Abstract
Background
Dietary habits are associated with obesity which is a risk factor for coronary heart disease. The objective is to estimate the change of lipoprotein(a) and other lipoprotein classes by calorie restriction with obesity index and Framingham risk score.
Methods
Sixty females (56 ± 9 years) were recruited. Their caloric intakes were reduced during the six-month period, and the calorie from fat was not more than 30%. Lipoprotein profiles were estimated at baseline and after the six-month period of calorie restriction. Cholesterol levels in six lipoprotein classes (HDL, LDL, IDL, VLDL, chylomicron and lipoprotein(a)) were analysed by anion-exchange liquid chromatography. The other tests were analysed by general methods. Additionally, Framingham risk score for predicting 10-year coronary heart disease risk was calculated.
Results
Body mass index, waist circumference, insulin resistance, Framingham risk score, total cholesterol, LDL-cholesterol and IDL-cholesterol were significantly decreased by the calorie restriction, and the protein and cholesterol levels of lipoprotein(a) were significantly increased. The change of body mass index was significantly correlated with those of TC, VLDL-cholesterol and chylomicron-cholesterol, and that of waist circumference was significantly correlated with that of chylomicron-cholesterol. The change of Framingham risk score was significantly correlated with the change of IDL-C.
Conclusion
Obesity indexes and Framingham risk score were reduced by the dietary modification. Lipoprotein profile was improved with the reduction of obesity indexes, but lipoprotein(a) was increased. The changes of obesity indexes and Framingham risk score were related with those of triglyceride-rich lipoproteins, e.g. IDL, VLDL and CM.
Introduction
Numerous clinical studies indicated that increased LDL cholesterol (LDL-C) and triglyceride (TG) and decreased HDL cholesterol (HDL-C) were risk factors for coronary heart disease (CHD).1–5 Recently, several clinical studies showed that non-HDL-C has more predictive potential to estimate CHD risk than LDL-C.6–8 The guideline for National Cholesterol Education Program (NCEP)-Adult Treatment Panel-III indicates that LDL-C, HDL-C and TG are important targets for primary and secondary prevention of CHD, and non-HDL-C is an established secondary therapeutic target for CHD prevention. 9 The non-HDL contains LDL and TG-rich-lipoproteins, i.e. IDL, VLDL and chylomicron (CM). The VLDL cholesterol (VLDL-C) was indicated to be associated with CHD risk in Framingham Heart Study, independently of LDL-C. 10 The association between IDL-C and the severity of CHD was shown. 11
Body weight (BW), body mass index (BMI) and waist circumference (WC) are reduced by the change of lifestyle, diet and exercise.12–14 The lifestyle change improves lipid and lipoprotein profiles.12–14 BMI and WC of type 2 diabetic patients with insulin resistance (HOMA) are higher than those with insulin sensitivity, and the insulin-resistant patients have decreased HDL-C and increased VLDL-C. 15 We thought that BMI, WC, HOMA, LDL-C, TG-rich lipoproteins are decreased, and HDL-C is increased with decrease of CHD risk by improvement of dietary composition. Increased lipoprotein(a) (Lp[a]) concentration is associated with the risk of CHD.16–18 Diabetes was reported to increase the risk of atherosclerotic diseases, e.g. CHD, intermittent claudication. 19 However, it has been reported that Lp(a) concentration was associated inversely with the risk of type 2 diabetes. 20 Therefore, the change of Lp(a) concentration with the improvement of dietary composition could not be expected. Furthermore, in most of the studies, the Lp(a) protein concentration is estimated, and there are only a few studies that include the estimation of Lp(a) cholesterol.
HDL, LDL, IDL, VLDL, CM and Lp(a) in serum can be separated by anion-exchange chromatography (AEX-HPLC).21,22 Lipoprotein profile can be accurately and conveniently estimated by AEX-HPLC. We studied the changes of cholesterol concentrations in Lp(a) and other lipoprotein classes obtained by AEX-HPLC in females with the dietary modification by calorie restriction, and the relationship between the change of BMI, WC, HOMA and Framingham risk score (FRS) and the change of the lipoprotein profiles.
