Abstract
Background
Oral prochlorperazine, a dopamine D2 receptor antagonist, is largely metabolized to sulphoxide, 7-hydroxylate and N-desmethylate by cytochrome P450s (CYPs). This study evaluated the influence of CYP genotype on the plasma dispositions of prochlorperazine and its metabolites and their relationships with antiemetic efficacy and prolactin elevation in cancer patients.
Methods
Forty-eight cancer patients treated with oral prochlorperazine were enrolled. Plasma prochlorperazine and its metabolites concentrations and serum prolactin concentration were determined at 12 h after the evening dosing. The genotypes of CYP2C19, CYP2D6 and CYP3A5 and the incidences of nausea and vomiting were investigated.
Results
The plasma concentrations of the prochlorperazine metabolites were weakly correlated with that of the parent drug. The CYP genotypes did not affect the plasma concentrations of prochlorperazine and its metabolites. The plasma concentrations of prochlorperazine and its metabolites were not associated with the incidences of nausea and vomiting. The incidence of vomiting was significantly higher in females than in males. The serum prolactin concentration was weakly correlated with the plasma concentrations of prochlorperazine and its metabolites. The plasma concentrations of prochlorperazine metabolites rather than the parent drug had a weaker relation to serum prolactin concentration.
Conclusions
The CYP genotypes did not affect the plasma dispositions of prochlorperazine and its metabolites. The prochlorperazine metabolites did not have a strong effect on antiemetic efficacy, while they were slightly associated with prolactin secretion in cancer patients.
Introduction
Prochlorperazine (PCZ) is a dopamine D2 receptor antagonist belonging to the phenothiazine group. PCZ is commonly used for the treatment of nausea and vomiting in clinical settings. 1 The pharmacokinetics and pharmacodynamics of PCZ showed a large variation in cancer patients. 2 PCZ is extensively metabolized in the intestine and liver by cytochrome P450 (CYP) and has low oral bioavailability due to a large first-pass effect. 3
Recent studies have demonstrated that CYP2C19, CYP2D6 and CYP3A4/5 are the major CYP isozymes involved in the metabolization of PCZ. 4 The major metabolites in humans are identified as prochlorperazine sulphoxide (PCZSO), 7-hydroxy prochlorperazine (PCZOH), and N-desmethy prochlorperazine (NDPCZ).3,4 Our previous study demonstrated there were interindividual variations in the predose plasma concentrations of PCZ metabolites in cancer patients treated with oral PCZ. 5 The plasma concentration of PCZSO was higher than that of the other metabolites. 3 The plasma concentration–time profile of PCZ was similar to that of NDPCZ. 6 However, the pharmacokinetic profiles of PCZ metabolites still need to be clarified in cancer patients.
There are several genetic variants of CYP2C19, CYP2D6 and CYP3A4/5, the main metabolizing enzymes of PCZ. CYP2C19*2 showed a splicing defect in exon 5, resulting in early termination of protein synthesis, and CYP2C19*3 is a premature stop codon single nucleotide polymorphism, resulting in a truncated protein. The CYP2C19 genotype is correlated with the plasma disposition of omeprazole and the cure rates for Helicobacter pylori infection and peptic ulcer.7,8 CYP2D6*5 causes the complete loss of the CYP2D6 gene and CYP2D6*10 results in a substantial reduction in CYP2D6 activity.9,10 CYP3A5*3 was found to affect the dispositions of tacrolimus and fentanyl as a result of the almost complete absence of CYP3A5 protein by splice site mutation.11–13 The CYP3A5*3 allele frequencies are 77% in Japanese and 91% to 95% in whites. Although PCZ is markedly metabolized by CYPs, the contribution of these gene variants to the plasma disposition of PCZ and its metabolites have not been fully evaluated in clinical settings.
The clinical responses to PCZ vary in patients receiving opioid analgesics. Our previous report demonstrated that DRD2 Taq IA polymorphism and female gender affected the antiemetic effect of PCZ. 5 PCZ increases the serum prolactin concentration by inhibiting dopamine D2 receptors.14,15 Hyperprolactinaemia is an adverse effect of dopamine D2 receptor antagonist. It causes some issues such as leakage of milk and sexual dysfunction and influences quality of life. The contribution of PCZ concentration to serum prolactin elevation has been partially clarified.5,6,16 In addition, the plasma PCZ concentration, OPRM1 A118G polymorphism and female gender affected serum prolactin concentrations. 5 Opioids also cause the blockade of dopaminergic pathways through the binding to OPRM1 on the hypothalamus and elevate the serum prolactin in humans.17,18 However, the relationships between the PCZ metabolites and antiemetic efficacy and prolactin secretion have not been fully evaluated in patients receiving opioid analgesics.
The aim of this study was to evaluate the influence of CYP genotype on the plasma dispositions of PCZ and its metabolites and their relationships with the antiemetic efficacy and prolactin elevation in cancer patients.
