Abstract
Background
The Japan Statin Treatment Against Recurrent Stroke (J-STARS) is a clinical trial that administered pravastatin for secondary stroke prevention. Lipid concentrations are a well-established risk factor for cerebrovascular diseases. Elevated high-sensitivity C-reactive protein (hs-CRP) indicates a high risk of inflammatory reactions. In clinical trials, internationally approved standardization is essential for obtaining study results that are comparable with those from overseas. Therefore, total cholesterol (TC), high-density lipoprotein cholesterol (HDL-C), low-density lipoprotein cholesterol (LDL-C), triglycerides (TG) and hs-CRP were standardized throughout a 10-year study period.
Methods
J-STARS specified a single clinical laboratory for blood analyses. Four lipids were evaluated by calculating the total error: accuracy (absolute mean %bias vs. reference value) + precision (1.96 among-run coefficient of variation [CV, %]). Accuracy for hs-CRP was ensured using a calibrator traceable to the international plasma protein reference material and precision was evaluated by CV.
Results
Average total errors (standard deviation, %) throughout the study period were as follows: TC 1.35% (0.290%), HDL-C 2.45% (1.087%), LDL-C 2.65% (0.956%) and TG 3.70% (0.559%). Four lipids met the performance criteria of the US Centers for Disease Control and Prevention (CDC). The precision of hs-CRP was 3.28% (0.627%), which met the performance criterion established by the American Heart Association/CDC.
Conclusions
Based on standardization, the results of J-STARS appear to be comparable with those of similar intervention-based clinical studies on statins overseas. These study results will contribute to the establishment of preventive measures against recurrent stroke in Japanese patients. J-STARS is registered in ClinicalTrials.gov under NCT00221104.
Introduction
A large number of intervention studies have been performed on the initial and secondary prevention of arteriosclerotic cardiovascular events by statins (3-hydroxy-3-methylglutaryl-coenzyme A reductase inhibitors).1–5 The Stroke Prevention by Aggressive Reduction in Cholesterol Levels (SPARCL) 6 study was the first clinical trial to investigate the secondary preventive effects of statins on cerebrovascular diseases, such as stroke. In SPARCL, atorvastatin was administered at a high dose (80 mg/day) to patients with a medical history of stroke. Atorvastatin is a potent lipid-lowering statin that is used to prevent recurrent stroke. The effects of atorvastatin were investigated by comparisons between statin and placebo groups in a double-blind study. In the SPARCL study, the statin dose was markedly higher than that approved in Japan; therefore, the findings obtained in the SPARCL study may not be applicable to Japanese patients.
The Japan Statin Treatment Against Recurrent Stroke (J-STARS) 7 (this study was registered in ClinicalTrials.gov under NCT00221104) is an investigator-initiated 10-year clinical trial that involved disease subtype at registration and the administration of pravastatin at a low dose (10 mg/day) to patients aged between 45 and 80 years old with a medical history of non-cardioembolic ischaemic stroke such as lacunar and atherothrombotic infarctions. In this study, the usefulness of statins for the secondary prevention of non-cardioembolic ischaemic stroke in Japanese patients was investigated, and the significance of statins for each disease subtype was also examined.
Serum lipid concentrations have been identified as a risk factor for ischaemic cardiovascular events1–5 and elevated high-sensitivity C-reactive protein (hs-CRP) indicates a high risk of inflammatory reactions.8–10 In clinical trials or epidemiological studies, internationally approved standardization is essential for obtaining high-quality study results that are comparable with those from overseas. Therefore, the standardization of four lipids including total cholesterol (TC), high-density lipoprotein cholesterol (HDL-C), low-density lipoprotein cholesterol (LDL-C) and triglycerides (TG) was performed and hs-CRP concentrations were investigated throughout the 10-year study period. The clarification of measurement performance for these items through the standardization preceded the analytical process of J-STARS.7,11
Materials and methods
A total of 123 regional core hospitals throughout Japan collected blood samples from patients. J-STARS specified a single clinical laboratory in Tokyo, Japan, named SRL, for blood analyses throughout the study period. Eight blood chemistry markers including TC, HDL-C, LDL-C, TG and hs-CRP were analysed. SRL used enzymatic methods to measure TC and TG concentrations and homogeneous methods to assay HDL-C and LDL-C concentrations. The method of hs-CRP is described in the section of hs-CRP standardization. As an internal quality control, in-house pooled serum prepared at SRL was used in lipid and hs-CRP assays for precision control.
