Abstract
Background
This retrospective longitudinal study was performed to determine whether tafamidis treatment leads to improvements in commonly used blood data for transthyretin familial amyloid polyneuropathy (TTR-FAP).
Methods
Commonly used blood data (complete blood count [including a haemogram], total protein, albumin, blood urea nitrogen, creatinine, aspartate aminotransferase, alanine aminotransferase, alkaline phosphatase, lactate dehydrogenase, γ-glutamyl transpeptidase, total bilirubin [T-Bil], creatine kinase, choline esterase, high-density lipoprotein cholesterol, low-density lipoprotein cholesterol, estimated glomerular filtration rate [eGFR], serum amyloid A protein, TTR, haemoglobin A1c, free triiodothyronine [FT3] and free thyroxine [FT4]) were investigated in 33 TTR-FAP patients. These values included longitudinal data at three time points: six months before or after tafamidis treatment and one year after tafamidis treatment. Longitudinal changes in each blood item were examined using a linear mixed model, adjusting for age at starting tafamidis, sex, TTR-FAP stage and value before tafamidis treatment.
Results
Our results show elevated TTR concentrations after tafamidis treatment. In contrast, haemoglobin, mean corpuscular haemoglobin, mean corpuscular haemoglobin concentration, mean platelet volume, platelet distribution width, T-Bil, eGFR, FT3 and FT4, gradually decreased through a reference range. There were no characteristic observations in any other items. TTR binds to thyroid hormone; therefore, FT3 and FT4 decreased in inverse proportion to increased TTR concentrations.
Conclusion
Unfortunately, progression to anaemia may occur regardless of tafamidis treatment. Because anaemia is sometimes present in TTR-FAP, attention should be paid to longitudinal changes in commonly used blood data, irrespective of tafamidis treatment.
Introduction
Tafamidis has recently been used in familial amyloid polyneuropathy (FAP) as a stabilizer for the circulating tetrameric transthyretin (TTR).1,2 Tafamidis inhibits conformational changes and fibril formation of TTR by preventing dissociation from heterotetrameric TTR into monomeric wild-type and mutated TTR and delaying progression of neurological impairment.1,2
Moreover, besides these neurological symptoms, TTR-FAP manifests additional clinical abnormalities (e.g. bad nutrition or anaemia)3–5 that are reflected by commonly used blood data. However, the utility of tafamidis against these symptoms is not well understood. Therefore, in the current study, we compared blood data changes before tafamidis treatment and one year after tafamidis treatment to determine if tafamidis has more wide-ranging effects in TTR-FAP patients.
Methods
Patients
A retrospective longitudinal study was performed at Kumamoto University Hospital, Japan. Clinical information including blood data was used to examine 33 non-liver transplantation TTR-FAP patients (15 V30M, one V30A, one V30M/K80R, one F33V, one A36D, two G47V, one T49I, two S50I, one T60A, one G83R, two E89K, one I107V and four Y114C). Detailed clinical information on the patients is provided in Table 1. All TTR-FAP patients initiated administration of tafamidis (20 mg/day) by 2015. Written informed consent was obtained from TTR-FAP patients for their participation in this study.
Clinical features of FAP patients.
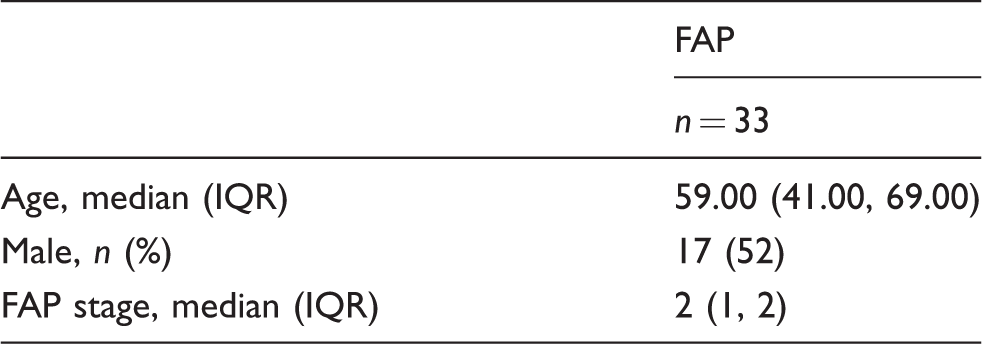
IQR: interquartile range; FAP: familial amyloid polyneuropathy.
