Abstract
Background
The peptide hormone oxytocin acts in the central nervous system and plays an important role in various complex social behaviours. We report the production of a high affinity and specificity antibody for oxytocin and its use in a highly sensitive enzyme immunoassay. Biotin that was chemically bound to oxytocin derivative containing zero to six lysines as bridge was the labelled antigen. Seven labelled antigens were used to develop a highly sensitive enzyme immunoassay.
Methods
Antioxytocin antiserum was obtained by immunization of oxytocin-bovine thyrogloblin conjugate to rabbit. Oxytocin sample was added to the second antibody-coated microtitre plate and allowed to react overnight at 4℃, then biotinylated oxytocin was added 1 h at 4℃, and horseradish peroxidase-labelled avidin was added and incubated for 1 h at room temperature. The plate was then washed. Horseradish peroxidase activity was measured by a colorimetric method using o-phenylenediamine (490 nm).
Results
The sensitivity of the enzyme immunoassay improved as the number of lysine residues increased; consequently, biotinylated oxytocin bridged with five lysines was used. A standard curve for oxytocin ranged from 1.0 to 1000 pg/assay. The detection limit of the assay was 2.36 pg, and the reproducibility was 3.6% as CV% (n = 6). Cross-reactivity with vasopressin and vasotocin was less than 0.01%.
Conclusion
The sensitivity of the enzyme immunoassay could be improved by increasing the number of lysine residues on the biotin-labelled antigen. The proposed method is sensitive and more specific than conventional immunoassays for oxytocin and can be used to determine plasma oxytocin concentrations.
Keywords
Introduction
Oxytocin, which has a molecular weight of 1007 Da, is synthesized in the hypothalamus and secreted from the posterior lobe of the pituitary. The molecule consists of nine amino acids linked with a [1–6] disulfide bond and a semi-flexible carboxyamidated tail.
Oxytocin promotes the action of uterotonic and galactopoiesis in peripheral organs1,2 and is used as a delivery accelerator. In addition, oxytocin acts in the central nervous system of males and females and plays an important role in a variety of complex social behaviours including affiliation, sexual behaviour, social recognition, stress buffering, aggression and trust.3–12 Researchers have an increased interest in measuring peripheral oxytocin concentrations to better understand the role of this peptide in human social behaviour. Thus, the measurement of oxytocin is important in clinical practice.
The concentrations of oxytocin in plasma are typically measured by radioimmunoassay (RIA) and enzyme immunoassay (EIA). Oxytocin can also be measured in body fluids such as cerebrospinal fluid, saliva or urine. 13 Sample preparation often involves extraction,14–17 but several reports have used direct measurement.11,18 The reported concentrations of plasma oxytocin vary from 1 to 300 pg/mL in humans and other mammalian species. 13 Significant differences exist between values observed in similar populations and are likely due to methodological differences, making comparison between studies difficult. 13 In addition, many reported methods lack sufficient sensitivity and specificity for oxytocin.
The current oxytocin immunoassay is done by EIA using horseradish peroxidase (HRP) or alkaline phosphatase as a label enzyme. The specific activity of the enzyme-labelled antigen is higher than isotopes due to amplification of the enzyme. Therefore, in many cases, EIA is more sensitive than RIA.
However, the sensitivity of EIA for low-molecular weight antigens (haptens) is often reduced when the same hapten derivative is present on the immunogen for the carrier protein conjugate and the labelled antigen with a probe (e.g. enzyme or biotin), because the affinity of the antibody for the labelled antigen is thought to be stronger than to a non-labelled antigen.
In EIAs of haptens, a system in which the same hapten derivative is used both for preparing the immunogen and the hapten-enzyme conjugate is referred to as ‘homologous’, in contrast to ‘heterologous’ systems in which different hapten derivatives are used.
In this paper, homologous and heterologous systems were established by utilizing different bridge lengths (lysine residues) between the antigen and carrier protein or biotin probe. We developed an EIA with sufficient sensitivity to measure oxytocin in the blood by using a heterologous bridge system.
