 3-MeO-PCP (c,d), MXE (e) and ketamine (f) in the indicated concentration were analysed on Siemens VIVA E analyzer. (d) Dose–response curve of 3-MeO-PCP near the positive cut-off, shown as the area in square in (c). The dashed line represents positive cut-off of absorbance rate equivalent to 25 ng/mL PCP.
We also tested cross-reactivity of other PCP and ketamine analogues with the EMIT-II PCP immunoassay. A concentration of 3-MeO-PCP at 25 ng/mL produced cross-reactivity similar to 25 ng/mL PCP, whereas a concentration of 10 ng/mL failed to produce cross-reactivity equivalent to 25 ng/mL PCP (see Figure 3(c) and (d)). Thus, 3-MeO-PCP has similar cross-reactivity to PCP on the EMIT-II assay. An MXE concentration of 400 µg/mL and above produced cross-reactivity stronger than 25 ng/mL PCP, but that of 200 µg/mL failed to produce cross-reactivity equivalent to 25 ng/mL PCP (see Figure 3(e)). Thus, MXE resulted in a positive PCP immunoassay test at a concentration of approximately 400 µg/mL and above.
Ketamine concentrations of up to 1000 µg/mL failed to produce cross-reactivity equivalent to 25 ng/mL PCP. Thus, ketamine resulted in negative PCP immunoassay testing up to 1000 µg/mL (see Figure 3(f)). This is consistent with the manufacturer’s provided documentation of compounds resulting in negative tests (ketamine non-reactive at 100 µg/mL). Of note, all other tested compounds are not included on the package insert for the EMIT-II assay. A summary of these results can be seen in Table 2.
Summary of spiking study results with cross-reactivity data and 2D similarity calculations.
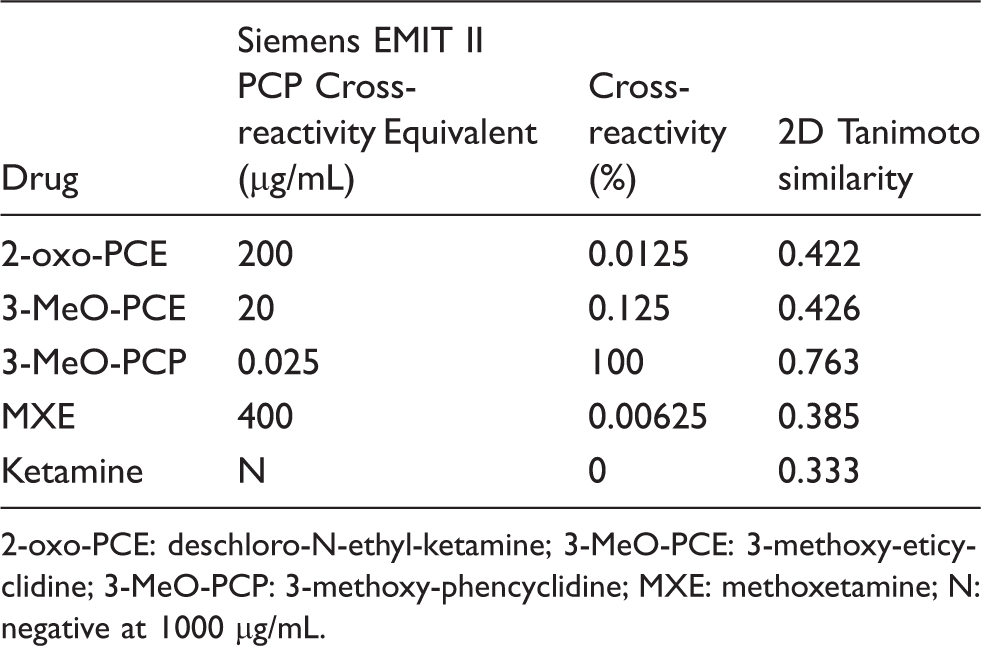
2-oxo-PCE: deschloro-N-ethyl-ketamine; 3-MeO-PCE: 3-methoxy-eticyclidine; 3-MeO-PCP: 3-methoxy-phencyclidine; MXE: methoxetamine; N: negative at 1000 µg/mL.
Overall, the relative cross-reactivity for each compound on the PCP immunoassay, from greatest to least, was as follows: 3-MeO-PCP > 3-MeO-PCE > 2-oxo-PCE > MXE > ketamine. This confirms that the false positive PCP immunoassay result in our patient was most likely due to 2-oxo-PCE, which was also suspected to be the causative agent of acute intoxication in this case.
