Abstract
Background
Antiphospholipid syndrome is an autoimmune disease characterized by thrombosis, fetal losses and thrombocytopenia associated to antiphospholipid antibodies. They are directed to phospholipids, such as cardiolipins (anticardiolipin) and lupus anticoagulant or to complexes formed by phospholipids and protein cofactors, such as β2 glycoprotein 1 (a-β2GP1) and annexin V (a-annexin V). These auto-antibodies may be considered as a family of antibodies involved in thrombotic events and antiphospholipid activity. On the other hand, some proangiogenic factors are involved in the normal development of placental vasculature, such as the vascular endothelial growth factor. Overexpression of vascular endothelial growth factor receptor in its soluble form (sVEGFR-1) has been associated to a higher antiangiogenic activity. Our aim was to analyse the association between anticardiolipin, lupus anticoagulant, a-β2GP1, a-annexin V and sVEGFR-1 with recurrent miscarriage before week 10 of gestation in females with antiphospholipid syndrome.
Methods
We studied 24 females (primary or secondary antiphospholipid syndrome), who were divided into two groups: females with recurrent miscarriage before week 10 of gestation (M; n = 12) and females with no history of fetal loss (NM; n = 12). Anticardiolipin, a-β2GP1, a-annexin V and sVEGF-R1 concentrations were assessed by ELISA, while lupus anticoagulant was assessed by screening and confirmatory tests.
Results
A significant association was observed between the number of positive biomarkers and the belonging group (P < 0.05). Besides, a positive result for lupus anticoagulant and a-β2GP1 was found to be significantly associated to the M group (P < 0.05).
Conclusions
Lupus anticoagulant and a-β2GP1 may be implicated in pregnancies complicated by recurrent miscarriage in females with antiphospholipid syndrome.
Introduction
Antiphospholipid (APL) antibodies (Abs) constitute a heterogeneous group of Abs that can appear associated to autoimmune diseases, infectious diseases, cancer and some lupus-inducing drugs. The probability of miscarriage in patients bearing these Abs, without treatment, is of 91%, 1 and those pregnancies that do not have an abortion outcome, have a higher risk of preeclampsia, low weight of the newly born, prematurity and fetal distress. 2 Although placental thrombotic events may play an important role, 3 it is nowadays assumed that non-thrombotic mechanisms would be the main cause for the defective placentation observed in patients with antiphospholipid syndrome (APS).4–6 APS is an autoimmune disease characterized by arterial/venous thrombosis and/or obstetric complications, associated to the persistent presence of anticardiolipins (ACA) and/or lupus anticoagulant (LA) and/or anti-β2 glycoprotein 1(a-β2GP1). 7 APS is either a primary disease or it can be associated to another autoimmune disease, such as systemic lupus erythematosus (SLE). For the diagnosis of APS, it is required: the presence of moderate/high levels of ACA; the presence of LA; the presence of a-β2GP1 higher than percentile 99, together with a clinical event of vascular thrombosis or pregnancy complications. APS is present if at least one of the clinical criteria and one of the laboratory criteria (on 2 or more occasions, at least 12 weeks apart) are met.8–10
In 1990, it was reported that the main antigenic target of APL Abs is β2GP1; consequently, the study of this protein in the pathogenesis of APS became of great interest for the scientific community. 11 Among the known cofactors, β2GP1 is the most studied, since it would play an anticoagulant role in vivo; the presence of Abs against this molecule would interfere with its normal physiological function. β2GP1 is a highly glycosylated polypeptidic chain with five domains: domain I reacts with APL Abs, while Abs associated to non-APS diseases are directed to domains II to V. The site of interaction of β2GP1 with negatively charged phospholipids, such as cardiolipins, phosphatidylserine and phosphatidylinositol, is located in domain V. Domain I is normally hidden and it is exposed when in contact with membrane phospholipids. When APL Abs bind to endothelial cells forming complexes with β2GP1, they induce the activation of such cells, and therefore, the initiation of the coagulation process. 