Criteria for the measurement performance of TC, HDL-C, LDL-C, TG and hs-CRP.
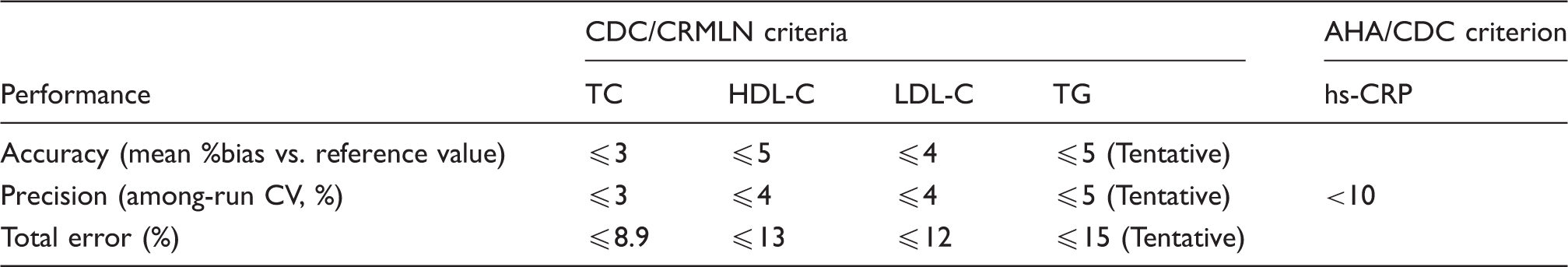
CDC: Centers for Disease Control and Prevention; CRMLN: Cholesterol Reference Method Laboratory Network; AHA: American Heart Association; TC: total cholesterol; HDL-C: high-density lipoprotein cholesterol; LDL-C: low-density lipoprotein cholesterol; TG: triglycerides; hs-CRP: high-sensitivity C-reactive protein; CV: coefficient of variation (%).
Note: TG criteria are tentative.
Total error = accuracy (absolute mean %bias vs. reference value) + precision (1.96 among-run CV, %).
TC standardization
Measurement performance of TC, HDL-C, LDL-C, TG and hs-CRP assayed at SRL between 2004 and 2014.
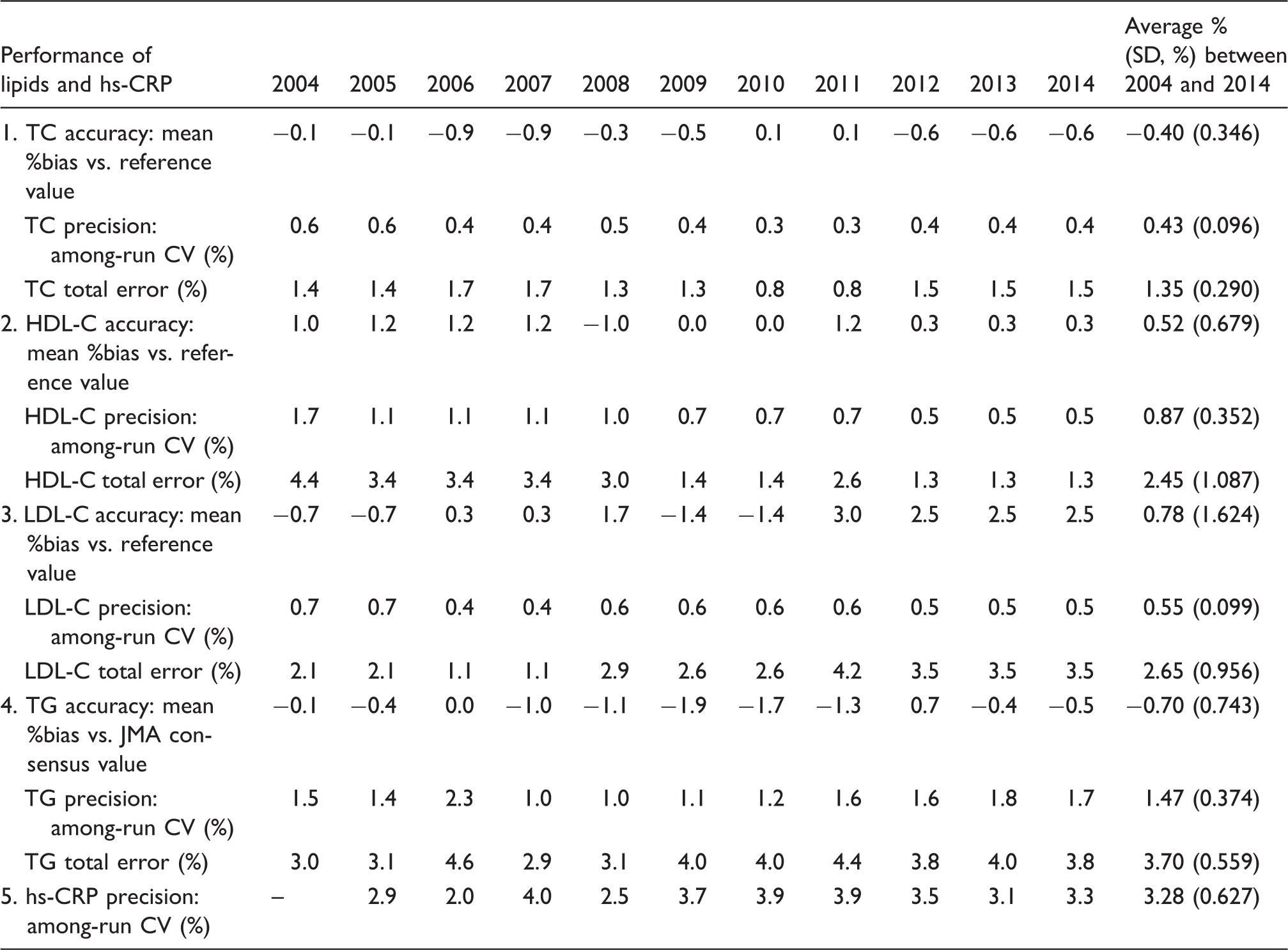
SD: Standard deviation; CV: coefficient of variation (%); JMA: Japan Medical Association; SRL: A clinical laboratory in Tokyo for blood analyses specified by J-STARS; TC: total cholesterol; HDL-C: high-density lipoprotein cholesterol; LDL-C: low-density lipoprotein cholesterol; TG: triglycerides; hs-CRP: high-sensitivity C-reactive protein.
Note: TG criteria are tentative. Total error = accuracy (absolute mean %bias vs. reference value) + precision (1.96 among-run CV, %).
HDL-C standardization
