Abstract
Background
The national programmes for the harmonization of haemoglobin A1c measurement in the US and Japan are based on differently designated comparison methods. The future basis for international standardization is expected to be the reference system developed by the International Federation of Clinical Chemistry (IFCC) Working Group on haemoglobin A1c Standardization. The aim of the present study is to compare the relationship between the IFCC reference method (RM) and Japanese Diabetes Society (JDS) RM used for the conversion to the National Glycohemoglobin Standardization Program (NGSP) values.
Methods
Three different method-comparison studies were performed. All blood samples were measured at the National Cerebral and Cardiovascular Centers (Lipid Reference Laboratories) that serve as Level 1 reference laboratories of the NGSP Network. Regression equations were calculated for the IFCC RM and JDS RM for the conversion to NGSP values.
Results
Differences were found between the haemoglobin A1c values of the IFCC RM and those of JDS. However, in all cases, the relationships of the IFCC RM and JDS RM were linear and commutable. The relationship is described by the following regression equations: NGSP-HbA1c = 0.915(IFCC-HbA1c) + 2.15% (r2 = 0.998); JDS/JSCC-HbA1c = 0.927(IFCC-HbA1c) + 1.73% (r2 = 0.997).
Conclusion
There is a firm and reproducible link between the IFCC and JDS-HbA1c values. However, the values calibrated by JDS RM were consistently and significantly higher than the IFCC values (0.1–0.2%) when used for conversion to the NGSP values.
Keywords
Introduction
The haemoglobin A1c (HbA1c) reading reflects a time-averaged blood glucose concentration during the previous two to three months, and is used as the gold standard for long-term follow-up of glycaemic control. The same HbA1c method was used in both the Diabetes Control and Complications Trial (DCCT) study published in 1993 1 and the UK Prospective Diabetes Study (UKPDS) published in 1998. 2 Following the development of a reference measurement procedure (RMP) by the International Federation of Clinical Chemistry and Laboratory Medicine (IFCC), 3 it was stipulated by an international consensus that all manufacturers should make their routine methods traceable to this RMP. 4 A second consensus meeting was held in 2009, where the main issue was how the results should be presented to clinicians and patients and how they should be reported in scientific journals. 5
The HbA1c values in Japan have been calibrated and published in the Japanese Clinical Laboratory Use Certified Reference Material (JCCRM). The certified values are determined by high-resolution type ion-exchange high performance liquid chromatography (HPLC) (i.e. the KO500 method) and certified using the designated comparison method (DCM) of the Japan Society of Clinical Chemistry as well as the Japan Diabetes Society (JDS). 6 After incorporating a proportional bias correction to the value anchored to the peptide mapping method of IFCC, the JDS measures b-N-mono-deoxyfructosyl haemoglobin and has an intercept approximately equal to zero against the peptide mapping method of IFCC in terms of measuring fresh raw human blood samples.7,8
The standardization of HbA1c in Japan was initiated in 1993, and to date, the serial reference materials from JDS Lot 1 to JDS Lot 5 have been certified using the DCM. 9 In the criteria, the cut-point of HbA1c (JDS) for the diagnosis of diabetes mellitus was 6.1%, which is essentially equivalent to the internationally used HbA1c (National Glycohemoglobin Standardization Program [NGSP]) of 6.5%, as HbA1c (NGSP) (%) is reported to be equivalent to the 1.019 HbA1c (JDS) % in the ‘Criteria of Diabetes Mellitus’ published in the Journal of Diabetes Investigation. 10 and Diabetology International. 11 The new diagnostic criteria in Japan came into effect on 1 July 2010. According to the new version, HbA1c (JDS) 6.5% is now considered to indicate a diabetic criterion, but the previous diagnosis criteria of high plasma glucose (PG) concentrations in the control state at 0.3%, which is reasonably estimated by the equation of HbA1c (JDS) % + 0.4%, is within the margin of error of HbA1c measurements (2–3%).
In Japan, the conversion equation from the HbA1c (JDS) to HbA1c (NGSP) value was officially certified in a council meeting of the JDS, in which it was decided to use the HbA1c (NGSP) values in clinical practice starting on 1 April 2012, although the HbA1c (JDS) values were to be included until people become familiar with the new form of expression. Finally, it is important to emphasize that the new HbA1c (NGSP) values have been directly measured and printed out since 1 April 2012. However, both the new diagnostic reference values and target values for glycaemic control have been adjusted to these equivalent values of HbA1c (JDS) in Japan.