Materials
Oxytocin, [Arg8]-vasopressin, [Lys8]-vasopressin, [Arg8]-vasotocin, bovine serum albumin and o-phenylenediamine were obtained from Sigma Chemical Co (USA). HRP-labelled avidin was from Thermo Fisher scientific (USA). The oxytocin EIA kit was from Enzo Life Sciences, Inc (USA). The microtitre plates coated with secondary antibodies were from Eiken Chemical Co. (Japan). Sep-pack (C18, 200 mg) was from Nihon Waters K.K.(Japan). Oxytocin-(Lys)n derivatives (n = 0, 1, 2, 3, 4, 5, 6) were prepared by the peptide synthesizer in Immuno-Biological Laboratories Co. Ltd (IBL, Japan).
Reagents
Preparation of biotinylated oxytocin
Oxytocin derivatives containing zero to six lysine residues were chemically labelled with N-succinimidyl D-biotin at the N-terminal domain. The obtained biotinylated oxytocin probes were purified by HPLC using C18 RT column (14.6 mm× 250 mm, 5 μm, buffer A; 0.1% trifluoroacetic acid (TFA) in water, buffer B; 0.1% TFA in acetonitrile, gradient linear = 10–40% buffer B in 20 min). The structural analysis of synthesized biotinylated oxytocin probes was carried out by electrospray-mass spectrometry (MS). The MS spectral analysis exhibited expected molecular weight for each biotinylated oxytocin probe (biotinylated-(Lys)n-oxytocin; n = 0: MW 1233.5, n = 1: MW 1361.7, n = 2: MW 1489.9, n = 3: MW 1618, n = 4: MW 1746.2, n = 5: MW 1874.4, n = 6: MW 2002.6). These biotinylated oxytocin was stored at 1 mg/mL in H2O at −20℃ and diluted with assay buffer (see below) before use.
Antioxytocin antiserum
Antioxytocin antiserum was obtained by immunizing rabbits with oxytocin-(Lys) n =2-bovine thyrogloblin conjugate prepared by the glutaraldehyde method in IBL. Purification of the antibody was performed from the immunoglobulin G (IgG) fraction using saturated ammonium sulphate and with affinity chromatography. The column for affinity chromatography was prepared with the same immunogen (oxytocin-(Lys) n =2-bovine thyrogloblin conjugate) immobilized on a Formyl caro fine column. Acetate buffer (10 mmol/L, pH 2.5) was used as the eluent. The eluted antibodies were stored frozen at 0.5 mg/mL and diluted with assay buffer before use.
Buffer and reagent solutions
Assay buffer
A 10 mmol/L phosphate buffer (pH 7.4) containing 0.9% NaCl and 0.1% bovine serum albumin was used.
Wash buffer
A 10 mmol/L phosphate buffer (pH 7.4) containing 0.9% NaCl and 0.1% polyoxyethylene (20) sorbitan monolaurate was used.
o-Phenylenediamine solution:
Anhydrous citric acid (5.5 g), 6.2 g trisodium citrate dehydrate, and 0.5 mL hydrogen peroxide (30%) were dissolved in 1000 mL H2O.
Stop solution for the colorimetric method: 2 mol/L sulphuric acid was used.
Apparatus
The WALLAC ARVOTM SXd 1420 MULTILABEL COUNTER (PerkinElmer) was used.
Human plasma sample
Human plasma sample was obtained from Showa University Hospital. The study was approved by the Ethics Committees of the Showa University Hospital, Reference number 123.
Methods
Assay method
Assay method: the oxytocin sample (100 μL) and 50 μL antioxytocin antibody were added to a microtitre plate coated with secondary antibody and incubated overnight at 4℃. Biotinylated oxytocin (50 μL) was then added and incubated for 1 h at room temperature. The plate was washed three times with 350 μL wash buffer, followed by addition of 200 μL HRP-labelled avidin and incubation for 1 hour at room temperature. The plate was washed three times in the same way, and 200 μL o-phenylenediamine solution was added and reacted for 1 h at room temperature. Finally, 50 μL stop solution was added, and the absorbance was measured at 490 nm.