Although SRL is a clinical laboratory, they have participated in the CDC HDL-C standardization programme for reagent manufacturers. In this programme (HDL Cholesterol Certification Protocol for Manufacturers, November 2002, http://www.cdc.gov/labstandards/crmln_manufacturers.html#HDL), at least 40 fresh individual human serum samples were collected at reagent manufacturers. Samples were prepared to include at least five samples each within concentration range of 0.517–0.750, 0.776–1.009, 1.034–1.267, 1.293–1.526 and 1.552–1.784 mmol/L for HDL-C concentrations. They measured these samples in duplicate in five runs over a one-month period using a routine homogeneous method at SRL. The lipid reference laboratory at Osaka measured each sample in duplicate using the HDL-C reference method for CRMLN, the designated comparison method (DCM).15,16 Specimens with a TG concentration of >2.258 mmol/L were excluded from measurements by the DCM 15 because elevated TG concentrations interfere with the precipitation reactions. The reference value serving as an accuracy index was measured by HDL-C DCM. At SRL, the among-run CV of a single quality control serum value measured from 20 separate runs as an internal precision control was assessed and used as a precision index. The reference value and assayed value were both input into CDC comparative evaluation software and statistically analysed. The results obtained were sent to the CDC and evaluated based on the assessment criteria (Table 1). The performance of SRL for HDL-C was then obtained (Table 2).