Clinical information
The following commonly used blood data were measured by central clinical laboratory in Kumamoto University Hospital: red blood cells (RBC), white blood cells (WBC), neutrophil frequency, lymphocyte frequency, monocyte frequency, eosinophil frequency, basophil frequency, haemoglobin (Hb), haematocrit (HCT), mean corpuscular volume (MCV), mean corpuscular haemoglobin (MCH), mean corpuscular haemoglobin concentration (MCHC), red cell distribution width (RDW), platelets (PLT), plateletcrit (PCT), mean platelet volume (MPV), platelet distribution width (PDW), total protein (TP), albumin (ALB), blood urea nitrogen (BUN), creatinine (Crea), estimated glomerular filtration rate (eGFR), aspartate aminotransferase (AST), alanine aminotransferase (ALT), alkaline phosphatase (ALP), lactate dehydrogenase (LDH), γ-glutamyl transpeptidase (γ-GTP), total bilirubin (T-Bil), creatine kinase (CK), choline esterase (ChE), high-density lipoprotein cholesterol (HDL-C), low-density lipoprotein cholesterol (LDL-C), serum amyloid A protein (SAA), transthyretin (TTR), haemoglobin A1c (HbA1c), free triiodothyronine (FT3) and free thyroxine (FT4). Average blood data values were calculated in each TTR-FAP patient at three periods: six months before tafamidis treatment, six months after tafamidis treatment and one year after tafamidis treatment. All values for each item before and after tafamidis treatment are shown in Table 2.
Longitudinal data for each blood item.
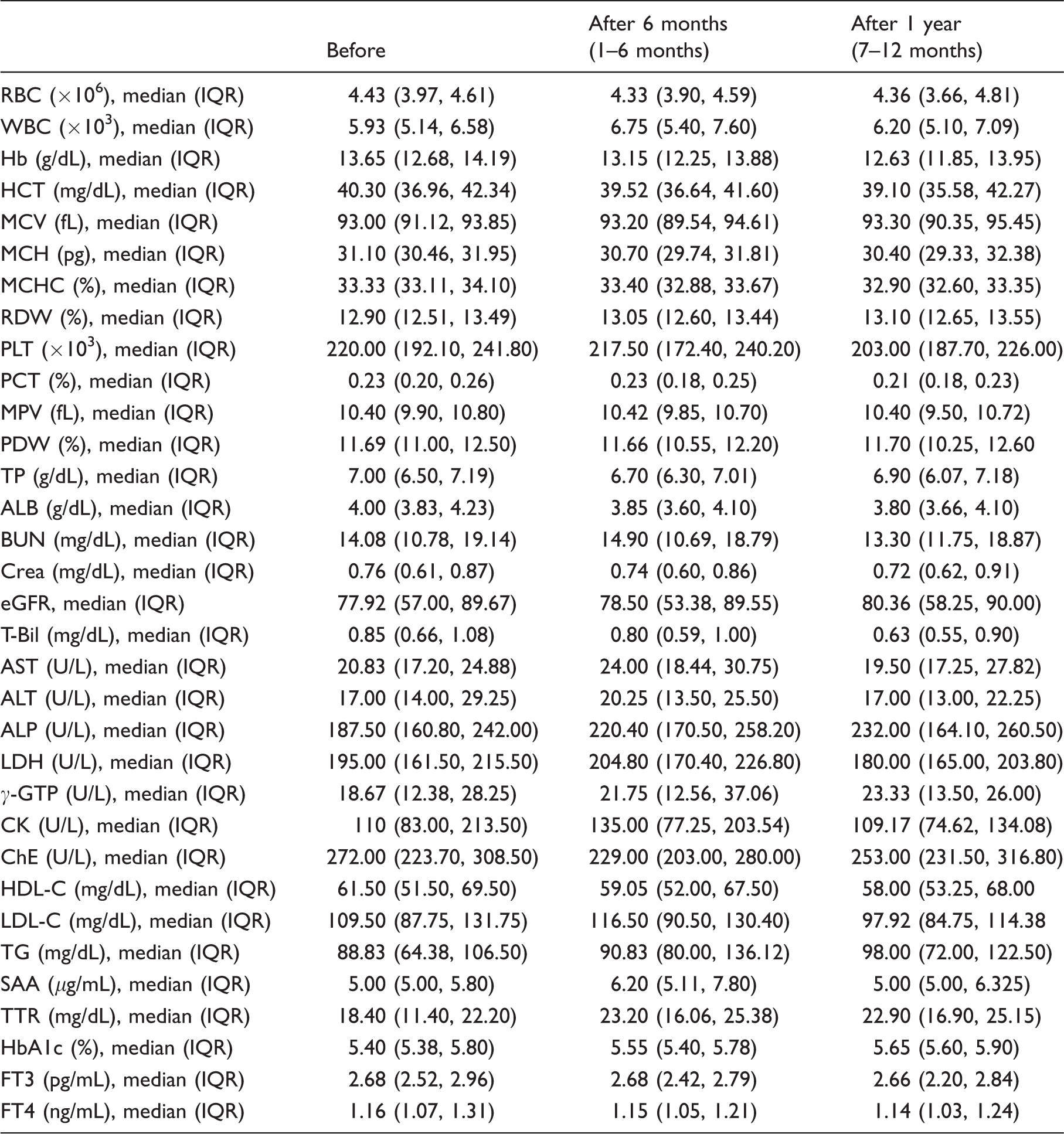
IQR: interquartile range; RBC: red blood cells; WBC: white blood cells; Hb: haemoglobin; HCT: haematocrit; MCV: mean corpuscular volume; MCH: mean corpuscular haemoglobin; MCHC: mean corpuscular haemoglobin concentration; RDW: red cell distribution width; PLT: platelets; PCT: plateletcrit; MPV: mean platelet volume; PDW: platelet distribution width; TP: total protein; ALB: albumin; BUN: blood urea nitrogen; Crea: creatinine; eGFR: estimated glomerular filtration