12 Evidence also exists for APL Abs to promote platelet activation and aggregation via binding of a-β2GP1 complexes to glycoprotein Ib on platelet surface. Abs that recognize a cryptic epitope (Gly40-Arg43) in domain I of β2GP1 are closely related to APL and strongly correlate with thrombosis and obstetric complications. β2GP1-dependent APL Abs can recognize their own antigen on the trophoblast and decidual cells. It has been suggested that cellular functions of the trophoblast would be altered, thus leading to defective placentation and playing a crucial pathogenic role in obstetric complications in the APS, by non-thrombotic mechanisms.5,13–15 At the same time, the formation of immune complexes a-β2GP1 can activate the classical pathway of complement system and induce localized immune damage. 16 It has been demonstrated in vitro that binding of a-β2GP1 to the syncytiotrophoblast affects its invasive capacity 17 and decreases human chorionic gonadotrophin synthesis.18,19
Another cofactor studied in APS is annexin V. Annexins are glycoproteins that bind phospholipids in a calcium-dependent manner. They present a wide range of biological actions, including an anticoagulant effect. This effect would be explained by their ability to sequester calcium from the phospholipid matrix, where it interacts with proteins of the coagulation cascade. Then it would form a protective cover that would isolate the apical surface of placental villi from circulating coagulation proteins. 20 It has been postulated that displacement of annexin V from the cellular surface by a-annexin V Abs found in the serum samples of patients with APS, would be responsible for the generation of a thrombogenic environment and the consequent fetal loss. 21 On the other hand, the proper development of placental vasculature requires the action of proangiogenic factors, such as vascular endothelial growth factor (VEGF), a factor involved in vasculogenesis and angiogenesis. In the angiogenesis, VEGF stimulates the survival, proliferation and motility of endothelial cells, thus initiating the genesis of a new capillary. This factor is found either soluble in plasma or bound to heparin sulphate, depending on the isoform. The surface receptors of VEGF (VEGFRs1, 2 and 3) are expressed mainly in endothelial cells and placenta. 22 Different studies have clearly demonstrated that receptors 1 and 2 are involved in capillary formation and vascular development during embryogenesis. 23 The soluble isoform of VEGF R1 (sVEGF R1) retains the ability to bind VEGF and forms inactive homo- and heterodimers, thus impeding its binding to the cell surface receptor and therefore acting as an inhibitor. 24
Available data regarding the role of ACA, LA, a-β2GP1, a-annexin V and sVEGF R1 in the recurrent miscarriage in females with APS are scarce. Besides, if a-β2GP1 were found to be involved in recurrent miscarriage before week 10 of gestation, it would imply the additional utilization of anti-inflammatory and immunosuppressor drugs in such patients. Therefore, the aim of the present study was to analyse the association of ACA, LA, a-β2GP1, a-annexin V and sVEGF R1 with miscarriage before week 10 of gestation in females with APS.
Methods
Patients
A retrospective study was performed. Twenty-four female patients with an established diagnosis of APS (either primary or secondary to SLE), according to the International Consensus 2006 2 5 were studied. The APS clinical criterion present in all patients was vascular thrombosis. Twelve of these patients also presented three or more consecutive unexplainable miscarriages before week 10 of gestation, having already excluded maternal anatomical or hormonal abnormalities, as well as chromosomal alterations in both parents (group M). The other 12 patients, who had at least two previous normal pregnancies and no history of obstetric complications constituted the control group (NM). Patients were classified for SLE according to the criteria of the American College of Rheumatology 1997. 26 Patients were derived from the Rheumatology and Systemic Autoimmune Diseases Section of the Hospital J. B. Iturraspe of Santa Fe. Samples used throughout the study were obtained during the first trimester of pregnancy. The study was approved by the Bioethics Committee of Facultad de Ciencias Bioquímicas y Farmacéuticas (Universidad Nacional de Rosario), and all participants signed the corresponding informed consent.
