Abstract
Background
To promote glycated albumin standardization, the Committee on Diabetes Mellitus Indices, Japan Society of Clinical Chemistry published the recommended reference method and is supplying the reference standard material (JCCRM 611). In this study, we evaluated the basic performance of ‘standardized enzymatic method’ that is traceable to JCCRM 611 and established a unit conversion formula from standardized glycated albumin value (mmol/mol) to the routinely used glycated albumin value (%).
Methods
To evaluate the accuracy and within-run reproducibility of the ‘standardized enzymatic method’, JCCRM 611 was measured 20 times. To establish the unit conversion formula, serum specimens with known HbA1c concentrations were collected, and their glycated albumin (%) and glycated albumin (mmol/mol) were measured.
Results
The accuracy of glycated albumin value of the ‘standardized enzymatic method’ was 100.8–103.0%. The within-run reproducibility CV (coefficient of variation, %) of glycated albumin value was 0.6–0.9%. In total, 240 serum specimens were collected, and the distribution of their HbA1c values was HbA1c ≤ 4.9% (n = 23), 5.0–5.9% (n = 50), 6.0–6.9%, (n = 44), 7.0–7.9% (n = 40), 8.0–8.9% (n = 37) and ≥9.0% (n = 46). The unit conversion equation was defined as: GA (%) = 0.05652 × GA (mmol/mol)–0.4217 (r = 0.999). Glycated albumin (%) calculated by the equation was in close agreement (96.6–104.2%) with the actual measured glycated albumin (%).
Conclusions
The glycated albumin unit conversion formula was established from mmol/mol to %. This formula provides an easy method to convert the unit and is expected to be useful for the standardization of glycated albumin measurement.
Introduction
The number of patients with diabetes was announced to be 425 million worldwide in 2017, which has become a large social problem. 1 Continuous blood glucose control is highly important in the treatment of diabetes.2–4 In addition to blood glucose concentration, haemoglobin A1c (HbA1c) and glycated albumin (GA) are used as glycaemic control markers.
HbA1c reflects the average glycaemic concentration during the previous two to three months. Based on the results of large-scale clinical trials, such as the Diabetes Control and Complications Trial (DCCT), 2 the United Kingdom Prospective Diabetes Study (UKPDS) 3 and the Kumamoto Study, 4 HbA1c is used as the index for glycaemic control. In addition, HbA1c is also widely used for the diagnosis of diabetes and the risk prediction of diabetes complications. 5 Therefore, HbA1c is the gold standard of glycaemic control index, and the standardization of HbA1c is considered to be important. It has been proceeded with the National Glycohemoglobin Standardization Program (NGSP) and the International Federation of Clinical Chemistry and Laboratory Medicine (IFCC). 6
GA reflects the average blood glucose over the previous two to three weeks. As an intermediate-term glycaemic control index, GA is believed to be useful to confirm the effectiveness of diabetes treatment when it is started or modified and to replace HbA1c when HbA1c values are inaccurate. 7 Moreover, evidence indicating the association of GA with diabetes complications has been accumulating based on the results of large-scale clinical trials, such as the DCCT/Epidemiology of Diabetes Interventions and Complications, 8 Atherosclerosis Risk in Communities study, 9 the Hisayama Study 10 and the study by Jia et al. 11
While GA is measured by the high performance liquid chromatography method, 12 enzymatic method, 13 immunological methods,14,15 chemical methods 16 and others, the obtained values vary. Under these circumstances, in 2002, the Committee on Diabetes Mellitus Indices (cDMI), Japan Society of Clinical Chemistry started the GA standardization project. In 2008, cDMI published the ‘JSCC Recommended Method for Glycated Albumin Measurement in Serum’ and started to supply the reference standard material JCCRM 611 with assigned GA value expressed in international units (mmol/mol) from the Reference Material Institute for Clinical Chemistry Standards (ReCCS).17,18
The JSCC-recommended method for measuring GA stipulates that GA was initially defined as albumin (ALB) containing lysine residue irreversibly bound to glucose. In this method, ALB is separated from the serum, and stable isotopes of glycated lysine and lysine (Lys) were added as the internal standard. After hydrogenation, the ALB solution undergoes hydrolysis into its constituent amino acids using hydrochloric acid. Next, the resulting Nε-(1-deoxy-D-fructos-1-y1)-L-lysine (DOF-Lys) and Lys are quantified using mass spectrometry (MS) to obtain DOF-Lys concentration (μmol/L) and ALB concentration (μmol/L). Finally, the DOF-Lys concentration (μmol/L) is divided by the ALB concentration (μmol/L) to obtain the GA value (mmol/mol).17,18
On the other hand, the enzymatic method for GA is widely used in routine clinical settings. In this method, ALB is degraded into its amino acids or peptides using ALB-specific proteinase, and then only glycated amino acids or peptides are oxidized by ketoamine oxidase and generate hydrogen peroxide. The hydrogen peroxide is measured by colorimetric determination to obtain GA concentration (g/dL). ALB concentration (g/dL) is measured separately. Finally, the GA concentration (g/dL) is divided by the ALB concentration (g/dL) to obtain the GA value (%) 13 (Figure 1).