Chemical similarity studies
A computational technique known as 2D molecular similarity analysis has previously been used to predict cross-reactivity of compounds to the target molecules of drug of abuse immunoassays,2,5,6 including PCP immunoassays. 3 We compared the cross-reactivity data obtained above (Figure 3) with the calculated 2D molecular similarity values of these compounds (Table 2, Figure 4). The relative cross-reactivity based on both the spiking studies and predicted cross-reactivity based on 2D similarity follow an identical pattern when compared with PCP as the target molecule: 3-MeO-PCP > 3-MeO-PCE > 2-oxo-PCE > MXE > ketamine (greatest to least).
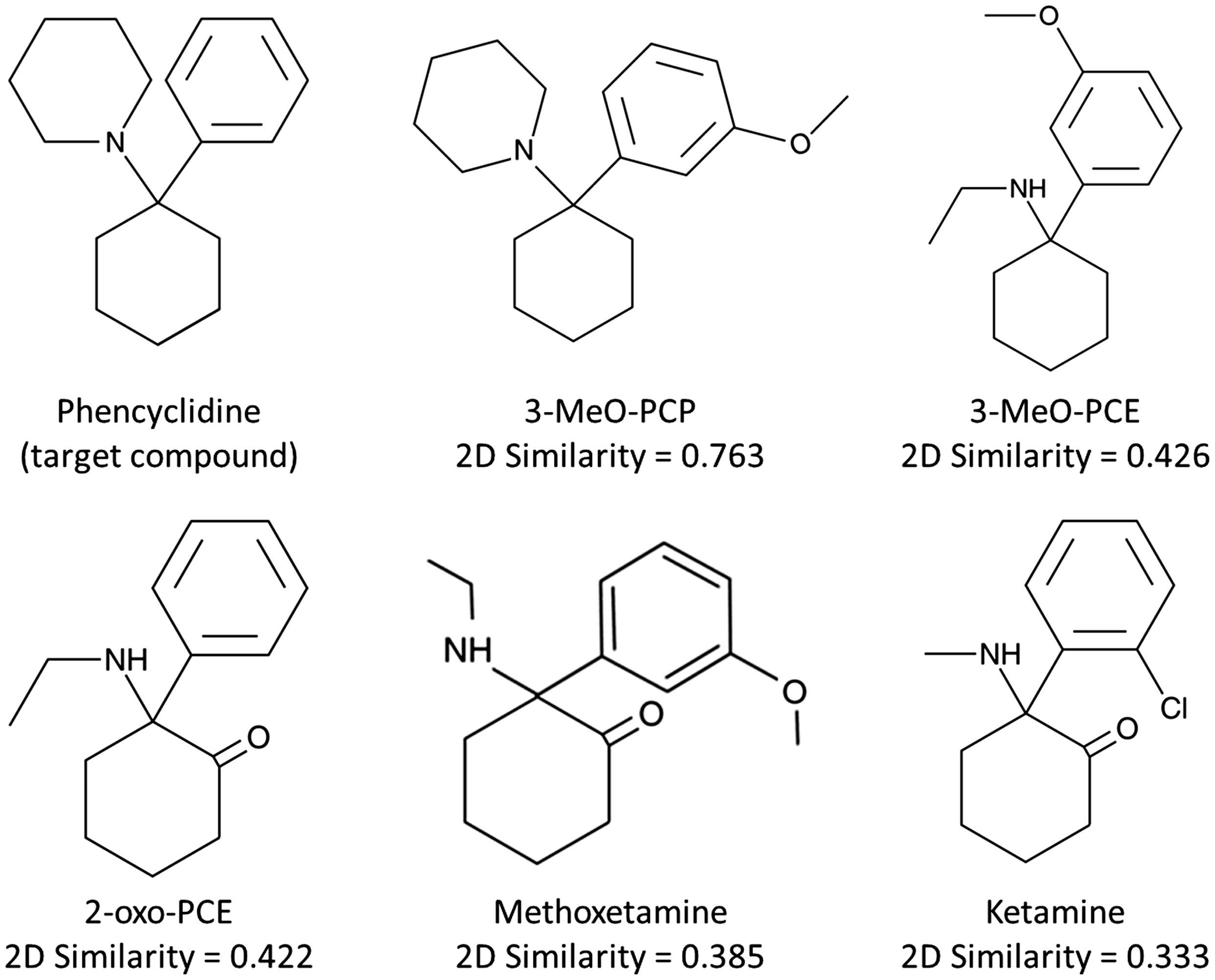
Chemical structures with 2D Tanimoto similarity scores to PCP using MDL keys.
