Abstract
Introduction
Hyperglycaemia increases succinate concentrations and succinate receptor activation in the kidney resulting in renin release. The aim of our study was to determine if there is an association between glycaemic control as evidenced by glycated haemoglobin values and activation of the renin-angiotensin-aldosterone system in patients with type 2 diabetes mellitus and hypertension.
Methods
A cross-sectional study was conducted at Galway University Hospitals between December 2014 and March 2015. Participants (n = 66) were identified following interrogation of the electronic database for patients with type 2 diabetes mellitus. Baseline clinical demographics, aldosterone, plasma renin activity, direct renin concentration, urea and electrolytes, glycated haemoglobin, cholesterol, urine sodium and albumin creatinine ratio were recorded.
Results
There was a significant positive linear correlation between glycated haemoglobin and renin (both plasma renin activity [P = 0.002] and direct renin concentration [P = 0.008]) and between serum creatinine and aldosterone measured using both radioimmunoassay (P = 0.008) and immunochemiluminometric assay (P = 0.008). A significant negative linear correlation was demonstrated between serum sodium and plasma renin activity (P = 0.005) and direct renin concentration (P = 0.015) and between estimated glomerular filtration rate and aldosterone measured using radioimmunoassay (P = 0.02) and immunochemiluminometric assay (P = 0.016). A significant negative linear correlation existed between urine sodium and plasma renin activity (P = 0.04) and aldosterone measured using radioimmunoassay (P = 0.045).
Conclusions
There is a direct positive association between glycaemic control and renin. We advocate for renin measurement to be part of the diabetologist's armamentarium to assess, guide and optimize therapeutic strategies in patients with diabetes.
Introduction
Diabetes mellitus represents a group of metabolic disorders characterized by hyperglycaemia resulting from defects in insulin secretion, insulin action or both. 1 In 2015, 415 (340–536) million people worldwide (aged 20–79 years) had a diagnosis of diabetes mellitus. 2 By 2040, this is expected to increase to 642 (521–829) million people. 2 Hyperglycaemia defines diabetes and is a causative factor for its complications that result in significant morbidity and mortality for millions of people worldwide. 3 Common microvascular complications include retinopathy, nephropathy and neuropathy. Diabetes is the commonest cause of chronic kidney disease (CKD) and end-stage renal disease (ESRD) worldwide.4–6 Epidemiological evidence suggests a correlation between glucose, atherosclerotic plaque burden, cardiovascular events and increased ill health and death.7–9
Glycaemic control as assessed through the measurement of HbA1c is central to the management of diabetes. HbA1c is haemoglobin modified by the covalent attachment of glucose to the amino terminus valine of the β-chain of adult haemoglobin (HbA). The concentration of HbA1c depends on the life span of red blood cells and the concentration of blood glucose.10,11 HbA1c reflects the average blood glucose over the previous 120 days.12–16 There is a direct association between HbA1c and mean blood glucose.17–23 The clinical utility of HbA1c as a marker of diabetic control was not fully appreciated until the publication of two seminal trials: the Diabetes Control and Complications Trial (DCCT) for people with type 1 diabetes mellitus in 1993 and the United Kingdom Prospective Diabetes Study for people with type 2 diabetes mellitus (T2DM) in 1998.24–26 These studies confirmed that there is a direct relationship between mean blood glucose and the risk of progression of complications of diabetes.26–29
The metabolic abnormalities associated with T2DM, especially insulin resistance, cause activation of the renin-angiotensin-aldosterone system (RAAS).3,30 Glucose metabolism generates adenosine triphosphate and pyruvate. Pyruvate is the substrate of the citric acid cycle. The G-protein-coupled receptor 91 (GPR91) is activated by succinate (an intermediate metabolite of the citric acid cycle). 31 GPR91 is highly expressed in the kidney and activation of this receptor triggers paracrine signalling, resulting in renin release; this suggests that there may be a direct link between hyperglycaemia and renin release from the juxtaglomerular apparatus (JGA);31–33 as glycaemic control decreases renin increases. 32 Gonçalves et al. 34 demonstrated that there is an association between HbA1c and renin (r = 0.20 [0.14–0.26]) measured using the proximity extension assay technique. Renin converts angiotensinogen into angiotensin I (ANG I). ANG I is converted into angiotensin II (ANG II) by angiotensin-converting enzyme (ACE). ANG II is a critical mediator of renal injury resulting from diabetes.35,36 Pharmacological inhibition of ANG II slows disease progression beyond that which would be expected by blood pressure-lowering effects alone, 30 suggesting the importance of non-haemodynamic pathways of ANG II in mediating disease. 37 Concentrations of ANG II in tubular ultrafiltrate are up to 1000-fold greater than concentrations in the systemic circulation, indicating intrarenal generation. 38 Podocyte injury and loss are cardinal features of diabetic kidney disease. 39 The podocyte is a direct target of ANG II.
The post hoc analysis of the AVOID study showed combination therapy (direct renin inhibitor and an ACE-i or ARB) resulted in a greater reduction in microalbuminuria in patients with poorly controlled diabetes (HbA1c: > 68 mmol/mol/>8.4%) compared with those with good control (HbA1c: < 54 mmol/mol/<7.1%). 40 RAAS blockade may have a greater effect on those with higher HbA1c and possibly higher renin.