LDL-C standardization
Although SRL is a clinical laboratory, they have participated in the CDC LDL-C standardization programme for reagent manufacturers. In this programme (LDL Cholesterol Certification Protocol for Manufacturers, June 2006, http://www.cdc.gov/labstandards/pdf/crmln/MFRLDLJune2006final.pdf), at least 40 fresh individual human serum samples were collected at reagent manufacturers. As specified by the standardization programme, 20% of individual human serum samples are expected to be <2.586 mmol/L, 30% within 2.586 to 3.362 mmol/L, 30% within 3.388 to 4.138 mmol/L and 20% within 4.163 to 10.344 mmol/L for LDL-C concentrations. They measured these materials in duplicate in five runs over a one-month period using a routine homogeneous method at SRL. The lipid reference laboratory measured each sample in duplicate using the LDL-C RMP, a beta-quantification method using ultracentrifugation. 17 The reference value serving as an accuracy index was assessed by the LDL-C RMP. At SRL, the among-run CV of a single quality control serum value measured from 20 separate runs as an internal precision control was assessed and used as a precision index. The reference value and measured value were both input into CDC comparative evaluation software and statistically analysed. The results obtained were sent to the CDC and evaluated based on the performance criteria (Table 1). The performance of SRL for LDL-C was then obtained (Table 2).
TG standardization
The external quality assessment of the clinical laboratories programme survey by the Japan Medical Association (JMA) is performed every year in October in Japan. In the JMA survey, TG samples with three concentrations were distributed to participating laboratories. They measured net TG (total glycerides−free glycerol) using a routine enzymatic method with an automatic free glycerol cancellation reagent, 18 which is commonly used in Japan. They were evaluated using a four-step (A, B, C and D) relative evaluation system uniquely established by the JMA representing the deviation from the adjusted mean net TG value obtained after removing statistical outliers from the measurement results of all participating laboratories. The four-step relative evaluation system is defined as follows: A ≤ adjusted mean net TG ± 1 standard deviation (SD) of all participating laboratories after removing statistical outliers; B ≤ adjusted mean net TG ± 2 SD; C ≤ adjusted mean net TG ± 3 SD and D > adjusted mean net TG ± 3 SD. Regarding TG, the adjusted mean from approximately 2700 clinical laboratories was regarded as a consensus value instead of a reference value as an accuracy index by the RMP. Precision as among-run CV was evaluated from SRL internal quality control serum values measured from 20 separate runs. Total error was calculated from accuracy (absolute mean %bias vs. JMA consensus value) + precision (1.96 among-run CV [%]). The performance of SRL for TG was then obtained (Table 2). Total glycerides and free glycerol by gas chromatography-isotope dilution mass spectrometry (GC-IDMS) at Osaka have both been standardized by GC-IDMS at the CDC since April 2011.19,20 The accuracy of net TG by GC-IDMS at Osaka in October 2013 showed −0.73% (n = 8) vs. CDC on average, which was in good agreement with that of the CDC reference value by GC-IDMS.
hs-CRP standardization
Sixty-nine hospitals participated in hs-CRP standardization in the J-STARS substudy. hs-CRP concentrations were measured at SRL. hs-CRP measurement conditions were as follows: measurement device – the BN ProSpec System of Siemens; measurement reagents – N-Latex CRP II CardioPhase hs-CRP, calibrator for instrument control and N-Rheumatology Standard SL. The accuracy of the nominal value of the standard substance for calibrations was tested based on the international plasma protein reference material (IRMM ERM-DA470), which has the highest ranking in the hs-CRP traceability system. In-house pooled serum from SRL was used for internal quality control. Precision was presented as the among-run CV of values measured for 20 separate runs in October every year. The precision performance criterion is shown in Table 1. hs-CRP concentrations were examined in a substudy (this study was registered in ClinicalTrials.gov as number NCT00361699) of J-STARS.
