Abstract
Haemoglobinopathies may interfere with the haemoglobin A1c (HbA1c) measurement, leading to incorrect diagnosis and inappropriate treatment. It is essential that HbA1c assays are capable of identifying haemoglobinopathies. We report two cases of haemoglobin New York (HbNY) discovered through HbA1c analysis using capillary electrophoresis (Capillarys 2 Flex Piercing [C2FP], Sebia). We used these samples to evaluate the ability of three other HbA1c assays to identify this variant: ion-exchange high-performance liquid chromatography (Variant II Turbo [VII-T], Bio-Rad); boronate affinity high-performance liquid chromatography (Ultra2, Trinity Biotech) and immunoassay (Cobas c501 Tina-quant Generation 3, Roche Diagnostics). Each method was used for HbA1c assay of in samples from two cases of heterozygous haemoglobinopathy: β0-thalassemia/HbNY (Case 1) and HbA/NY (Case 2). Only the C2FP system detected HbNY (an additional peak appeared between HbA1c and HbA0). Clinical laboratories should be aware of the limitations of their HbA1c assay methods especially in geographic areas, where haemoglobinopathy prevalence is high.
Keywords
Introduction
Glycated haemoglobin (HbA1c) is formed by a non-enzymatic interaction of glucose with the N-terminal valine residue of the β-chain of haemoglobin A (HbA). HbA1c is an important marker of long-term glycaemic control of diabetes and more recently has been proposed as a diagnostic test for type 2 diabetes.1,2 Reliable quantitation of HbA1c is therefore of clinical importance. Various methods have been used to measure HbA1c based on different physico-chemical principles: capillary electrophoresis, ion-exchange high-performance liquid chromatography (HPLC), boronate affinity chromatography and immunoassay.
Haemoglobinopathies may impact on the reliability of HbA1c measurement, leading to incorrect diagnosis and inappropriate treatment. For example, an alteration in erythrocyte lifespan will cause a spurious HbA1c result, regardless of the measurement method used. A net charge difference may interfere with capillary electrophoresis or ion-exchange HPLC results. Furthermore, boronate affinity chromatography or immunoassay may be affected by a discrepancy in glycation rate between HbA and haemoglobin variants. Thus, each specific method for determining HbA1c should be evaluated with regard to the effect of haemoglobinopathy.
We report two cases of haemoglobin New York (HbNY) identified by HbA1c analysis using capillary electrophoresis (Capillarys 2 Flex Piercing [C2FP], Sebia) during an epidemiology study. These samples were evaluated in three other common HbA1c methods: ion-exchange HPLC, boronate affinity HPLC and immunoassay.
Methods
Haemoglobin separation
Haemoglobin separation was performed with a Capillarys 2 Flex Piercing (C2FP; Sebia, France) system (haemoglobin program) and Variant II (VII; Bio-Rad, Japan) system (β-thalassemia program) in accordance with the manufacturers’ instructions.
Haemoglobin variant confirmation
Genotype analysis was undertaken at the clinical laboratory center of Beijing Genomics Institute (Shenzhen, China). DNA sequencing of haemoglobin subunit α1, subunit α2 and subunit β was performed to identify mutations of the haemoglobin α- and β-chain genes. Multiplex-PCR and real-time PCR assays were performed to detect α- and β-chain gene deletions.3–5
HbA1c measurement
HbA1c values were quantified using four different methods, each in accordance manufacturers’ instructions: (1) capillary electrophoresis (C2FP; HbA1c program); (2) ion-exchange HPLC (Variant II Turbo [VII-T], Bio-Rad, Japan); (3) boronate affinity HPLC (Ultra2, Trinity Biotech, Ireland) and (4) immunoassay (Cobas c501 Tina-quant Generation 3, Roche Diagnostics, Germany). These methods have been certified by the International Federation of Clinical Chemistry and Laboratory Medicine and the National Glycohemoglobin Standardization Program.