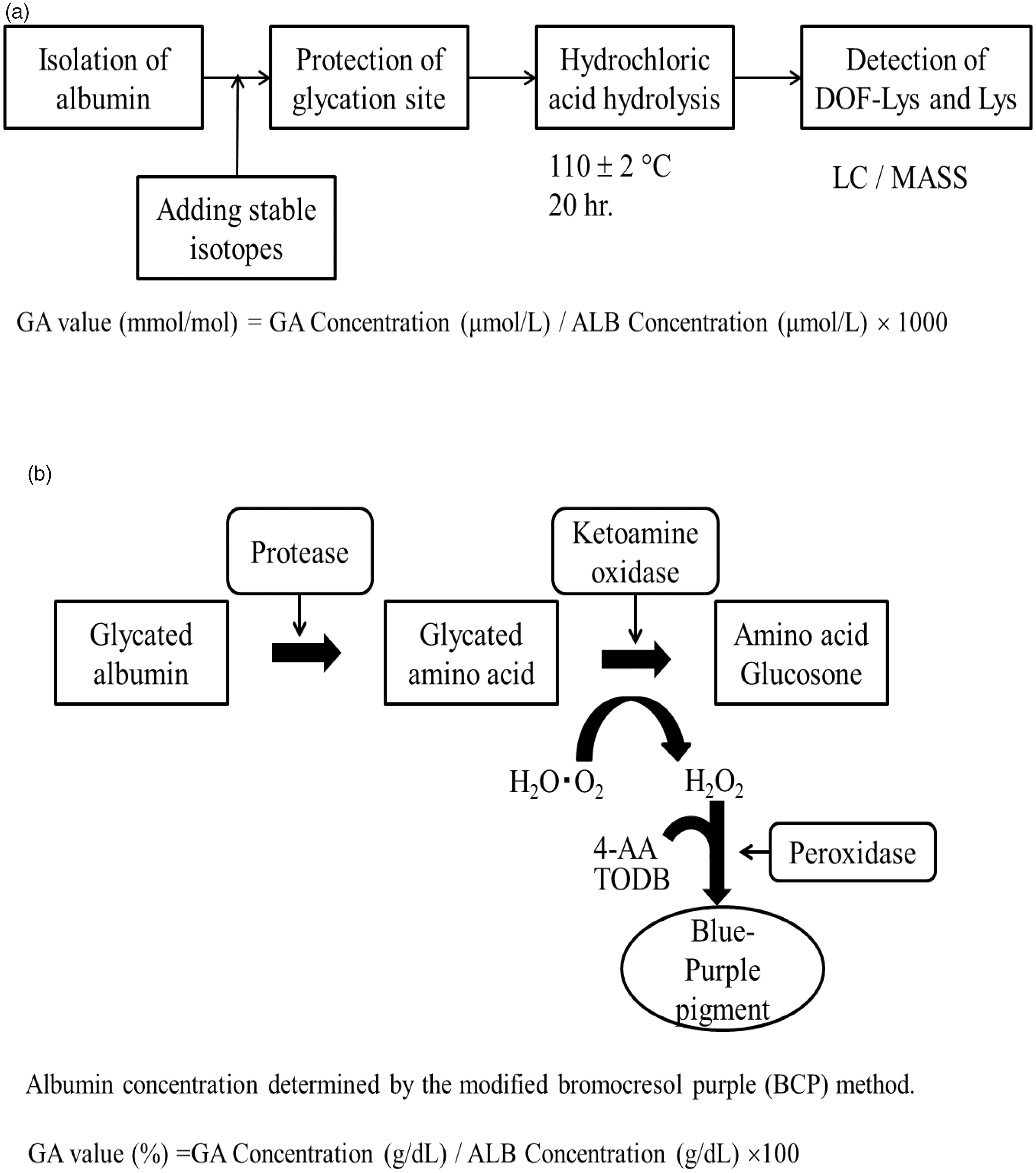
Principle of GA measurement. (a) JSCC-recommended method (ID MS method) and (b) enzymatic method.