Discussion
The scientific literature about 2-oxo-PCE intoxication is very limited, but 2-oxo-PCE seems to be more potent than ketamine. 18 Dehydration and rhabdomyolysis are the most frequent complications of ketamine intoxication. 35 Ketamine exerts the majority of its clinical effects through antagonism of the N-methyl-D-aspartic acid (NMDA) receptor, producing bilateral nystagmus and dissociated mental state, as seen in this patient. Due to its structural homology with ketamine and similar clinical effects, 2-oxo-PCE likely has a similar mechanism of action, although pharmacodynamic data are lacking. Data on the metabolism of 2-oxo-PCE are also lacking in the current literature. Both of these drugs were identified in this case, though the TIC peak for ketamine was much smaller than 2-oxo-PCE. The clinical symptomatology and laboratory findings, including urine drug testing (see Figures 1 and 2) and elevation of CPK and creatinine, all support the diagnosis of 2-oxo-PCE intoxication, with possible concomitant ketamine toxicity (see further discussion below). The urinary concentration of excreted 2-oxo-PCE should be at least 200 µg/mL based on the spiking studies, indicating the severity of the 2-oxo-PCE overdose (Figure 3). Quantification was not performed because this is not routine clinical practice at our institution and the patient sample was discarded per protocol before this was considered.
The spiking study results above demonstrate that all tested drugs except for ketamine can result in false positive PCP immunoassay results, particularly at higher concentrations. It can, therefore, be predicted that intoxication with any of the other analogues could result in a false positive PCP immunoassay, which has significant implications in the clinical and laboratory setting. The impact of metabolites of many of these analogues on the cross-reactivity is currently unknown due to the paucity of data about their metabolism. Additional studies are needed to establish common metabolites and allow for their clinical detection and evaluate their cross-reactivity individually. The 2D similarity analysis based on the molecular structures of these compounds further reinforces these cross-reactivity findings.
Previous studies have shown that strong cross-reactivity to PCP immunoassays generally occurred with compounds having 2D similarities of 0.6 or higher to PCP using MDL public keys as the molecular fingerprints.3,5 Of note, other studies that have looked at more general cut-offs have used a 2D similarity of 0.8, but this was to predict strong true positive reactions, whereas weakly true or false positive reactions tended toward lower values. 2 Compounds with 2D similarity reported in this range above 0.6 have generally been metabolites or analogues of PCP and produced cross-reactivity equivalent to 25 ng/mL PCP at test concentrations of less than 200 ng/mL. 3-MeO-PCP has a much higher 2D similarity (0.763) even compared with dextromethorphan (0.565), which is the most often encountered cross-reactant associated with positive screens with the EMIT-II PCP immunoassay kit at our institute. 3-MeO-PCP results in 100% cross-reactivity to PCP at 25 ng/mL (Figure 3(c)), which is consistent with the high 2D similarity of this molecule to PCP.
On the other hand, weaker cross-reactivity (e.g., equivalent cross-reactivity to 25 ng/mL PCP occurring only at urine concentrations of 10 µg/mL or higher) has been noted with compounds whose 2D similarity to PCP is as low as 0.3 to 0.4.2,3 Examples of drugs with well-documented cross-reactivity to multiple marketed PCP immunoassays include dextromethorphan (0.565), meperidine (0.538), mesoridazine (0.424), tramadol and its M1 metabolite, O-desmethyl tramadol (both 0.412), venlafaxine (0.400), and desmethylvenlafaxine (0.386) (Table 1). As seen in Table 1, these drugs show variability in their cross-reactivity concentrations, even among those with relatively similar 2D similarities, though the difference in platforms may be partially responsible. 2-oxo-PCE has 2D similarity to PCP of 0.422 and thus is in a 2D similarity range similar to mesoridazine and tramadol, which are both documented to produce cross-reactivity in multiple marketed PCP immunoassays. 3-MeO-PCE (0.426), 3-hydroxy-PCE (0.413), MXE (0.385), and MXE metabolites (0.370–0.373) also have a 2D similarity to PCP close to that of 2-oxo-PCE and thus would also be predicted to produce cross-reactivity with at least some PCP immunoassays. The cross-reactivity concentrations of dextromethorphan and meperidine are lower than those of 2-oxo-PCE and MXE, which have lower 2D similarities, but not 3-MeO-PCE. Mesoridazine, however, has a relatively high 2D similarity like that of the ketamine analogues but a much lower cross-reactivity concentration. Tramadol has both values in a similar range to the ketamine analogues. While the 2D similarity appears to be useful for predicting general cross-reactivity, its ability to predict exact concentrations is less robust. 2 Of note, MXE had the lowest 2D similarity of the tested compounds which were cross-reactive to PCP, and it also had the lowest relative cross-reactivity. Cross-reactivity of MXE, as well as another PCP analogue (4-methoxy-PCP) not evaluated in this study, to PCP has previously been documented. 36 Overall, our spiking data of 2-oxo-PCE, 3-MeO-PCE, and MXE (Figure 3) further support the validity of the 2D similarity-based predictions.