HbA1c analysis reportedly performed with HPLC at more than 70% of the hospitals in Japan. Currently, most of the uses of HbA1c as a primary standard value in Japan with the KO500 (JDS) method. 6 This HbA1c calibrator uses a freeze-dried extract of erythrocytes, which is a different matrix from the whole blood of the HbA1c calibrator provided by IFCC. The Health Care Technology Foundation (HECTEF) in Japan collaborated with the IFCC network and developed the two-dose IFCC calibrator (reference and abnormal range) named ‘HECTEF HbA1c calibrator set’, which was approved by the Japanese Committee for Clinical Laboratory Standards (JCCLS) as the ‘IFCC HbA1c calibrator’. Using the HECTEF HbA1c calibrator, we compared the analytical values obtained with the DCM-KO500 (JDS) and IFCC calibrator in blood sent from NGSP (the mean of the Secondary Reference Laboratory [SRL]) and along with the health check-up population and routine clinical practice results.
Materials and methods
The HbA1c concentration was measured with the HLC-723 G8 at the National Cerebral and Cardiovascular Center, that was calibrated by the HECTEF HbA1c calibrator (IFCC calibrator) and KO500 (JDS) (TOSOH Bioscience (Tokyo)) based HbA1c calibrator. The eight doses of blood used in the IFCC HbA1c calibrator were obtained from the IFCC Network coordinator Dr. Cas Weykamp [Streekziekenhuis Koningin Beatrix, The Netherlands]) as the original IFCC calibrator for comparison with HECTEF HbA1c calibrator. 3 Eighty blood samples (eight times at three-month intervals during the period 2015 to 2017) sent from NGSP with dry ice were kept frozen kept –80°C until analysis. Forty anonymous patient blood samples randomly selected were obtained at Hidaka Hospital and kept refrigerated until analysis.
The analysis of an anonymous Japanese health check-up population (n = 20,466) and outpatient samples (n = 13,322, n = 40) was performed just for the calculation of the data among the previously determined by JDS RM in de-identified those study population at the Hidaka Hospital. The correlation and commutability between the blood samples of domestic and international origin were evaluated by the two methods (IFCC and JDS) in order to convert them to NGSP values. The correlation co-efficiency and sy/x of the actual measurement value were also evaluated. A statement of institutional approval of the study in accordance with the Declaration of Helsinki was provided. The study was approved by the ethical committees of Hidaka Hospital (Ref. No. 127).
Statistical analysis
Statistical analysis was conducted with Statflex version 6 (Artech, Osaka, Japan). Paired t-test was used to compare the difference in bias among the three different calibrators. Pearson’s correlation analysis was conducted between the mean of SRL value and IFCC converted to NGSP or JDS converted to NGSP. McNemar test was used to compare the relationship between the IFCC and JDS calibrators when used to convert to NGS values. Results are expressed as the mean (SE), and P < 0.05 was considered significant.
Results
Comparison of the three HbA1c calibrators available for use in Japan
We obtained the three kinds of HbA1c calibrators available in Japan (Table 1). All were composed of two certified values (within the reference and diabetic range, respectively). One was the calibrator distributed by the IFCC (HECTEF HbA1c calibrator) (whole blood). The others were two kinds of calibrator obtained from TOSOH Bioscience (freeze-dried blood extract). One of the latter two was a calibrator designed for overseas use that was adjusted to the IFCC calibrator (the original eight-dose calibrator). The other was designed for domestic use and was adjusted to the JDS calibrator. HbA1c values of 40 anonymous blood samples that were randomly selected were determined by three different calibrators and the % of bias was calculated for the HECTEF HbA1c calibrator (IFCC calibrator) and TOSOH calibrator for overseas use (adjusted to IFCC), and also the % of bias between the IFCC and TOSOH calibrators was designed for domestic use (adjusted to JDS). The average % of bias between the IFCC calibrator and the TOSOH calibrator designed for overseas use was significantly smaller (1.61%) than that between the IFCC and TOSOH calibrators designed for domestic use (2.52%) (P < 0.001). This result indicated that the IFCC values were obtained by the IFCC calibrator (whole blood) and the TOSOH overseas calibrator (extracted and freeze-dried HbA1c), the values of which were adjusted by the IFCC calibrator and still displayed a difference because of the different matrixes used for HbA1c, although the difference was smaller than that of the domestic calibrators adjusted to the KO500 JDS calibrator.
Comparison among the three kinds of HbA1c calibrator.