Statistical analysis
All statistical analyses were conducted using the protocol EP9-A from the Clinical and Laboratory Standards Institute for bias estimation 21 and STATA12 analysis program. P-values for statistical tests were two-tailed, and the values of p < 0.05 were regarded as significant.
Results
Table 2 lists the measurement performance by routine analytical methods at SRL of accuracy, precision and total error for TC, HDL-C, LDL-C, TG and hs-CRP by year between 2004 and 2014 as well as the summarized average % (SD, %) throughout the study period. The average % (SD, %) of accuracy, precision and total error for TC for 10 years were −0.40% (0.346%), 0.43% (0.096%) and 1.35% (0.290%), respectively. Similarly, the average % of accuracy, precision and total error for HDL-C were 0.52% (0.679%), 0.87% (0.352%) and 2.45% (1.087%), respectively. The average % of accuracy, precision and total error for LDL-C were 0.78% (1.624%), 0.55% (0.099%) and 2.65% (0.956%), respectively. The average % of accuracy, precision and total error for TG were −0.70% (0.743%), 1.47% (0.374%) and 3.70% (0.559%), respectively; however, TG criteria are tentative (Table 1). The average % of precision for hs-CRP was 3.28% (0.627%). The performance of four lipids and hs-CRP over a 10-year period was stable every year within acceptable performance criteria (Table 2).
Comparison of adjusted mean net TG by JMA, assayed net TG by SRL and target net TG by GC-IDMS using survey samples in 2013.
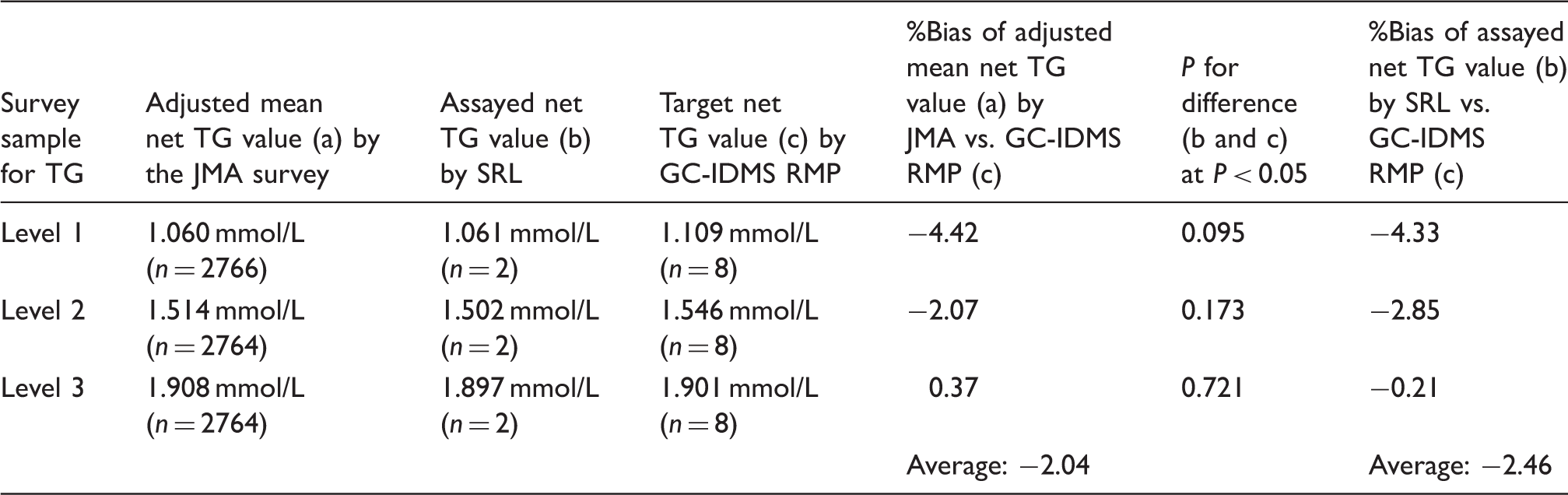
TG: triglycerides; JMA: Japan Medical Association; GC-IDMS: gas chromatography-isotope dilution mass spectrometry; RMP: reference measurement procedure; SRL: A clinical laboratory in Tokyo for blood analyses specified by J-STARS.