The aim of our study was to determine if there is an association between glycaemic control as evidenced by HbA1c and activation of the RAAS in patients with T2DM and hypertension.
Methods
Ethical approval for this study was granted by the Research Ethics Committee at Galway University Hospitals (GUH). The study was conducted in accord with the ethical principles outlined in the Declaration of Helsinki.
Study design
A cross-sectional study was conducted at the Centre for Endocrinology, Diabetes and Metabolism (CEDAM) at GUH between December 2014 and March 2015. Participants were recruited prospectively (Figure 1(a)). Participants were identified following interrogation of the electronic database (DIAMOND®) for patients with diabetes, using non-probability consecutive sampling. A letter of invitation to participate in this study was sent to eligible participants. Participants were invited to attend the CEDAM where further information was provided and informed written consent given. Consenting participants provided blood and urine samples and other clinical and laboratory measurements were recorded.
(a) Flow diagram of study design, (b) flow diagram of patient recruitment.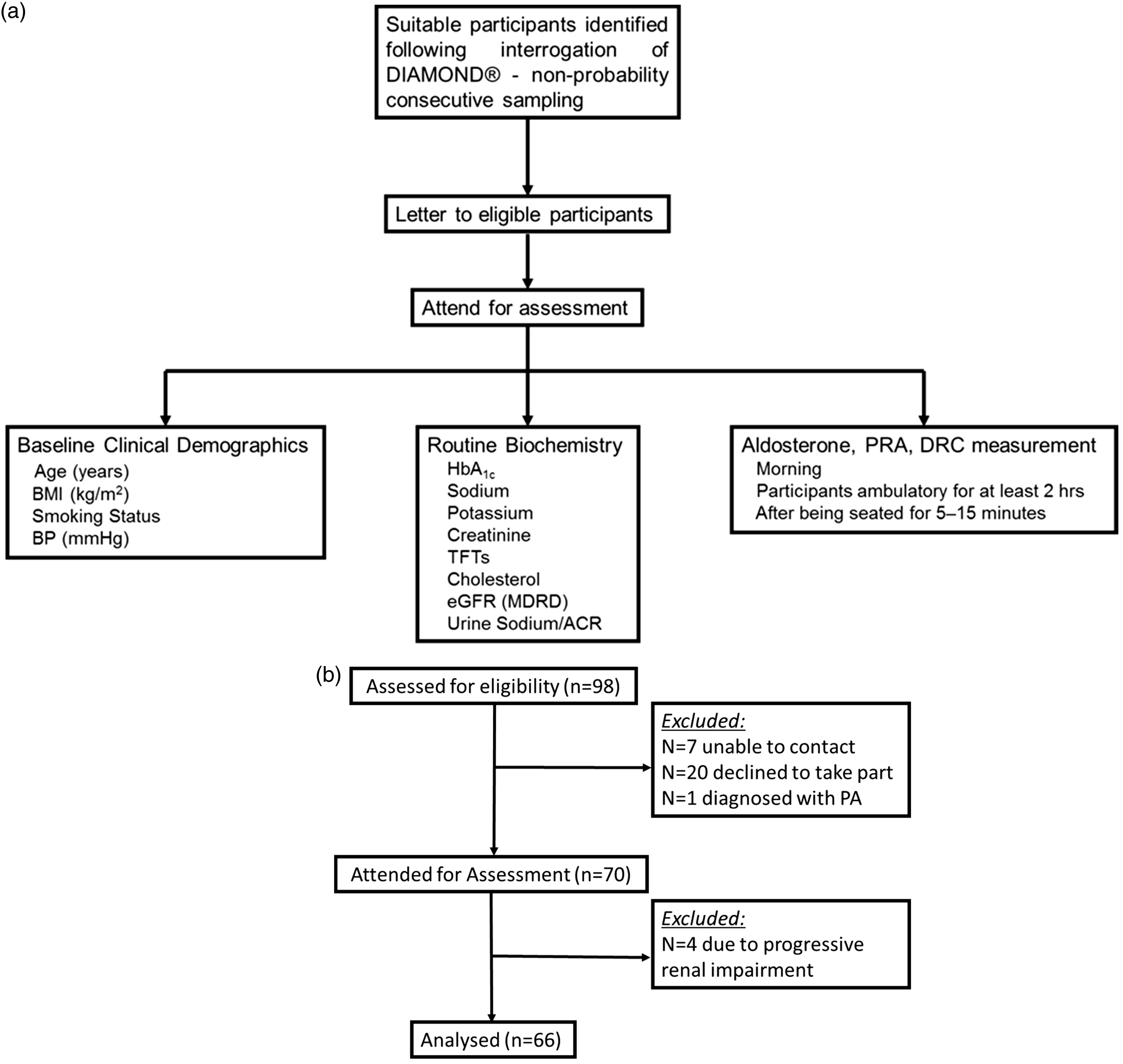
Inclusion criteria
The study inclusion criteria were: age ≥18 years and ≤80 years, Irish white population, unrestricted salt diet, normal serum potassium at baseline, alanine aminotransferase ≤3 times the upper reference limit, T2DM, chronic hypertension (on treatment for hypertension ≥1 year), unchanged antihypertensive medications in the previous three months, biochemically euthyroid and normocalcaemic.