In the present study, we evaluated the performance of the standardized enzymatic method for GA and established a unit conversion formula from standardized GA (mmol/mol) to the routinely used GA (%).
Methods
Basic performance of the standardized enzymatic method for GA
The standardized GA value (mmol/mol) was measured by a standardized GA kit (Asahi Kasei Pharma, Japan). The standardized GA kit is composed of a combination of Lucica® GA-L reagent (Asahi Kasei Pharma, Japan) and the standardized GA calibrator (Asahi Kasei Pharma, Japan) that is traceable to JCCRM 611. Reference standard material for GA measurement (JCCRM 611-1M, H, and HH, ReCCS, Japan), plasma protein reference material (ERM-DA470k/IFCC), standardized calibrator for Lucica® GA-L (L and H) and Controls for Lucica® GA-L (L and H, Asahi Kasei Pharma, Japan) were measured for the evaluation of the basic performance of the standardized GA kit on the automatic analyser LABOSPECT008 (HITACHI, Japan). Samples with known GA values were measured 20 times and the results were compared with the target values to evaluate the accuracy of the standardized GA kit. The coefficient of variation (CV%) of the 20 measurements was calculated to evaluate the within-run reproducibility. In addition, samples were measured using Modular P (Roche, Switzerland) and LABOSPECT008 (HITACHI, Japan) to evaluate the difference between analysers.
Study subjects
Patients who visited or were hospitalized at the Tokyo Women’s Medical University Hospital for the measurement of HbA1c were investigated. Subjects with a serum ALB concentration of 3.5–5.1 g/dL and an albumin to globulin (A/G) ratio of 1.00–2.00 were included in the study. The patients were divided into six groups consisting of patients with HbA1c values of (1) < 5%, (2) 5–5.9%, (3) 6–6.9%, (4) 7–7.9%, (5) 8–8.9% and (6) ≥9%, each of which were intended to include 20 patients or more. Patients with nephrosis, cirrhosis, thyroid disease and others who were determined to be unsuitable by the principal investigator were excluded. In addition, blood samples with extreme colours (for example, extreme haemolysis, chyle and hyperbilirubinaemia) were excluded from the test. This study was approved by the ethics committee of Tokyo Women's Medical University Hospital (Reference number: 3415).
Establishment of the unit conversion formula
Two hundred and forty serum specimens were measured using routinely used enzymatic method for GA (Lucica® GA-L kit: %) and the standardized enzymatic method for GA (mmol/mol). The automatic analyser LABOSPECT008 (HITACHI, Japan) was used for the measurement of GA.
Statistics analysis
The regression equation was calculated using a Passing-Bablok regression to establish the unit conversion formula between % and mmol/mol. Calculated GA (%) was obtained from the unit conversion formula, and differential analysis was performed between calculated GA (%) and actual measured GA (%). Statistical analysis was performed using SPSS (ver. 22, IBM, U.S.A.) and Stat Flex (ver. 6, Artec, U.S.A.). The numerical values were described using the mean ± SD, CV% and others.
Results
Basic performance of the standardized enzymatic method for GA
The total accuracy of GA concentration, ALB concentration and GA value of all measured samples was 98.6–101.0%, 95.6–98.1% and 100.8–103.0%, respectively. In particular, the accuracy of measured GA values of the reference standard material JCCRM 611-1 M, H and HH was 102.0%, 101.2% and 100.8%, respectively. The within-run reproducibility CV% of GA concentration, ALB concentration and GA value was 0.43–0.74%, 0.78–1.39% and 0.57–0.88%, respectively. The values obtained from LABOSPECT008 compared with those from Modular P were 98.2–101.2% for GA concentration, 95.4–98.5% for ALB concentration and 100.4–102.9% for GA value, respectively.
Study subjects
A total of 240 serum specimens (HbA1c concentration ≤4.9% [n = 23], 5.0–5.9% [n = 50], 6.0–6.9%, [n = 44], 7.0–7.9% [n = 40], 8.0–8.9% [n = 37] and ≥9.0% [n = 46]) were collected. The mean HbA1c, ALB concentration and A/G ratio were 7.34%, 4.27 g/dL and 1.52, respectively.