Determination of ACA (immunoglobulin M and immunoglobulin G)
Serum samples were collected and ACA were determined by an ‘in house’ ELISA method using cardiolipin from bovine heart as antigen (Sigma, St. Louis, USA). Results are expressed as UMPL/ml and UGPL/ml for M and G isotypes, respectively. The reference (normal) value obtained in our laboratory was < 10 (either for IgM or IgG) and was considered negative. The positive values were classified as: low titre, 10 to 40; moderate titre, 40 to 80; high titre, >80. We considered as positive criterion the finding of values in the high and moderate ranges. 25
Determination of total a-β2GP1 (immunoglobulin G, immunoglobulin A and immunoglobulin M)
Serum samples were collected and total a-β2GP1 was assessed by a quantitative ELISA (ORG 521S Anti-beta-2-Glycoprotein I Screen, Orgentec Diagnostika GmbH Mainz – Germany). The cut-off value for our laboratory was 10 U/ml. This value was obtained as a result of media ± 3 SD, from serum samples of 50 healthy subjects.
Determination of a-annexin V (immunoglobulin G and immunoglobulin M)
Serum samples were collected and determination of a-annexin V Ab was performed by means of an ‘in house’ ELISA, utilizing annexin V form human placenta as antigen (Sigma, St. Louis, USA). The reference interval for our laboratory was 0 to 6 U/ml. 27
Detection of LA
Study of LA was performed in patients not receiving oral anticoagulants or heparin. Samples were collected with sodium citrate as anticoagulant and then, subjected to double centrifugation in order to obtain platelet-poor plasma. Protocol for LA study followed the criteria of the International Society on Thrombosis and Haemostasis 2009. As screening tests, we used aPTT Actin FSL (Siemens, Munich, Germany), PTT-LA (Stago, Asnières-sur-Seine, France), dilute Russell's viper venom time dRVVT (Stago, Asnières-sur-Seine, France) and mixtures with normal plasma for the determination of correction or no correction of altered tests. In the case of positive screening tests, we performed confirmatory tests using reagents with high concentrations of phospholipids as the test of neutralization with hexagonal phospholipids Staclot-LA (Stago, Asnières-sur-Seine, France).
Detection of sVEGF R1
Samples were collected with EDTA as anticoagulant. Detection of sVEGF R1 was performed using a quantitative ELISA (R&D Systems, Minneapolis, MN, USA). The cut-off value for our laboratory was 123 pg/mL. This value was obtained as a result of media ± 3 SD, from serum samples of 50 non-pregnant healthy subjects. The cut-off value for the first trimester of pregnancy was <1453 pg/mL, and it was obtained by processing 30 plasma samples of pregnant females with no associated disease and media ± 3 SD was calculated.
Statistical analysis
Statistical analysis was performed using the Chi square test, the Score exact test or the Fisher exact test and odds ratio (OR) estimates and their 95% confidence intervals (CI), depending on the case. A P value < 0.05 was considered as statistically significant.
Results
APS is an autoimmune disease characterized by thrombosis, fetal losses and thrombocytopenia associated to APL Abs. The aim of this study was to analyse the association between ACA, LA, a-β2GP1, a-annexin V and sVEGFR-1 with recurrent miscarriage before week 10 of gestation in females with APS.
Relative value of each individual assay in both groups of patients studied.