Exclusion criteria
The study exclusion criteria were: known secondary causes of hypertension, stage 4 or 5 CKD, 41 renal replacement therapy or transplantation, congestive cardiac failure and patients taking the following prescribed medications: direct renin inhibitors, mineralocorticoid receptor antagonist, amiloride therapy, centrally acting antihypertensive therapy, oral contraceptive pill, hormone replacement therapy or self-reported poor medication adherence.
Measurement of clinical and laboratory variables
Baseline clinical demographics were recorded (Figure 1(a)). Blood pressure was recorded as previously described using a UM-101 mercury-free manual sphygmomanometer. 42 Pulse rate was recorded at the radial artery in the dominant arm as beats/min.
Venipuncture was performed as per the Endocrine Society Clinical Practice Guidelines for measuring aldosterone, PRA and DRC. 43 Blood (20 mL) was drawn from each eligible participant and collected in appropriate specimen tubes (Becton Dickinson® plastic vacutainer): potassium ethylenediaminetetraacetic acid (EDTA) plasma for the measurement of aldosterone/DRC using Immunodiagnostics Systems (IDS/iSYS®) immunochemiluminometric assay (ICMA) platform and PRA using RADIM/MAIA® radioimmunoassay (RIA) kit and plain (serum) for the measurement of aldosterone by RADIM/MAIA® RIA (Figure 1(a)). The modification of diet in renal disease 4-variable study equation was used to calculate estimated glomerular filtration rate (eGFR). 44
As outlined in previous studies,45,46 DRC and aldosterone were measured in EDTA plasma using the IDS/iSYS® ICMA. In brief, the DRC assay is calibrated to the WHO International Standard 68/356. The inter-assay precision expressed as coefficient of variation (CVA %) over DRC concentrations from 14 mIU/L to 390.2 mIU/L ranged from 4.9% to 8.4%. The IDS-iSYS® aldosterone assay is referenced to LC-MS/MS. The between-run CVA% for aldosterone concentrations from 238 pmol/L to 1648 pmol/L was < 9.71%. 47
For PRA, a modified method proposed by Sealey et al. 48 was used. The RADIM/MAIA® aldosterone method is a competitive RIA. Aldosterone in serum samples competes with iodine125 (I125)-labelled aldosterone for a limited number of aldosterone antibody sites. The amount of aldosterone I125 bound by the antibody is inversely proportional to the amount of aldosterone present in the sample. Between-assay precision for PRA gave CVA% of <10% at PRA of 0.69 ng/mL/h and <5% at PRA of 2.96–6.33 ng/mL/h. For aldosterone, the inter-assay precision gave a CVA% of <5% at concentrations of between 244 and 3805 pmol/L.
HbA1c was assayed using the Menarini® HA 8160 automated haemoglobin analyser. The method was calibrated according to International Federation of Clinical Chemistry (IFCC) standardization. Calibrator values were assigned by two approved IFCC Secondary Reference Methods in one approved network laboratory. The system operates on the principle of reverse phase cation exchange chromatography. The CVA% at a mean HbA1c of 41.6 mmol/mol (Derived DCCT: 6%) and 100.5 mmol/mol (Derived DCCT: 11.4%) was 2.0 and 1.3%, respectively. The mean bias of the assay as determined by proficiency testing through the Irish External Quality Assessment Scheme was −2.1% when compared with values assigned by the IFCC reference laboratory.
Statistical analysis
Statistical analysis was performed using R® V3.2.0 (R Foundation for Statistical Computing, Austria; accessible at www.r-project.org). Summary statistics for normally distributed continuous variables were represented by mean (standard deviation [SD]) and median (interquartile range) for data which were not normally distributed. Categorical data were summarized with frequencies (percentages). Comparisons of proportions were performed using a chi-square test. Relationships between HbA1c and urea, electrolytes and aldosterone/renin parameters were assessed using Pearson's correlations. Multiple linear regression models were used to explore the relationships between HbA1c and PRA, DRC and aldosterone taking various potential confounders into account including gender, age, blood pressure, serum sodium, serum potassium, eGFR and the number and type of antihypertensives. This approach permits an estimate to be made of the association between a given independent variable and the outcome holding all other variables constant. Stepwise regression analysis – a method that involves carrying out multiple regression a number of times, each time removing the weakest correlated variable until you are left with the variables that best explain the distribution – was also used. Stepwise regression requires that the residuals of the data be normally distributed and that there is no collinearity between the independent variables. Stepwise regression was used to identify which of the baseline characteristics and types of antihypertensives needed to be included in the model. For statistical analysis, adherence to medication was assumed to be 100%. A P-value less than 0.05 was deemed statistically significant.