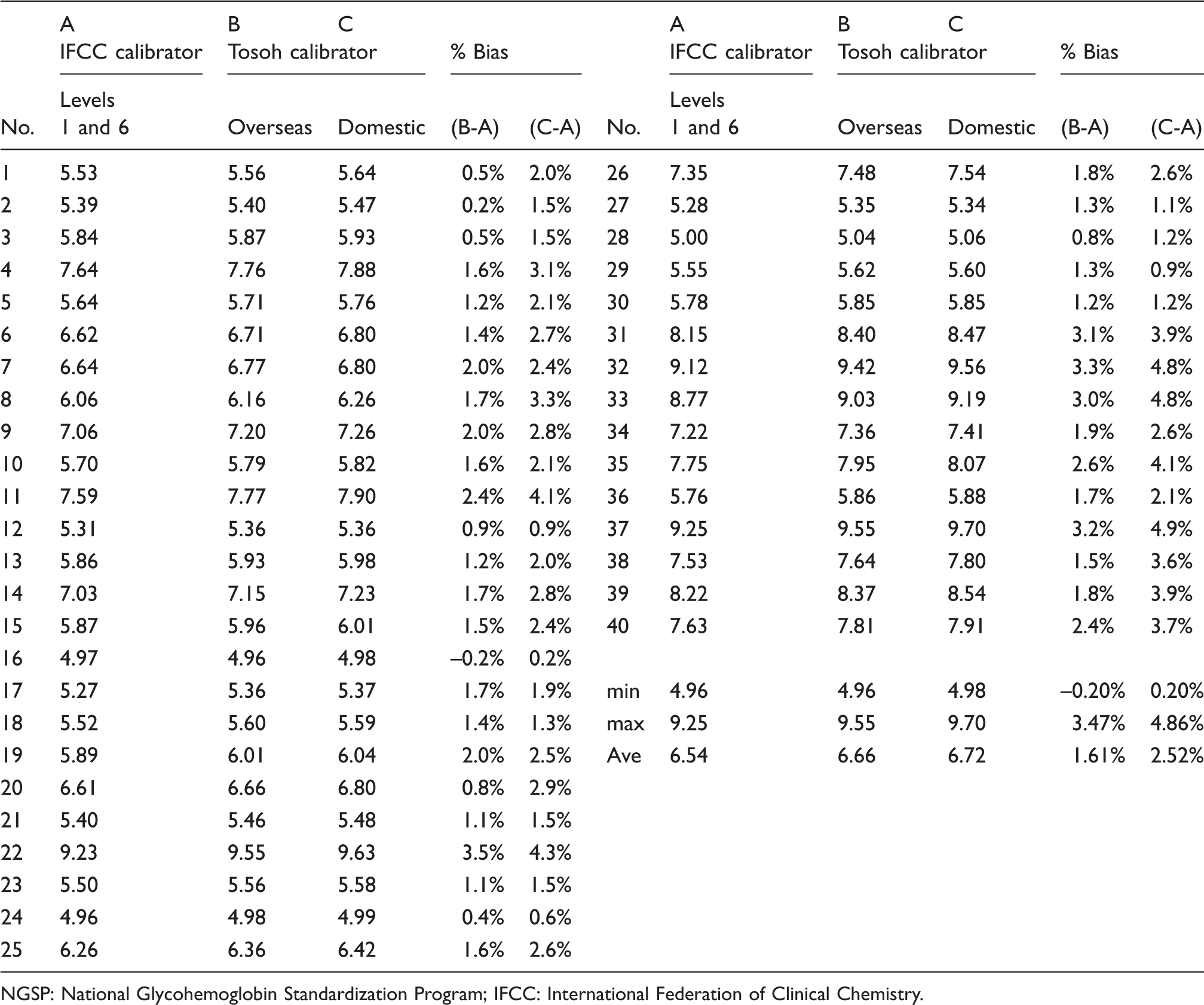
NGSP: National Glycohemoglobin Standardization Program; IFCC: International Federation of Clinical Chemistry.
NGSP values obtained by the JDS calibrator
The HbA1c values of the 80 blood samples from NGSP were determined with the JDS calibrator at eight different time points and compared with the NGSP values measured at the Secondary Reference Lab of the University of Missouri (NGSP-SRL) (Figure 1). We measured 10 blood samples every three months during the period 2015–2017. The plots show the values of the blood samples assayed at the eight different times. The 10 plots of the different HbA1c concentrations obtained with the JDS calibrator were linear but widely dispersed when compared with the NGSP (mean of the SRL) values.