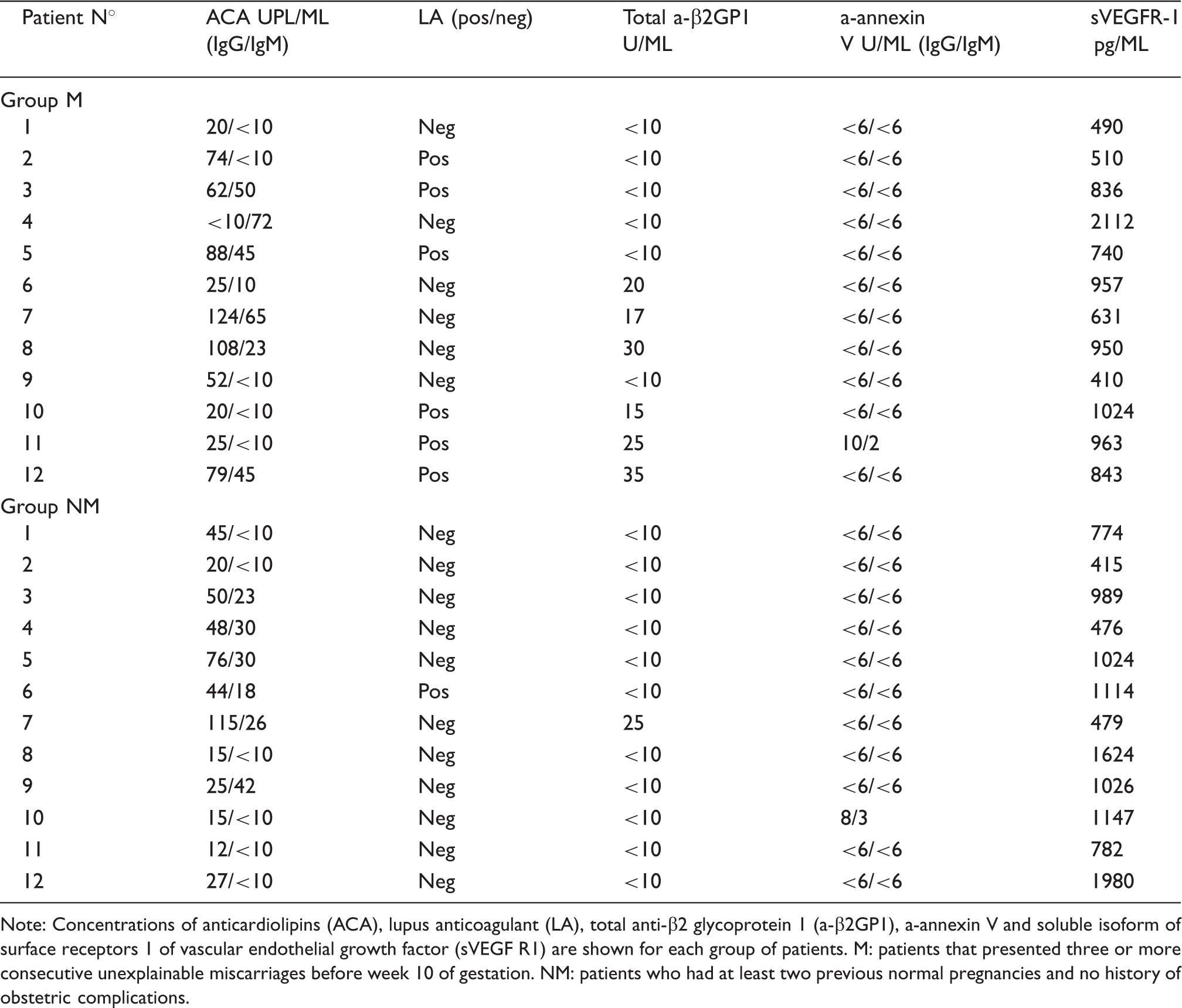
Note: Concentrations of anticardiolipins (ACA), lupus anticoagulant (LA), total anti-β2 glycoprotein 1 (a-β2GP1), a-annexin V and soluble isoform of surface receptors 1 of vascular endothelial growth factor (sVEGF R1) are shown for each group of patients. M: patients that presented three or more consecutive unexplainable miscarriages before week 10 of gestation. NM: patients who had at least two previous normal pregnancies and no history of obstetric complications.