Materials and methods
Ethics
The study was performed in accordance with the Declaration of Helsinki and its amendments, and the protocol was approved by the Ethics Committee of Hamamatsu University School of Medicine. The patients received information about the scientific aim of the study and each patient provided written informed consent.
Patients and blood sampling
This observational study enrolled 48 cancer inpatients at Hamamatsu University Hospital. They received 5 mg oral PCZ (Novamin® tablet, Shionogi & Co., Ltd, Osaka, Japan) three times daily for the prophylaxis of oxycodone-induced nausea and vomiting. Exclusion criteria were as follows: Patients who (1) were being co-treated with another emetic drug or antiemetic drug; (2) did not undergo chemotherapy, radiotherapy and surgery in observation period; (3) were being co-treated with a strong inducer or inhibitor of CYPs, including triazole antifungal agents, macrolide antibiotics, rifampicin and carbamazepine; 19 (4) had a serum creatinine concentration >2 mg/dL or total bilirubin concentration >2 mg/dL; (5) were being co-treated with drugs acting on prolactin secretion; (6) had a disease-influencing prolactin secretion and (7) with poor medication adherence based on pharmacist interview and medical records. Blood sampling was performed on day 6 or later after starting the PCZ treatment. Blood specimens were collected in tubes containing EDTA dipotassium salts at 12 h after the evening dosing.
Determination of plasma PCZ and its metabolites
PCZ maleate and amitriptyline hydrochloride as an internal standard were purchased from Sigma Aldrich (St. Louis, MO, USA). PCZSO and PCZOH were obtained from BDG Synthesis (Wellington, New Zealand). NDPCZ dimaleate was purchased from Toronto Research Chemicals (Toronto, Ontario, Canada). Plasma was separated by centrifugation of the EDTA-treated blood samples at 1670 × g at 4℃ for 10 min. Plasma concentrations of PCZ and its metabolites were determined by LC-MS/MS as previously described. 2 The calibration curves in human plasma were linear over the concentration ranges of 0.01–40 ng/mL for PCZ (r > 0.999), NDPCZ (r > 0.999), PCZOH (r > 0.999) and 0.05–80 ng/mL for PCZSO (r > 0.999). Their intra- and inter-assay precisions and accuracies were within 5.8% and 100–104% and within 7.8% and 99%, respectively. The lower limits of quantification in human plasma were 10 pg/mL for PCZ, PCZOH and NDPCZ, and 50 pg/mL for PCZSO.
CYP genotyping
Genomic DNA was extracted from the peripheral whole blood of each patient using a DNA Extractor WB Kit (Wako Pure Chemicals, Osaka, Japan). CYP2C19*2, CYP2C19*3, CYP2D6*10 and CYP3A5*3 were determined using polymerase chain reaction (PCR) restriction fragment length polymorphism (RFLP) procedures. The CYP2D6*5 allele was identified using a long-PCR procedure. Detection of CYP2C19*2, CYP2C19*3, CYP2D6*5, CYP2D6*10 and CYP3A5*3 alleles was performed as described previously, with some modifications.20–23 With respect to CYP2C19, the enrolled patients were grouped into two genotype groups based on the genetic variants (*1, *2 and *3): *1 carrier (*1/*1, *1/*2 and *1/*3) and *1 non-carrier (*2/*2, *2/*3 and *3/*3). With respect to CYP2D6, the enrolled patients were grouped into three genotype groups based on the genetic variants (*1, *5 and *10): *1 carrier (*1/*1, *1/*5 and *1/*10), *1 non-carrier (*5/*10 and *10/*10), and *5/*5. With respect to CYP3A5, the enrolled patients were grouped into two genotype groups based on the genetic variants (*1 and *3): *1 carrier (*1/*1 and *1/*3) and *3/*3.
Evaluation of antiemetic efficacy
The antiemetic efficacy of PCZ for opioid-induced nausea and vomiting was monitored for two weeks after starting the oral PCZ treatment. The antiemetic efficacy was evaluated by the incidences of nausea and vomiting. The incidences and severities of nausea and vomiting in each patient were assessed using their medical records and the grading system of the Common Terminology Criteria for Adverse Events (CTCAE v4.0).
Determination of serum prolactin
Serum prolactin concentration was measured using chemiluminescent immunoassay according to the manufacturer’s protocol (Architect® Prolactin, Abbott Japan, Tokyo) using an automated immunoassay analyser (Architect® i1000 SR, Abbott Japan). The intra- and inter-assay precisions and accuracies were within 1.8% and 99–100% and within 2.6% and 97–100%, respectively. The serum prolactin concentration was calculated from the calibration information stored in the instrument memory. The lower limit of quantification in human serum was 0.6 ng/mL.