Extraction of oxytocin from human plasma samples
The plasma sample was added to an equal volume of 0.1% TFA in water. The solution was centrifuged at 17,000 g for 15 min at 4 ℃ to clarify the solution, and the supernatant was saved. The Sep-pack column (C18, 200 mg) was equilibrated with 1 mL acetonitrile, followed by 10.5 mL 0.1% TFA in water. The supernatant was applied to the Sep-pack column, which was washed with 10.5 mL TFA in water. We eluted the sample slowly by applying 3 mL acetonitrile: 0.1% TFA in water (95:5). The eluant was collected in a tube, evaporated to dryness using a centrifugal concentrator under vacuum and reconstituted with assay buffer. This method was done with an oxytocin EIA kit.
Statistics
Statistics used in this study were the standard deviation (SD) and coefficient of variation (CV) and a correlation coefficient.
Results and discussion
List of biotinylated oxytocin derivative and its structure.
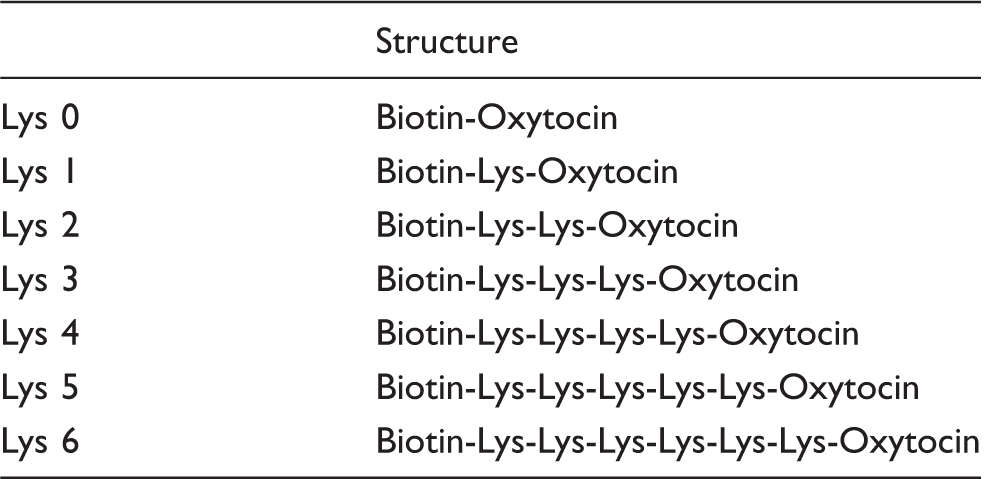
Lys: lysine.
(1) Reactivity of the labelled antigen for the antioxytocin antibody
We examined the reactivity of the seven biotinylated oxytocin derivatives for the antioxytocin antibody using the following dilutions: antioxytocin 1:1000, HRP-labelled avidin 1:4000 and biotinylated oxytocin 1:25,000. The bound reactivity of biotinylated oxytocin for the antibody was increased in proportion to the number of lysine residues used as the bridge. Maximum binding was obtained when using five lysine residues as the bridge, as shown in Figure 1. Increasing the binding reactivity allowed a greater dilution of the antibody and labelled antigen. As a result, the development of a highly sensitive EIA can be expected. Labelled antigen with five lysine residues was considered the optimal bridge.