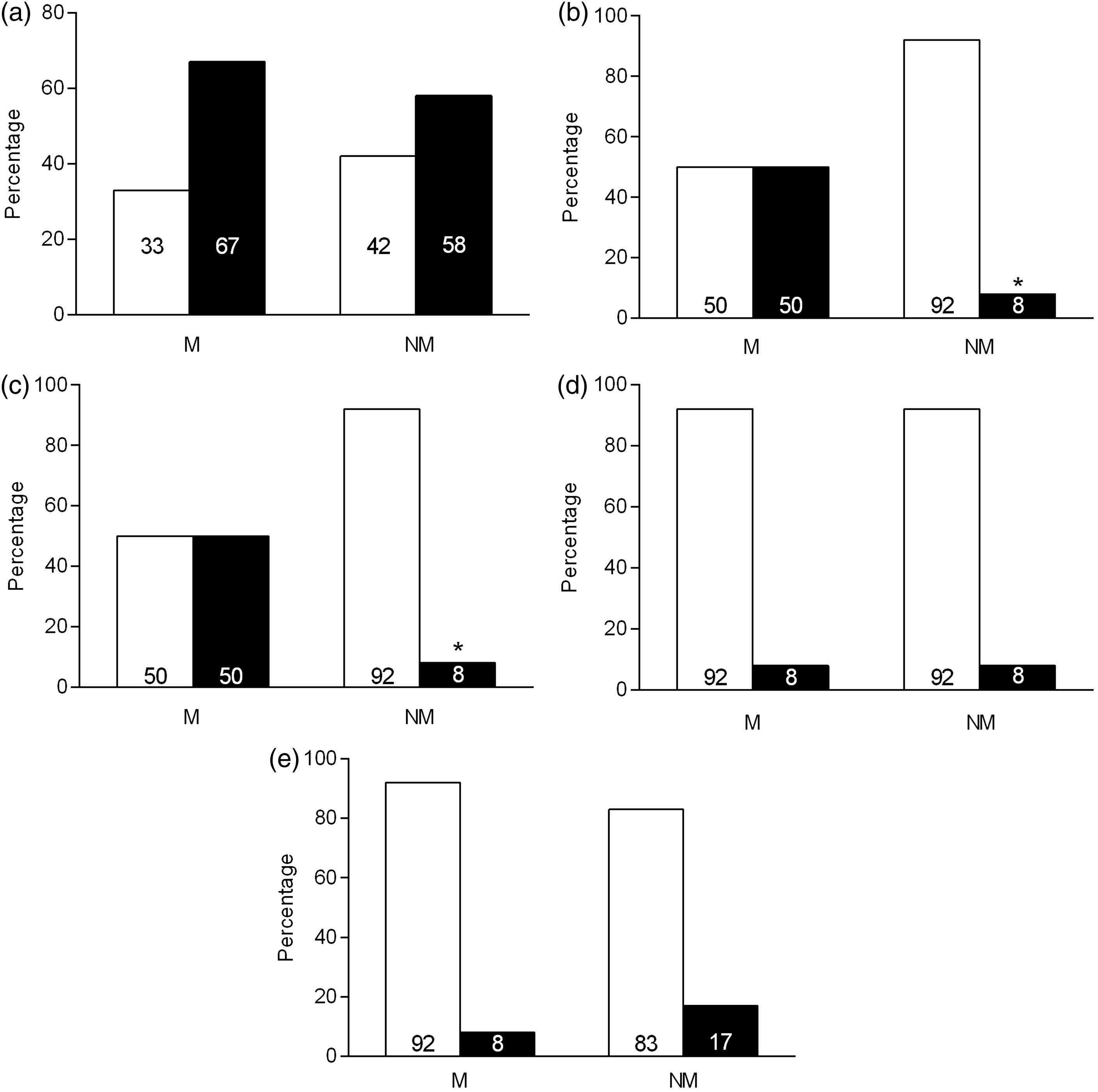
Concentrations of anticardiolipin (ACA), lupus anticoagulant (LA), anti-β2 glycoprotein 1 (a-β2GP1), antiannexin V (a-annexin V) and soluble vascular endothelial growth factor receptor 1 (sVEGF R1) in females with antiphospholipid syndrome (APS). The following parameters were evaluated in females with APS with recurrent miscarriage before week 10 of gestation (M; n = 12) and with no history of fetal loss (NM; n = 12). (a) ACA; (b) LA; (c) a-β2GP1; (d) a-annexin V and (e) sVEGFR-1. Black and white bars represent the percentage of positive and negative results for each parameter, respectively. ACA were considered positive for titres > 40. *P < 0.05 vs. M.