Unit conversion formula
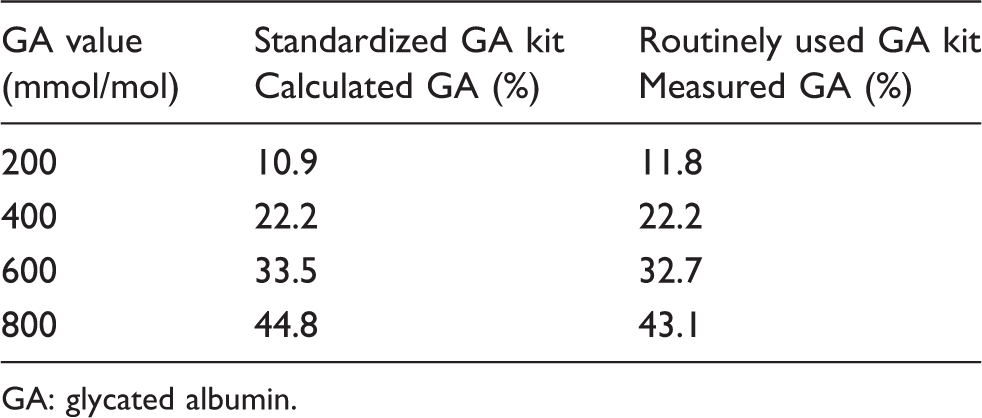
The correlation equation between GA (%) and GA (mmol/mol) was GA (%) = 0.05652 × GA (mmol/mol)–0.4217 (r = 0.999, Figure 2), which produced an excellent linear relationship from 198 to 937 mmol/mol and was set as the unit conversion formula. Table 1 shows the relationship between GA (mmol/mol) and GA (%). GA (%) calculated