Results
Recruitment of subjects
The patient recruitment strategy is outlined in Figure 1(b). In brief, patient information leaflets were sent to 98 patients following interrogation of our patient database, DIAMOND®. It was not possible to contact seven patients. Twenty patients declined to take part in the study and one patient had a new diagnosis of primary aldosteronism (PA) since review in diabetes clinic. On review, four patients had experienced a decline in renal function below the threshold for inclusion in the study. In total, 66 subjects with T2DM were enrolled in the study.
Baseline patient demographics
Demographics, clinical details, renal indices, metabolic parameters, aldosterone and renin measurements.
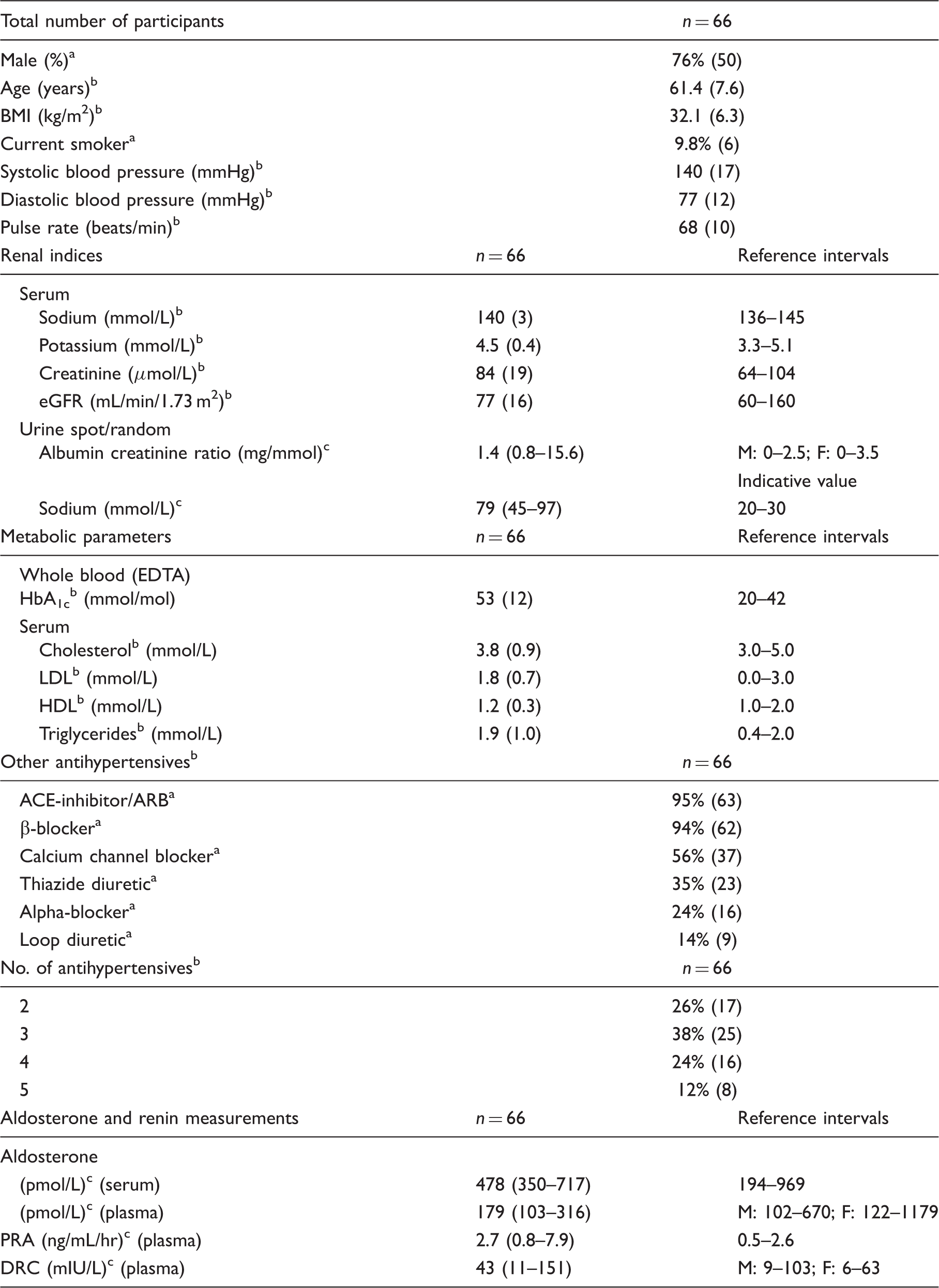
BMI (kg/m2): body mass index (kilogram per metre2); eGFR: estimated glomerular filtration rate; EDTA: ethylenediaminetetraacetic acid; ACE: angiotensin-converting enzyme; ARB: angiotensin receptor blocker; DRC: direct renin concentration; PRA: plasma renin activity; M: male; F: female.
Percentage (number effected/total number).
Mean (standard deviation).
Median (IQR).
Relationships between variables
Correlation between renin aldosterone parameters and HbA1c, serum sodium, serum potassium, creatinine, eGFR, urine sodium, urine ACR and cholesterol.