(2) The effect of sensitivity using various labelled antigens on the competitive immunoassay
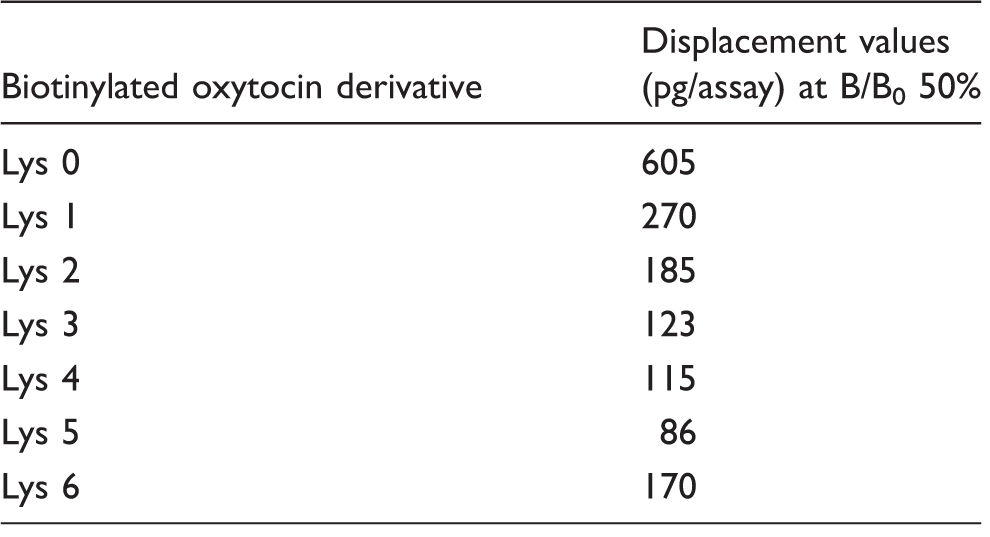
We examined the effect of the labelled antigen on the competitive immunoassay under optimal conditions which the concentrations of antibody and labelled antigen is set to be about 1.0 as OD value of B0%. The high OD value is needed in order to improve the reproducibility of the assay. And we investigated the immunoassay sensitivity using a combination of each labelled antigen and antioxytocin antibody on bridge homologous and heterologous combination system. In this study, since the antibody is prepared by immunogen of oxytocin – (Lys) n =2 – bovine thyrogloblin conjugate with two lysine, that is, the use of biotinylated-(Lys)2-oxytocin is homologous system and other probes with different bridge lengths (0,1,3,4,5,6) in lysine number are correspond to heterologous system. The sensitivity was compared with the oxytocin value that is required to displace 50 % of the B0. As this value decreases, the immunoassays show high sensitivity. The B/B0 50% bound value decreased when the number of lysine residues as a bridge increased, and a minimum value was obtained with five lysines. With more than five lysines, the B/B0 50% bound value increased, as shown in Table 2. Measuring a small amount of oxytocin was impossible.
The results of the above (1) and (2) can be considered as follows. Figure 1 shows the affinity of biotinylated-(Lys)
n
=0–6-oxytocin to the antibody.
Antibody binding ability B0 values (signal/noise ratio).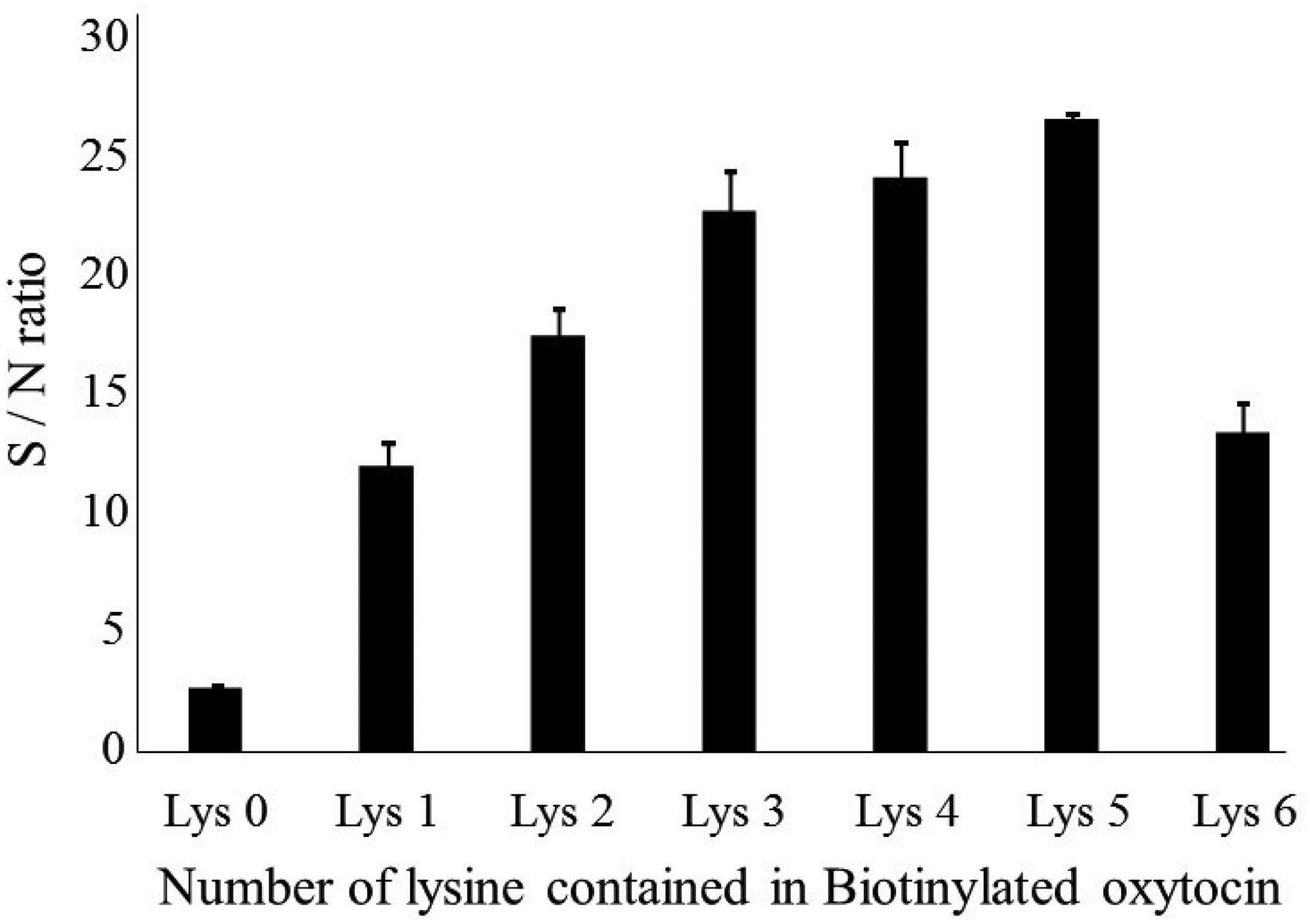