from GA (mmol/mol) using the unit conversion formula was 96.6–104.2% of the actual measured GA (%) (Figure 3).
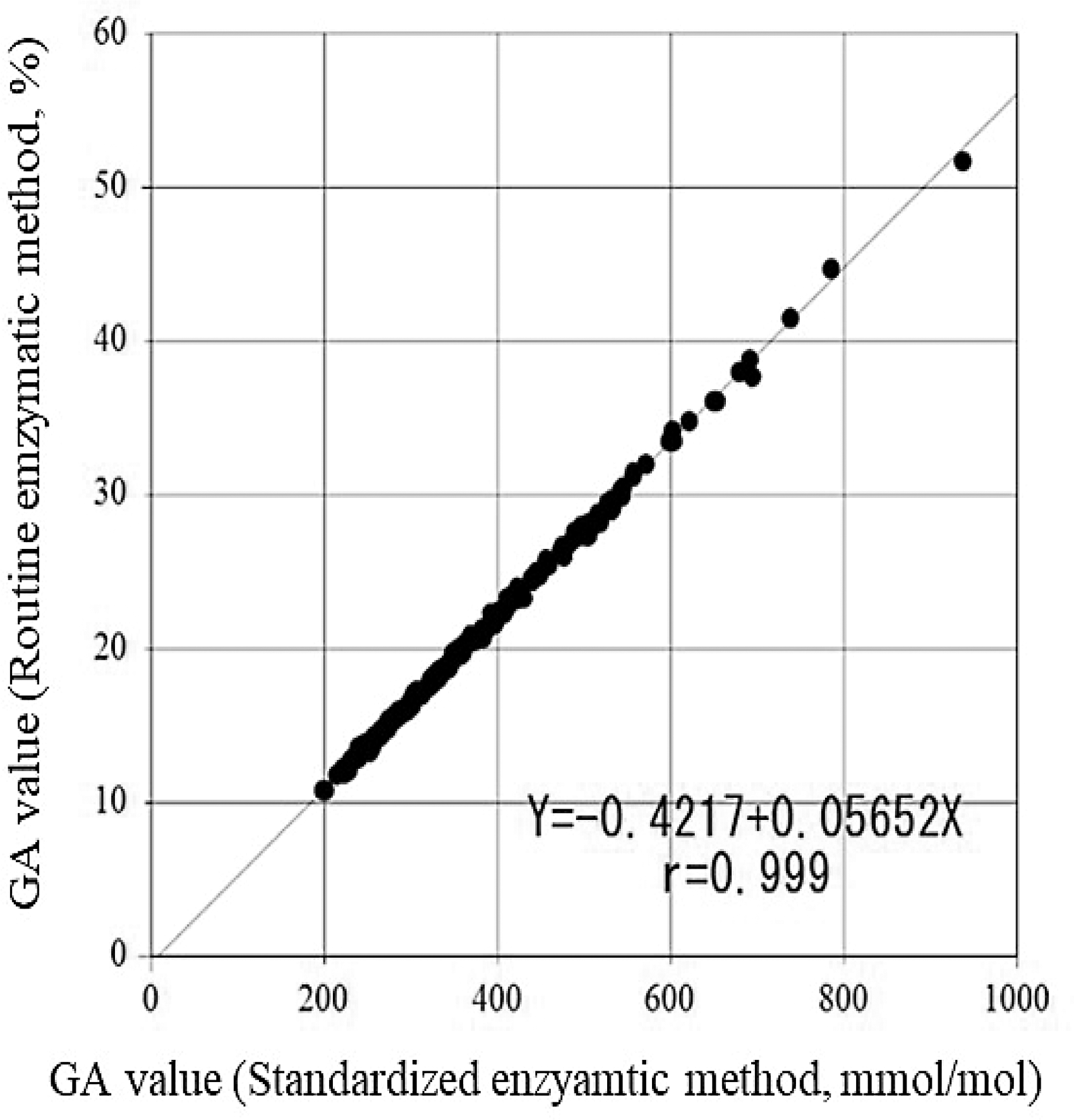
Relationship between GA (mmol/mol) and GA (%).
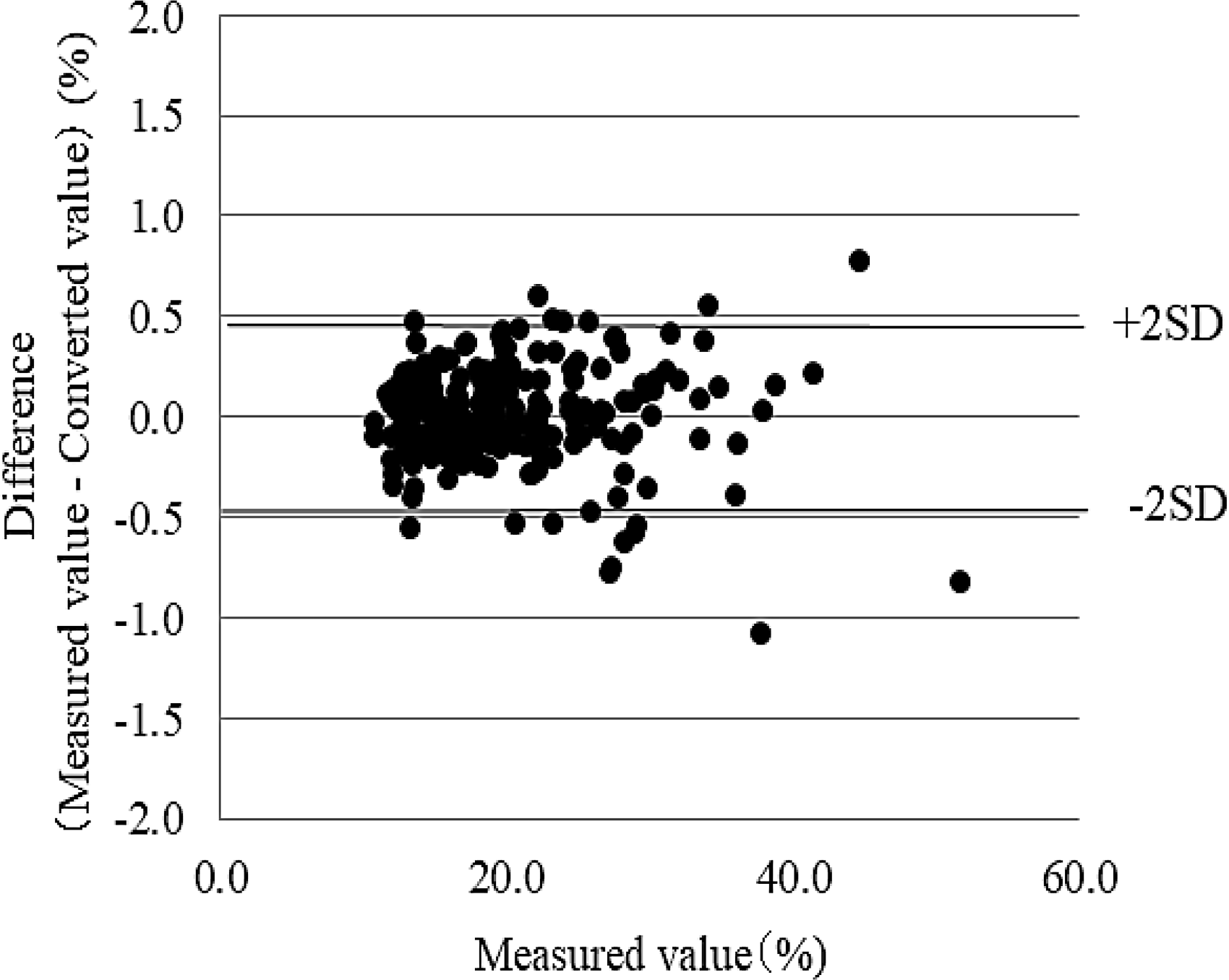
The difference between GA (%) obtained from GA (mmol/mol) using the GA unit exchange formula and GA (%) obtained from the routine method.