Statistical analysis
All statistical analyses were performed using SPSS (21.0 J, IBM, Tokyo). The plasma concentrations of PCZ and its metabolites were evaluated as absolute values and the evening dose and body weight-normalized values. PCZ metabolism was estimated by the predose plasma concentration ratio of the metabolites to PCZ as the metabolic ratio. The correlations between the plasma concentrations of PCZ and its metabolites were tested by Spearman’s rank correlation coefficient analysis. The influence of gender and each genotype groups of CYP2C19, CYP2D6 and CYP3A5 on the plasma concentrations of PCZ and its metabolites was tested using the Mann-Whitney U test, as were the influences of the plasma concentrations of PCZ and its metabolites on the incidences of nausea and vomiting. Multiple regression analysis was performed to evaluate the influences of each genotype groups of CYP2C19, CYP2D6 and CYP3A5, age, serum albumin, aspartate aminotransferase, alanine aminotransferase and total bilirubin on the plasma concentrations of PCZ and its metabolites. The influences of gender on the incidences of nausea and vomiting were tested using Fisher's exact test. Stepwise multiple logistic regression analysis was performed to evaluate the influences of the plasma concentrations of PCZ and its metabolites and gender on the incidences of nausea and vomiting. The changes in serum prolactin concentration before and after PCZ administration were tested by the Wilcoxon signed rank test. The correlations between the plasma concentrations of PCZ and its metabolites and serum prolactin concentration were tested by Spearman’s rank correlation coefficient analysis. The influences of gender on the serum prolactin concentration were tested using the Mann-Whitney U test. Multiple regression analysis was performed to evaluate the influences of the plasma concentrations of PCZ and its metabolites and gender on the serum prolactin concentration. All values are expressed as the median and interquartile range (IQR) unless otherwise stated. A P < 0.05 was considered to indicate statistical significance.
Results
Study populations
Demographics of patients.
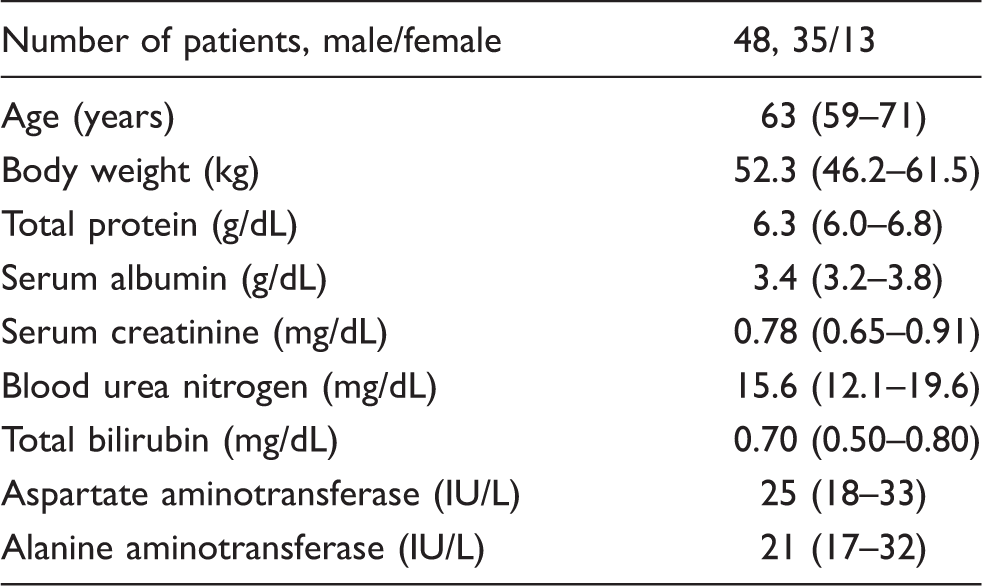
Note: Data are expressed as median with interquartile range in parentheses.
Genetic variants and allele frequencies in this study population.
