Abstract
Neuroblastoma is the most common solid extracranial malignancy diagnosed in childhood. Clinical presentation is variable, and metastatic disease is common at diagnosis. Analyses of urinary catecholamines and their metabolites are commonly requested as a first-line investigation when clinical suspicion exists. Levodopa (L-Dopa) therapy is utilized as a treatment for a number of disorders in childhood, including Dopa-responsive dystonia. Neuroblastoma may mimic some of the clinical features of this disorder. L-Dopa can interfere with analysis of urinary catecholamines and their metabolites and complicate the interpretation of results. We present the cases of three children who were prescribed L-dopa at the time of analysis of urinary catecholamines and metabolites as a screen for neuroblastoma, but who did not have the disease. Comparison of their results with those from cases with true neuroblastoma reveal that it is impossible to reliably distinguish true neuroblastoma from L-Dopa therapy using these tests. We recommend that patients should be off L-dopa therapy, if possible when these tests are performed. These cases illustrate the importance of providing clinical details and drug history to the laboratory in order to avoid diagnostic confusion.
Introduction
Neuroblastoma is an embryonal tumour of the autonomic nervous system. It is the most common tumour diagnosed in the first year of life and the most common solid extracranial malignancy diagnosed in childhood. 1 Median age at diagnosis is 17 months, 2 and around half of cases present with metastases. 1 Tumours arise in tissues of the sympathetic nervous system, most commonly in the adrenal medulla, and often have the capacity to secrete catecholamines directly into the circulation. Clinical presentation is dependent on the site of the primary tumour and metastases. Abdominal pain, nausea, weight loss and anorexia may be present. Tumours along the spinal column can expand through the intraforaminal spaces and cause spinal cord compression with neurological symptoms. Neuroblastoma typically metastasizes to regional lymph nodes and to bone marrow via the haematopoietic system and can infiltrate cortical bone. A child with neuroblastoma may present with bone pain, limping or lower limb weakness. 1 Approximately 2% of patients present with opsoclonus and myoclonus, a paraneoplastic syndrome characterized by the presence of myoclonic jerking and random eye movements. These patients often have low-grade disease and a good long-term prognosis. 3 Unfortunately, neurological abnormalities can persist or progress and can be devastating. 4
Dopamine metabolism and therapeutic implications
Elevated concentrations of the urinary catecholamines noradrenaline (NA), adrenaline (AD) and dopamine (DA) and their metabolites vanillylmandelic acid (VMA), homovanillic acid (HVA), free metadrenaline (fMA), free normetadrenaline (fNMA) and free 3-methoxytyramine (f3MT) can be detected in urine.
Sulphate-conjugated metanephrines and catecholamines are formed from the free amines by the actions of the sulfotransferase isoenzyme, monoamine-preferring sulfotransferase (SULT1A3). SULT1A3 is found predominantly in the gastrointestinal tract, which is the source of most of the sulphate-conjugated catecholamines produced in the body. It seems likely that sulphate-conjugated metanephrines are formed predominantly in gastrointestinal tissue. 5 In assays of urinary metanephrines, free metanephrines represent a small proportion (<3%) of the total measured as free plus conjugated metanephrines 5 and are most commonly measured when screening for tumours arising from the sympathetic nervous system.
Abnormalities of the urinary catecholamine profile are frequently encountered in patients harbouring neuroblastoma, with over 90% of cases noted to have at least one analyte elevated above the reference interval in one series. 6 Urinary catecholamine metabolites are often requested as a first-line investigation when the diagnosis of neuroblastoma is clinically suspected. A number of drugs can interfere with urine catecholamine analysis and complicate interpretation of results, risking erroneous diagnosis. For example, low concentrations of paracetamol contamination can produce patterns of results which may be easily confused with those found in predominantly AD or MA producing phaeochromocytomas, 7 and tricyclic antidepressants can cause false-positive MA and NMA results in both plasma and urine. 8 Another drug with the potential to interfere with analysis is Levodopa (L-Dopa).
L-Dopa is the precursor to the neurotransmitters dopamine, NA and AD. L-Dopa is metabolized to dopamine by DOPA decarboxylase and requires vitamin B6 as a co-factor. In adults, L-Dopa is used to treat Parkinson’s disease, resulting from degeneration of dopaminergic neurons in the substantia nigra and manifesting with the classic triad of bradykinesia, rigidity and tremor. 9 L-Dopa crosses the blood–brain barrier, whereas dopamine does not; therefore, the former is used to increase dopamine concentrations in the central nervous system. An extra-cerebral dopa-decarboxylase inhibitor is commonly administered concomitantly, which reduces peripheral conversion of L-dopa to dopamine and limits systemic side-effects such as nausea and cardiovascular effects.