There are compounds with even lower 2D similarity to PCP that have historically been shown to produce cross-reactivity including diphenhydramine (0.364) and ketamine (0.333), but it is worth noting that these are older case reports, one of which used a now-outdated method (FPIA) and the other whose screening method was not defined.34,37 Additionally, these compounds have been reported as non-reactive in the majority of currently marketed PCP immunoassays. 2 The antibodies used in immunoassays are constantly evolving and becoming more specific for their intended targets. The specific epitope used for the antibodies also varies based on the manufacturer. These phenomena have likely contributed to the current absence of cross-reactivity for these compounds. Our spiking data of ketamine (Figure 3(e)) are compatible with the current understanding and manufacturer data. 2
In this case, we identified ketamine by GC-MS in addition to 2-oxo-PCE (Figure 1), but the origin of the ketamine remains in question. The metabolism of 2-oxo-PCE is currently unknown, but chlorination of 2-oxo-PCE is unlikely to take place in the body. Ketamine was not given clinically in this case. Therefore, ketamine was most likely a component or contaminant of the drugs purchased online or ingested separately by the patient rather than synthesized through metabolism in the body. Samples of the drugs that the patient ingested were not available for further analysis to confirm this theory.
Clinical decisions for acute overdose cases are often made in conjunction with the drug testing with immunoassays because of their rapid turnaround time. 1 The ability to detect these newly emerging drugs of abuse, as well as knowledge about their reactivity on traditional assays, is critical in the laboratory and clinical setting, but it is often unknown, as exemplified in this clinical case study. Thus, the cross-reactivity data presented in this current study will aid the clinician in identifying a possible drug exposure and guide medical decision making in a timely manner. The ever-increasing list of cross-reactive drugs also highlights the importance of performing confirmatory testing by more specific methods, particularly in unexpected cases. Our study also further supports the validity of the 2D molecular similarity calculation for prediction of cross-reactivity of these molecules in the appropriate clinical context. While this method may not be able to definitively predict a cross-reactivity concentration, the ability to predict general cross-reactivity is likely more important in a clinical scenario where a false positive is being considered. This may be particularly important in the current climate of the emerging novel, synthetic drug analogues in order to predict their potential for cross-reactivity with immunoassays.
Conclusions
Several ketamine and PCP analogues can result in false-positive PCP immunoassay testing based on our spiking study results. Knowledge of potentially cross-reactive compounds is important for both laboratories and clinicians. Additionally, 2D molecular similarity analysis correlates well with the tested drugs, indicating that this method could be helpful in predicting cross-reactivity of novel drugs on available immunoassays for more traditional drugs in certain clinical scenarios. The application of this analysis could help toxicology laboratories in evaluating unexpected results, particularly when novel synthetic drugs are being considered. The accurate interpretation of toxicology testing is crucial for both the laboratory itself and the clinicians that the laboratory supports.
Footnotes
Acknowledgements
SE kindly acknowledges Biovia for providing Discovery Studio.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SE is CEO of Collaborations Pharmaceuticals, Inc. and Phoenix Nest, Inc. The other authors have no conflicts of interest to disclose.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Guarantor
KT.
Contributorship
JMS performed the manuscript drafting and figure development. KT was involved with the study design, supervision and manuscript writing. JMS, JAR and SG performed the spiking studies. AS and AFP provided clinical toxicology expertise. MDK and SE provided expertise on computational chemical similarity calculations and performed the molecular similarity analysis. All authors reviewed and edited the manuscript and approved the final version.