As a result, this antibody showed the strongest affinity to biotinylated oxytocin with five lysines. On the other hand, Table 2 shows the affinity of the antibody for non-labelled oxytocin. It shows the displacement effect in the competitive reaction. In this experiment, biotinylated oxytocin and antibody were used with the concentration optimized for each combination. That is, for the strong affinity combination of antibody and biotinylated oxytocin, a greater dilution can be used. As a result, biotinylated oxytocin with five lysine showed the highest sensitivity.
We concluded that a labelled antigen with five lysines on bridge heterologous combination was the most suitable for EIA.
In the following experiments, labelled antigen with five lysine residues was used as the optimal bridge.
(3) Standard curve
A standard curve for oxytocin was plotted under the optimal conditions using biotinylated (Lys)5 oxytocin as follows: antioxytocin 1:2000, HRP-labelled avidin 1:4000 and labelled antigen 1:50,000. The range of the standard curve for oxytocin was 1.0 to 1000 pg/assay (10 to 10,000 pg/mL), as shown in Figure 2. The detection limit of the assay was 2.36 pg/assay (B0 – 3SD%). The B/B0 50% bound value of the standard curve was 63 pg/assay. The reproducibility of each point in the standard curve was shown as a coefficient variation (n = 6); blank at absence of antibody: 4.0%, B0: 6.3%, B1pg: 1.9%, B2.5 pg: 5.0%, B5pg: 2.5%, B10pg: 2.5%, B25pg: 3.0%, B50pg: 1.6%, B100pg: 5.7%, B250pg: 3.8%, B500pg: 3.1%, B1000pg: 3.7%, with an average of 3.6%.