Methods
Subjects and study protocol
Sixty Japanese females (age: 56 ± 9 years, postmenopausal females;
All volunteers attended nutrition education session and individual guidance during the six-month period, which were provided once a week for the first one month and once two weeks after that. In the education session, we showed the target of dietary composition as follows: caloric intake from diet is 1600 kcal/day, and the percentages of calories from protein, fat and carbohydrate are 13–20%, 20–30% and 55–65%, respectively. The target of the caloric intake was reduced 16% from the caloric intake 1900 kcal/day of females (50–69 years) reported in 2015 by Japanese Ministry of Health, Labour and Welfare (JMHLF). The percentages of calories were also according to the dietary reference intake of females (50–69 years) reported in 2015 by JMHLF. In the individual guidances, the records of eaten foods in each day by themselves were checked. The check points were reduction of caloric intake from diet and percentages of calories from fat as follows: the caloric intake <1600 kcal/day, <1800 kcal/day, <1900 kcal/day, <2000 kcal/day and ≥2000 kcal/day in the start point of study are reduced by >0%, ≥5%, ≥10%, ≥15% and ≥20%, respectively, and the percentages of calories from fat is not more than 30%. All volunteers could meet the demand of the check points during the study period, and did not drop out.
Comparison between baseline diet composition and diet composition after six months of lifestyle intervention in females.
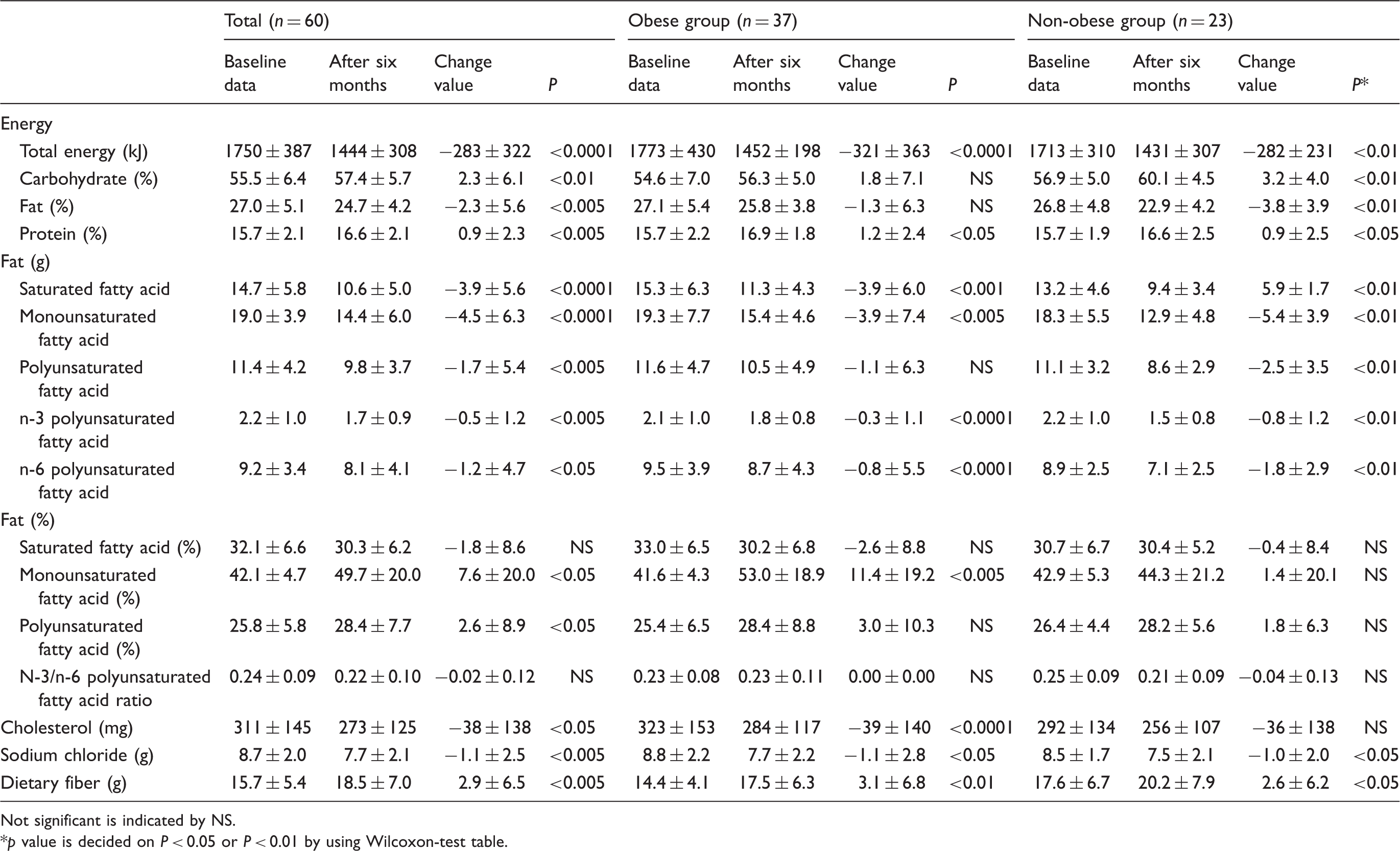
Not significant is indicated by NS. *
Measurement
GA08 (A and T Corporation), HLC-723G9 (Tosoh Corporation), Chemilumi-Insulin-Centaur (Siemens Healthcare Diagnostics) and Cholestest-CHO, Cholestest-NHDL and Cholestest-TG(Sekisui Medical), and Lp(a)-latex and sd-LDL-C (Denka Seiken) were used to measure fasting blood glucose (FBG), glycated haemoglobin A1c (A1c), Insulin, total cholesterol (TC), HDL-C, TG, Lp(a) protein (Lp(a)-P) and small dense LDL cholesterol (sdLDL-C), respectively. LDL-C was calculated by Friedewald equation (LDL-C = TC−[HDL-C]−[TG÷5]). Non-HDL-C was calculated using the equation: (non-HDL-C = TC−[HDL-C]). Insulin resistance (HOMA) was calculated by insulin and glucose concentrations in fasting blood: HOMA = insulin concentration × glucose concentration÷405. 23 FRS for predicting 10-year CHD risk was calculated by using gender, age, LDL-C, HDL-C, blood pressure, diabetes and smoking details. 24