In children, L-Dopa is used to treat dopa-responsive dystonia. Also known as Segawa’s disease, this is a hereditary progressive movement disorder that manifests in early childhood and is caused by a mutation which disrupts the production of tetrahydrobiopterin, a co-factor in the production of endogenous dopamine. 10 Diagnosis is frequently delayed and children are often misdiagnosed as cerebral palsy or a neurodegenerative disorder. Response to L-dopa can be dramatic, with marked improvement in symptoms. 11 A number of other mutations have been described which result in tetrahydrobiopterin deficiency and hyperphenylalaninaemia. These disorders are treated with tetrahydrobiopterin replacement, diet, folinic acid supplementation and neurotransmitter replacement therapy with L-Dopa and 5-hydroxytryptamine. 12 L-dopa is also utilized in early-onset generalized dystonia, an autosomal dominant disorder which is characterized by involuntary muscle contractions beginning in the limbs, presenting most commonly in childhood or adolescence. Some rare inborn errors of dopamine metabolism (including juvenile Parkinsonism and spinocerebellar atropy type 3) may manifest as hypokinetic rigid syndrome, and respond to a trial of L-Dopa therapy. 13 L-Dopa therapy has also been trialled in other disorders in childhood, including restless leg syndrome, periodic limb movements in sleep and attention deficit hyperactivity disorder.14,15 The dopa-decarboxylase inhibitor Carbidopa is commonly used in paediatric cases, administered as a combined preparation with L-Dopa (Co-careldopa) in divided doses three to four times daily. Dosage is dependent on indication and is usually calculated per kilogram body weight and titrated to response.
Case presentations
We present three paediatric cases that were investigated to exclude neuroblastoma, but were undergoing L-Dopa therapy at the time of urine collection for catecholamines and metabolites (Figure 1). However, this relevant drug history was not conveyed to the laboratory. In all three cases, laboratory staff contacted the responsible clinician to request a drug history and communicated the results and potential for interference from L-Dopa therapy via telephone and laboratory report.