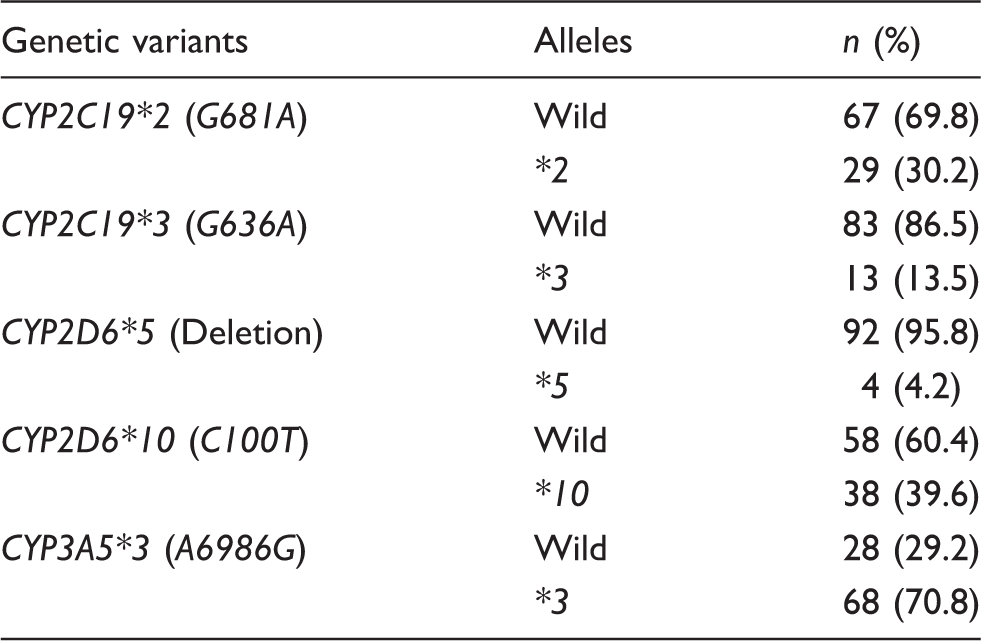
Plasma concentrations of PCZ and its metabolites
The median plasma concentrations of PCZ, PCZSO, PCZOH and NDPCZ at 12 h after the evening dosing were 2.11 ng/mL (IQR: 0.61–4.66 ng/mL), 2.86 ng/mL (1.26–5.34 ng/mL), 0.45 ng/mL (0.32–1.16 ng/mL) and 1.80 ng/mL (0.90–3.23 ng/mL), respectively. Weak correlations were observed between the plasma concentrations of PCZ and PCZSO (ρ = 0.766, P < 0.001), PCZOH (ρ = 0.708, P < 0.001) and NDPCZ (ρ = 0.762, P < 0.001) (Figure 1).
The relationships between the plasma concentrations of prochlorperazine (PCZ) and of prochlorperazine sulphoxide (PCZSO) (a), or 7-hydroxy prochlorperazine (PCZOH) (b), or N-desmethyl prochlorperazine (NDPCZ) (c) in cancer patients.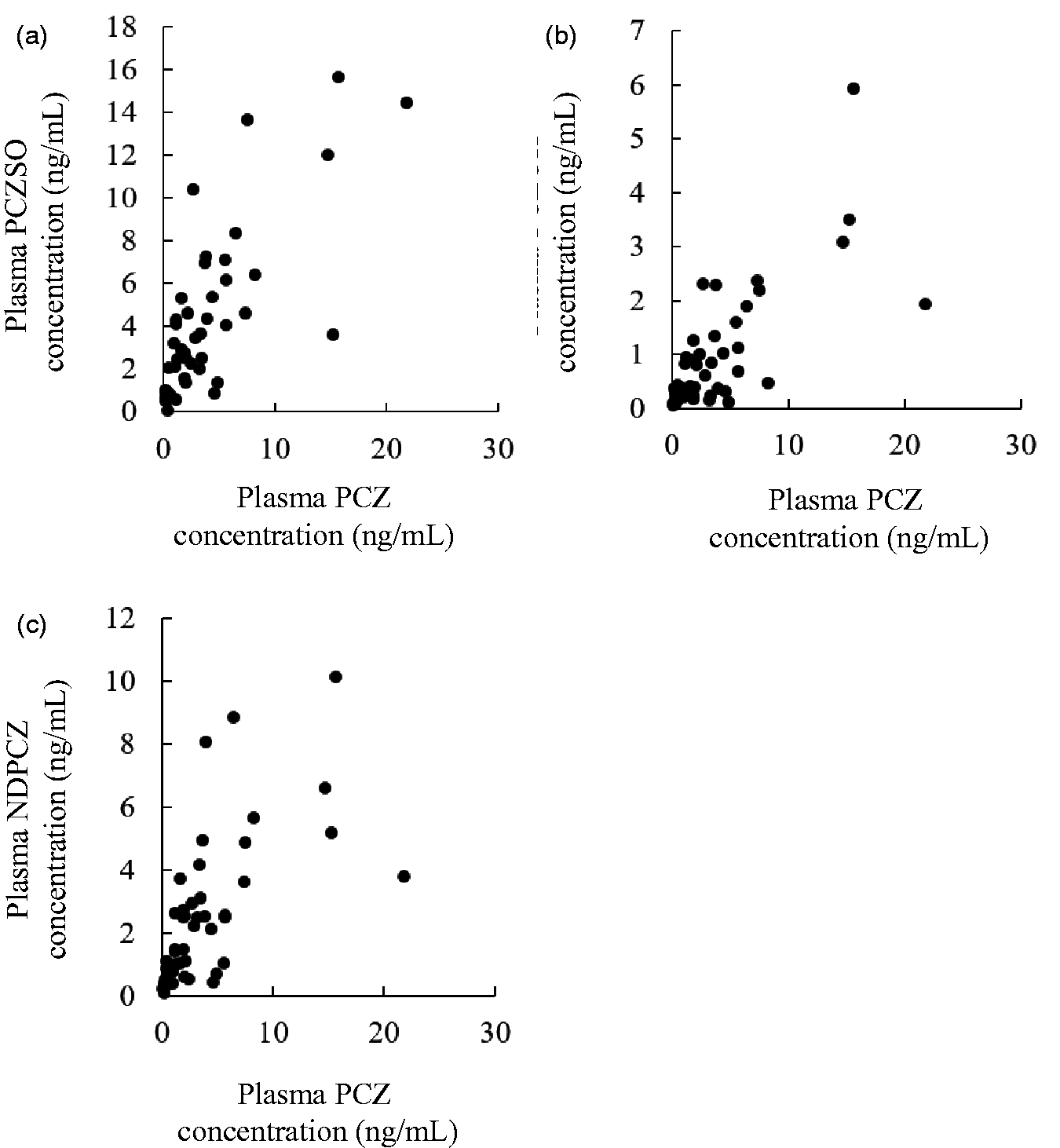
Influences of CYP genotype
Influences of CYP2C19 (a), CYP2D6 (b), and CYP3A5 (c) genotype on plasma concentrations of PCZ and its metabolites in cancer patients.
