Abstract
Background
Familial amyloid polyneuropathy is caused by a variant transthyretin, which is a serum protein secreted by the liver. We previously reported that mutated transthyretins were detected in serum samples by surface-enhanced laser desorption/ionization time-of-flight mass spectrometry (SELDI-TOF MS). The aim of this study was to evaluate the clinical usefulness of SELDI-TOF MS for diagnosis of transthyretin-related amyloidosis.
Methods
We used 106 serum samples obtained from patients who were clinically suspected of having amyloidosis between February 2011 and April 2014. SELDI-TOF MS allowed analysis for transthyretin via a 3-h one-step procedure.
Results
Of the 106 patients, 51 are transthyretin amyloidosis. Mutated transthyretins were detected in serum samples from 30 of 51 patients with transthyretin amyloidosis. The results of genetic analysis showed that all of those patients had mutations in the transthyretin gene. For all 18 patients with senile systemic amyloidosis of 51 patients with transthyretin amyloidosis, SELDI-TOF MS detected only wild-type transthyretin peaks, not mutated transthyretin peaks.
Conclusion
SELDI-TOF MS is a clinically useful tool for diagnosis of transthyretin-related amyloidosis.
Keywords
Introduction
Transthyretin (TTR)-related familial amyloid polyneuropathy (FAP) is an inherited disorder characterized by amyloid deposits derived from mutated TTR. 1 To date, about 130 different mutations in the TTR gene have been reported. 2 On the other hand, senile systemic amyloidosis (SSA) is a non-hereditary disorder caused by amyloid fibrils derived from wild-type (WT) TTR. 3 Because the molecular mass of WT TTR differs from the mass of mutated TTRs, which have mutations causing amino acid exchanges, mass spectrometry (MS) has been reported to be a powerful tool for detecting mutated TTRs in serum samples. However, in those previous methods, it is essential to purify TTR from serum by immunoprecipitation before MS analysis. To detect mutated TTRs more rapidly and easily, it is needed to develop novel techniques.
Surface-enhanced laser desorption/ionization time-of-flight MS (SELDI-TOF MS) provides rapid protein expression profiles from various biological samples including serum, cerebrospinal fluid and saliva. 4 We previously reported a convenient SELDI-TOF MS method that does not require an immunoprecipitation step to detect several types of variant TTR. 5 In this study, we evaluated the clinical usefulness of SELDI-TOF MS for diagnosis of TTR-related amyloidosis.
Materials and methods
Between February 2011 and April 2014, 106 patients with clinically suspected amyloidosis were examined after having given informed consent.
We used SELDI-TOF MS to measure variant TTRs in serum samples as previously described. 5 In brief, 2.2 μL samples were diluted 50-fold with 50 mM phosphate buffer, pH 7.0. Aliquots of 100 μL of the diluted samples were applied to a ProteinChip array (Bio-Rad Laboratories, Hercules, CA, USA), which is a strong anion exchange array (Q10), and were incubated on a shaker for 90 min at room temperature. For reduction of proteins, samples were applied to chips with 5 μL of 50 mM dithiothreitol solution in 50 mM NH4HCO3, and then samples were air dried at 70℃. Sinapinic acid (1 μL; Bio-Rad Laboratories) was applied twice to the ProteinChips. The samples were analysed with the PCS 4000 SELDI-TOF MS instrument (Bio-Rad Laboratories).
We also performed genetic testing of the TTR gene using 3130 Genetic Analyzer (Life Technologies, Carlsbad, CA, USA), as previously described. 4 The statistical analysis was performed with JMP version 8 (SAS Institute Inc, Cary, NC). A P-value of <0.05 was regarded as statistically significant.
Results and discussion
Of the 106 patients, 51 patients are TTR amyloidosis and 55 patients are non-TTR amyloidosis. In these patients with TTR amyldoidosis, mutated TTRs were detected within 3 h in serum samples from 30 patients (Figure 1(a) and (b)). Genetic analysis revealed that all patients had TTR mutations including Val30Met, Val30Ala, Ala45Asp, Gly47Arg, Ser50Arg, Ser50Ile, Leu55Pro, Thr59Arg and Ser77Tyr. In this system, the specificity and sensitivity are 100% and 90.9%, respectively. We also measured the mass shifts of variant TTRs by SELDI-TOF MS and compared them with theoretical mass changes. Our analysis revealed a highly significant correlation between measured and theoretical mass shifts from WT TTR in serum samples from patients with FAP (P < 0.0001; r2 = 0.9993; Figure 1(c)). These data indicate that SELDI-TOF MS may be useful for rapid detection of mutated TTRs in serum from most patients with FAP. To avoid misdiagnosis of FAP, we need a convenient, accurate system for diagnosis and for double-checking results, which SELDI-TOF MS provides.