(4) Cross-reactivity
The cross-reactivities of three oxytocin-like peptides, [Arg8]-vasopressin, [Lys8]-vasopressin and [Arg8]-vasotocin, were examined with cross-reactivity tests at a range of 0 to 10,000 pg/assay. The cross-reactivity for all three compounds was less than 0.01%, indicating that this antiserum is highly specific for oxytocin. As described above, because our antibodies showed very high specificity, we tried to directly measure oxytocin in plasma without extraction. A low correlation (r = 0.2, n = 20) was found between the extraction method using the Sep-pack column and the direct method (no extraction). The likely reason is the presence of multiple immunoreactive products in addition to oxytocin in plasma samples. 13 Therefore, a pretreatment was used for oxytocin immunoassay. In general, Sep-pack C18 column method is used. As another extraction method, a method using an organic solvent can be used, but the extraction rate is reported to be low. 13 Therefore, we used Sep-pack C18 column as the extraction method of oxytocin.
(5) Recovery study
The recovery % was calculated by dividing the value read from the standard curve with the theoretical amount. A known amount (theoretical amount) of oxytocin was added to pool plasma samples containing endogenous oxytocin. As shown in Table 3, the recoveries for 500 pg /ml and 1000 pg/ml spiked were 104.5% and 95.2%, respectively.
(6) Precision
Plasma samples were pretreated using a solid-phase extraction method as recommended by the manufacturer of the oxytocin EIA kit (Enzo Life Sciences). Intra- and inter-assay precision was evaluated by testing three pretreated plasma samples A, B and C. The average amount of oxytocin was 16.0 pg/assay in sample A, 37.3 pg/assay in sample B and 33.5 