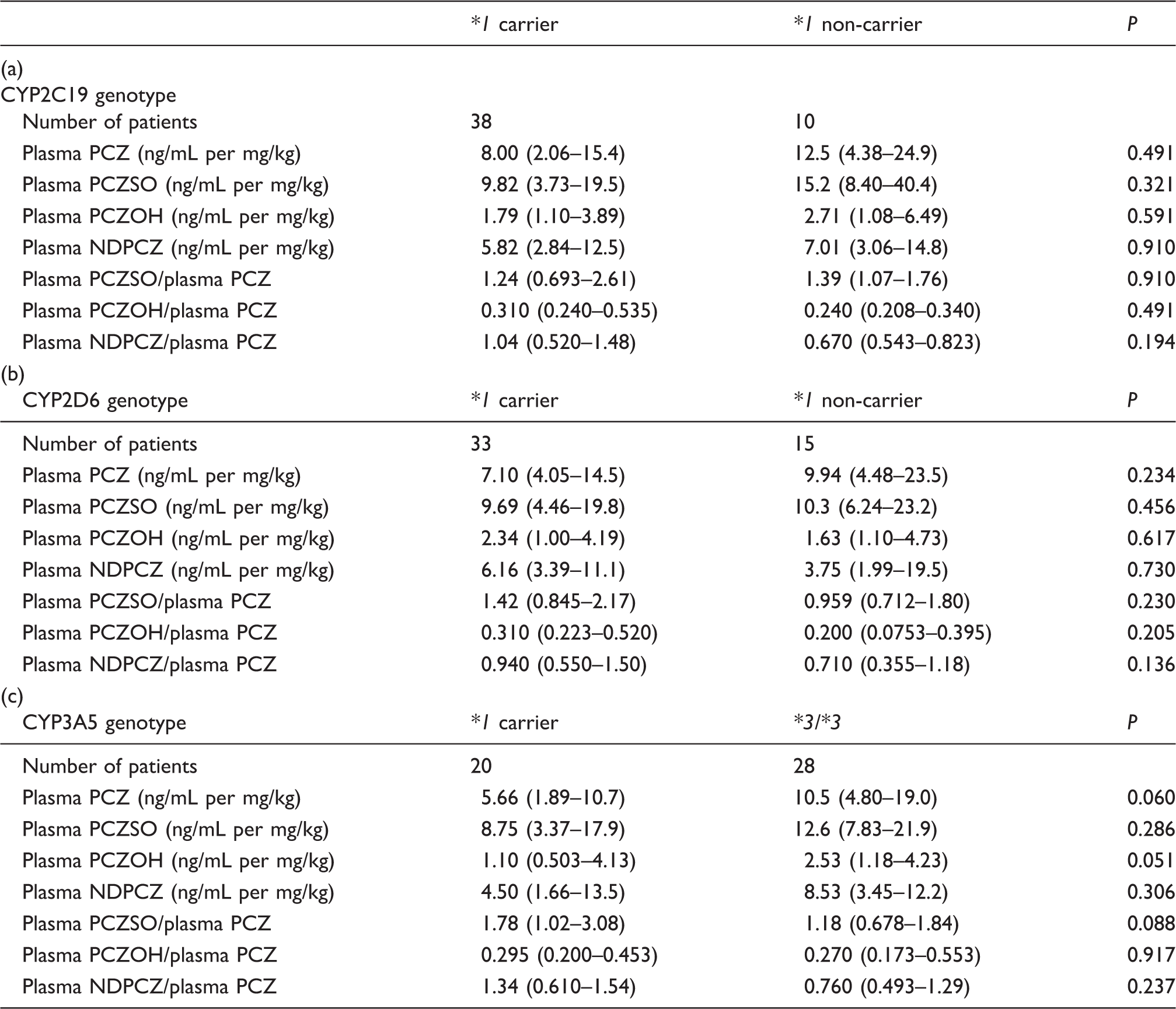
Note: Data are expressed as median with interquartile range in parentheses. The influences of CYPs phenotype on the plasma dispositions of PCZ and its metabolites were tested using the Mann-Whitney U test.