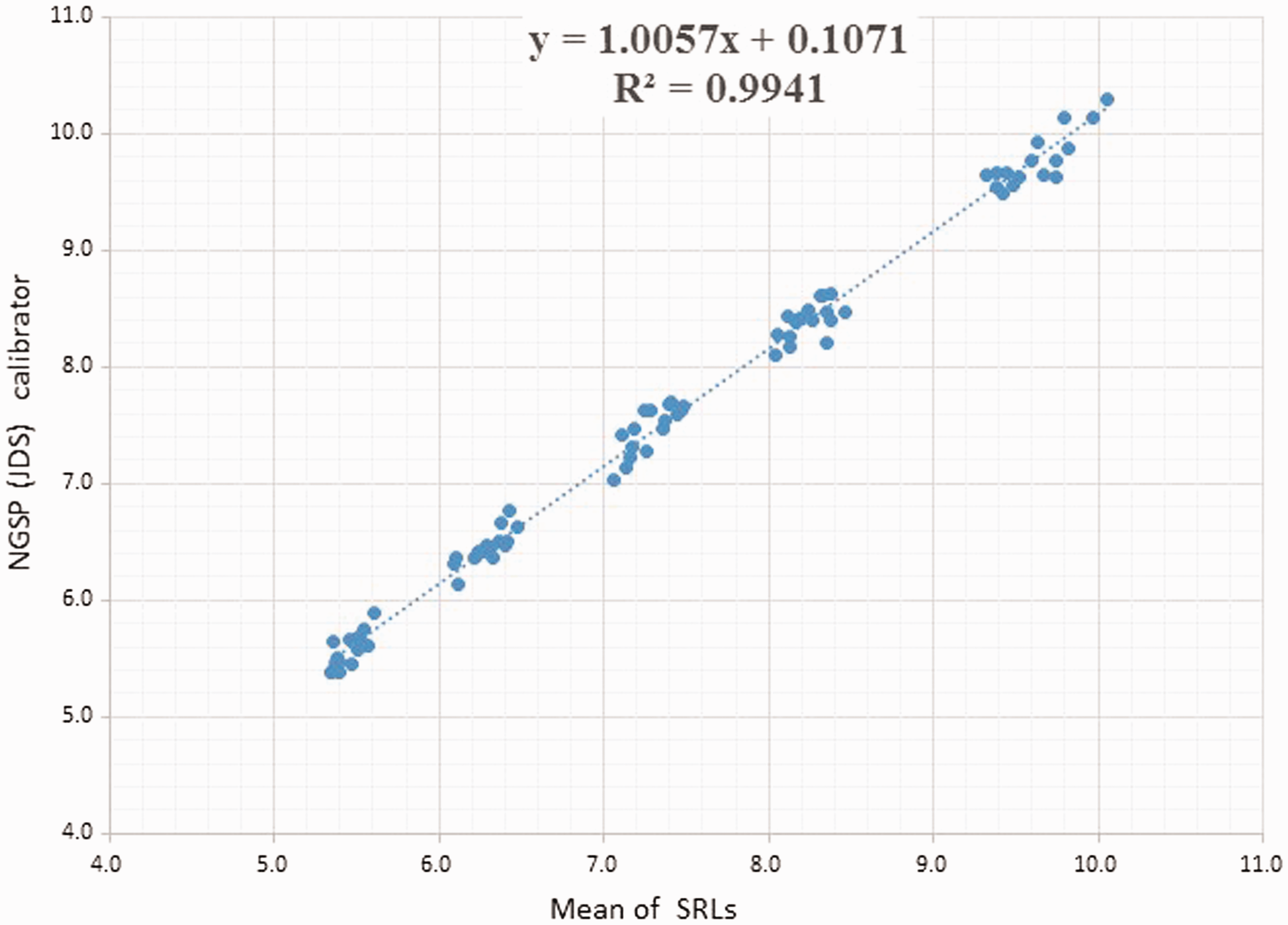
The HbA1c values of the 80 blood samples from NGSP were determined with the JDS calibrator at eight different time points and compared with the NGSP values measured at the Secondary Reference Lab of the University of Missouri (NGSP-SRL).
NGSP values obtained with the IFCC calibrator
The HbA1c values of 80 blood samples obtained from NGSP were determined with the IFCC calibrator (Figure 2). The plots show the values of the blood samples assayed at eight different times with the same manner as the JDS calibrator and compared with the NGSP (mean of the SRL) values. The 10 plots of the different HbA1c concentrations measured at eight time points using the IFCC calibrator were linear and less widely dispersed when compared with the NGSP (mean of the SRL) values than those of the NGSP-JDS values.