Note: TG criteria are tentative. The adjusted mean net TG value by the JMA survey is the net TG value (total glycerides−free glycerol) by the routine enzymatic methods of participating laboratories using automatic free glycerol cancellation reagent.
Baseline profiles of cardiovascular risk factors in J-STARS (n = 1578).
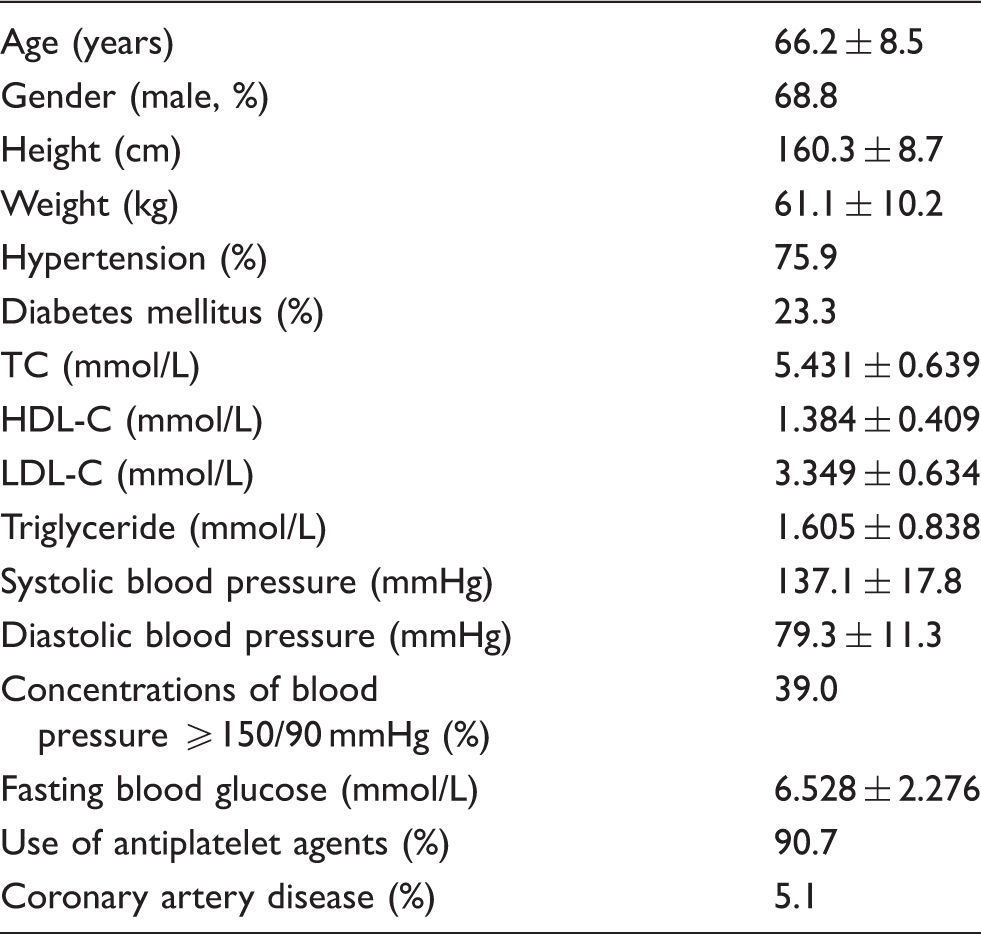
Note: This table is adapted from Nagai et al. 7 in the baseline article of J-STARS.