CYP2C19*1 carrier (*1/*1, *1/*2, and *1/*3), CYP2C19*1 non-carrier (*2/*2, *2/*3, and *3/*3), CYP2D6*1 carrier (*1/*1, *1/*5, and *1/*10), CYP2D6*1 non-carrier (*5/*10 and *10/*10), CYP3A5*1 carrier (*1/*1 and *1/*3), and CYP3A5*3/*3.
PCZ: prochlorperazine; PCZSO: prochlorperazine sulphoxide; PCZOH: 7-hydroxy prochlorperazine; NDPCZ: N-desmethyl prochlorperazine;.
Relationships with antiemetic efficacy
Influences of gender and plasma concentrations of PCZ and its metabolites on the incidences of nausea (a) and vomiting (b) in cancer patients.
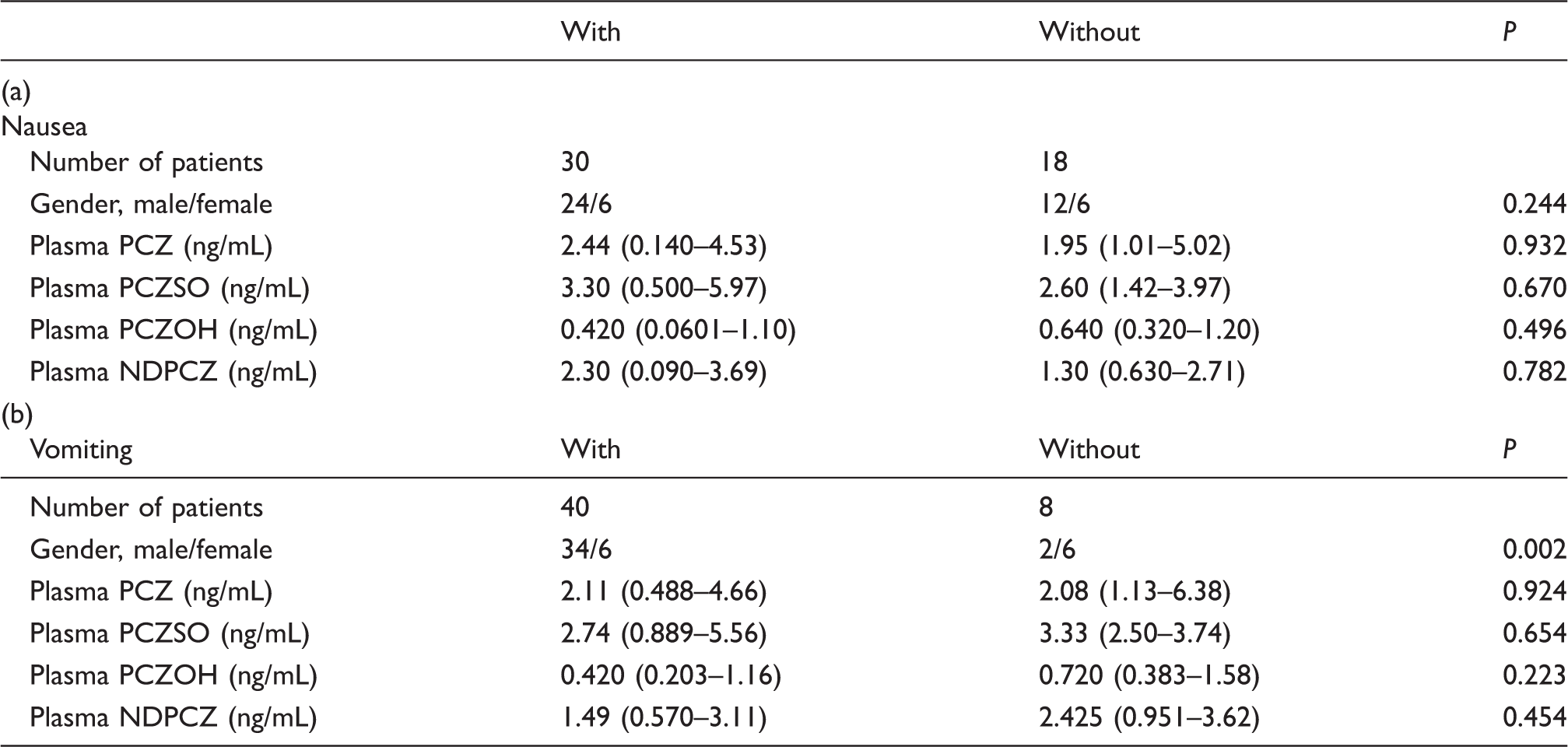
Note: Data are expressed as median with interquartile range in parentheses. The influence of gender on the incidences of nausea and vomiting was tested using Fisher's exact test. The influences of plasma concentrations of PCZ and its metabolites on the incidences of nausea and vomiting were tested using the Mann-Whitney U test.