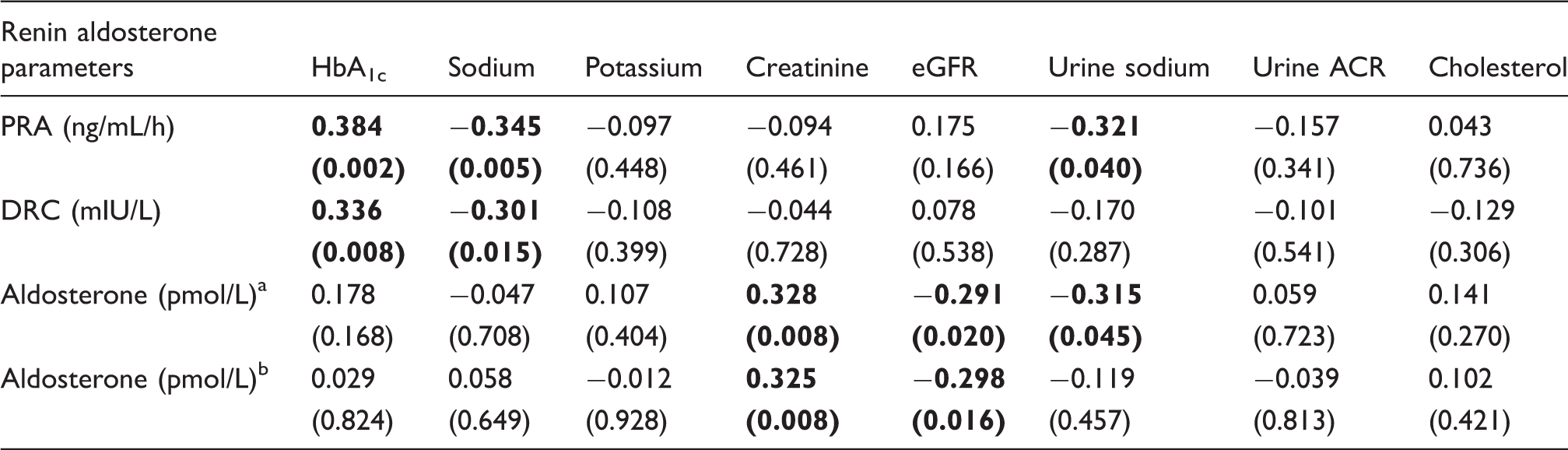
eGFR: estimated glomerular filtration rate; ACR: albumin to creatinine ratio; PRA: plasma renin activity; DRC: direct renin concentration.
Aldosterone measured in serum using RIA platform.
Aldosterone measured in plasma using IDS/iSYS® ICMA platform. Statistically significant values are in bold.
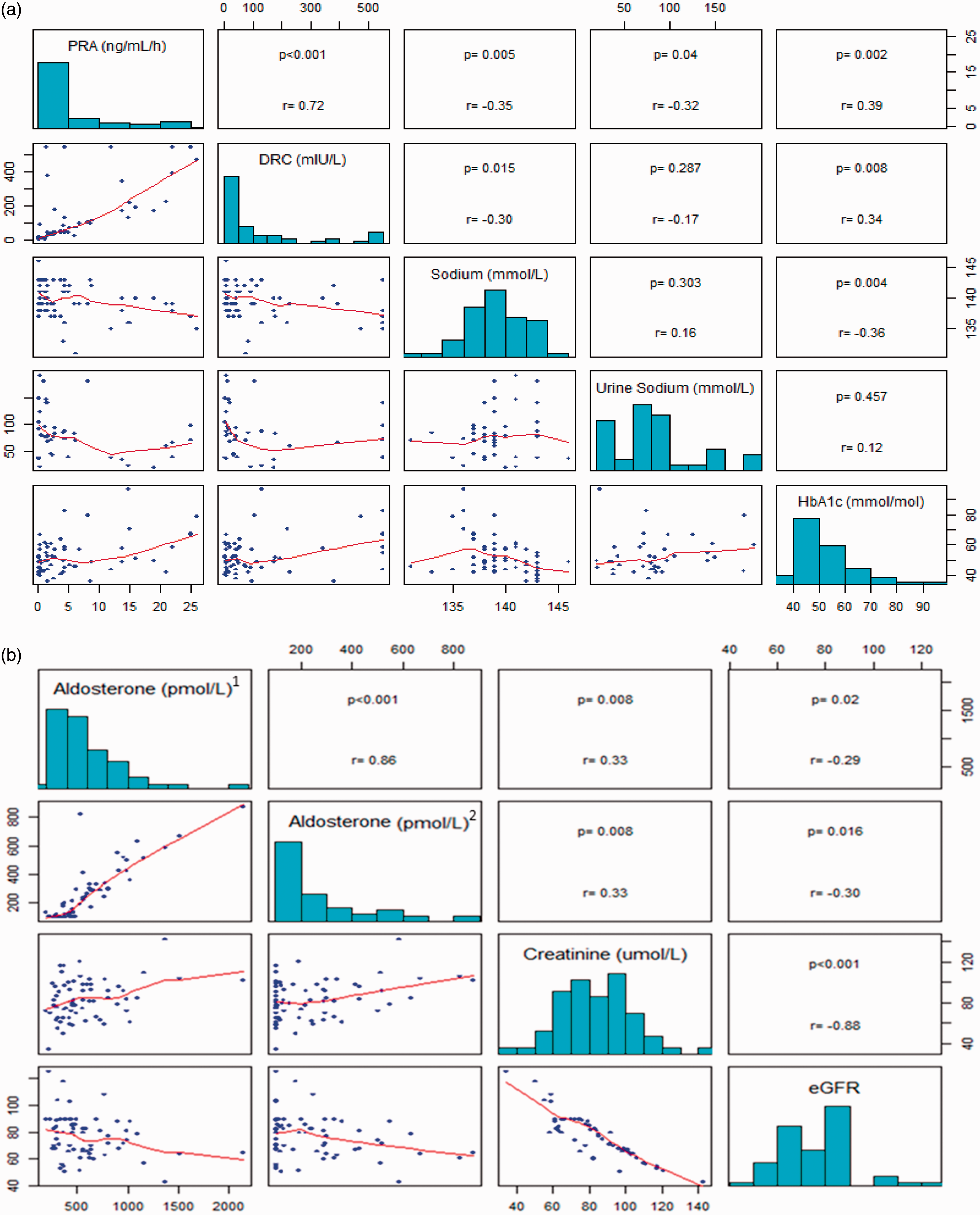
(a) Correlation between plasma renin activity (PRA) (ng/mL/h), direct renin concentration (DRC) (mIU/L), sodium (mmol/L), urine sodium (mmol/L) and HbA1c (mmol/mol). (b) Correlation between Aldosterone (pmol/L)a, Aldosterone (pmol/L)b, creatinine (µmol/L) and eGFR (mL/min/1.73 m2).a Aldosterone measured in serum using RIA platform. bAldosterone measured in plasma using IDS/iSYS® ICMA platform.
Multivariate models
Multivariate model of the association between (a) PRA and HbA1c and (b) DRC and HbA1c.
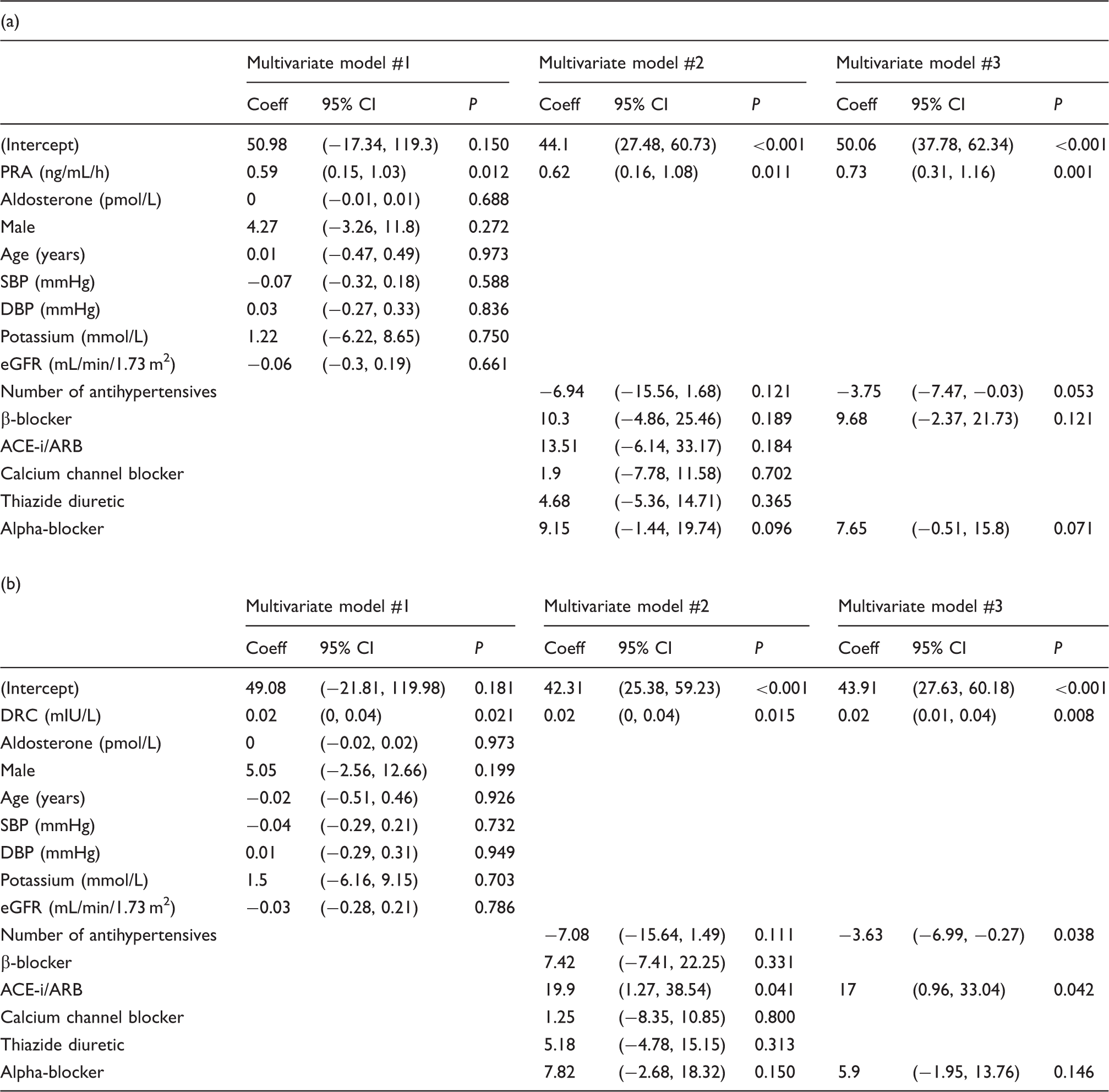
Coeff: the regression coefficient; CI: confidence interval; eGFR: estimated glomerular filtration rate; ACR: albumin to creatinine ratio; PRA: plasma renin activity; DRC: direct renin concentration; ACE: angiotensin-converting enzyme; SBP: systolic blood pressure; DBP: diastolic blood pressure.