Data are the mean ± standard deviation (SD) of continuous variables.
TC: total cholesterol; HDL-C: high-density lipoprotein cholesterol; LDL-C: low-density lipoprotein cholesterol.
Discussion
Selection for the measurement of lipids and hs-CRP at a single clinical laboratory and the significance of standardization
A total of 123 regional hospitals throughout Japan provided blood samples from 1578 patients to a single clinical laboratory, SRL in Tokyo. Numerous epidemiological and interventional studies have been conducted with measurements of markers in a single laboratory center. This is also described in relevant studies and textbooks. If measured at multiple laboratories, the reproducibility and accuracy of assayed values may not be maintained or assured at constant quality control for a prolonged time, resulting in misjudgments in the interpretation of J-STARS results. Therefore, J-STARS adopted a measurement system in a single laboratory during the study period in order to minimize annual changes in measurement results. In clinical trials and epidemiological studies on cardiovascular diseases, lipids, such as TC, TG and HDL-C, are analysed as important risk factors. However, different measurement methods, reagents, primary standards, analytical instruments and quality control systems are selected in various laboratories, with the reproducibility and accuracy of measurements differing among them; therefore, compatibility-assured results are not always obtained. We considered ‘standardization’, in which the accuracy of measurements is standardized in order to assure compatibility in accordance with internationally accepted criteria when comparing values among studies or reviewing annual changes, to be a basic and important requirement for maintaining the homeostasis of research and performed it accordingly. Thus, the results of this study suggest the importance of internationally accepted standardization for assuring the compatibility of clinical trials at the international level.
Lipid standardization excluding TG
In large-scale clinical trials or epidemiological studies on the inhibitory effects of statins on cerebro- and cardiovascular events conducted in Western countries, risk factors are generally detected from primary and secondary preventive measures by assaying lipid concentrations and investigating the relationship between major events including cerebral haemorrhage, stroke and myocardial infarction.1–5 Since the risk factors vary depending on ethnicity, the duration of the study, types and doses of statins, duration of statin administration and the effects of statins on cerebro- and cardiovascular events typically show various patterns. We concluded that a demonstration of the high-level reliability of study results through the standardization of blood chemistry findings was necessary for J-STARS in order to receive affirmative approval for the J-STARS study results. 11 The central clinical laboratory of J-STARS, SRL, participated in the same lipid standardization programmes applied to reagent manufacturers conducted by the CDC and its international network, the CRMLN. The NCVC is an international member of the CRMLN, and the RMP for lipids at the NCVC have been standardized by the CDC since July 1992. The NCVC standardized all lipids measured in SRL, except for TG.
Although the standardization of blood chemistry results is generally only briefly or not described at all in clinical study reports, the standardization period (10 years) in this study was 2–3-fold longer than that of normal clinical studies. Fewer clinical studies in Japan were standardized through the CDC. Evidence from Asia, in which the prevalence of dyslipidaemia is generally lower than that in Western populations, has been limited. In order to obtain J-STARS results that were comparable with those of similar clinical studies performed in other countries, the establishment of reference values using the RMP was performed to ensure that the measured blood chemistry values were accurate and reliable. More than 40 clinical and epidemiological studies have participated in the WHO-CDC Cooperative Cholesterol and Triglycerides Standardization Program 22 and CDC-National Heart, Lung, and Blood Institute Lipid Standardization Program 23 conducted by the CDC and CRMLN. J-STARS study is also one of the participating clinical trials in the standardization conducted by the CDC and the CRMLN.