PCZ: prochlorperazine; PCZSO: prochlorperazine sulphoxide; PCZOH: 7-hydroxy prochlorperazine; NDPCZ: N-desmethyl prochlorperazine.
Relationships with serum prolactin
The median serum prolactin concentration was 33.1 ng/mL (IQR: 29.8–47.3 ng/mL) after PCZ administration. The serum prolactin concentration increased after the treatment in 26 patients who donated serum specimens before starting PCZ administration (median and IQR: 12.7 and 9.6–17.8 ng/mL, P < 0.001). The serum prolactin concentration was weakly correlated with the plasma concentrations of PCZ (ρ = 0.398, P = 0.005), PCZSO (ρ = 0.299, P = 0.039), PCZOH (ρ = 0.548, P < 0.001) and NDPCZ (ρ = 0.332, P = 0.021). The serum prolactin concentration was significantly higher in females than in males after PCZ administration (P = 0.006). Multiple regression analysis demonstrated that plasma PCZ concentration (standardized partial regression coefficient, β = 0.332, P < 0.001) and female gender (β = 0.428, P = 0.005) were significantly associated with the serum prolactin concentration.
Discussion
This study evaluated the relationships between the plasma dispositions of the PCZ metabolites and antiemetic efficacy or prolactin secretion based on CYP2C19, CYP2D6 and CYP3A5 genotypes in cancer patients. These CYP genotypes did not alter the plasma concentrations of PCZ and its metabolites in this study. The plasma concentrations of the PCZ metabolites together with the parent drug were not associated with the incidence of nausea and vomiting, while they were weakly correlated with serum prolactin concentration. The plasma concentrations of the PCZ metabolites have a weaker effect on the secretion of prolactin. These findings suggest that the influences of the PCZ metabolites on antiemetic effect and prolactin secretion were similar to that of PCZ. To the best of our knowledge, this is the first report that has evaluated the relationships between CYP genotype, PCZ metabolite pharmacokinetics, antiemetic efficacy and prolactin secretion in cancer patients.
Interindividual variations were observed in the plasma concentrations of the PCZ metabolites together with the parent drug in this study population. PCZSO, PCZOH and NDPCZ are identified as major PCZ metabolites in humans. 4 The present study showed that the plasma concentration of PCZSO was higher than that of the other metabolites, followed in order by those in NDPCZ and PCZOH. Finn et al. 3 reported that the major metabolite of PCZ was PCZSO in healthy subjects. Weak correlations were observed between the plasma concentrations of PCZ and its metabolites in this study population. This result indicates that the plasma exposure of the PCZ metabolites is largely determined by that of PCZ. This study has demonstrated that the plasma concentrations of the PCZ metabolites can be predicted from that of PCZ.
The CYP2C19 and CYP2D6 genotype did not affect the plasma exposure and metabolism of PCZ in this study. However, the plasma concentrations of PCZ and PCZOH in the CYP3A5*3/*3 group tended to be higher than those in the CYP3A5*1 carrier group (P = 0.060 and P = 0.051). These results indicated that CYP3A5 is potentially involved in the metabolic pathway of PCZ to PCZSO or to NDPCZ. In addition, the metabolic ratio of PCZ to PCZSO in the CYP3A5*3/*3 group was slightly lower than that in the CYP3A5*1 carrier group (P = 0.088). Although multiple regression analysis also demonstrates the CYP3A5 genotype did not affect the plasma exposure of PCZ, CYP3A5 may contribute to the metabolic pathway of PCZ to PCZSO. The oral bioavailability of PCZ is very low (10–20%), and the various CYPs may be involved in PCZ metabolism in the intestine and liver.6,16 CYP2C19 and CYP2D6 expression in the intestine is very low, while CYP3A5 expression is detectable. 24 Thus, CYP3A5 may have an effect on the first-pass effect in the intestine. In addition, plasma PCZSO may be produced during the first pass through the intestine. The correlations between total bilirubin and plasma concentration PCZ, PCZSO or NDPCZ were observed in this study. Although the present study did not enrol the liver dysfunction patients with a total bilirubin concentration >2 mg/dL, the biliary excretion efficiency may affect the pharmacokinetics of PCZ and its metabolites.