Concentrations of urinary VMA, fNMA, DA, f3MT and HVA from the three cases discussed in the text, and from 10 children aged between 1 and 10 years of age with true neuroblastoma (International Neuroblastoma Staging System stages 2–4). All analyses were undertaken using high-performance liquid chromatography with electrochemical detection at Crosshouse hospital, Kilmarnock, Scotland. Measurements are expressed as multiples of the age-related upper reference range value. The 10 cases of true neuroblastoma have been selected, as they exhibit normal or lower than anticipated VMA values at diagnosis (less than three times the age related upper reference interval).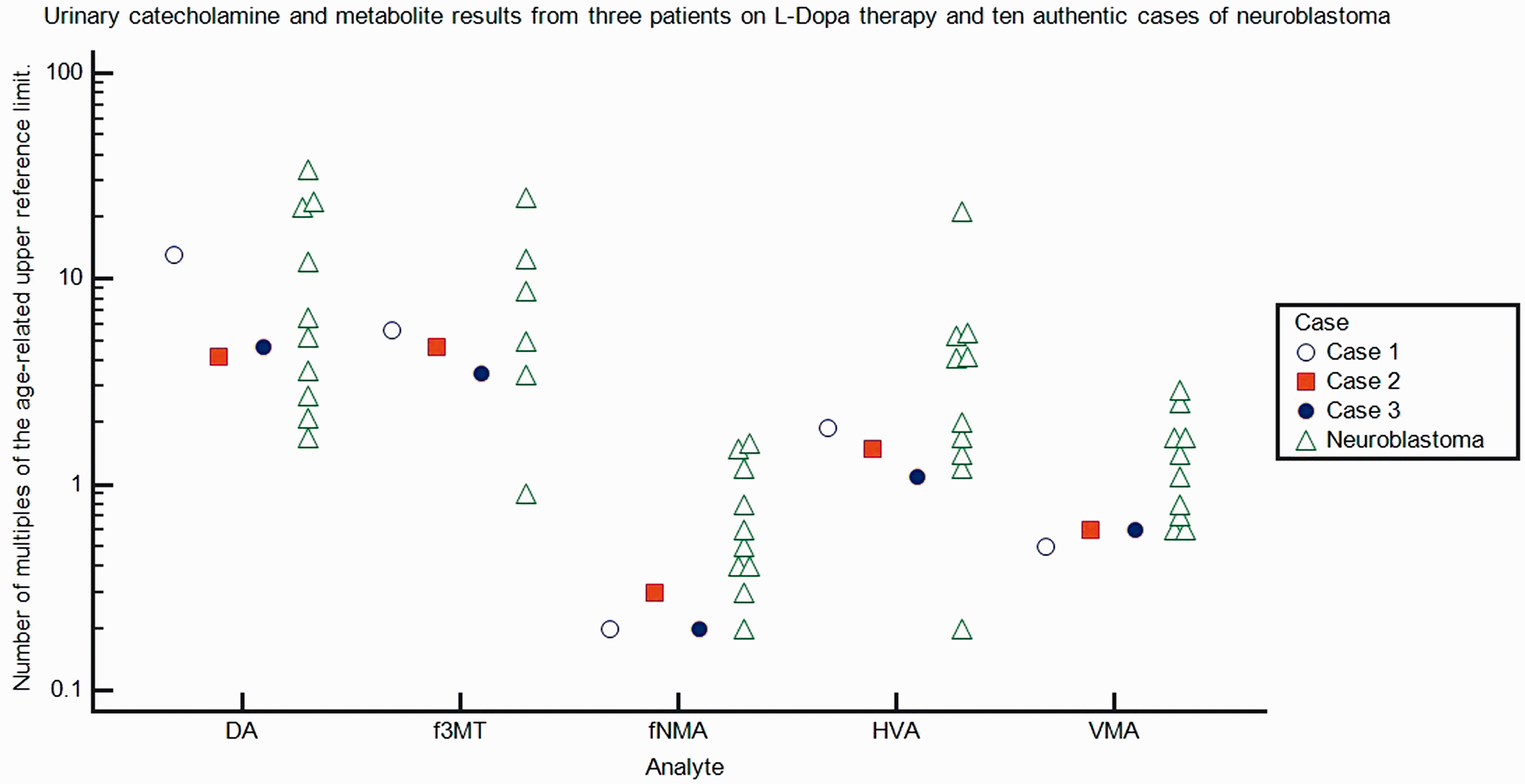
Case 1 is a child referred to neurology at the age of three years because of right leg pain, stiff gait and reluctance to walk. Examination revealed no bony tenderness and a full range of passive movement in the lower limbs. Routine blood tests, inflammatory marker analyses and MRI of the brain and spine as well as nerve conduction studies did not identify any abnormality. A provisional diagnosis of dopa-responsive dystonia was made and a trial of L-Dopa (as Co-careldopa) commenced. Lack of clinical improvement prompted analysis of urinary catecholamine metabolites to screen for skeletal neuroblastoma. Results noted elevated HVA and f3MT and marked elevation of DA. Three months later, the child developed a clinically apparent inflammatory arthropathy. Co-careldopa was stopped and steroid joint injections and methotrexate commenced with significant clinical improvement.
Case 2 is a baby presenting at four weeks of age. The child was noted to have daily episodes of twitching of the left leg associated with poor feeding and irritability. No abnormality was identified on routine blood tests (including plasma lactate) and CSF analysis. Urinary amino and organic acid analysis was carried out to look for a metabolic cause to explain the baby’s symptoms but did not reveal any abnormalities. EEG demonstrated normal wave patterns and MRI excluded structural abnormalities of the brain or spine. Analysis of the SLC2A1 gene (found in families with paroxysmal exertion-induced dyskinesia) showed no rearrangement. L-Dopa therapy (as Co-careldopa) was commenced for a potential dopa-responsive dystonia. The child was admitted to hospital at 17 months of age with vomiting and tachycardia, with a heart rate persistently above 160 bpm, prompting request for urinary catecholamine metabolites to screen for neuroblastoma, paraganglioma or phaeochromocytoma. The results showed significant elevations of DA, f3MT and HVA. The child’s condition improved spontaneously. L-Dopa was weaned at 2.5 years of age without ill effect, and a diagnosis of non-Dopa-responsive transient movement disorder was made.