Cholesterol concentrations of six lipoprotein classes (HDL, LDL, IDL, VLDL, CM, and Lp[a]) were measured by using AEX-HPLC, 22 which consists of a column filled with diethylaminoethyl-ligand non-porous gel, two eluents for step gradient of sodium percorolate, and a reagent containing cholesterol esterase and cholesterol oxidase for postcolumn reaction.
Statistical analysis
The data were presented as mean ± standard deviation (SD). Wilcoxon test was used to compare between baseline data and that after six months. Non-obese group was small (
Results
Comparison between baseline data and data after six months of lifestyle intervention in females.
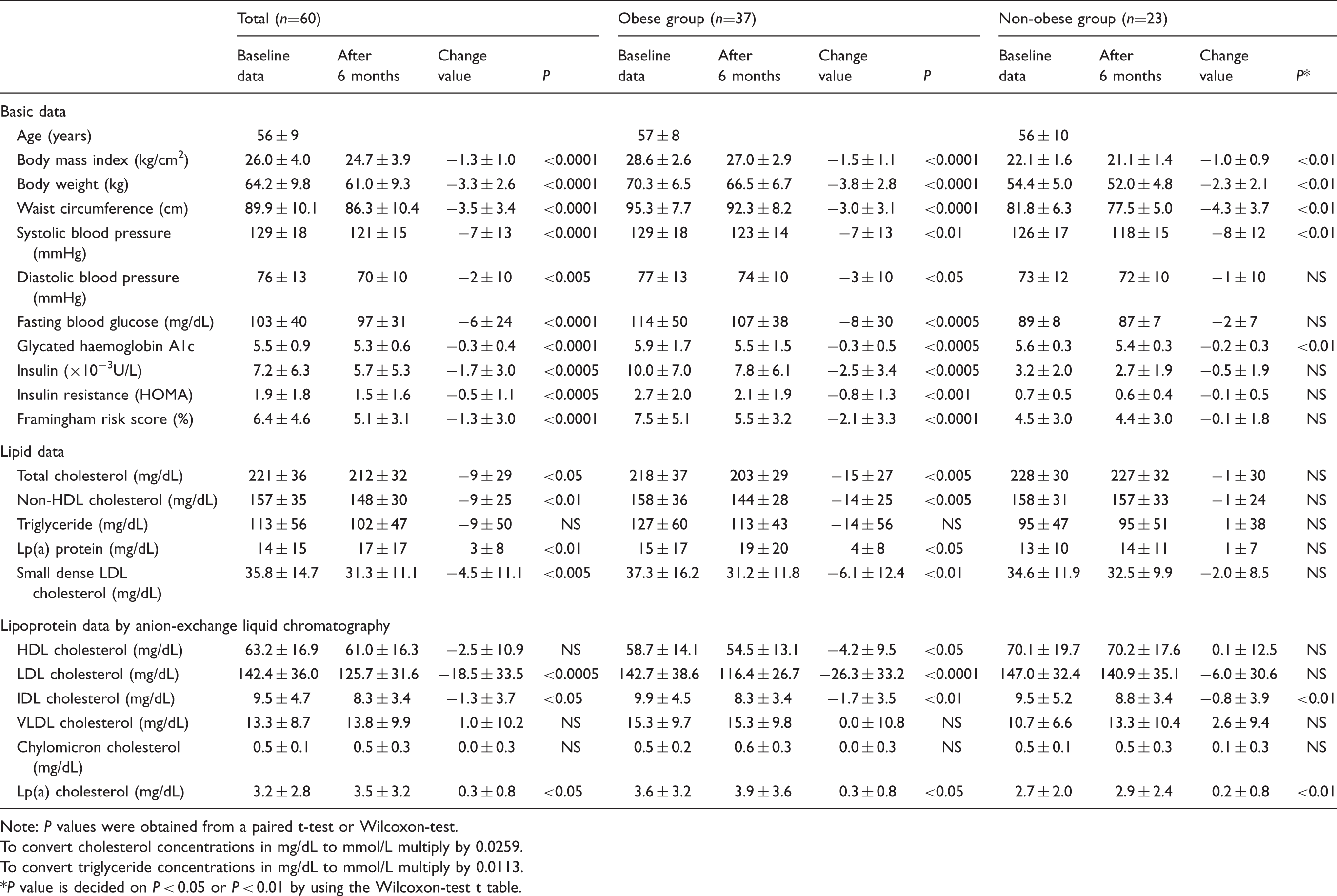
Note:
To convert cholesterol concentrations in mg/dL to mmol/L multiply by 0.0259.
To convert triglyceride concentrations in mg/dL to mmol/L multiply by 0.0113.
Comparison between changes in basic data and lipid profile in females.
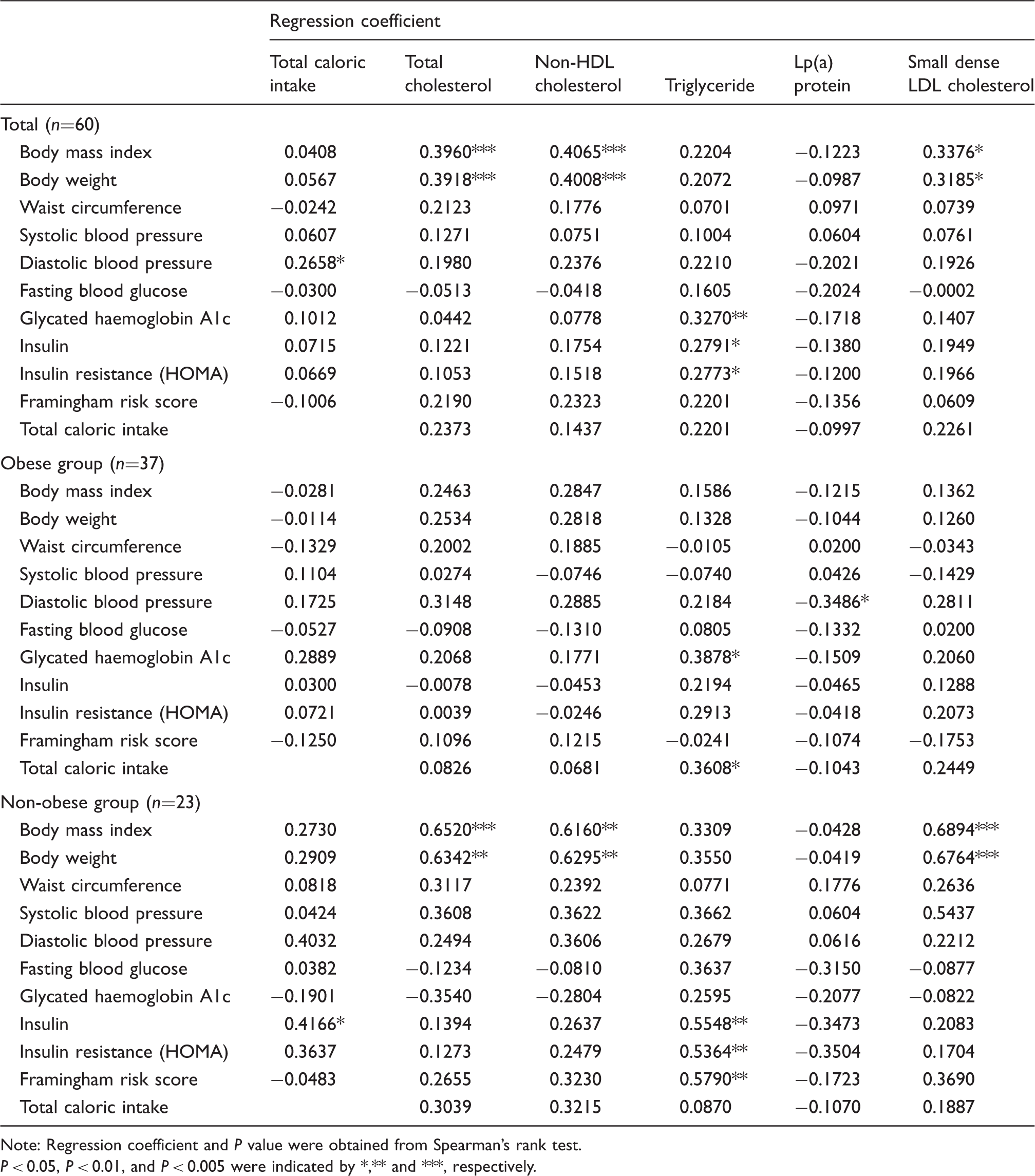
Note: Regression coefficient and
Comparison between changes in basic data and lipid profile obtained by AEX-HPLC method in females.
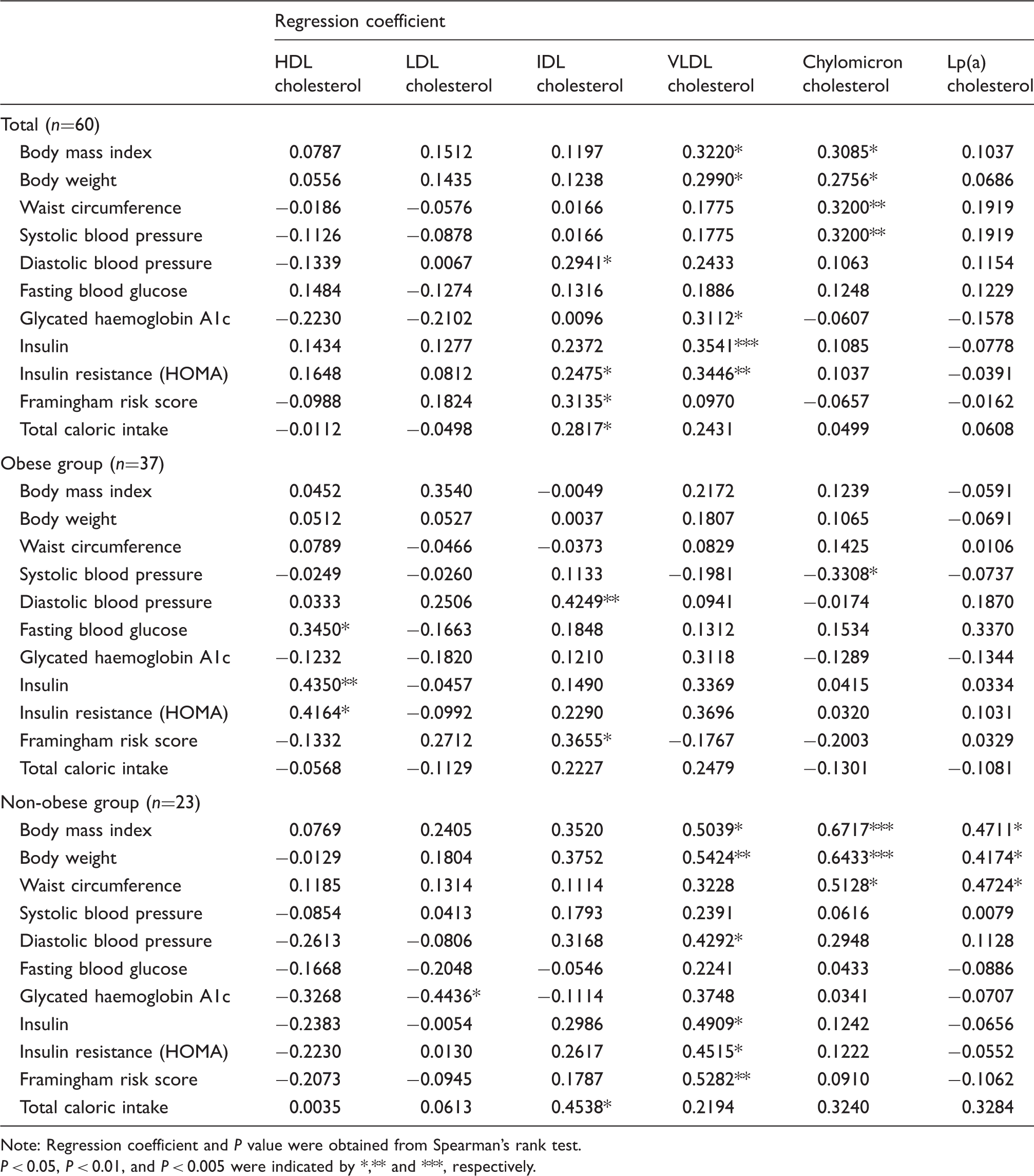
Note: Regression coefficient and