Results
Case 1
A sample from a 19-year-old (non-diabetic) male participating in an epidemiological study was analysed for HbA1c values on the C2FP system. The absence of HbA1c and HbA0 peaks and the presence of two major unidentified peaks (93.6% and 6.4%) were noted (Figure 1). The results of other relevant tests were: haemoglobin, 130 g/L (reference range 130–175); mean corpuscular volume (MCV), 60.6 fL (82–100); mean corpuscular haemoglobin (MCH), 19.5 pg (27–34); red cell distribution width (RDW), 15.1% (11.6–14.6) and glucose 4.40 mmol/L (3.9–6.1).
The HbA1c analysis by the Sebia C2FP HbA1c program and haemoglobin analysis by Sebia C2FP haemoglobin program. Reference: HbA/A. Case 1: heterozygous β0-thalassemia/HbNY. Case 2: heterozygous HbA/NY.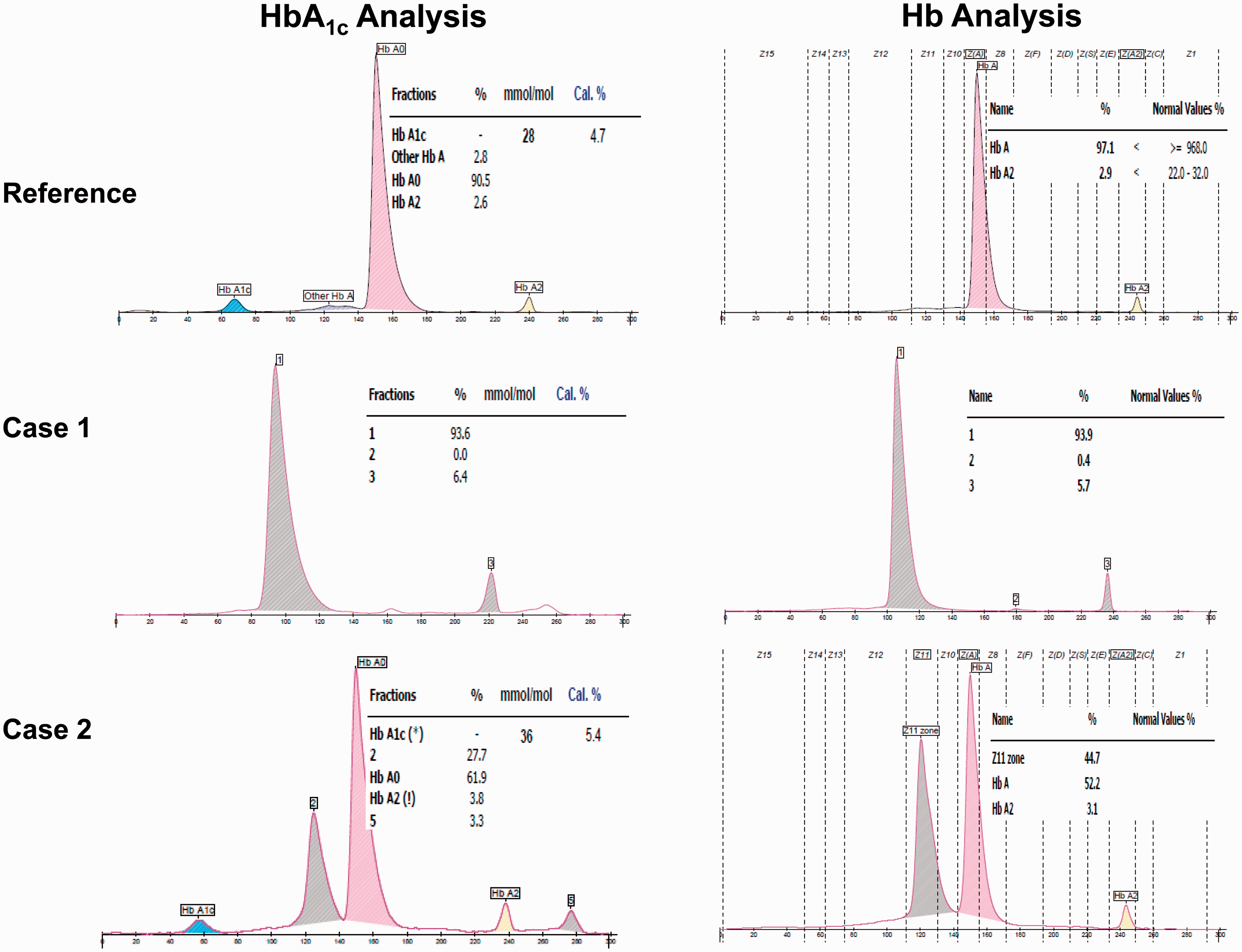
Haemoglobin electrophoresis result (C2FP) showed two main unidentified peaks (93.9% and 5.7%) and the absence of an HbA peak (Figure 1). However, haemoglobin analysis performed by the VII revealed a normal pattern, with HbA0 quantified at 83.2% and HbA2 at 5.5%; HbA1c analysis performed by VII-T showed an HbA1c peak of normal appearance quantified at 31 mmol/mol (5.0%; Figure 2). Given the reduced MCV and MCH concentrations and elevated HbA2 (VII), the possibility that the subject was a β-thalassemia carrier was investigated. Haemoglobin electrophoresis performed by C2FP indicated the presence of an unidentified haemoglobin. DNA sequencing revealed