Number of positive biomarkers in each of the groups of patients studied.
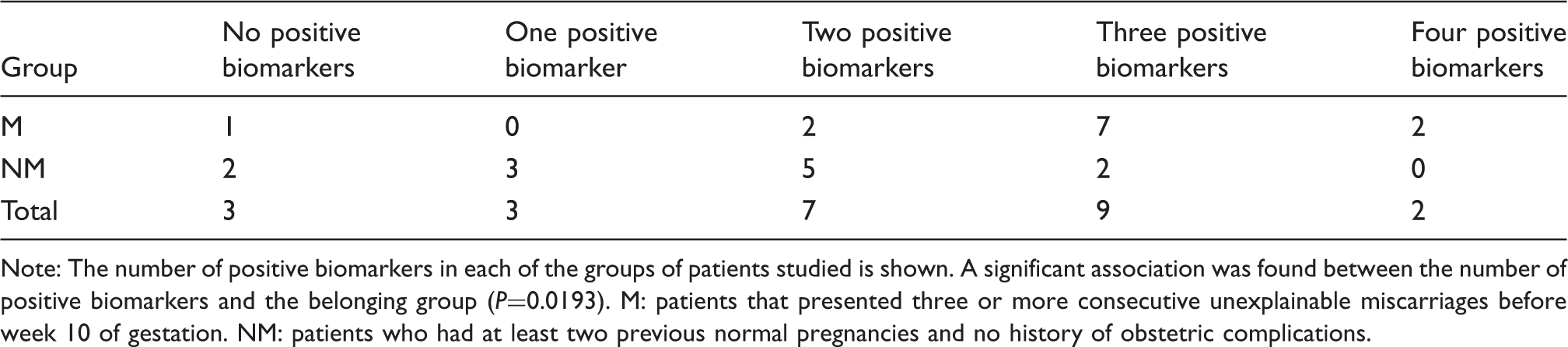
Note: The number of positive biomarkers in each of the groups of patients studied is shown. A significant association was found between the number of positive biomarkers and the belonging group (P=0.0193). M: patients that presented three or more consecutive unexplainable miscarriages before week 10 of gestation. NM: patients who had at least two previous normal pregnancies and no history of obstetric complications.
Odds ratios (OR) estimates for lupus anticoagulant (LA) and anti-β2 glycoprotein 1(a-β2GP1).

Note: OR and their 95% confidence intervals (CI) were evaluated for the variables LA and a-β2GP1, which were significantly associated to the belonging group.
Discussion
The mechanism of fetal loss related to the presence of APL Abs is poorly understood yet. Although these Abs are directly involved in thrombosis leading to placental infarction and fetal death, nowadays other non-thrombotic a-β2GP1-mediated mechanisms have been postulated. The non-thrombotic hypothesis suggests that binding of these Abs to β2GP1 on the trophoblastic membrane would perturb these membranes and alter placentation.