Discussion
This study found that there is a positive association between renin and HbA1c and a negative association between renin and serum/urine sodium. There is a positive association between creatinine and aldosterone and a negative association between eGFR and aldosterone. After adjusting for multiple confounders, the association between PRA and HbA1c (0.62; 95% CI: 0.16, 1.08) and DRC and HbA1c (0.02; 95% CI: 0, 0.04) remained significant. There was a stronger association between HbA1c and PRA than HbA1c and DRC. PRA reflects the activity of the plasma enzyme renin. It does not reflect the actual concentration of renin.49,50 It is plausible that PRA may better reflect the cellular actions of the RAAS.
Hyperglycaemia leads to the formation of succinate and the activation of its receptor (GPR91) 31 in renal, cardiac, pulmonary and other tissues. This results in renin release and links the local effects of elevated glucose with the intrarenal and the systemic activation of RAAS.31–33 It is worth noting that Toma et al. 31 in their rabbit and murine models of diabetes exploring the link between high blood glucose and renin, induced diabetes using streptomycin. Induction of diabetes was deemed successful if blood glucose concentrations were >22.2 mmol/L/>400 mg/dL. 31 The succinate receptor is a key mediator in the development of hypertension and possibly fibrosis in diabetes mellitus. 51 Durvasula and Shankland 30 found that exposure to high glucose resulted in a 2.1-fold increase in ANG II mediated through increased renin activity which could promote progressive podocyte injury and loss in diabetic kidney disease. Escalating doses of glucose (25 mmol/L/450 mg/dL and 40 mmol/L/721 mg/dL) were used in vitro to mimic diabetic conditions. The concentrations of glucose employed in the studies of Durvasula and Shankland 30 and Toma et al. 31 are far greater than those routinely seen in clinical practice. In our study, the mean HbA1c was 53 mmol/mol and equivalent to a mean plasma glucose of 8.6 mmol/L/155 mg/dL. 52 Despite participants in our study having lower mean glucose concentrations, we demonstrated that HbA1c is associated with PRA and DRC but not aldosterone. This confirms the findings of Gonçlaves et al. 34 who demonstrated an association between HbA1c and renin measured using a different platform. Unlike these authors, we did not find an association between age, BMI, SBP, cholesterol or eGFR and renin. Our data suggest that as glycaemic control deteriorates, renin increases but aldosterone stays constant. It is surprising that this increase in renin was not associated with an increase in aldosterone. The majority of participants in this study were on an ACE-i or ARB reducing the conversion of ANG I to ANG II. This prevents the increased renin stimulating aldosterone secretion.
Renin may be more responsive to changes in blood glucose. Changes in HbA1c (reflecting glycaemia [weighted average] over the previous two to three months) may underestimate the strength of this association. Persistently elevated HbA1c and insulin resistance are associated with more micro- and macro-vascular complications of diabetes. Based on our observations, it is possible that elevated renin is associated with worse outcomes in patients with T2DM. Our results suggest that patients with higher HbA1c and thus higher renin may benefit more from RAAS blockade (as a greater reduction in renin could be expected with optimal RAAS inhibition) than patients with lower HbA1c. Parving et al. 53 noted that there was no benefit from dual RAAS blockade in terms of cardiovascular or renal events and that such a treatment strategy may be harmful to some study subjects. Measurement of renin/aldosterone parameters was not part of their study protocol. A prospective trial with longitudinal measurements of renin/aldosterone parameters would be required to explore this hypothesis.