pg/assay in sample C. The intra-assay coefficient of variation (CV%, n = 10) was 2.9% in sample A, 3.2% in sample B and 3.8% in sample C. The inter-assay CV% (n = 5) was 17.2% in sample A, 16.8% in sample B and 12.0% in sample C. From the above result, this method was highly reproducible.
(7) Correlation analysis
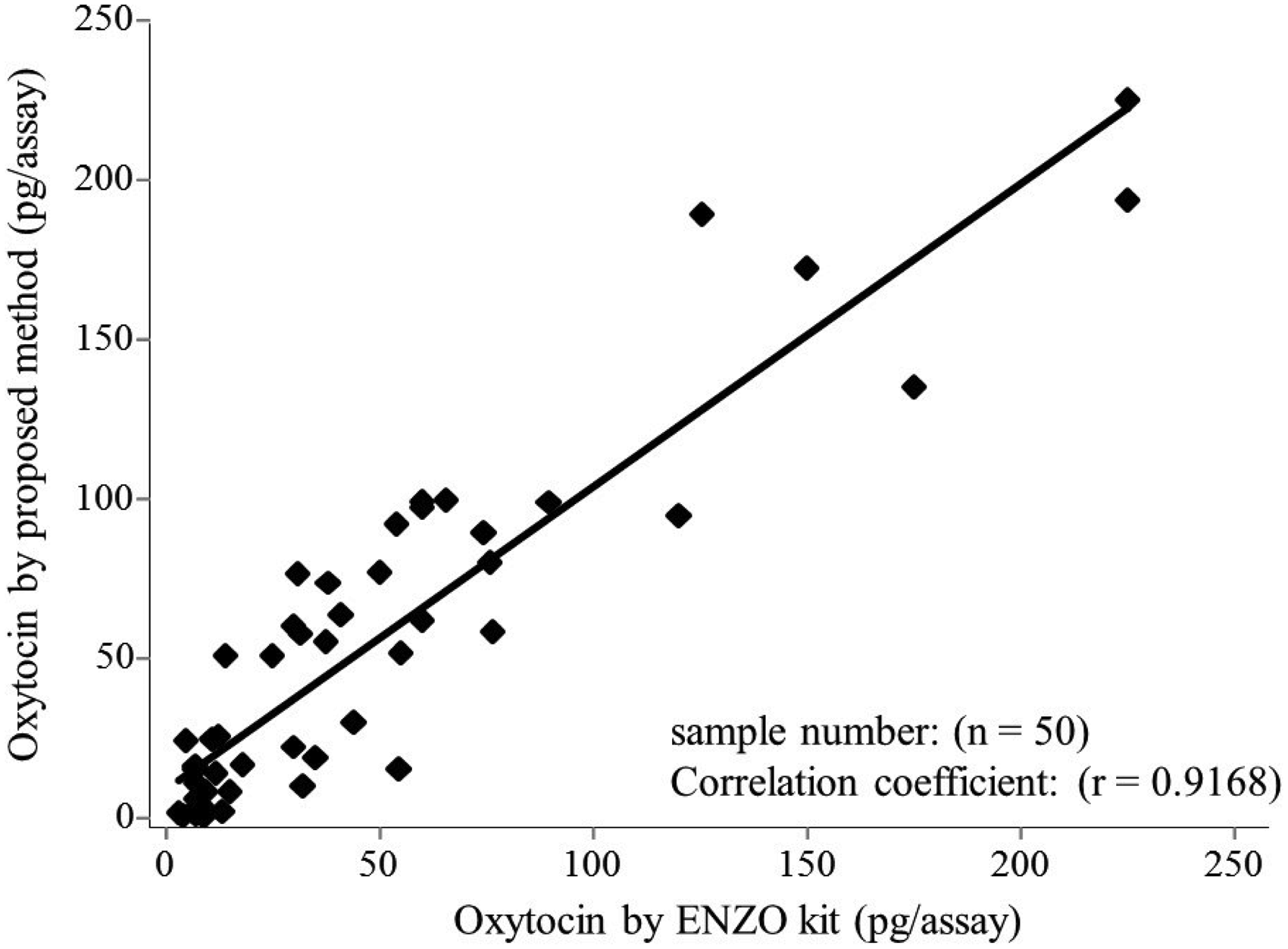
Using the present EIA and Enzo kit EIA, we assayed 50 plasma samples in duplicate. The linear correlation coefficient between the oxytocin values determined by these two methods was 0.917 (y = 0.950 x + 8.584), as shown in Figure 3. Therefore, this method was found to be sufficiently applicable to measurement of clinical samples.
Conclusion
This method is based on the principle of competitive EIA using an antioxytocin antibody. The reactivity of seven biotinylated oxytocin derivatives to the antioxytocin antibody was examined to develop a highly sensitive oxytocin EIA.
The sensitivity of the EIA could be improved by increasing the number of lysine residues on the biotin-labelled antigen. The sensitivity was 2.36 pg/assay (B0 – 3SD%) as detection limit under the optimal condition. This was corresponding to that (1.5 pg/assay) of the oxytocin enzyme-linked immunosorbent (Enzo) kit. And cross-reactivity of proposed method is less than 0.01% with respect to Arg8-vasotocin. In contrast, the cross-reactivity of the kit showed 7.5%. Therefore, this proposed method is sensitive enough to measure oxytocin in blood also showed a higher specificity than conventional immunoassay.
Moreover, the sensitivity of the immunoassay using the bridge heterologous combination system devised in this study indicates that a similar method is likely to be useful as a highly sensitive immunoassay for other small peptides.