rate; AST: aspartate aminotransferase; ALT: alanine aminotransferase; ALP: alkaline phosphatase; LDH: lactate dehydrogenase; γ-GTP: γ-glutamyl transpeptidase; T-Bil: total bilirubin; CK: creatine kinase; ChE: choline esterase; HDL-C: high-density lipoprotein cholesterol; LDL-C: low-density lipoprotein cholesterol; SAA: serum amyloid A protein; TTR: transthyretin; HbA1c: haemoglobin A1c; FT3: free triiodothyronine; FT4: free thyroxine.
Statistical analysis
FAP is a rare neurodegenerative disease. Therefore, sample size was determined with consideration to the number of outpatients or inpatients to Kumamoto University Hospital during the survey period. To examine normal distribution of variables, Shapiro–Wilk’s test was performed. A linear mixed model with a variance-component covariate covariance structure was used to examine the effect of tafamidis on the time course of commonly used blood data at six months or one year after tafamidis treatment compared with baseline blood data at six months before tafamidis treatment. Age at starting tafamidis, sex and FAP stage were included in the model as covariates. Analyses were performed using R version 3.3.1 (The R Foundation for Statistical Computing, Vienna, Austria) or SAS Version 9.4 (SAS Institute Inc., Cary, NC, USA). A value of P < 0.05 was considered statistically significant.
Results
Except for TTR, blood data values were within the reference range at all phases (Table 2). Next, we checked longitudinal changes in blood data (Table 3). TTR concentrations after tafamidis treatment were elevated compared with baseline concentrations (i.e. 6 months before tafamidis treatment). In contrast, compared with baseline, Hb, MCH, MCHC, MPV, PDW, T-Bil, eGFR, FT3 and FT4 gradually decreased from linear mixed model estimated values (Table 3). There were no characteristic observations in any other items, namely frequency of neutrophils, lymphocytes, monocytes, eosinophils, and basophils, AST, ALT, ALP, LDH, γ-GTP and ChE.