Case 3 presented at the age of two months with recurrent episodes of multifocal polymyoclonus (myoclonus in several muscle groups simultaneously in rapid succession) lasting for many hours. Features were felt to be in keeping with an episodic fragmentary movement disorder. Fixed upward movements of the eyes and roving multiplanar eye movements were apparent during some attacks. EEG recordings were unremarkable and a number of biochemical and genetic tests failed to elicit a diagnosis. L-Dopa therapy was trialled with little effect. Urinary catecholamine metabolites were requested to exclude an occult neuroblastoma associated with opsoclonus myoclonus syndrome as a possible diagnosis. The results noted elevated HVA, fMA and f3MT and grossly elevated DA. Further investigation revealed a mutation in the TBC 1D24 gene, associated with familial infantile myoclonic epilepsy. The patient receives ongoing follow-up and treatment for multifocal polymyoclonus and chronic ataxia.
Discussion
Figure 2 illustrates the normal metabolism of tyrosine to catecholamines and their metabolites. Neuroblastomas produce the adrenal catecholamines NA, AD and DA. They are commonly characterized biochemically by elevated catecholamine metabolites, particularly VMA and HVA in urine. The proportion of neuroblastoma patients with elevated concentrations of urinary VMA and HVA varies with disease stage, with low-stage tumours less likely to exhibit abnormal levels.
16
The addition of urinary fNMA with either VMA or HVA significantly enhances the diagnostic sensitivity,
17
and inclusion of 3-methoxytyramine (f3MT) in the profile has been suggested to further increase sensitivity up to 100% in patients harbouring neuroblastoma.
18
In a study of 55 patients with neuroblastic tumours, HVA, f3MT, VMA and DA were the most frequently elevated analytes at the time of diagnosis, with HVA elevated in 54/55 and VMA in 45/53 cases.
19
Neuroblastoma has also been demonstrated to be the most common cause of elevated urinary DA in children, however it is a poorer marker than its metabolites in very young patients.
20
Metabolism of tyrosine to catecholamines and their metabolites.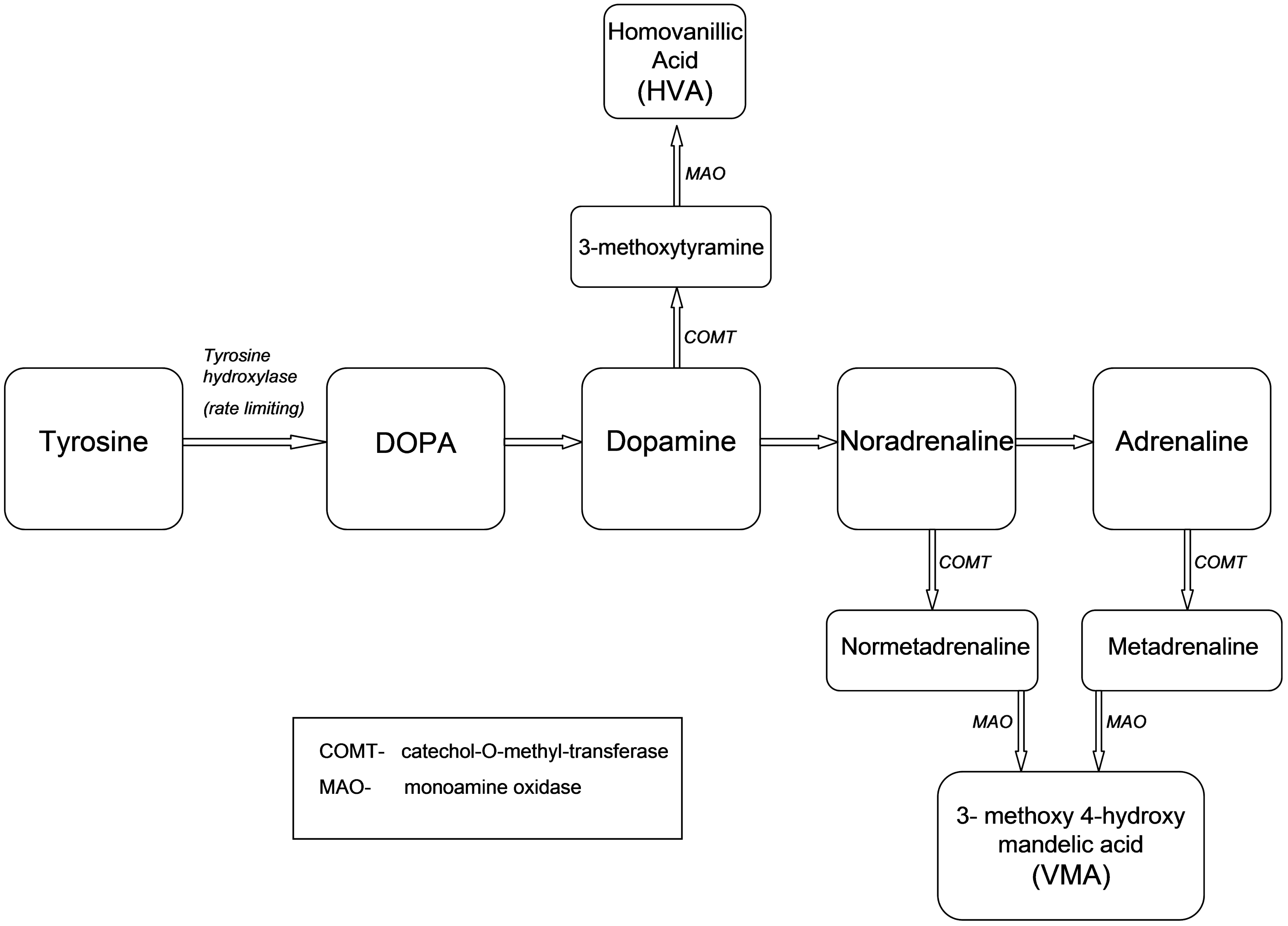
Neuroblastomas often exhibit high expression of tyrosine hydroxylase, which catalyses the conversion of tyrosine to DOPA. DOPA is metabolized to DA and then f3MT and VMA. 16 While NA and AD can be overproduced, the lack of storage granules in neuroblastic tumour cells has been hypothesized to explain the rapid conversion to the metabolite VMA, 21 which is frequently noted to be elevated in urine, whereas elevations of NA and AD are less common. 19