Relationship between GA (mmol/mol) and GA (%).
GA: glycated albumin.
Discussion
In the present study, we evaluated the basic performance of the standardized enzymatic method for GA, established the unit conversion formula and obtained the following results:
The basic performance of the standardized enzymatic method for GA was maintained at an acceptable level. Unit conversion formula: GA (%) = 0.05652 × GA (mmol/mol)–0.4217 (r = 0.999) was obtained. Calculated GA (%) using the unit conversion formula was in agreement with the actual measured GA (%).
The standardized enzymatic method for GA was proven to have good basic performance, such as good reproducibility, accuracy and no differences between analysers. The method is believed to be applicable to daily medical practice.
Currently, GA is measured clinically and reported as GA (%). Meanwhile, the GA value recommended by JSCC is reported using the unit of mmol/mol (International System of Units). In order to facilitate the conversion of GA value from % to the standardized value (mmol/mol), it is vital to establish a conversion formula that converts GA (mmol/mol) into GA (%).
To date, in the ‘JSCC Recommended Method for Glycated Albumin Measurement in Serum’ reported by cDMI, 17 42 samples were measured using the JSCC-recommended reference method and the routine enzymatic method (Lucica® GA-L). According to the report published by cDMI, 17 the regression line between the routinely used enzymatic method (%) and the JSCC-recommended reference method (mmol/mol) was GA (%) = 0.0523 × GA (mmol/mol) + 1.306 (r = 0.996), which produced a good linear relationship from 162 to 836 mmol/mol. However, the study was conducted on a relatively small number of samples, and the obtained regression line was developed using the least squares linear regression method, which was inappropriate to be used as a conversion formula. The present study used a relatively large number of samples (n = 240). A new unit conversion formula that can provide useful and robust information for the value conversion was developed using a Passing-Bablok regression. The GA (%) calculated from mmol/mol using the conversion formula fell between 96.6 and 104.2% of the actual measured GA (%) (Table 1). Therefore, the use of the unit conversion formula in the present study has proven applicable for use in daily medical practice.
Although HbA1c is the gold standard measurement for glycaemic control and is well established, it is reported in two different units: the NGSP value (%) and the IFCC value (mmol/mol), which may cause massive confusion in a clinical setting. 6 In the case of GA, GA (%) is prevalent in actual clinical practice, while GA (mmol/mol) is ideal for the standardization of GA measurement and should become the global reference standard. Therefore, the unit conversion formula obtained in the present study is useful for the standardization of GA.
There was a limitation to the present study. It was a single-site study so that the findings of the present study need to be confirmed by multicentre study in the future.
Conclusion
The basic performance of the standardized GA kit was maintained at an acceptable level, and the unit conversion formula GA (%) = 0.05652 × GA (mmol/mol)–0.4217 has been established. Converted values by the formula were in agreement with the actual measurement using the routine method. This formula can be used to convert values for the standardization of GA measurement in the future.
Footnotes
Acknowledgements
The authors appreciate members of Committee on Diabetes Mellitus Indices, Japan Society of Clinical Chemistry for the planning of glycated albumin standardization project.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was partially supported by Asahi Kasei Pharma Corporation.
Ethical approval
This study was approved by the ethics committee of Tokyo Women's Medical University Hospital (Reference number: 3415).
Guarantor
AS.
Contributorship
All authors designed the study, conducted the study, wrote and reviewed the article and contributed to the discussion.