that the subject was a compound heterozygous of β0-thalassemia (Codon 17 A→T) and HbNY β-globin mutation (β113 GTG→GAG; Val→Glu). The HbA1c values obtained via the Ultra2 and Cobas c501 were 26 mmol/mol (4.5%) and 31 mmol/mol (5.0%), respectively.
HbA1c analysis by the Bio-Rad VII-T and haemoglobin analysis by the Bio-Rad VII β-thalassemia program. Case 1: heterozygous β0-thalassemia/HbNY. Case 2: heterozygous HbA/NY.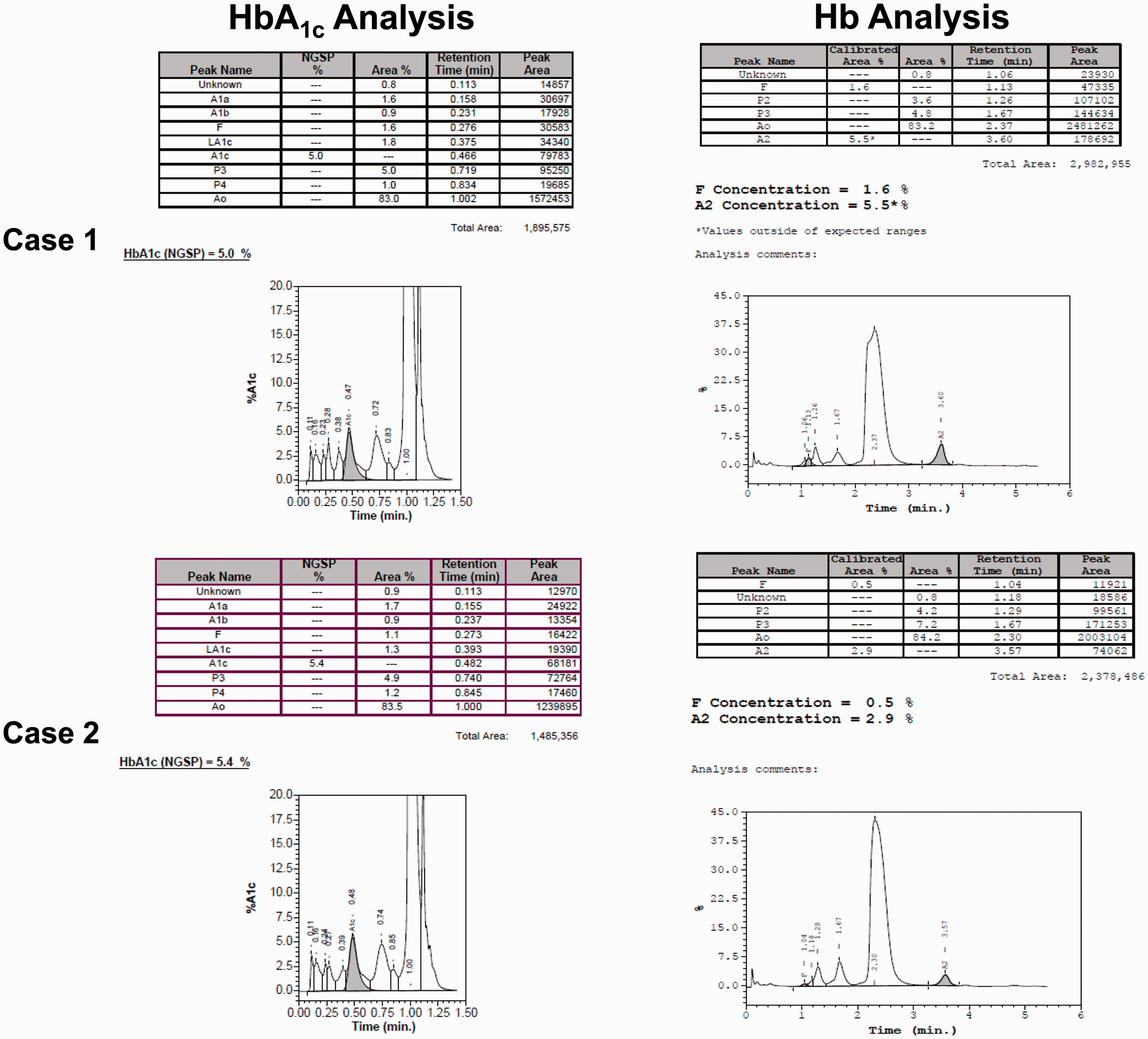
Case 2
A sample from a 69-year-old (non-diabetic) male also participating in the epidemiological study was analysed for HbA1c on C2FP system. This showed the presence of an additional peak, completely separate from the HbA0 and HbA1c peaks, with the HbA1c measured at 34 mmol/mol (5.4%; Figure 1). The results of the relevant tests were: haemoglobin 154 g/L; MCV 94.4 fL; MCH 30.6 pg; RDW 12.7% and glucose 5.4 mmol/L.
The chromatogram on the VII for haemoglobin analysis showed a normal pattern (HbA0 84.2% and HbA2 2.9%); the chromatogram on VII-T for HbA1c analysis showed an HbA1c peak of normal appearance quantified at 36 mmol/mol (5.4%; Figure 2). The presence of a haemoglobin variant was confirmed by capillary electrophoresis with an abnormal peak noted in the Z11 zone (44.7%), HbA 52.2% and HbA2 3.1% (Figure 1). There are 15 specific zones in the Phoresis software of the C2FP haemoglobin program. Common Hb fractions appear in the 15 zones as following: HbCS = Z2, HbA2 = Z3, HbE = Z4, HbF = Z7, HbA = Z9, HbNY = Z11, HbBart’s = Z12 and HbH = Z15. 6 The possibility that the subject was a HbNY carrier was investigated. DNA sequencing revealed that this subject was heterozygous HbA/NY. HbA1c measurement on the C2FP, VII-T, and Cobas c501 all gave a result of 34 mmol/mol (5.4%), while that on the Ultra2 was 32 mmol/mol (5.1%).
Discussion
Haemoglobinopathies may be divided into two subgroups: those causing decreased expression (thalassemia) or a haemoglobin structural defect (haemoglobin variants). Either subgroup may impact on HbA1c measurement either by direct interference in the analytical system or indirectly, through an effect on reduced red cell survival or an altered glycation with HbA. It is therefore important that HbA1c assays are capable of identifying both thalassemia and haemoglobin variants, especially in populations in which haemoglobinopathies are prevalent.
HbNY is a β-globin variant in which valine replaces glutamate at position 113. 7 HbNY was first identified in 1967. Although HbNY is one of the most common β-globin variants in Southern China, subjects who are heterozygous for HbNY are asymptomatic with normal haemoglobin, MCV and MCH concentrations. Most of these subjects are undetected, while a small number may be identified incidentally if they co-inherit thalassemia or other variants. Given that HbNY heterozygosity is asymptomatic, it is likely that the global prevalence is underestimated.