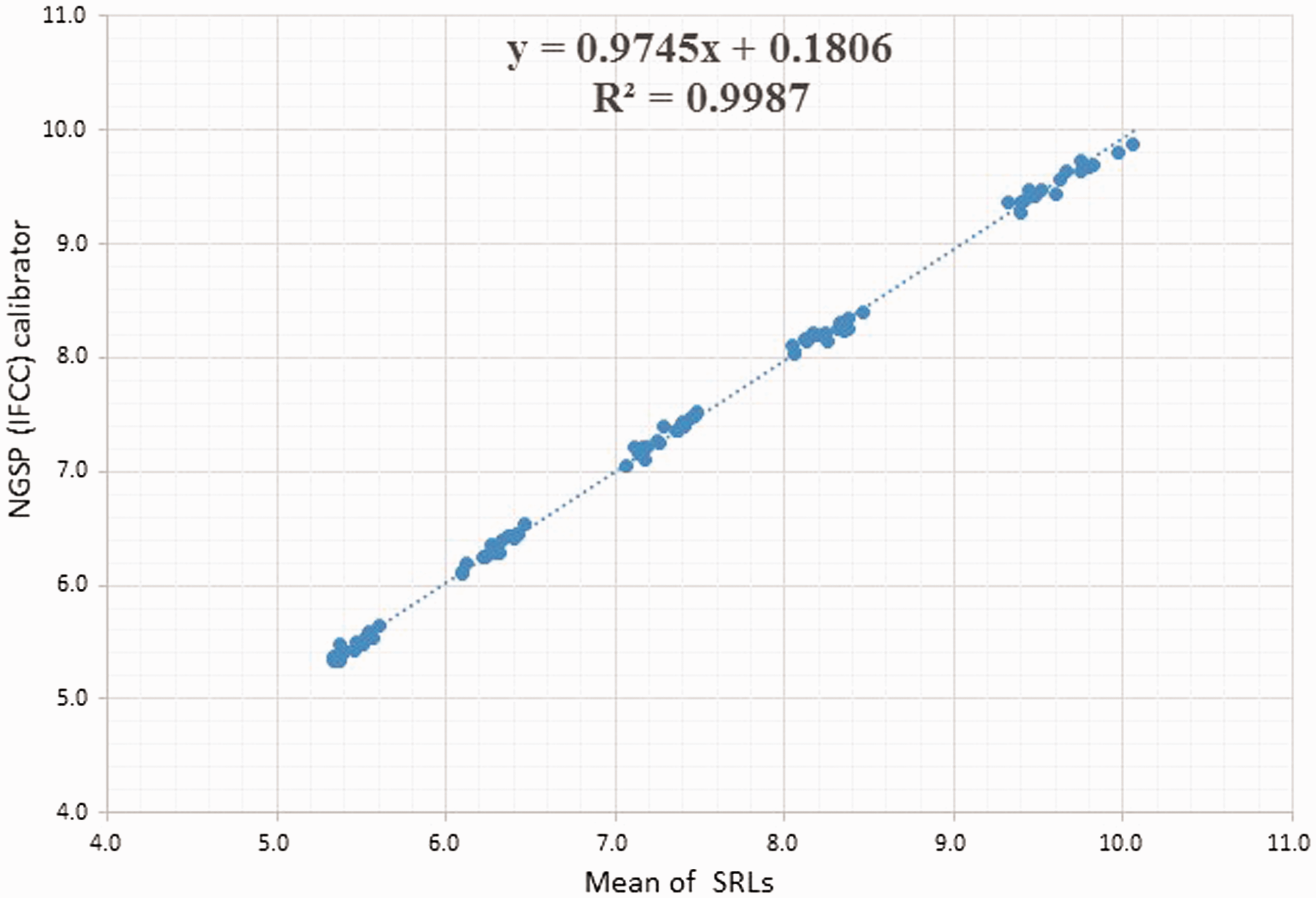
The HbA1c values of 80 blood samples obtained from NGSP were determined with the IFCC calibrator.
Comparison of the eight time point assay results obtained with the IFCC and JDS calibrators
The HbA1c values of the blood samples received from NGSP (n = 80) were determined with the IFCC calibrator (Figure 3(1)) and JDS calibrator (Figure 3 (2)), and compared directly with the NGSP (mean of the SRL) values. The plots show the eight times assay results of the 10 blood samples every three months during the period 2015–2017. When the IFCC value was 6.5%, the NGSP (mean of SRL) value was almost 6.5%, indicating high commutability. However, when the JDS values were 6.5%, the IFCC value was significantly lower and dispersed in a range of 6.2–6.5%. This indicates that the JDS values converted to NGSP values were higher than the IFCC values, and dispersed significantly when assayed at different times.