Results of the linear mixed model.
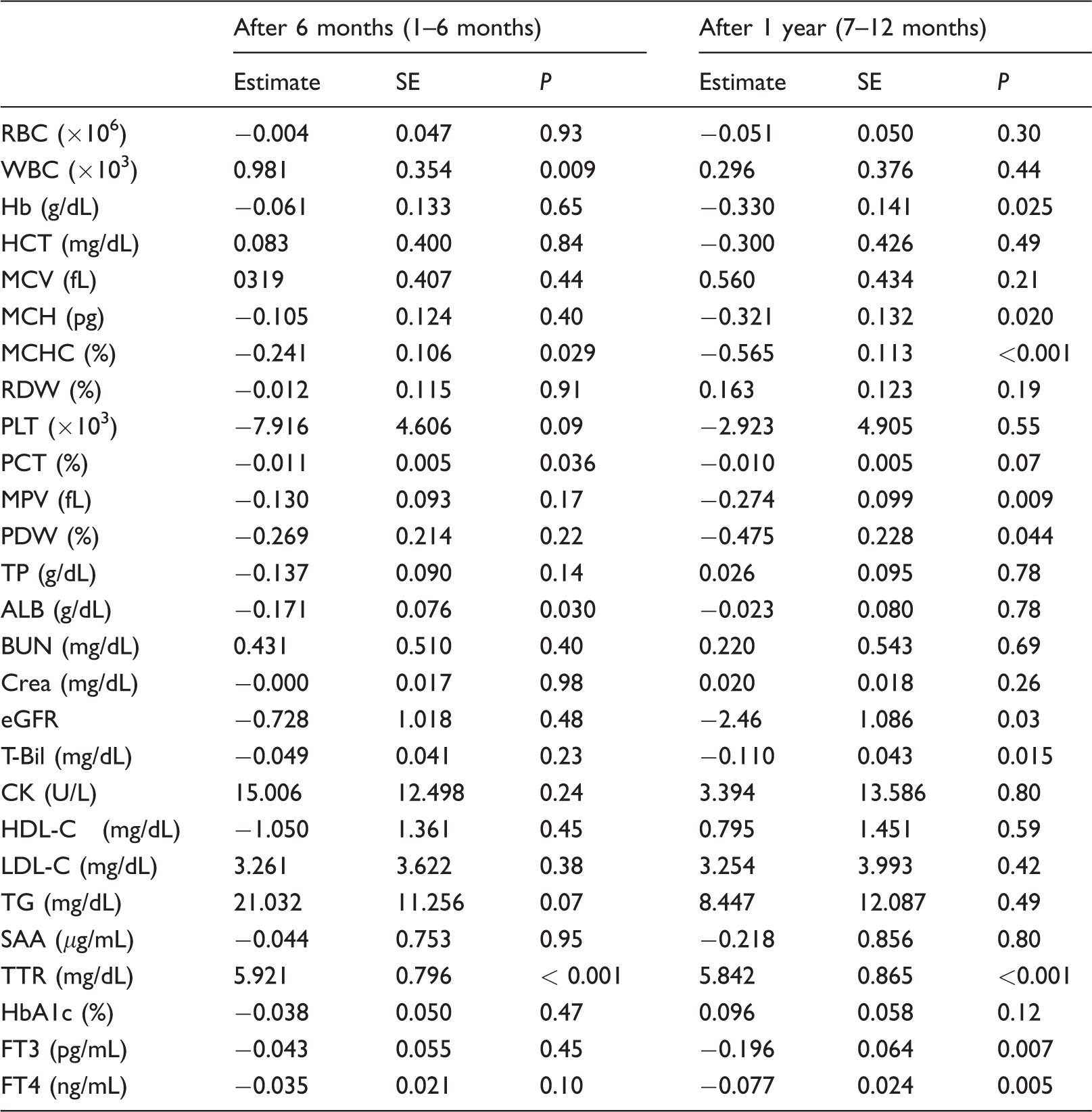
Note: Adjusted factors were ‘age at starting tafamidis’, ‘sex’, ‘FAP stage’ and ‘each item value before tafamidis treatment’.