Detection of mutated TTRs by using SELDI-TOF MS. (a) Schematic overview of TTRs obtained by using SELDI-TOF MS. (b) Mass peaks of TTRs in serum samples obtained by SELDI-TOF MS. Arrows indicate mutated TTR peaks. (c) Correlation between measured and theoretical mass shifts (P < 0.0001; r2 = 0.9993).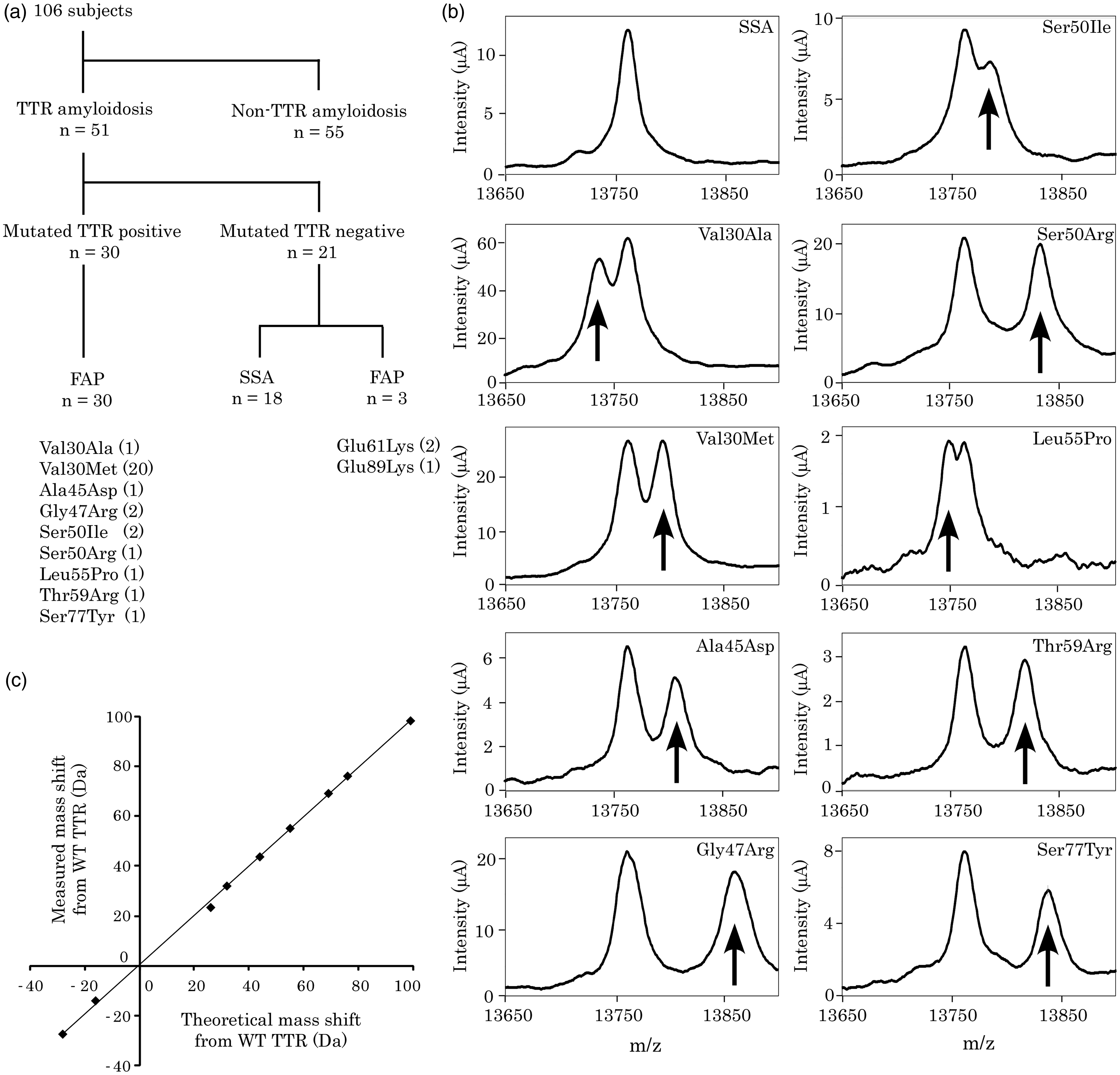
We detected only WT TTR in 76 of 106 (71.7%) patients: 3 of 33 (9%) with FAP, 18 of 18 (100%) with SSA and 55 of 55 (100%) with non-TTR amyloidosis. SELDI-TOF MS could not detect mutated TTRs from patients with Glu61Lys and Glu89Lys mutations. The theoretical mass shift difference between Glu61Lys and Glu89Lys TTRs and WT TTR was 0.94 m/z, which was insufficient to separate those mutated TTRs from WT TTR by SELDI-TOF MS. The lower limit of separation minimum mass difference between two molecules could be separated in this system is thought to be about 15 Da, based on the present and previous studies. 5 We were able to separate Leu55Pro and Ala25Ser mutants, which theoretically have a mass shift of 16 Da from WT TTR but were not able to separate Lys35Asn and Ile107Val mutants, which theoretically have a mass shift of 14 Da from WT TTR. 5 SELDI-TOF MS could not separate the atypical TTR variants, whereas succeeded in detecting most variant TTRs, including the common Val30Met TTR. For reliable clinical diagnosis of TTR-related amyloidosis, histopathological and genetic tests are also required. We believe that this MS system can be used in genetic testing for screening of FAP and double-checking TTR variants.
Conclusions
SELDI-TOF MS is a useful tool to detect variant and WT TTRs and should therefore be a sufficient tool to double check the diagnosis of FAP.
Footnotes
Acknowledgements
The authors are indebted to Ms Judith B Gandy for providing professional English editing of the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported in part by grants from the Japan Society for the Promotion of Science KAKENHI grant numbers 26860372, 25870541 and 24249036; Japanese Society of Laboratory Medicine Fund for the Promotion of Scientific Research; and the Amyloidosis Research Committee, the Pathogenesis, Therapy of Hereditary Neuropathy Research Committee, the Surveys and Research on Specific Disease, the Ministry of Health and Welfare of Japan.
Ethical approval
The Human Ethics Review Committee of Kumamoto University approved the study (Reference number: 903).
Guarantor
MU.
Contributorship
MT, MU and YA were involved in study design and getting ethical approval. KO, HM, YK, GS, AY, RT, YM, TM and TY enrolled patients and collected samples. MT, HM and YK analysed all samples and performed the data analysis. MT and MU wrote the first draft of the manuscript. All authors reviewed, edited and approved the final draft of the manuscript.