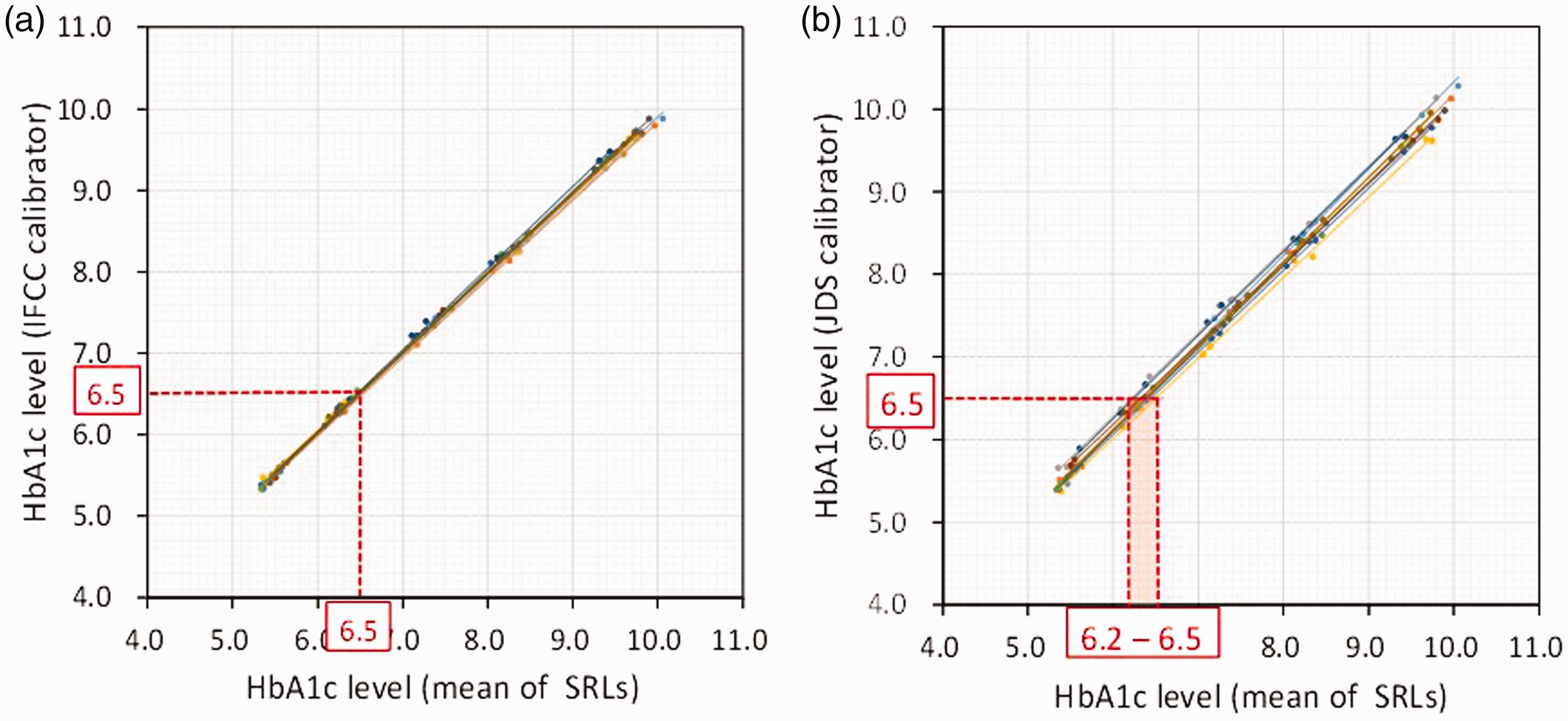
The HbA1c values of the blood samples received from NGSP (n = 80) were determined with the IFCC calibrator and JDS calibrator, and compared with the NGSP-mean of the SRL.
Clinical results; comparison of the NGSP-IFCC calibrator and NGSP-JDS calibrator in population studies and hospital outpatient cases
The following two different clinical data previously reported as JDS values were re-calculated by the IFCC calibrator and JDS calibrator. The following is the regression equation calculated using the 80 samples in order to convert to NGSP values.
The HbA1c values of the health check-up population and the outpatient cases at the Hidaka Hospital were examined as NGSP (JDS) and NGSP (IFCC) data. The average value of the health check-up population showed that the JDS values were significantly higher than the IFCC values (5.36% by the JDS and 5.26% by IFCC calibrator; P < 0.001) in the health check-up population (Table 2). In the cases above, the result of HbA1c ≤6.5 by JDS was 8.0% higher than the IFCC value. Among the four classifications, NGSP (IFCC) was approximately 10% lower in total, and the 16–34% cases that were above 5.6% in NGSP (JDS) were shifted to a lower classification by NGSP (IFCC). The lower NGSP (JDS) cases were shifted to a lower NGSP (IFCC).
The comparison between NGSP (IFCC) and NGSP (JDS) HbA1c values among the four classifications in health check-up population.