HPLC and capillary electrophoresis are currently the two most commonly used methods for detection of haemoglobinopathy. A previous report 6 and our present haemoglobin electrophoresis results indicate that HbNY can be separated from HbA easily and quantified accurately with a capillary electrophoresis C2FP haemoglobin program. Capillary electrophoresis is a relatively new method for HbA1c measurement that may allow separation of common and rare haemoglobin variants from the HbA0 fraction. In addition, the C2FP analyser provides a rapid and reliable separation of HbA2 during HbA1c measurement, thus allowing for the detection of β-thalassemia. 8 In our present study, HbA1c analysis using the C2FP revealed a complete separation of HbNY from HbA0 or HbA1c (Case 2), and for this heterozygous HbA/NY sample, it could generate an accurate HbA1c result. For the compound heterozygous β0-thalassemia/HbNY sample (Case 1), C2FP was the only method to report no measurable HbA1c in the sample.
Ion-exchange HPLC is a recognized technique in screening for haemoglobinopathy. However, it is not able to separate HbNY from HbA, since they have identical migration profiles. 6 Because in the ion-exchange HPLC β-thalassemia program, the retention times of HbNY and HbA were identical, we hypothesized that the ion-exchange HPLC system could not distinguish HbNY0 from HbA0 or HbNY1c from HbA1c. On the VII-T system, this hypothesis was confirmed when the samples from Cases 1 and 2 showed normal patterns with no additional peak. Thus, the ion-exchange HPLC system VII-T also could not report the possible presence of HbNY during the HbA1c analysis. As HPLC relies on retention time data, previous studies also found that some variants could not be observed, or significantly affect the HbA1c value when using ion-exchange HPLC.9,10 The capillary electrophoresis C2FP system has advantages over the HPLC VII-T for HbA1c analysis, at least for the detection of thalassemia and some variants such as HbNY.
Using ion-exchange HPLC, the glycation rate for a heterozygous HbA/NY sample is the sum of HbNY1c and HbA1c. With boronate affinity methods, the HbA1c value is the ratio of the glycated fraction to the non-glycated fraction; this method measures total glycated haemoglobin (i.e. HbA1c and haemoglobin glycated at other sites). In immunoassay methods, HbA1c antibodies recognize the N-terminal glycated amino acids of the haemoglobin β chain. As long as the mutation is far from the antigen–antibody binding site, it is unlikely to interfere in the assay results. It is unfortunate that not all of these three methods could report the possible presence of this variant. The influence of the HbNY variant on erythrocyte lifespan or glycation rate in comparison with HbA is unknown. Consequently, HbA1c cannot be used for the diagnosis of diabetes in subjects with HbNY variant and further studies are required.
In this study, we have shown that the capillary electrophoresis Sebia C2FP system allows the detection of HbNY during haemoglobin electrophoresis and HbA1c assay, while this variant is not detected by ion-exchange HPLC Bio-Rad VII-T, boronate affinity HPLC Biotech Ultra2 or immunoassay Roche Cobas c501 Tina-quant Generation 3 during HbA1c assay. Our study stresses the necessity for clinical laboratories to be aware of the limitations of their HbA1c assay methods, especially for populations in which haemoglobinopathies are common.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research was supported by grants from the National Key Technologies R&D Program of China (2012BAI37B01), the National Special R&D Program of Major New Drugs of China (2012ZX09303009-003), the National Natural Science Foundation of China (81572088), the Natural Science Foundation of Guangdong Province (2015A030313340) and the Foundation of Guangdong Provincial Hospital of Chinese Medicine (2014KT1593).
Ethical approval
This study was approved by the Research and Ethics committees of the first hospital affiliated to China Medical University (2013-172-2). The subjects were informed and signed the study contents.
Guarantor
XZH.
Contributorship
YC drafted the manuscript; YC, ZMW and XBW collected data; YC, ZMW, XBW, FQ and YXW performed laboratory detection; XZW, PFK, JHX and JHZ helped in designing the study; XZH designed the study. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