TG standardization
Since the RMP was not employed in the JMA survey, the accuracy of the adjusted means of samples reported in the JMA summary is unclear. The assayed values of each participating laboratory are evaluated based on the adjusted mean values obtained through statistical analyses of values from all participating laboratories. The adjusted mean is a consensus value instead of accuracy by the RMP. Concerns were expressed regarding whether the adjusted mean has high-level reliability for accuracy. In order to overcome this issue, TG samples of the JMA survey in 2013 were measured only once using GC-IDMS RMP at the end of the J-STARS study period and its values were compared to assess accuracy.19,20 In the average %bias of the three samples, the adjusted mean net TG value by JMA vs. GC-IDMS indicated −2.04% (Table 3). As a result, the adjusted mean as a consensus value was investigated where available because it met the CDC accuracy performance criteria for TG (≤5% [Tentative]) (Table 1). After the end of the J-STARS study, the measurement of TG using the JMA survey samples has been continued by the GC-IDMS RMP every year in 2014, 2015 and 2016. We are observing TG accuracy in JMA survey samples with the GC-IDMS RMP every year. Meanwhile, in the average %bias of three samples, the assayed net TG value by SRL vs. GC-IDMS was −2.46% (Table 3). Although the assayed net TG values by SRL were slightly lower than the target net TG values by GC-IDMS, they all met the performance criteria by CDC/CRMLN for TG accuracy (≤5% [tentative]) (Table 1). As a result, the assayed TG values by SRL provide reliable data in the J-STARS study.
hs-CRP standardization
No criteria are currently available for assessing the precision and accuracy of hs-CRP values. Furthermore, no reliable standard material for calibrations or RMP has been established.24,25 However, hs-CRP reference material, IRMM ERM-DA470, exists whereby values measured by several laboratories with high-level analytical techniques are evaluated as a highly reliable subject. This international reference subject is used for practical purposes as the primary reference material for the standardization of hs-CRP. Accuracy is ensured by the calibration of analytical instruments with standard serum originating from IRMM ERM-DA470. The AHA/CDC Scientific Statement 8 proposed a CV of <10% within a concentration range of 300–10,000 μg/L for the precision performance criteria (Table 1). Therefore, the precision target was set at this criterion in J-STARS. The precision of hs-CRP by SRL throughout the study period was within the acceptable criteria established by the AHA and CDC (Table 2). hs-CRP is a marker of low-grade inflammation and its presence indicates an increased risk of vascular disorders such as cardiovascular disease, arteriosclerosis, diabetes mellitus, obesity and hypertension.8–10 Serum lipid concentrations are also reported to be risk factors for these diseases. Therefore, lipid concentrations and hs-CRP were measured in J-STARS. The details of this substudy for hs-CRP will be reported elsewhere.
Conclusions
The results of the 10-year standardization for lipids and hs-CRP in J-STARS involving the administration of pravastatin suggest that the results of this clinical study are comparable with those of similar clinical trials performed in other countries. This also suggests the importance of the international standardization of measurements for a long period in a single laboratory in a clinical trial and markers as risk factors. Reliable results will contribute to the establishment of preventive measures against recurrent stroke in Japanese patients.
Footnotes
Acknowledgments
The authors would like to thank Hiroyasu Iso, M.D., Ph.D. at the Osaka University Graduate School of Medicine and Hubert W. Vesper, Ph.D. at the CDC for their reviews and comments. The authors also wish to thank Ms. Yukari Ichikawa for her excellent assistance in providing references and assisting with the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was initially supported by a grant from the Ministry of Health, Labour and Welfare, Japan. After governmental support expired, it was conducted in collaboration with Hiroshima University and the Foundation for Biomedical Research and Innovation.
Ethical approval
The ethics committee of the Hiroshima University Graduate School of Biomedical and Health Sciences approved the study. Ethical approval: 30002 for the main study (J-STARS) and 30003 for the substudy (hs-CRP).
Guarantor
MN.
Contributorship
All authors confirmed they have contributed to the intellectual content of this manuscript and have met the following three requirements: (a) significant contributions to the conception and design, acquisition of data or analysis and interpretation of data, (b) drafting or revising the article for intellectual content, and (c) final approval of the published article.