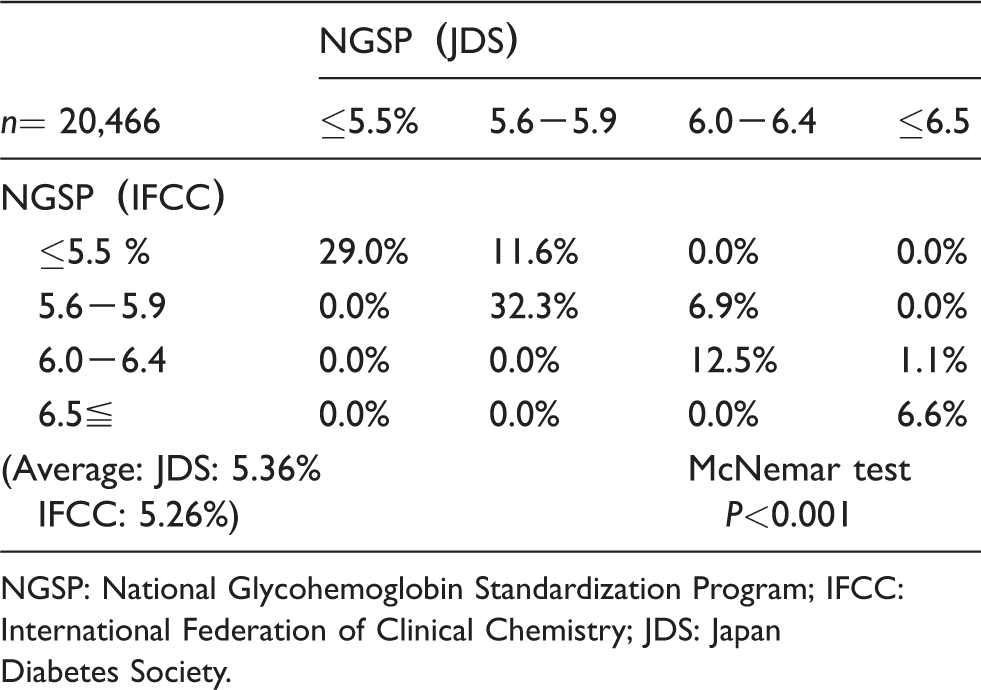
NGSP: National Glycohemoglobin Standardization Program; IFCC: International Federation of Clinical Chemistry; JDS: Japan Diabetes Society.
The outpatient cases showed the average HbA1c value was 7.07% by the JDS calibrator and 6.92% by the IFCC calibrator. Among the four classifications, NGSP (IFCC) was approximately 10% lower in total, and 16–38% of the cases above 5.6% in NGSP (JDS) were shifted to a lower classification by NGSP (IFCC) (Table 3). The lower NGSP (JDS) cases were shifted to a lower NGSP (IFCC). The percentage of border zone cases was 19.4% (6.0–6.4%) by NGSP (IFCC) in the outpatient cases. There were 21% of the NGSP (JDS) cases above 6.5% among them.
The comparison between NGSP (IFCC) and NGSP (JDS) HbA1c values among the four classifications in outpatient study.
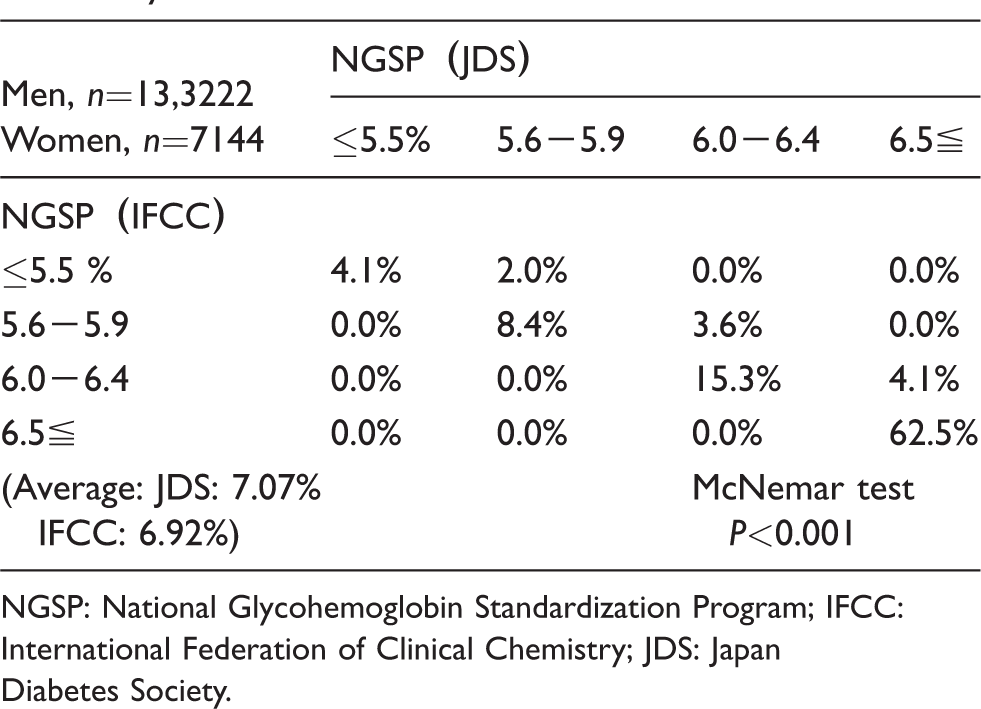
NGSP: National Glycohemoglobin Standardization Program; IFCC: International Federation of Clinical Chemistry; JDS: Japan Diabetes Society.