Standard curve for oxytocin. Correlation between proposed method and Enzo kit values of oxytocin in plasma. The effect of the number of lysine residues as a bridge on immunoassay sensitivity. Lys: Lys,0,1,2,3,4,5,6: number of lysine residues used as a bridge between oxytocin and biotin. Recovery of oxytocin from plasma (n = 5).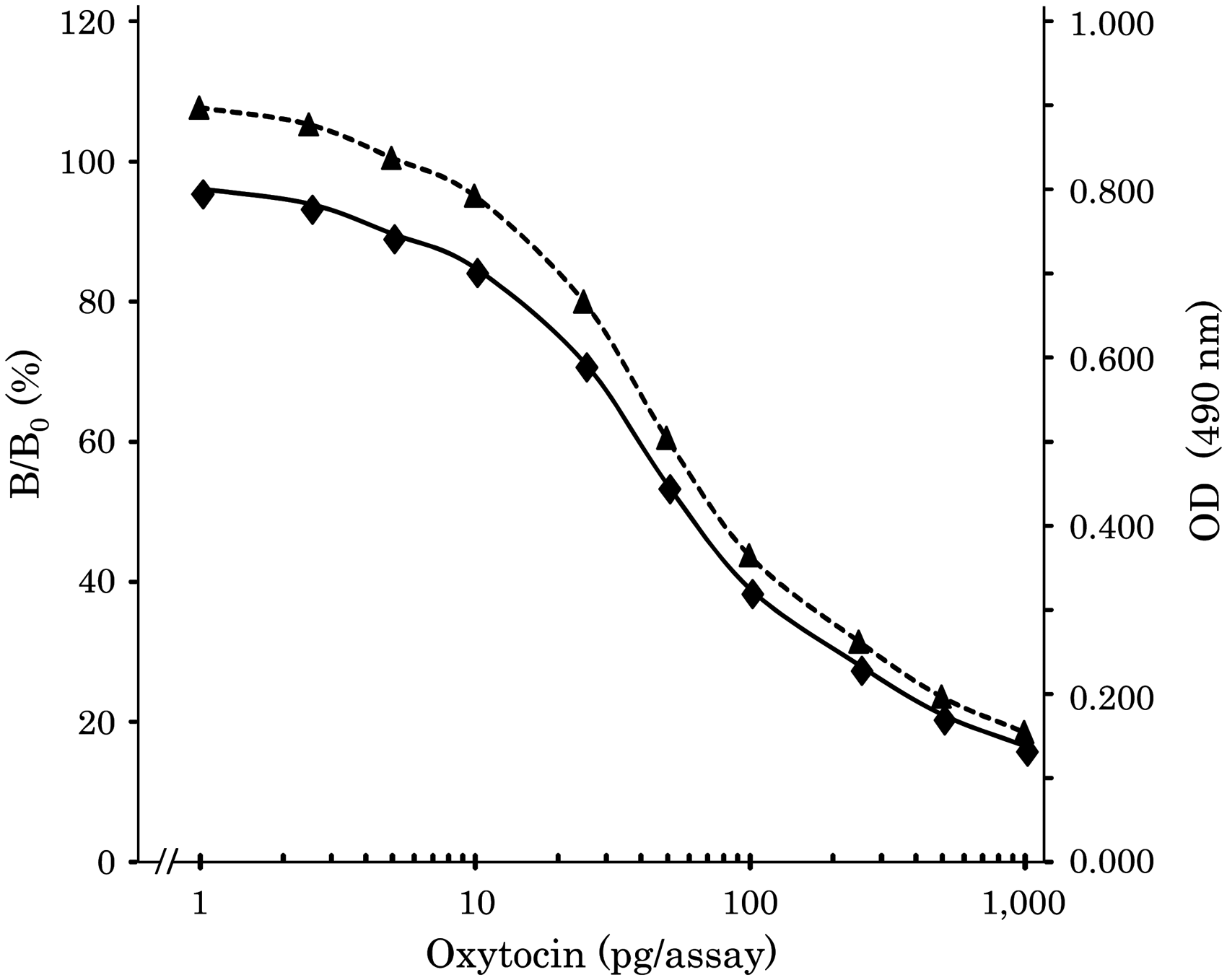

Footnotes
Acknowledgements
This research was supported by JST, CREST.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The study was approved by the Ethics Committees of the Showa University Hospital, Reference number 123.
Guarantor
HA.
Contributorship
HA designed this study. SH and HA wrote this manuscript. KK, YS and KO were also involved in the EIA development. NK was involved in gaining ethical approval and plasma sample. All authors reviewed the manuscript and approval the final version.