In comparison between the baseline data of basic data and those of lipids and lipoproteins, FRS and WC were highly correlated with TG and VLDL-C, and CM-C, respectively ( Correlation of Lp(a) cholesterol with the protein concentration. Data of baseline and after six months of dietary modification were indicated by closed and open circles, respectively.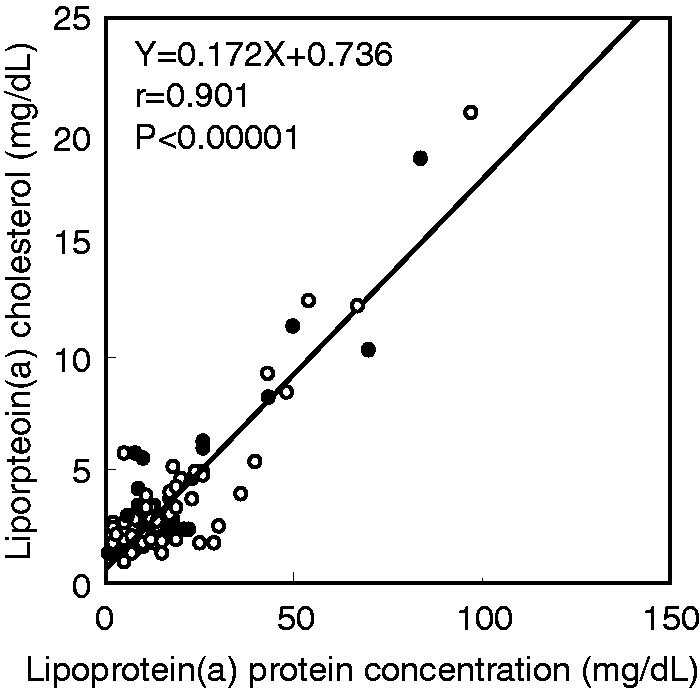
Discussion
We have estimated the change of lipid and lipoprotein profiles in Japanese females with six-month period of dietary modification by the calorie restriction and the change of caloric percentage from fat. Those changes during the six-month period were −283±322 kcal/day (−16.1±13.1%) and −2.3±5.6%, respectively, in the total group. Then, both BMI and WC decreased by 5% in the total group (Table 2). LDL-C, non-HDL-C and IDL-C decreased by 10%, 4% and 4%, respectively in the total group (Table 2). Additionally, those changes in the obese group were −321±363 kcal/day (−16.0±13.3%) and −1.3±6.3%, respectively. LDL-C, non-HDL-C and IDL-C decreased by 15%, 7% and 10%, respectively. The several previous papers showed the effect of diet, exercise and diet-plus-exercise on lipoprotein profile.25–27 Stefanick et al. reported that LDL-C of males and females significantly decreased by 12% and 9% in a one-year programme of diet-plus-exercise (National Cholesterol Education Program [NCEP] Step2 diet and aerobic exercise), respectively, but not significantly decreased in only diet programme. 25 They additionally showed that HDL-C did not significantly change by diet-plus-exercise or only diet programme. 25 Beard et al. reported that total cholesterol, LDL-C, HDL-C and glucose decreased by 20%, 20%, 17% and 16% in a three-week programme of diet-plus-exercise, respectively. 26 The diet programme was that the ratios of caloric intake from fat, protein and carbohydrate were <10%, 10–20% and 70–80%, respectively, and the exercise programme was that the exercise classes were held five days per week, and included 15–20 min of stretching and 45 min of aerobic exercise. 26 Varady et al. reported that LDL-C decreased by 10% and 8% in 12-week diet programmes, 75% and 25% energy restriction, respectively and HDL-C increased by 16% with three days per week of aerobic exercise. 27 Additionally, Varady et al. indicated that the proportion of small LDL particle size decreased in the programme of 75% energy restriction. 27 We indicated that small dense LDL-C decreased by 13% in the six-month period (Table 2). The cause for not increasing HDL-C (Table 2) may be that lipoprotein profile was improved only by dietary modification, not using exercise, in this study.