SE: standard error; RBC: red blood cells; WBC: white blood cells; Hb: haemoglobin; HCT: haematocrit; MCV: mean corpuscular volume; MCH: mean corpuscular haemoglobin; MCHC: mean corpuscular haemoglobin concentration; RDW: red cell distribution width; PLT: platelets; PCT: plateletcrit; MPV: mean platelet volume; PDW: platelet distribution width; TP: total protein; ALB: albumin; BUN: blood urea nitrogen; Crea: creatinine; eGFR: estimated glomerular filtration rate; T-Bil: total bilirubin; CK: choline esterase; HDL-C: high-density lipoprotein cholesterol; LDL-C: low-density lipoprotein cholesterol; SAA: serum amyloid A protein; TTR: transthyretin; HbA1c: haemoglobin A1c; FT3: free triiodothyronine; FT4: free thyroxine.
Discussion
Concentration of TTR was increased by tafamidis, as described previously.1,2 In theory, serum TTR forms heterotetramers. 6 Consequently, increased serum TTR concentration in our data may reflect increased heterotetrameric TTR. Indeed, this is likely because tafamidis prevents dissociation from heterotetrameric TTR. In addition, heterotetrameric TTR reportedly binds and transports thyroid hormone.6,7 Therefore, it is possible that heterotetrameric TTR-bound thyroid hormone increases, while unbound thyroid hormone, FT3 and FT4 concentration paradoxically decrease.
Anaemia is sometimes present in TTR-FAP, with its frequency related to disease duration.4,5 In our data, Hb, MCH and MCHC values remained within the reference range, which may reflect the mild case of our examined TTR-FAP patients. In contrast, linear mixed model estimated values of these anaemia-related parameters gradually decreased from each time point compared with six months before tafamidis treatment. In this regard, because most TTR-FAP patients were promptly administrated tafamidis after the initial visit, almost none had blood data from more than six months before tafamidis treatment. Consequently, we could not evaluate the degree of time-dependent changes in anaemia-related parameters during the period without tafamidis therapy within the same individual. Therefore, although it is possible that tafamidis counteracts progression of anaemia-related parameters, progression to anaemia may continue in some measures, regardless of tafamidis treatment.
Anaemia in TTR-FAP is also reported after liver transplantation and is responsible for bone marrow dysfunction and lower serum erythropoietin (EPO), which is produced in the kidney.4,5,8 In our other data, although the cause of reduced T-Bil is unknown, the decrease in not only Hb, MCH, and MCHC but also MPV and PDW suggest a bone marrow abnormality, while that of eGFR supports renal function degradation.9,10 Recently, we reported elevated interleukin-6 (IL-6) concentration in TTR-FAP carriers and patients. 11 Additionally, IL-6 interrupts production of EPO and erythroid progenitor cells in bone marrow.12,13 Therefore, IL-6 may be involved in our results. Moreover, because the effectiveness of recombinant human EPO was previously reported in TTR-FAP patients with anaemia, 4 complex treatments aside from tafamidis may also be required depending on the patient’s situation.
Our study has limitations with respect to the number of TTR-FAP patients. In addition, our TTR-FAP patients exhibited a variety of phenotypes. Thus, whether gradual decrease of Hb, MCH and MCHC is a common phenomenon for all phenotypes is not known. Additionally, evaluation of longitudinal data in our study was only for one year, and longer term follow-up is needed. Nonetheless, greater focus should be given to longitudinal changes of not only neurological symptoms but also typical blood data, irrespective of tafamidis treatment.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
All investigations using patient information were conducted in accordance with the Declaration of Helsinki and under the approval of the Institutional Review Board of Kumamoto University (permit number: 1195).
Guarantor
TI.
Contributorship
TI and MU contributed equally to this article. TI conceived and designed the study, performed the statistical analysis. TI and MU wrote the first draft of the manuscript. TI, TM, MU, TA, YM, SS and YA contributed to data interpretation and reviewed and edited the manuscript.