Discussion
This is a study performed for the international clinical harmonization of HbA1c in Japan, comparing JDS RM and IFCC RM to determine when to adjust to NGSP standardization.
We have shown that there is an appropriate commutability between the HECTEF HbA1c calibrator (IFCC) and the KO500 method (JDS) (TOSOH calibrator) for the determination of HbA1c values. However, the values calibrated by the KO500 method (JDS) were consistently higher and more dispersed than the IFCC values (0.1–0.2%) when converted to NGSP values. Because the values of TOSOH calibrator for overseas use and for domestic use are different in spite of the same calibrator specimens, overseas values are adjusted to IFCC calibrator (eight concentrations) and domestic values are adjusted to JDS calibrator which express the higher value than overseas value. Also, the reason for the dispersion among the values obtained by the KO500 method may be associated with the three-sided adjustment among the JDS, IFCC and NGSP values. Conversion factor may change among the lot of JDS and the matrix difference between IFCC calibrator (whole blood) and JDS calibrator (freeze-dried blood extract).
The Reference Material Institute for Clinical Chemistry Standards was certified as an Asian Secondary Reference Laboratory using the KO500 method which became the source for the reference materials JCCRM411-2 (JDS Lot 4) in Japan after the completion of the NGSP network laboratory certification. Therefore, the JDS HbA1c value may be said to be traceable to the DCCT reference method. 1 The comparison was carried out at the Central Primary Reference Laboratory in the University of Missouri School of Medicine. 12 The conversion equation from the HbA1c (JDS) to HbA1c (NGSP) value is officially defined as follows: NGSP (%) = 1.02 × JDS (%) +0.25%; conversely, JDS (%) = 0.980 × NGSP (%)–0.245%. Based on this equation, in the range of JDS values below 4.9%, NGSP (%) = JDS (%) + 0.3%; in the range of JDS 5.0–9.9%, NGSP (%) = JDS (%) +0.4%; and in the range of JDS 10–14.9%, NGSP (%) = JDS (%) + 0.5%. These results indicate that the previous equation of NGSP (%) = JDS (%) + 0.4% is confirmed in the present equation, considering a 2–3% error of HbA1c measurement. On the contrary, the conversion of IFCC to NGSP is defined as a master equation as follows: NGSP (%) = 0.09148 ×IFCC (mmol/mol) + 2.152.
After the conversion to the NGSP values with the IFCC and JDS calibrators, we compared the distribution differences of health check-up population and the outpatient cases. The frequency of the cases with an HbA1c value above 6.5% was 2–7% in the health check-up population study, but more than 60% were above HbA1c 6.5% among the outpatient cases. In particular, in the results for the hospital outpatients, the HbA1c concentrations valued by the JDS calibrator showed that approximately 20% of the cases whose HbA1c concentration was more than 6.5% were determined to be 6.0–6.4% by the IFCC calibrator. In order to adjust to the global standard of HbA1c measurement, it may be necessary to use the IFCC HbA1c calibrator for clinical studies in Japan, especially epidemiological studies and health check-up purposes. By using the IFCC calibrator, we could reduce the number of patients diagnosed as diabetes because of an HbA1c above 6.5% by approximately 10%, because most of the HbA1c results are currently obtained by the JDS calibrator in Japan.
In conclusion, we have shown that there is a firm and reproducible link between the IFCC and JDS- HbA1c values. However, the values calibrated by JDS RM consistently significantly higher than the IFCC values (0.1–0.2%) when converted to the NGSP values. These results may affect the important difference among the epidemiological studies when compared with the studies calibrated by IFCC calibrator and the diagnosis of diabetes at an HbA1c 6.5% concentration.
Footnotes
Acknowledgements
The authors thank Dr. Naotaka Hamasaki (JCCLS) and Dr. Cas Weykamp (IFCC) for their generous discussions and suggestions for this research.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethical Committee of Hidaka Hospital, approval Ref. No. 127.
Guarantor
KN.
Contributorship
IK performed experiments, analysed the data and contributed the scientific discussion. MW analysed the data and contributed the scientific discussion. MN and YM contributed the scientific discussion. YT, KS and SIY analysed the data and contributed the scientific discussion. YS contributed the scientific discussion. KN designed the study, analysed the data and wrote the article.