There is little published data on the effects of L-DOPA therapy on urine catecholamine excretion in children. Urinary excretion of DA and its major metabolite, HVA, is elevated in adult patients treated with L-dopa for Parkinson’s disease. In a study by Davidson et al., 22 elevations in both DA and HVA concentrations exhibited significant positive correlation with daily dose of L-dopa. Excretion of VMA, NA and AD were not affected. A further study of 20 patients with Parkinson’s disease on L-DOPA therapy 23 demonstrated a statistically significant 22- to 148-fold increase in plasma and urinary free and deconjugated 3MT and urinary DA in patients receiving L-DOPA when compared with the reference group. Urinary NA and urinary-free and deconjugated NMA concentrations were not elevated.
Signs and symptoms of neuroblastoma are varied and often vague. Limb pain, delay in walking and muscle myoclonus may be present both in neuromuscular disorders treated with L-Dopa and in the presence of occult neuroblastoma. Urine catecholamines are commonly requested as a first-line test when suspicion of neuroblastoma exists. The urinary catecholamines and metabolites commonly elevated due to a neuroblastic tumour (HVA, f3MT, VMA and DA 19 ) show significant cross-over with those likely to be elevated due to L-DOPA therapy (HVA, f3MT and DA 22 ) While it could be hypothesized that VMA might be utilized as a marker of neuroblastoma in these patients, significant elevations of VMA are not always seen in the urine of children who do harbour a neuroblastic tumour, such as in the 10 cases illustrated in Figure 1. Based on these results alone, it would be impossible to reliably distinguish a case of true neuroblastoma from that of a child on L-DOPA therapy. While a raised concentration of VMA may heighten concern regarding the presence of neuroblastoma in a child on L-DOPA in whom there is already clinical suspicion, it should not be regarded as a confirmatory test. Plasma metanephrines and imaging studies may provide further useful information.
Conclusion
Our data illustrate that the analysis of urinary catecholamines and their metabolites cannot reliably distinguish between patents on L-Dopa therapy and patients with neuroblastoma. When screening for neuroblastoma using these tests, patients should be off L-Dopa therapy if possible. While there is no published data regarding an optimum time frame, the authors suggest repeating analysis after one week off treatment. Many clinicians will not have detailed knowledge of the interpretation of urinary catecholamine results, and the cases presented illustrate the importance of providing full clinical details and drug history to the laboratory in order to avoid diagnostic confusion and unnecessary investigation.
Footnotes
Acknowledgements
We would like to thank Peter Galloway for his assistance and guidance.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Informed consent was obtained from the parents/guardians of all cases.
Guarantor
RS.
Contributorship
DFD and RS researched literature and conceived the study. DFD and ED collected data. Data interpretation was performed by DF D. AK wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version.