In our study, TG, VLDL-C and CM-C were not significantly decreased by the intervention of calorie restriction and the change of caloric percentage from fat. Stefanick et al. reported that TG was not significantly decreased in the one-year programme of diet-plus-exercise, and the baseline of TG was 159 mg/dL. 25 Beard et al. reported that TG decreased by 17% in a three-week programme of diet-plus-exercise, and the baseline of TG was 221 mg/dL. 26 Varady et al. reported that TG significantly decreased in a 12-week diet programme of 75% energy restriction, but not significantly changed in the 12-week diet programme of 25% energy restriction. 27 We thought that TG can be significantly decreased by extreme energy restriction, or in the case of high TG concentrations of subjects. In this study, the calorie restriction was 16±13%, and the baseline TG concentration was normal (113±56 mg/dL). Therefore, the change of TG was small (Table 2), and the correlations with BMI, BW, WC and FRS were not significant (Table 3). A recent paper reported that the reference range for VLDL-C was 19.8±23.8 mg/dL, 28 whereas in our study this was 13.3±8.7 mg/dL. The CM-C concentration in our study was low, 0.5±0.1 mg/dL, as shown in in Table 2. No decrease of VLDL-C and CM-C in our study might be caused by these low baseline concentrations.
In the Framingham Heart Study cohort, the correlation between lifestyle and the risk factor change of CHD in young adults during an eight-year follow-up period was reported.
13
The report showed that the change of BMI was significantly correlated with those of total cholesterol, LDL-C and VLDL-C, and significantly inversely correlated with that of HDL-C.
13
We indicated strong correlations of the change of BMI and BW with those of TC and non-HDL-C (
In our study, LDL-C, non-HDL-C and IDL-C decreased in the six-month period of dietary modification, but the cholesterol and protein concentrations of Lp(a) increased (Table 2). Many studies indicated that increased Lp(a)-P concentration is associated with the risk of CHD.16–18 It was shown that the risk of clinical atherosclerotic disease, e.g. CHD, intermittent claudication, congestive heart failure, increased by twofold to threefold with diabetes in the Framingham cohort study. 19 Prospective studies in US and Danish, however, showed that an increased Lp(a)-P concentration is associated inversely with a risk of incident type 2 diabetes in subjects without CHD. 20 Recently, a paper reported that increased Lp(a) was associated with worse outcomes in type 2 diabetic patients with CHD. 29 Seman et al. reported that an increased Lp(a)-C (>10 mg/dL) was an independent CHD risk in males with a relative risk of more than 2, and the Lp(a)-C concentrations were measured by using lectin affinity binding method. 30 A previous report indicated that high concentration of insulin suppressed apolipoprotein(a) synthesis in monkey hepatocytes. 31 In our study, insulin concentration decreased in the six-month period of dietary modification by calorie restriction (Table 2). Therefore, Lp(a)-C and Lp(a)-P in each of subjects might increase with the decreases of insulin and HOMA by the intervention of calorie restriction in our study. We thought that high Lp(a) concentration is the risk factor for CHD, but the Lp(a) concentration increases with the decreases of insulin and HOMA in individuals.