This study detected CYP2C19*2 and *3, CYP2D6*10 and CYP3A5*3 using a PCR-RFLP and CYP2D6*5 using a long-PCR. Other functional genetic variants such as CYP2C19*4 and *5 and CYP2D6*2, *3 and *4 were not determined. CYP2C19*4 and *5 and CYP2D6*3 and*4 are little observed in Japanese.25,26 Allele frequency of CYP2D6*2 with little CYP2D6 activity was 0.13. 23 PCR-RFLP analysis is not suitable for the detection of non-target genotypes and haplotypes. Newer techniques such as allele-specific primer PCR and single-strand conformation polymorphism analyses can determine the haplotypes and multiple genotypes. Further studies with the analyses of haplotype or multiple genotypes with newer techniques would be needed to clarify the impact of these genetic variants.
The plasma concentrations of the PCZ metabolites were not associated with the incidences of opioid-induced nausea and vomiting in cancer patients in this study. Our previous study demonstrated that no difference was observed in the plasma concentrations of PCZ between patients with or without nausea and vomiting. 5 In the present study also, the plasma concentrations of PCZ and its metabolites were not strongly associated with the antiemetic effect of PCZ. However, the plasma concentration of PCZSO was higher than those of PCZ, NDPCZ and PCZOH. There was no correlation between PCZSO and the incidence of nausea and vomiting. In contrast, the PCZ metabolites with higher polarity may have a low degree of permeability through the blood–brain barrier. The plasma concentrations of the PCZ metabolites as well as the parent drug have little influence on the antiemetic effect in cancer patients.
The plasma concentrations of the PCZ metabolites were weakly correlated with the serum prolactin concentration in the present study. Our previous study also demonstrated that the plasma PCZ concentration was weakly correlated with the serum prolactin concentration. 5 The plasma PCZ metabolites may have little effect on the serum prolactin concentration. In addition, our study confirmed that gender was a major factor for determining the serum prolactin concentration. In a previous study, OPRM1 A118G polymorphism and female gender had impacts on the serum prolactin concentration. 5 Our results indicated that one factor alone could not determine the serum prolactin concentration in cancer patients.
This study has several limitations. First, it analysed only CYP2C19, CYP2D6 and CYP3A5 for the evaluation of plasma exposure of PCZ and its metabolites. The flavin-containing monooxygenase 3 has a role in sulphoxidation and N-oxidation of the drug in the liver. 27 This enzyme may be involved in the production of PCZSO. Since human intestines have a low level of flavin-containing monooxygenase expression, its impact on the first-pass effect of PCZ would be small in first pass metabolism. Second, the liver dysfunction caused by cancer itself or liver metastasis has the potential to affect the metabolic process of PCZ in cancer patients. However, this study did not enrol cancer patients with a total bilirubin concentration >2 mg/dL; hence, the patients with liver dysfunction were excluded according to the criteria. The liver dysfunction based on cancer conditions would have little impact on our results. A recent study reported that inflammation reduces the expression of CYPs in the liver. 28 Future studies should consider CYP reduction caused by cancer inflammation such as cachexia. Third, the present study evaluated the plasma disposition of PCZ metabolites in cancer patients receiving oral PCZ. Since intramuscular PCZ is not influenced by first pass in the intestine and liver, the CYP genotype may have a different impact on the pharmacokinetics of PCZ and its metabolites in cancer patients receiving intramuscular PCZ. Fourth, opioid analgesics elevate the serum prolactin concentration.17,18 Although oxycodone have a lower effect for serum prolactin, our results related to serum prolactin need to be applied to the patients receiving both oxycodone and PCZ.
The clinical implications of monitoring the plasma dispositions of PCZ metabolites and the CYP genotyping in PCZ treatment have not been fully clarified in cancer patients. In this study, the PCZ metabolites did not have an effect on the antiemetic effect of PCZ. Our data indicated that the therapeutic monitoring of plasma PCZ is useful for the prediction of serum prolactin in cancer patients. In contrast, vomiting and serum prolactin elevation are seen females compared with male in cancer patients receiving oxycodone and PCZ. The clinical consideration of CYP genotype may not be necessary when using PCZ for opioid-induced nausea and vomiting in cancer patients.
Conclusions
The CYP2C19, CYP2D6 and CYP3A5 genotypes did not affect the plasma dispositions of PCZ and its metabolites. In addition, the PCZ metabolites did not have a strong effect on the antiemetic effect of PCZ, while they were slightly associated with prolactin secretion in cancer patients.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by JSPS KAKENHI Grant Numbers 23790181 and 25927005.
Ethical approval
This study was approved by the Ethics Committee of Hamamatsu University School of Medicine (24-233). All of the patients signed an informed consent form.
Guarantor
TN.
Contributorship
MT and TN conceptualized and designed the study. MT performed laboratory detection, drafted the initial manuscript, analysed data and performed statistics; NT, YK, and JK reviewed and revised the manuscript and approved the final manuscript as submitted.