Decrease in trophoblastic proliferation and growth, and defective placentation, both induce a proinflammatory phenotype interfering with physiological implantation. 18 In accordance with our results, Sailer et al. 28 reported an association between pregnancy loss and the presence of LA and a-β2GP1 Ab. On the other hand, Arnold et al. 29 found no significant differences in a-β2GP1 Abs between females with recurrent miscarriage with or without APS. Other authors 30 observed that the simultaneous presence of ACA, LA and a-β2GP1 Abs confers a higher risk of developing severe manifestations of APS. Our results particularly associate the presence of LA and/or a-β2GP1 Abs, but not that of ACA, with recurrent miscarriage. According to Meroni et al., 7 patients with APS should be divided into four categories: category I includes females with more than one positive test in any combination and category II includes those females with only one positive test (IIa: positive LA; IIb: positive ACA and IIc: positive a-β2GP1 Abs). Triple positivity constitutes the most predictive profile of clinical manifestations and recurrences despite conventional treatment. Our results show a similar behaviour, since group M bears a higher number of positive biomarkers than group NM. However, some females with persistent elevated concentrations of ACA and LA and history of thrombotic events do not have fetal complications, while in some cases, females without ACA or LA and without a known cause, undergo recurrent miscarriage. Some authors postulate that a-annexin V Abs would be involved in reproductive failures. 31 Our results are coincident with those of Alijotas-Reig et al., 32 who reported that a-annexin V Abs should not be considered as a risk factor for miscarriage and inexplicable fetal death. In line with this, Bizzaro et al. 33 conclude that a-annexin V Abs determined in healthy females do not bear a predictive value on involuntary abortion and that they are not useful to evaluate the risk at the beginning of pregnancy. On the other hand, Galli et al. 34 demonstrated that a-annexin V IgG but not those of IgM type are related to fetal loss. Although annexin V concentrations are decreased in the placenta of females with miscarriage and APL Abs, and although a-annexin V Abs could interfere with the normal function of the syncytiotrophoblast, our results are not those expected according to the physiology of annexin V and the pathophysiology of a-annexin V Abs. Lastly, different studies have demonstrated that placental expression and maternal circulating levels of sVEGFR-1 are elevated in preeclampsia. In vitro experiments showed that sVEGFR-1 induces vasoconstriction and endothelial dysfunction; therefore, this protein would be involved in the interaction between decreased placental perfusion and deep maternal endothelial function alteration. In light of these findings, 35 we investigated the likely association between this parameter and recurrent fetal losses in APS. Our findings in the sample studied, would however, discard a role for sVEGFR-1 in the recurrent miscarriage in females with APS.
Conclusion
Traditionally, ACA and LA have been considered serologic markers of choice in the classification of APS, and a-β2GP1 Abs has been recently incorporated as a laboratory criterion. We conclude that LA and a-β2GP1 Ab would be good indicators of miscarriage risk in females with APS, thus reinforcing the role of a-β2GP1, not only in the diagnosis but also as a likely marker of recurrent miscarriage in that disease. The optimal treatment in patients with APS is controversial, and it is under continuous revision due to the fact that the low number of patients makes it difficult to perform adequate prospective studies that allow drawing definitive conclusions. Although the association between the presence of APL Ab and thrombosis seems clear, the therapeutic attitude should not be primarily directed towards elimination or reduction of the levels of these Abs through plasmapheresis, high doses of intravenous immunoglobulins and immunosuppressors (except in catastrophic APS), since a clear correlation does not exist between Ab titres and thrombotic episodes. Treatment of these patients is based on the use of platelets antiaggregants and anticoagulants aimed to prevent placental thrombotic events that would ultimately lead to miscarriage. Nevertheless, the participation of β2GP1-dependent non-thrombotic events that would be responsible for the defective placentation observed in APS,4–6 would lead to reformulation of the treatment currently applied to these patients. It is worth noting that, although the number of patients included in the present study may seem somewhat low, this is due to the fact that we have been very strict as regard the inclusion criteria applied, thus partly explaining the amplitude of the confidence interval obtained. Recruitment of patients with APS and no history of obstetric complications (NM group), and with APS undergoing fetal losses before week 10 (M group), made it difficult to gather a larger group of patients.
Further work investigating the effect of anti-inflammatory and immunosuppressor therapy in patients with recurrent miscarriage before week 10 of gestation and persistent positive a-β2GP1 will be required for considering these drugs as an alternative therapeutic strategy besides the conventional anticoagulant therapy.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was financially supported by Universidad Nacional de Rosario (UNR), Argentina.
Ethical approval
The study was approved by the Bioethics Committee of Facultad de Ciencias Bioquímicas y Farmacéuticas (Universidad Nacional de Rosario). Res N 076/2008. All participants signed the corresponding informed consent.
Guarantor
SMMA.
Contributorship
HFP performed most assays and analysis of results, and wrote the paper. EP and CB performed some assays and collaborated in the analysis of results. JM performed patients selection for the study and aided in medical matters. MB, MJS and SMD performed sample collection and conditioning, as well as some laboratory determinations. HB performed statistical analysis. SMMA coordinated and supervised the whole study.