It is known that atherosclerotic lesion of early stage appears as chronic inflammation. 32 In the atherosclerotic vascular inflammation, platelet is activated and adhered onto the endothelial cells. 33 The endothelial cells expresse P-selectin glyocoprotein ligand-1 (PSGL-1) under an inflammation condition, and P-selectin which is up-regulated on activated platelets can be bound to the PSGL-1. 33 Apolipoprotein(a) was reported to be strongly bound to platelet activated with strong agonist, i.e. thrombin, arachidonic acid (AA), etc. 34 It was also reported that LDL and Lp(a) were found in atherosclerotic lesion, and Lp(a) was increased in advanced lesions. 35 In our study, blood pressures, LDL-C, FGB and A1c decreased during the six-month period of dietary modification and FRS decreased (Table 2). Therefore, the activated platelet and the bound Lp(a) might decrease with the improvement of inflammation in atherosclerotic lesions during the six-month period. For this reason, the cholesterol and protein concentrations of Lp(a) in serum might increase in the six-month period of dietary modification (Table 2). Increased Lp(a) concentration might be related to not only CHD risk but also insulin concentration or atherosclerotic vascular inflammation, but this remains to be elucidated. Nauck et al. indicated that the ratio of Lp(a)-C to Lp(a)-P was low in CHD patients with immunoassay for the measurement of Lp(a)-P and electrophoresis with ultracentrifugal separation for Lp(a)-C. 36 Konerman et al. indicated that HDL-C in the subjects with high Lp(a)-C and normal Lp(a)-P and high Lp(a)-C and high Lp(a)-P was higher than in the subjects with normal Lp(a)-C and high Lp(a)-P and normal Lp(a)-C and normal Lp(a)-P. 37 In our study, the ratios of Lp(a)-C to Lp(a)-P in baseline data and data after six months of dietary modification were similar.
It was reported that the increment from monounsaturated fatty acid (MUFA) and polyunsaturated fatty acid (PUFA) was inversely associated with CHD.
38
This study showed that the dietary intakes of saturated fatty acid (SFA), MUFA and PUFA significantly decreased by the calorie restriction and the change of caloric percentage from fat (<30%) in total subjects, and the ratio of MUFA and PUFA significantly increased (Table 1). Additionally, FRS significantly decreased in the intervention period (Table 2), but the change of FRS was not correlated with those of MUFA and PUFA. It was also reported that MUFA was positively correlated with TG and VLDL particle concentration, and n-6 PUFA was inversely correlated with those.
39
This study showed that MUFA and n-6 PUFA significantly decreased by the intervention (Table 1), but TG and VLDL-C were not significantly changed (Table 2). The change of MUFA was positively correlated with that of VLDL-C (
The study is limited by the small number of subjects and not including males and should be interpreted with caution. Additionally, the study is a one-armed design and does not contain the group without intervention by calorie restriction. We need a large scaled and randomized study containing the subjects with and without intervention by calorie restriction to further clarify the results obtained in our study.
In conclusion, this study showed that BMI, WC, HOMA and FRS were reduced by dietary modification with calorie restriction during the six-month period, and lipid and lipoprotein profiles were improved, but Lp(a) concentration increased. The high Lp(a) concentration is the risk for CHD, but the Lp(a) concentration may increase with the decreases in insulin and HOMA in individuals, but remains to be elucidated. It was additionally shown that the changes of BMI, WC and FRS were significantly correlated with those of TG-rich-lipoprotein; IDL, VLDL and CM.
Footnotes
Acknowledgements
We would like to thank Ms. Syuko Onodera for the excellent technical assistance.
Declaration of conflicting interests
YH was an employee of TOSOH Corp. (Tokyo, Japan) till 31 March 2015. DM is an employee of TOSOH Corp. (Tokyo, Japan).
Funding
Research funds were provided in part by the Kagawa Nutrition University Research Fund (to KK and AT) and in part by the Tosoh Research Fund (to DM).
Ethical approval
The study protocol was approved by the ethical committees of Kagawa Nutrition University (No. 222-G) and Tosoh Corporation (No. 10-02).
Guarantor
YH.
Contributorship
YH, DM and KH corrected data. YH contributed to the discussion and wrote the manuscript. YH and AT designed the study protocol. KK and AT reviewed and edited the manuscript.