The inclusion of renin in the diagnostic work-up and the monitoring of patients with T2DM may be beneficial, both as an objective indicator of patient compliance with medication regimens and as an aid to clinicians in optimizing treatment strategies. PRA- or DRC-guided antihypertensive therapy has the potential to identify antihypertensive medications matched to the patient's underlying pathophysiology. 54 With this strategy, pretreatment renin concentrations are used to determine the relative contribution of excess sodium-volume content or renin-dependent vasoconstriction in each individual. 55 Patients with low renin may have hypertension mainly due to excess body sodium-volume content, whereas those with medium to high renin may have hypertension due to excess plasma renin-angiotensin vasoconstriction. 56 Patients with low renin may benefit more from antihypertensives like thiazide diuretics and calcium channel blockers. Those with high renin concentrations may benefit more from ACE-i, ARB and β-blockers. Thus, measuring renin will help guide the personalized treatment strategy for our patients. A clinical trial comparing blood pressure control and cardiovascular outcomes in patients' whose therapy was and was not guided by renin measurement will be required to explore this.
The relationship between renin and serum/urine sodium is of interest. A potential explanation for this relationship is that decreased delivery of sodium to the macula densa results in increased renin secretion. 57 Thus, lower serum and urine sodium would stimulate higher renin production and higher serum and urine sodium would suppress renin release. Low plasma renin is a marker of sodium excess. Ekinci et al. 58 has demonstrated that lower 24-h urine sodium excretion was associated with increased all-cause and cardiovascular mortality. This may be associated with elevated renin concentrations and is worthy of further investigation.
Previous studies of hypertensive patients with preserved renal function have demonstrated a modest negative correlation between plasma aldosterone concentrations and eGFR (r = −0.196; P < 0.01). 59 Patients with lower renal function values have a higher plasma aldosterone. 59 This is confirmed in our study. Aldosterone has been shown to be increased in diabetes and is associated with the development of diabetic kidney disease. 60 It has been observed that an increase in potassium ion concentration in the extracellular fluid causes a marked increase in aldosterone secretion. 61 Unsurprisingly, there were no subjects with hypo or hyperkalaemia in our sample. Of import and not unexpectedly, there was no association between aldosterone and serum potassium in our sample.
Participants in our study were on a minimum of two antihypertensive agents. The majority (95%) were on an ACE-i or ARB – which is considered the cornerstone of blood pressure management in diabetes.62–64 Interestingly, 94% of our participants were on β-blocker therapy. Current treatment algorithms would favour calcium channel blockers and thiazide diuretics as second- and third-line agents. Potential explanations include other indications for β-blockers including ischaemic heart disease or tachycardia.65,66 Treatment algorithms and the role of β-blocker therapy in the management of hypertension have changed over time from AB/CD algorithm 67 to the recent JNC 8. 65 Patients established on β-blocker therapy with well-controlled blood pressure would not have their β-blocker changed to an alternative agent as part of clinical practice at our institution.
The findings of this study suggest the potential value of routine measurement of renin in patients with diabetes to assess, guide and optimize therapeutic strategies.
Limitations/strengths of this study
This study adds to the current available literature as it confirms the association between renin and HbA1c using two different clinically available platforms (RIA and ICMA). To our knowledge, this association had not previously been confirmed when renin was measured as either PRA or DRC. Similar associations were observed using both measurements which adds to the strength of our findings. This work establishes the foundation for future work that would explore longitudinal trends in renin and aldosterone in patients with diabetes and hypertension.
We acknowledge the small sample size of our study. Our work is descriptive and explores associations between different markers of the renin angiotensin aldosterone system and glycaemic control. It does not prove any causal pathway between poor glycaemic control and elevated renin. Antihypertensive medications have the potential to affect the interpretation of renin and aldosterone.68,69 We have previously shown that β-blocker therapy is associated with a reduction in renin, an increase in the aldosterone renin ratio (ARR) (measured using PRA or DRC) with no significant change in aldosterone.45,46,70 Calcium channel blockers such as amlodipine have been associated with a slight increase in PRA and reduction in aldosterone and ARR. 71 Alpha-blockers such as doxazosin are associated with minimal decreases in aldosterone and ARR.71–73 Thiazide diuretics can also cause a reduction in the ARR.74,75
Conclusions
There is a direct association between glycaemic control and renin. Patients with poor glycaemic control have higher renin concentrations than patients with good glycaemic control. The findings of our current study suggest that in clinical practice and in the context of personalized medicine, renin measurement may help to guide and optimize antihypertensive therapeutic strategies and act to aid the assessment of medication compliance in patients with diabetes. Renin measurement may also serve as a prognostic indicator, as patients who are poorly compliant with prescribed medication have higher all-cause mortality.76–78 Further longitudinal studies are required to evaluate this theory.
Footnotes
Acknowledgements
We wish to express our gratitude to all volunteers who made this study possible. Special thanks to the scientific, nursing and medical staff at the Centre for Endocrinology, Diabetes and Metabolism, and the Department of Clinical Biochemistry at Galway University Hospitals and Saolta Hospital Group.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval for this study was granted by Galway University Hospitals (GUH) Clinical Research Ethics Committee (Ref. No.: CA1075).
Guarantor
PM O'Shea.
Contributorship
TPG and PMOS – conception, design, data collation and assembly. TPG, GAB and MCD – recruitment, consenting of patients and sample collection. PMOS –quality and accuracy of laboratory methods. DW: statistical analysis. TPG wrote first draft of manuscript. All authors reviewed, edited and approved the final version of the manuscript.
